Abstract
Background:
Arterial calcification due to deficiency of CD73 (ACDC; OMIM 211800) is a rare genetic disease resulting in calcium deposits in arteries and small joints causing claudication, resting pain, severe joint pain, and deformities. Currently, there are no standard treatments for ACDC. Our previous work identified etidronate as a potential targeted ACDC treatment, using in vitro and in vivo disease models with patient-derived cells. In this study, we test the safety and effectiveness of etidronate in attenuating the progression of lower-extremity arterial calcification and vascular blood flow based on the computed tomography (CT) calcium score and ankle–brachial index (ABI).
Methods:
Seven adult patients with a confirmed genetic diagnosis of ACDC were enrolled in an open-label, nonrandomized, single-arm pilot study for etidronate treatment. They took etidronate daily for 14 days every 3 months and were examined at the NIH Clinical Center bi-annually for 3 years. They received a baseline evaluation as well as yearly follow up after treatment. Study visits included imaging studies, exercise tolerance tests with ABIs, clinical blood and urine testing, and full dental exams.
Results:
Etidronate treatment appeared to have slowed the progression of further vascular calcification in lower extremities as measured by CT but did not have an effect in reversing vascular and/or periarticular joint calcifications in our small ACDC cohort.
Conclusions:
Etidronate was found to be safe and well tolerated by our patients and, despite the small sample size, appeared to show an effect in slowing the progression of calcification in our ACDC patient cohort.
Keywords
Background
Arterial calcification due to deficiency of CD73 (ACDC) is an autosomal recessive ectopic mineralization syndrome caused by loss-of-function (LOF) disease-causing variants in the 5′-nucleotidase ecto (NT5E, HGNC:8021) gene encoding for CD73. 1 ACDC leads to de novo vascular calcifications developing in the media of medium to large size arteries with massive calcifications at levels not often observed in other vascular calcification diseases, affecting mostly the lower extremities, although most recently described in the upper extremities as well,2,3 but also the periarticular area of multiple joints. ACDC is considered a very rare disease with fewer than 20 patients identified worldwide and an estimated prevalence of less than 1 in 1,000,000. 4 The clinical symptoms of ACDC include lower-extremity claudication, chronic ischemic pain of the feet at rest with threat of potential limb loss, and also manifests with debilitating arthritic episodes in multiple joints that have been treated with multiple agents without complete resolution. Patients with ACDC present with joint symptoms at a mean age of 17 and develop claudication symptoms at a mean age of 29. 5 Figure 1 shows radiographs highlighting the typical calcium deposits that develop within the popliteal artery and around the hand joints of patients with ACDC because of the CD73 deficiency. Although exercise improves claudication symptoms to a certain degree without achieving symptom resolution, patients with ACDC are currently treated with aggressive management of cardiovascular risk factors, use of antiplatelets, and different revascularization procedures (bypass graft, angioplasty, endarterectomy, and/or stenting) when indicated. Since the identification of NT5E variants that lead to ACDC, we have continued to study the natural history, clinical presentation, and pathological mechanisms of this condition.
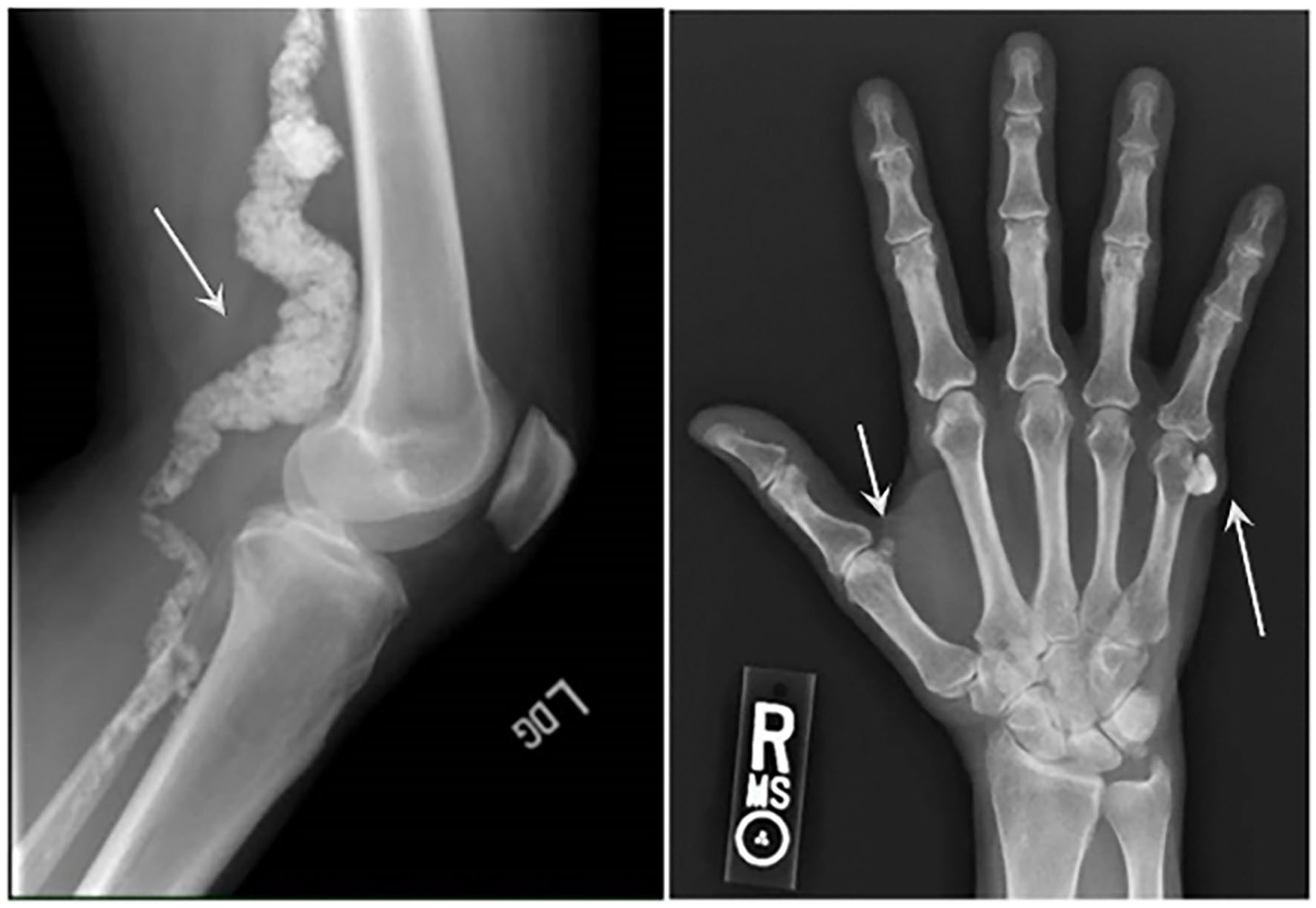
Radiographs of the popliteal artery
Mechanistically, CD73 is an enzyme that converts extracellular adenosine monophosphate (AMP) and pyrophosphate (PPi) to adenosine and inorganic phosphate (Pi).6–8 PPi, which is metabolized by the enzyme tissue-nonspecific alkaline phosphatase (TNAP), is also a well-described antimineralization factor.9,10 CD73 deficiency promotes tissue calcification and disrupts extracellular matrix homeostasis by reducing extracellular adenosine and producing an increase in TNAP activity that subsequently leads to a rise in PPi hydrolysis and decreases the PPi/Pi ratio.11–13 The imbalance of the PPi/Pi ratio from its normal physiologic levels drives calcium phosphate complex precipitation in the vasculature and connective tissues. 14 This mechanism is summarized in online supplemental Figure 1.
Though there are currently no treatment options available for patients with ACDC, preliminary in vitro studies using patient-derived fibroblasts suggested that bisphosphonate administration is a potential therapeutic approach for stopping the progression of lower-extremity vascular calcification because of its potent effects in inhibiting TNAP activity. 11 In fact, bisphosphonates are stable PPi analogs since their P-C-P backbone (similar to the P-O-P backbone in PPi) is not hydrolysable, thus inhibiting ectopic mineralization. 15 Bisphosphonates have also been shown to reduce soft tissue calcifications in rats even before their effect on bone resorption was known. 16 Of the currently available bisphosphonates, etidronate may have the largest potential to delay ectopic mineralization given its predominant inhibition of calcium precipitation and hydroxyapatite binding. 17 This is different from newer bisphosphonates, such as alendronate, which predominantly inhibit osteoclasts.18,19 Additionally, etidronate is a strong inhibitor of calcification and is approved for the treatment of conditions such as heterotopic ossification and Paget’s disease. Several nonrandomized and uncontrolled reports describe the beneficial effects of etidronate in patients with ectopic mineralization resulting from impaired PPi homeostasis. In generalized arterial calcification of infancy (GACI; OMIM 208000), etidronate has been used since the nature of the mineral deposit was proven to be calcium hydroxyapatite in the late 1970s. 20 In this disease, etidronate reduces arterial calcification and is associated with improved survival when started in early life, or even administered to the pregnant mother to treat GACI in utero.21,22 In pseudoxanthoma elasticum (PXE; OMIM 264800), another genetic disease of ectopic mineralization with considerable genotype and phenotype overlap with GACI,23,24 treatment with etidronate in PXE mouse models results in the prevention of ectopic mineralization and in alterations in bone microarchitecture.25–27 Further, in the Treatment of Ectopic Mineralization in Pseudoxanthoma elasticum (TEMP) trial, etidronate halted arterial calcification as assessed by computed tomography (CT) in all vascular beds except for the coronary arteries.15,28 Further, etidronate was identified for ACDC as a mechanistically targeted treatment using an in vitro disease model and confirmed in a murine model to halt the progression of calcification in patient-derived cells as a translational medicine bench-to-bedside approach. 11
In this study, we describe the results of an open-label, nonrandomized, single-arm pilot study in patients with genetically confirmed ACDC to evaluate the effectiveness of etidronate in attenuating the progression of lower-extremity arterial calcification and improving vascular blood flow (online supplemental Figure 1). The primary objective of the treatment included: (1) attenuation of the progression of lower-extremity arterial calcification as assessed by a CT calcium score; and (2) improvement in vascular blood flow as assessed by ankle–brachial index (ABI) measurements. The secondary objectives included: (1) functional improvement measured by stress test with the Gardner protocol; (2) changes in periarticular calcification of hand joints assessed by radiography; (3) decreased hand and foot pain based on a rheumatoid arthritis assessment tool; and (4) analysis of the composition of collected surgical tissue with calcification/inflammation.
Methods
Patient population
Seven adult patients (five patients from one family; two patients from two other families) with a confirmed genetic diagnosis of ACDC and evidence of lower-extremity arterial calcifications were enrolled in a National Institutes of Health treatment study with etidronate (ClinicalTrials.gov Identifier 01585402). Additionally, baseline and follow-up assessments were performed under other disease discovery/natural history NIH protocols (NCT03538639 and NCT01143454). All protocols were reviewed and approved by the NHLBI Institutional Review Board and patients provided written informed consent before participation per the ICH E6 Guidelines for Good Clinical Practice originating from the Declaration of Helsinki. 29 As shown in Figure 2, seven patients were screened for eligibility and enrolled in our studies. There were no dropouts during the study period and all seven patients received treatment in a single-arm, nonrandomized, open-label study design.
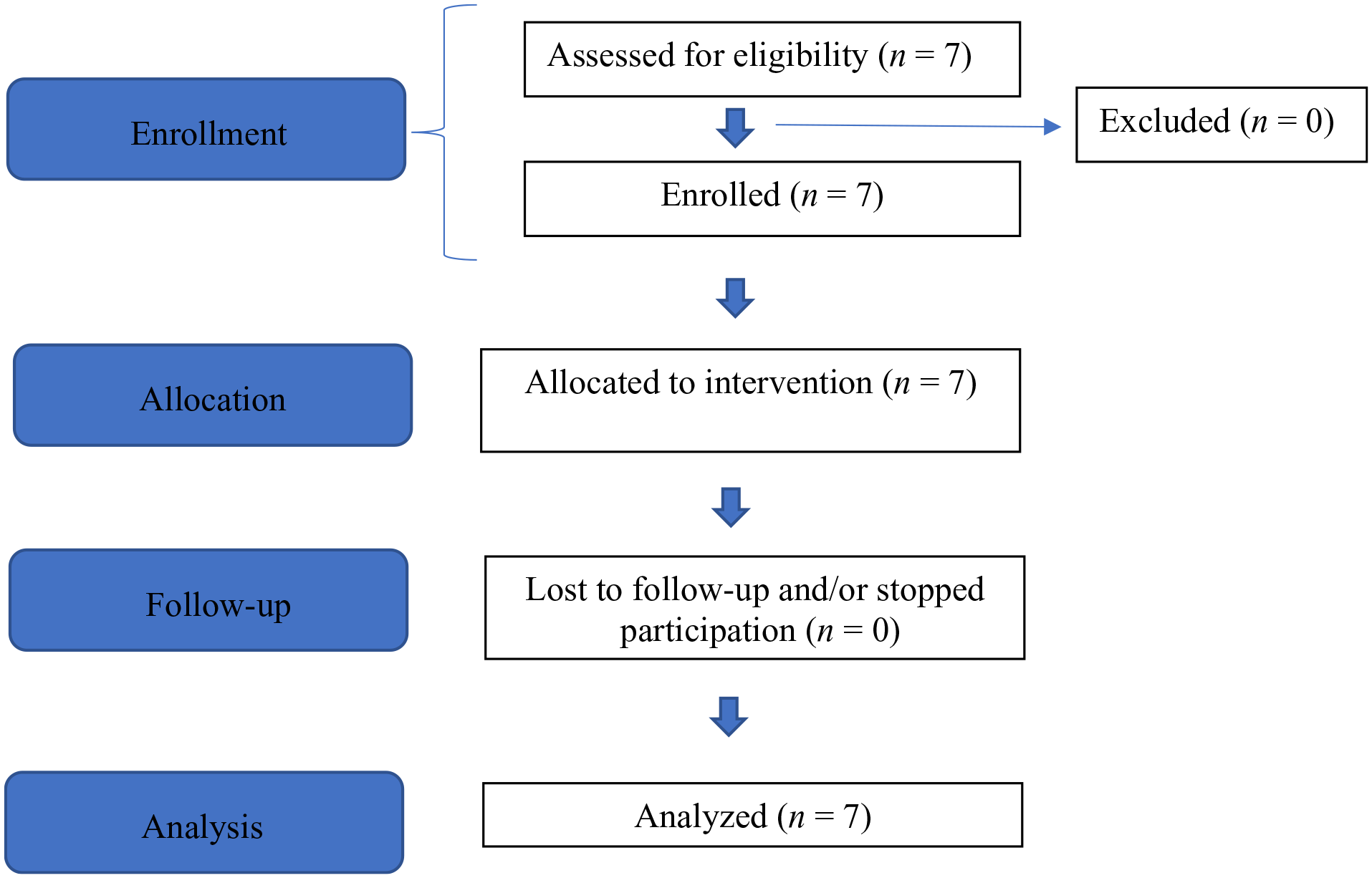
Modified CONSORT flow diagram for a single-arm, nonrandomized, open-label pilot study of etidronate treatment for arterial calcification due to deficiency of CD73 (ACDC).
Trial design and drug information
This was an open-label, nonrandomized, single-arm pilot study to evaluate the effectiveness of etidronate in attenuating the progression of lower-extremity arterial calcification in patients diagnosed with ACDC. Currently, a total of 13 patients have been diagnosed at our clinic, seven of whom were enrolled in this treatment study. Since ACDC is a very rare condition, the small patient population precluded us from conducting a larger clinical trial. Etidronate was administered orally as a 14-day treatment every 3 months over a 3-year period, with a total of 12 cycles as either 20 mg/kg daily or 10 mg/kg twice daily for 14 days. Patients had a period of 10 weeks off the study drug between drug cycles. One patient, however, received 11 drug cycles due to discontinuation of the drug by the manufacturer. We evaluated patients at the NIH Clinical Center at baseline (prior to treatment study enrollment), pretreatment (day 0), on treatment (months 6–36), and at follow up (yearly after treatment study completion), as summarized in Figure 3. While on treatment, patients were evaluated at the NIH Clinical Center approximately every 6 months and then yearly for follow ups after the last treatment cycle visit.
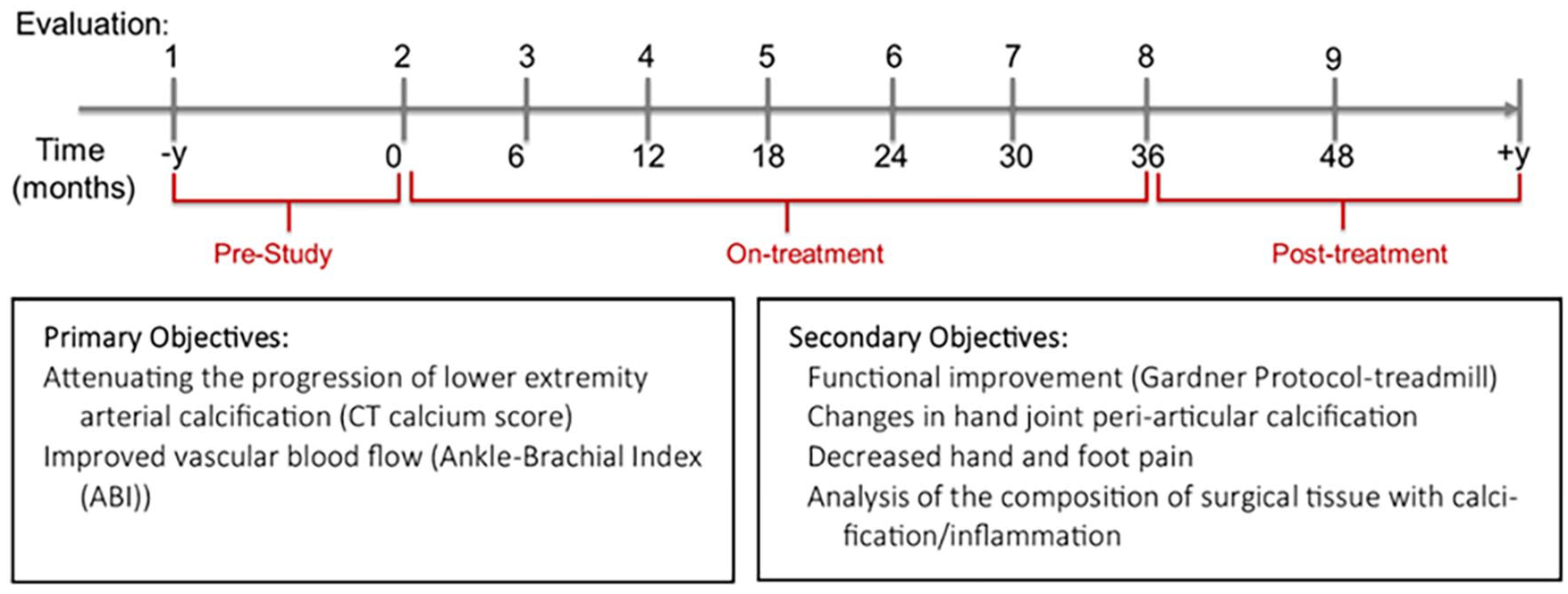
The treatment and visit (evaluation) schedule for patients with ACDC. Patients were evaluated prior to receiving etidronate for a period of 1–2 years. While on treatment, they were evaluated at the NIH Clinical Center every 6 months and every year after completion of treatment. Primary objectives focused on assessing the effect of treatment on lower-extremity arterial calcification assessed by CT and blood flow measured by ABI, whereas secondary objectives aimed at measuring functional capacity with the Gardner protocol and hand periarticular calcification.
Data collection
All patients underwent a complete medical history and physical exam at their initial visit to the NIH Clinical Center. The rate of disease progression before treatment was assessed with two lower-extremity CTs with calcium score measurements (Agatston score 30 ) and two ABI studies conducted between 1 and 5 years prior to initiation of treatment. The lower-extremity calcium scoring was performed by a board-certified CT cardiologist with over 20 years of experience. Once enrolled, but prior to receiving treatment, patients underwent clinical laboratory testing, CT calcium scoring of both the lower extremities (from the aortic bifurcation through the toes) and the coronary arteries, ABI measurements, 31 treadmill test using the Gardner protocol (a standardized graded treadmill exercise protocol), 32 magnetic resonance imaging (MRI) of the lower extremities, a 12-lead electrocardiogram (EKG), hand radiographs, a dual-energy X-ray absorptiometry (DXA) scan, as well as cardiovascular and dental consults. They were also evaluated with a Simplified Disease Activity Index (SDAI) scoring system, 33 which includes a tender/swollen joint count of 28 joints, patient/physician global assessment of disease activity, and C-reactive protein levels (mg/dL). Their ability to perform daily activity tasks was also evaluated by using a Duke Activity Status Index (DASI) questionnaire.34,35 Women of child-bearing potential were given a pregnancy test. Laboratory evaluations included a metabolic panel (Chem 14), lipid panel, total protein, high-sensitivity C-reactive protein (CRP), creatine kinase (CK), lactate dehydrogenase (LDH), fibrinogen, activated partial thromboplastin time (aPTT), prothrombin time/international normalized ratio (PT/INR), parathyroid hormone (PTH), 25-hydroxyvitamin D, and 1,25-dihydroxyvitamin D levels. Patients also had a complete CBC and differential as well as standard urinalysis with additional measurements for inorganic phosphorus and calcium. After starting treatment, patients were evaluated at the NIH Clinical Center every 6 months, as indicated in Figure 3. Research blood and skin biopsies were collected for laboratory assays and histology.
Safety assessment
The development of side effects, comorbidities, and hospitalizations was documented. The NIH’s Common Terminology Criteria for Adverse Events (CTCAE), version 4.0, was used to categorize abnormal results after enrollment. Vital signs, clinical laboratory tests, including a complete blood cell count with differential, renal and liver function, mineral panel, lipid profile, urinalysis, urine electrolytes, DXA scan, and dental evaluation including dental X-ray were performed throughout the protocol period.
Statistical analysis
Repeated measurements of variables during the study course were assessed for normality, and Box-Cox transformation was performed to make residuals closer to normal. We present the results with their original values because the transformation did not affect the significance of any results. Linear mixed-effects models were used to examine how study measurements were changing across study time (in days). The study time was also evaluated as three categorical periods: pretreatment, on-treatment (0–36 months), and posttreatment. Models with and without adjustment for age at baseline and sex of study subjects were both performed with similar findings. Results without adjustment are presented here. To assess disease progression by the CT score results, a red dotted line was added and represents the locally estimated scatterplot smoothing (LOESS) showing how the average data trended during the study. Statistical analyses were performed using SAS 9.4 (SAS Institute, Cary, NC, USA) and a 5% significance level was applied in all analyses.
Results
A total of seven patients with a confirmed genetic diagnosis of NT5E pathogenic variants resulting in ACDC were enrolled. Patients were examined at the NIH Clinical Center semiannually for 3 years under this treatment protocol for a total of seven visits as outlined in the study schedule in Figure 3. Baseline and follow-up evaluations were performed on different protocols outside of the treatment period. Patient demographics and health/risk factors are summarized in Table 1.
Patient demographics and risk factors at baseline for the ACDC cohort (N = 7).
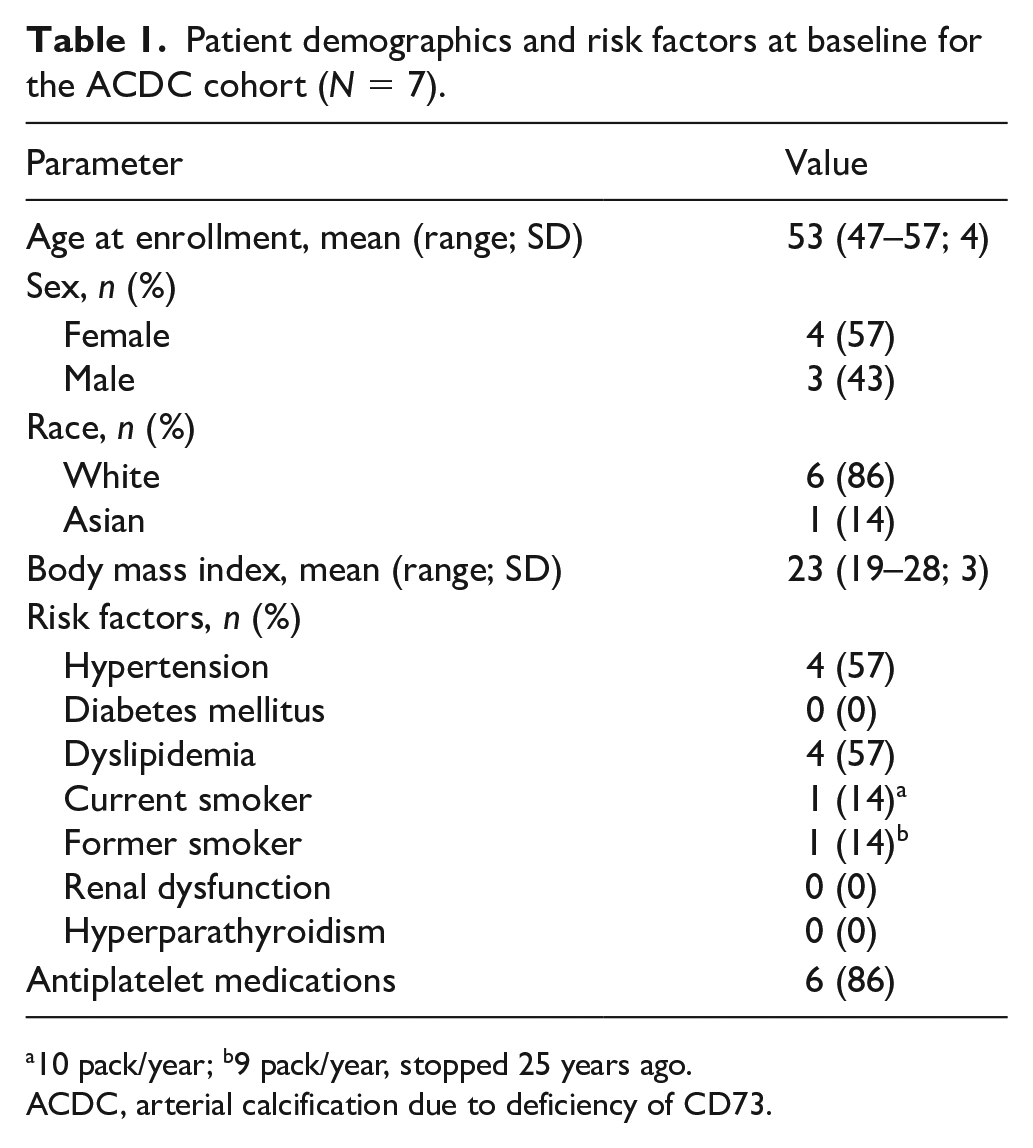
10 pack/year; b9 pack/year, stopped 25 years ago.
ACDC, arterial calcification due to deficiency of CD73.
Baseline lower-extremity CTs showed that all patients were affected by extensive arterial calcification typical of ACDC, starting at the level of the iliac arteries, but more pronounced in the femoral and popliteal arteries, then gradually decreasing toward the feet. We also reviewed all prior CT imaging results to confirm the typical ACDC phenotypes, which revealed massive bilateral external carotid calcification in two out of seven patients, mild calcification of aortic branches in four out of seven patients, and faint calcification of brachial arteries in two out of seven patients. Calcification of the external carotid arteries did not appear to provoke any obvious hemodynamic compromise or symptoms and remained stable during the study. Coronary calcifications were also present in three patients at the time of enrollment and four patients at the conclusion of the study and follow up.
At baseline, all patients presented with peripheral arthropathy and periarticular calcifications involving the hands, with the most affected joints being the proximal and distal interphalangeal as well as metacarpophalangeal, as evaluated by radiographs. A detailed description of this cohort’s radiological, histological, and arthritis features associated with their clinical presentation of ACDC has been previously described by our group. 5 Other predominantly affected joints were feet, shoulders, elbows, hips, and spine (online supplemental Table 1). Moreover, other nonvascular and ectopic calcifications, such as calcified oligodendroglioma, torus mandibularis, and calcification of the plantar surface of a foot, were identified in two out of seven patients (online supplemental Table 1).
All patients were experiencing activity-limiting claudication at the time of enrollment and two patients had undergone revascularization procedures due to progressive limb ischemia before starting treatment with etidronate. There was no significant worsening of symptoms requiring intervention during the study, though three patients required revascularization in the follow-up period between 2 and 4 years after completion of treatment. Revascularization procedures that were attempted/performed included bypass, angioplasty, endarterectomy, and/or stenting. For patients who underwent lower-limb revascularization procedures, measurements of ABIs and calcium scores of the lower extremities of the affected limb obtained after the procedure dates were excluded from analysis.
The primary objective of this clinical study was to test the effectiveness of etidronate in attenuating the progression of lower-extremity arterial calcification and improvement of vascular blood flow based on the CT calcium score (LECaS) and ABIs, respectively. Linear mixed-effects models were used to examine how these measurements were changing across the study period. The linear mixed-effect model of LECaS measurements shows no significant change over time (Figure 4), strongly suggesting that although the etidronate did not reverse or alter vascular calcifications that were already present, it may have slowed or halted their progression while patients were on treatment. The coronary calcium score, when available, remained stable during treatment and at follow up (mean Agatston score prestudy was 183 ± 43 (SD) and posttreatment was 226 ± 84). On a functional level, we found that ABI measurements at rest and after exercise (Figure 5), as well as treadmill peak metabolic equivalent of task (METS) (Figure 6A), followed a similar pattern as the LECaS measurements with no clear improvement in vascular function but, potentially, a trend of slower disease progression while on treatment. Further, the DASI scores also remained stable across treatment periods (Figure 6B).
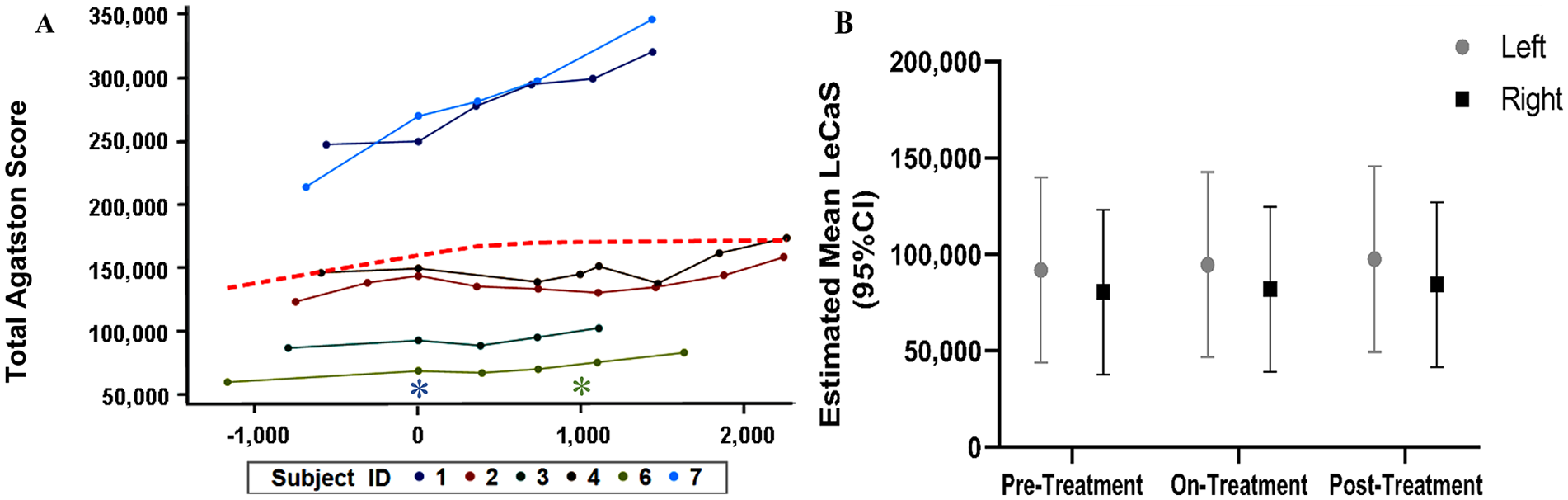
Stabilized lower-extremity calcium deposition determined by LECaS during the study time-course.
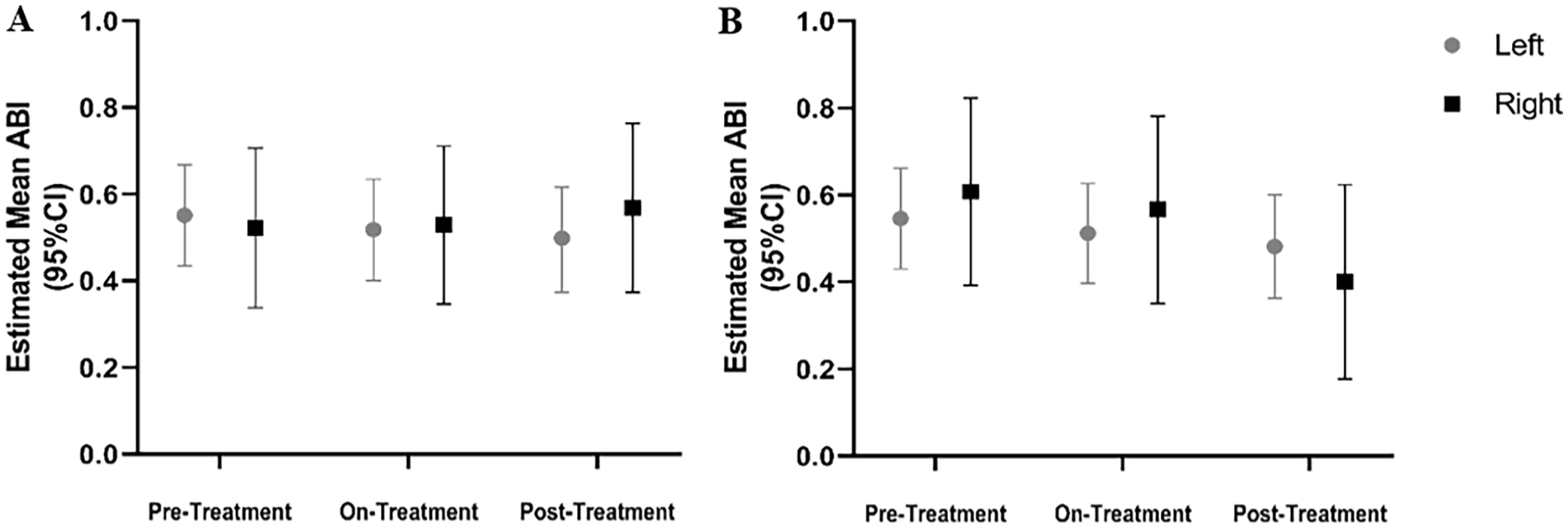
ABI measurements in patients with ACDC remained largely stable during treatment with etidronate. Estimated mean ABI measurements at rest
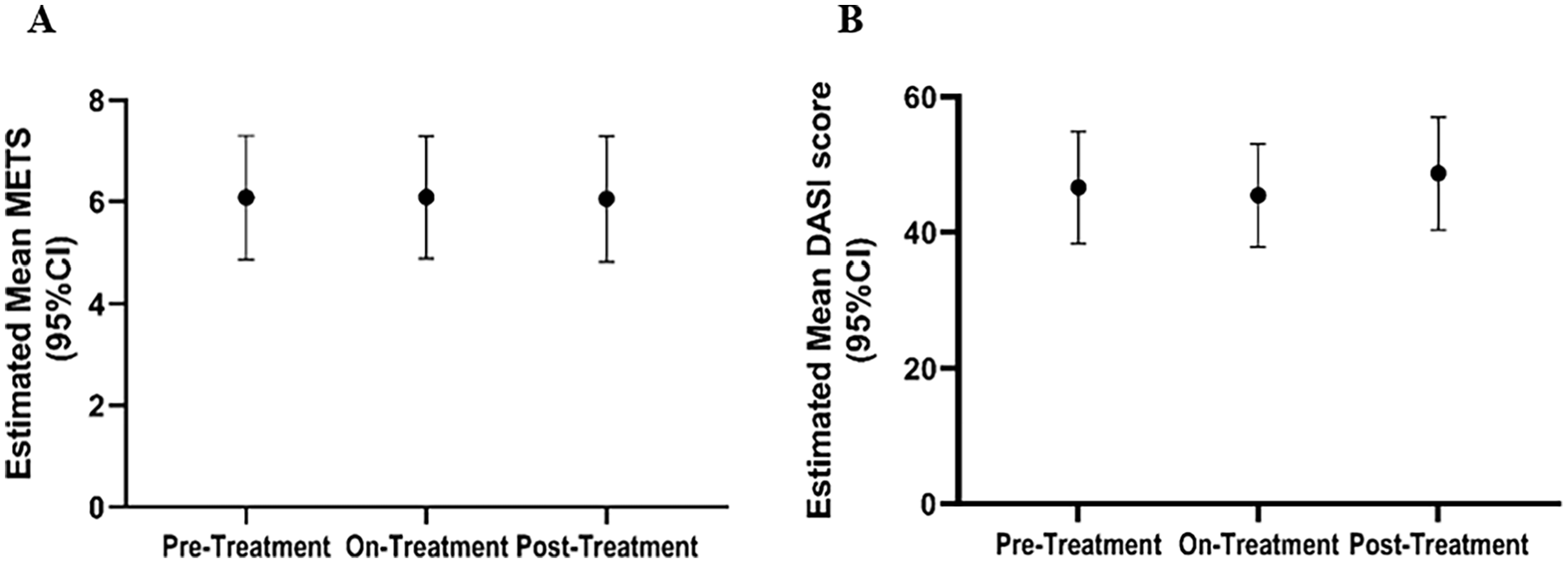
Treadmill peak METS and DASI vascular assessment remained stable across different treatment stages.
Lower-extremity magnetic resonance angiography (MRA) imaging was performed at three timepoints: prestudy (3–4 years before study enrollment), pretreatment (time 0 before starting drug), and after treatment. When qualitatively comparing MRA images over time, all patients appeared to be stable with very mild disease progression within the study timeframe, and no discernable change in the rate of progression between the prestudy and the treatment period. However, given the severity of the vascular disease and calcification with complete occlusion of most of the large lower-extremity arteries in all patients at baseline, it was difficult to assess small changes indicative of further disease progression.
Results from the SDAI questionnaires administered to the patients at each NIH Clinical Center visit showed significant improvement in the cohort scores going from a mean score of 18.5 (14.5–22.6) prior to etidronate, to 8.2 (5.1–11.4) during treatment, and to 5.0 (1.1–8.8) after treatment (Figure 7). This suggests that there was an improvement of symptoms, potentially due to etidronate treatment. The observed improvement of this score was driven primarily by the patient global assessment scores and physical exam, rather than inflammatory markers (CRP) that remained unchanged and evidence of synovitis.
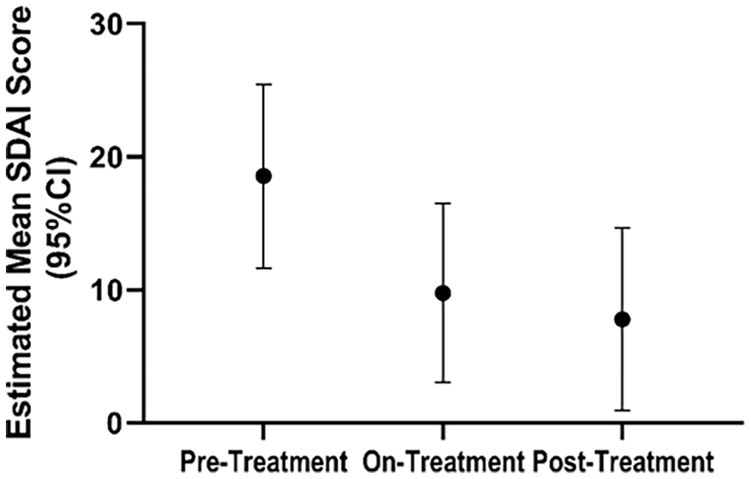
Rheumatological activity and pain scores reflect functional capacity improvement and decreased pain over time in patients with ACDC. The estimated mean SDAI score shows a significant improvement once patients start treatment, which continues even after treatment completion.
To assess the progression of periarticular calcifications in our patients with ACDC before, during, and after treatment, our team developed a scoring system (online supplemental Figure 2) on hand radiographs to quantify calcium deposits over time within the most frequently involved joints (15 total that included distal interphalangeal, proximal interphalangeal, metacarpophalangeal, and carpometacarpal joints). We assigned density scores for each visible calcification on a scale from 0 (no calcification) to 5 (dense calcification) based on visual assessment by two independent observers. The size (height and width in mm) of the calcifications was also measured and a total score for each deposit was obtained by multiplying the height, width, and density score (height × width × density). Total scores per patient per study period were then calculated as the sum of all individual calcification scores. Statistical analysis showed that etidronate did not affect the natural history of joint calcification (Figure 8).
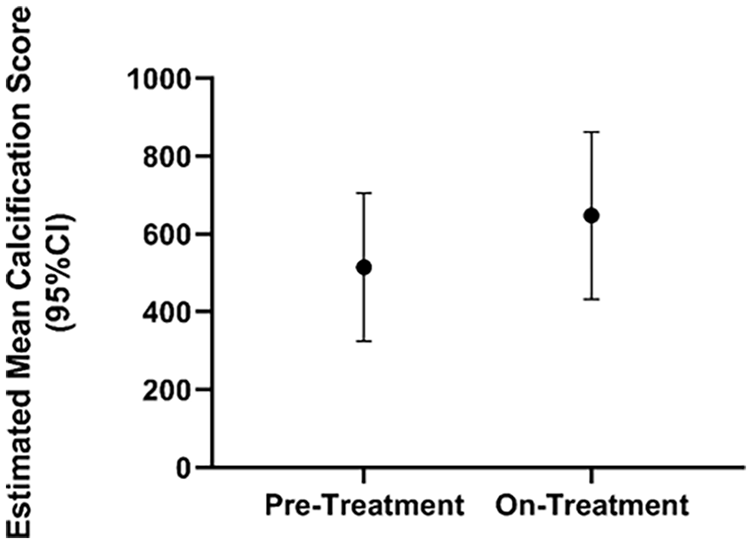
Hand radiograph scores show no treatment effect on the progression of joint disease. Estimated mean hand calcification scores (95% CI) show a trend toward disease progression in periarticular ACDC disease despite etidronate treatment.
Laboratory evaluations for several serum and urine biochemical markers were performed during the study period and were within normal ranges across time for all patients (select findings shown in online supplemental Figures 3 and 4). PTH was measured at baseline and was normal in all patients. Dental consultations were performed at baseline, halfway, and at the end of treatment to monitor for possible side effects, since etidronate has been associated with osteonecrosis. Panoramic dental X-rays were unremarkable, and no changes were detected for any of the patients. Similarly, DXA scans were performed to monitor bone mineral density before initiation of etidronate as well as yearly during treatment (12, 24, and 36-month visits). There were no significant changes in bone density over time that could be attributed to the treatment. Some patients reported gastrointestinal symptoms while taking the drug, but they were manageable and resolved quickly. Based on the common terminology criteria for adverse events (CTCAE) v4.0, we recorded two grade 3 adverse events possibly related to treatment (CTCAE v4.0 attribution terminology) for two patients, one being renal calculi (preexisting condition) and another being gastrointestinal (GI) symptoms. We also recorded 13 grade 2 adverse events in four patients, which largely involved GI symptoms or arthralgia/bone pain that were possibly or probably related to treatment (CTCAE v4.0 attribution terminology). Lastly, there were multiple grade 1 events, also GI manifestations and arthralgia/bone pain that were recurring during the on-treatment cycles.
Discussion
The study described here is the first of its kind on the largest cohort of patients with ACDC in the world, with the goal of assessing the efficacy and safety of an off-label treatment for vascular and periarticular calcification. The original accrual target for this study was enrollment of 20 patients and, despite this being the largest cohort of patients with ACDC in a clinical study to date, the disease is quite rare so only seven patients were enrolled. Additionally, six other patients were identified but three declined to participate and the three most recently diagnosed could not be enrolled because etidronate manufacturing was temporarily discontinued. For many measurements statistical significance was not achieved, but trends could be observed in the interpretation of the data showing that etidronate treatment may have slowed the progression of vascular calcification.
In this cohort, all seven patients had a similar clinical presentation at baseline, with extensive vascular calcification on their lower extremities and periarticular joint calcifications of the hands. Comparing the ABIs and the LECaS scores during the treatment and posttreatment phase to this baseline stage, the linear mixed-effect model results show a promising trend with etidronate potentially slowing the rate of disease progression. It is worth noting, however, that particularly for the CT measurements of the lower extremities, this test was adapted from its more typical application to measure coronary calcium scores, which typically cover a much smaller vascular region. As a result of measuring a larger vascular area and an extreme degree of calcification, the lower-extremity calcium scores fell on a different scale and well outside of what would be considered the upper level for high risk (a score above 400). The massive degree of vascular calcification in our patients with ACDC makes this population unique and made the identification of change in calcium score extremely challenging. The calcification in ACDC vessels appears to initiate at sites of broken and damaged elastin lamina 36 and this is reminiscent of the medial arterial calcification (MAC) in patients with peripheral artery disease. 37 Although PAD is commonly assumed to be an atherosclerotic pathology, recent pathological studies have identified that MAC is an independent disease state that itself leads to impaired wound healing and chronic and acute limb ischemia. 38 Indeed, signatures of ACDC are found in PAD patient vessels 12 and fully understanding ACDC pathogenesis and treatment strategies may likely benefit the greater PAD patient population.
We were generally able to use the LECaS measurements to determine that our cohort’s vascular disease remained stable with a potential slowing trend of disease progression due to etidronate treatment, but we need to identify more sensitive and sophisticated vascular calcium measurement methods to overcome the imaging issues we observed due to the baseline severity of vascular calcium deposition in our patients with ACDC. This is in contrast to the findings from the TEMP study, where PXE patients (n = 38) received etidronate treatment that resulted in a significant decrease in arterial calcification when compared to a placebo group. 15 However, the severity of lower-extremity calcification in the PXE cohort was lower than that of our patients with ACDC by several orders of magnitude and a larger patient population made the statistical analysis more reliable. However, considering these challenges and the small sample size of our ACDC patient cohort, the observed trend of slower disease progression shown by our ABI and LECaS results appears promising for this disease. In addition to the TEMP trial, our clinical trial showed that in patients with ACDC, etidronate treatment is well tolerated for a more extended period of time (3 years vs 1 year).
From a functional standpoint, the ABI and treadmill test results did not show an improvement or a slower disease progression rate, in comparison with the LECaS measurements, but remained in the moderate-to-severe disease range throughout the study, likely another effect of the severity of the large-vessel calcification and occlusion, with an extensive compensatory collateral vessel network. The DASI questionnaire is a clinically useful tool commonly used to assess functional capacity in multiple groups of patients with heart failure, coronary artery disease,39–41 peripheral artery disease, 42 chronic obstructive pulmonary disease, 43 and stroke, 44 but it had never been applied to an ACDC patient cohort. DASI results reflected a trend similar to that of other functional assessments we performed, suggesting that etidronate may have slowed the natural disease progression, although its applicability to ACDC needs to be further validated.
The periarticular joint calcifications of ACDC were not affected by treatment with etidronate. There was no evidence of slowing or reduced progression as measured by hand radiographs. However, we observed waxing and waning of joint calcifications over time, often with their spontaneous resolution after an acute arthritic flare. Nonetheless, our patients showed a statistically significant improvement on SDAI scores, moving from moderate/high disease activity to remission on a scale reflective of rheumatoid arthritis symptoms. When analyzing these data, the high scores observed in the pretreatment period were likely driven by an initial increase of global disease activity from patient self-assessments as well as a higher number of tender joints. While on treatment, swollen joints and inflammatory markers such as CRP remained relatively unchanged, but patients’ self-assessment scores improved. This trend could be reflective of the natural history of ACDC, since our patients reported that the most severe phase of the disease was at presentation in their late teens with a gradual improvement in symptoms and a decrease in the number and severity of arthritic flares over time, even prior to receiving etidronate treatment. The absence of a placebo group that could control for placebo-like effects of etidronate could have also confounded these results, although this study was designed for each patient’s baseline data to serve as his/her own pretreatment control. Additionally, expectation bias and patients’ awareness of the drug being administered could have been a cause of the observed reduction in SDAI score during the on- and posttreatment periods, particularly pertaining to patient-reported assessment. Further, the SDAI score is generally validated for patients with rheumatoid arthritis as a sensitive assessment of disease activity and treatment response but has not been validated in this disease. Nevertheless, it may be reasonable to employ the SDAI on this unique patient cohort owing to the inflammatory nature of the joint manifestations and to the lack of other ACDC-specific disease activity scores.
Our study demonstrated the tolerability and safety of etidronate when used at high dose for a 3-year period and results suggested a trend of slower disease progression in the arteries of the lower extremities of our patients, despite a small sample size. Future studies would need to be performed to identify and verify other treatments to target the auto-inflammatory pathways driving the vascular calcification in ACDC in in vitro disease or in vivo murine models.
It should be noted that aminobisphosphonates might also play a role in the inhibition of arterial mineralization. In the preclinical setting, alendronate and ibandronate have been shown to inhibit calcification of arteries and heart valves at doses comparable to those shown to inhibit bone resorption. 45 In the clinical setting, aminobisphosphonates have been associated with decreased prevalence of cardiovascular calcification in women older than 65 years of age, but with more prevalent calcification in those < 65 years old. 46 Data on the association of aminobisphosphonates with inhibition of ectopic calcification are inconsistent, whereas its association is more robust with etidronate.47,48 Current meta-analyses are underway to try to more consistently answer the question of whether bisphosphonates influence cardiovascular calcification. 49 In addition, new bisphosphonates continue to be developed; a novel bisphosphonate, FYB-931, was shown to inhibit vascular calcification more potently than etidronate, 50 specifically by interfering with the transformation of primary calciprotein particles to secondary ones. 51 Thus, it is possible that other bisphosphonates could be tried in the future for patients with ACDC.
Study limitations
The main limitation of this study is its small sample size due to ACDC being a rare genetic condition, and five of the seven patients were from the same family. It is hard to evaluate the shared genetic and environmental effects from these patients on etidronate treatment and the various repeated measurements conducted in this study. Although we have identified an additional three patients with ACDC, etidronate manufacturing has been discontinued in the United States since 2018 and those patients could not be enrolled. Another major challenge has been the extent of vascular calcification in the lower extremities of patients with ACDC, which are at levels well beyond many other known vascular calcification diseases and which made CT measurements somewhat challenging since looking for change in an endpoint at the high end of the scale is difficult. Further, we compared these changes while on treatment to the pretreatment phase for each patient (1–3 years) and concluded that although there was some disease progression in this small window of time, lower-extremity calcification ultimately showed stabilization during the treatment and posttreatment phases of the study. Unfortunately, we do not have older historical data of vascular calcification for this cohort. Similarly, we have no older historical data of the coronary calcium score. The MRA was used as a descriptive measure of vascular status rather than a quantitative measurement. Some measurements, such as ABI for example, may also have been impacted by environmental factors such as lifestyle changes. Lastly, though validated for patients with rheumatoid arthritis, the utility of SDAI questionnaires may have been limited in the absence of a placebo group. However, this study was designed for each patient’s baseline data to serve as his/her own pretreatment control to attempt to mitigate this effect. To conclude, the clinical evaluation tools used in the study are not specifically developed for ACDC, which although it has some similarities with other conditions, is also quite unique in the degree of vascular calcification present and joint manifestations and pain. In particular, though the Agatston score has been widely used for vascular calcification scoring in vascular beds other than the coronaries, 52 such as the aorta53–56 and lower extremities,57,58 these methods have not yet been validated for lower extremities as described here.
Conclusion
In conclusion, etidronate treatment did not appear to have a strong effect in reversing vascular and/or periarticular joint calcifications in our small cohort of patients with ACDC, but the LECaS scores suggested a slower rate of vascular disease progression. Furthermore, etidronate was found to be safe and well tolerated by our patients. Despite the small sample size, these results potentially suggest that etidronate, which is still available outside of the United States, could be considered for patients with progressive vascular calcification due to ACDC, particularly given the total lack of any other treatment options for this disease. In addition, the knowledge gained with respect to the natural history, pathology, and clinical presentation of ACDC has been invaluable and will inform the development of novel therapies and larger clinical trials for this and other calcifying vascular diseases.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241235669 – Supplemental material for Pilot study to evaluate the safety and effectiveness of etidronate treatment for arterial calcification due to deficiency of CD73 (ACDC)
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241235669 for Pilot study to evaluate the safety and effectiveness of etidronate treatment for arterial calcification due to deficiency of CD73 (ACDC) by Elisa A Ferrante, Cornelia D Cudrici, Mahmood Rashidi, Yi-Ping Fu, Rebecca Huffstutler, Katherine Carney, Marcus Y Chen, Cynthia St Hilaire, Kevin Smith, Hadi Bagheri, James D Katz, Carlos R Ferreira, William A Gahl, Manfred Boehm and Alessandra Brofferio in Vascular Medicine
Footnotes
Acknowledgements
We would like to thank our Patient Care Coordinator, Marta Cardenas, as well as the NIH Dental Clinic and the Radiology and Imaging Sciences teams at the NIH Clinical Center for their support during this study.
Data availability statement
The data may be shared upon reasonable request to the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by NIH-DIR HL006077-14 from the Division of Intramural Research (DIR) at the National Heart, Lung, and Blood Institute (NHLBI) at the National Institutes of Health (NIH).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
