Abstract
Erdheim–Chester disease (ECD) is a rare ‘L’ (Langerhans) group histiocytic neoplasm that affects a multitude of organ systems, causing osteosclerotic bone lesions, periaortic encasement (‘coated’ aorta), retroperitoneal fibrosis involving kidneys and ureters (‘hairy kidney’), and infiltration of the central nervous system. Cardiovascular involvement can occur in up to 70% of patients and is usually found during computed tomography/magnetic resonance imaging evaluation. When present, cardiovascular symptoms can have wide variability in presentation from asymptomatic to pericarditis, fatal cardiac tamponade, myocardial infarction, conduction abnormalities, heart failure, renal artery stenosis, and claudication. Cardiac involvement found on imaging includes right atrial pseudotumor, right atrioventricular groove infiltration, and pericardial effusions. ECD can involve the large- and medium-sized arteries, often seen as periarterial thickening (commonly coating the aorta) with stenosis/occlusion. Although more cardiovascular ECD cases have begun to be published in the literature, more data are needed on the outcomes of these patients, as well as how cardiovascular manifestations respond to treatment of ECD.
Introduction
Erdheim–Chester disease (ECD) is a rare ‘L’ (Langerhans) group histiocytic neoplasm that affects a multitude of organ systems, causing osteosclerotic bone lesions, periaortic encasement (‘coated’ aorta), retroperitoneal fibrosis involving kidneys and ureters (‘hairy kidney’), and infiltration of the central nervous system. 1 Clinic manifestations include long-bone pain, xanthelasma, exophthalmos, ataxia, and constitutional symptoms. 2 Prognosis of ECD is variable, but factors associated with poor prognosis include advanced age and disease involving the central nervous system, lungs, and retroperitoneum. 1 However, a study by Haroche et al. found that up to 60% of their patients died from cardiovascular involvement. 3 Cardiovascular involvement can occur in up to 70% of patients but may be asymptomatic, is underdiagnosed, and is usually found during imaging evaluation on computed tomography (CT) and/or magnetic resonance imaging (MRI). 1 We present two cases of ECD with cardiovascular involvement from our institution, and a narrative review of the cardiovascular manifestations of ECD.
Case histories
Case one is a 24-year-old woman with history of severe preeclampsia and acute heart failure at 31 weeks’ gestation requiring emergent cesarean section. Echocardiography at that time revealed severe mitral regurgitation with flail anterior leaflet due to chordae rupture and moderate aor-tic regurgitation. Valve repair evaluation with computed tomography angiogram (CTA) showed diffuse circumferential soft tissue density surrounding the aorta extending along the origin of the great vessels through the mediastinum down the descending aorta to the hiatus (Figure 1), for which she was referred to vascular medicine. Further imaging with MRI showed the soft tissue density also encased the coronary arteries, aortic branch vessels, central pulmonary veins, superior vena cava, bilateral pulmonary arteries, and both atria without arterial or venous luminal narrowing. She had patchy erythematous lesions on her face which were biopsied and nonspecific, revealing prominent lymphoplasmacytic and histiocytic proliferation filling the superficial dermis. Immunohistochemical staining revealed CD68, CD163, and S100 positive histiocytes, but was negative for CD1a, Langerin, and BRAF-V600E. A positron emission tomography (PET) scan revealed a sclerotic lesion on the right tibia which was biopsied and confirmed diagnosis of ECD. The patient was positive for MAP2K1 mutation and started targeted therapy.
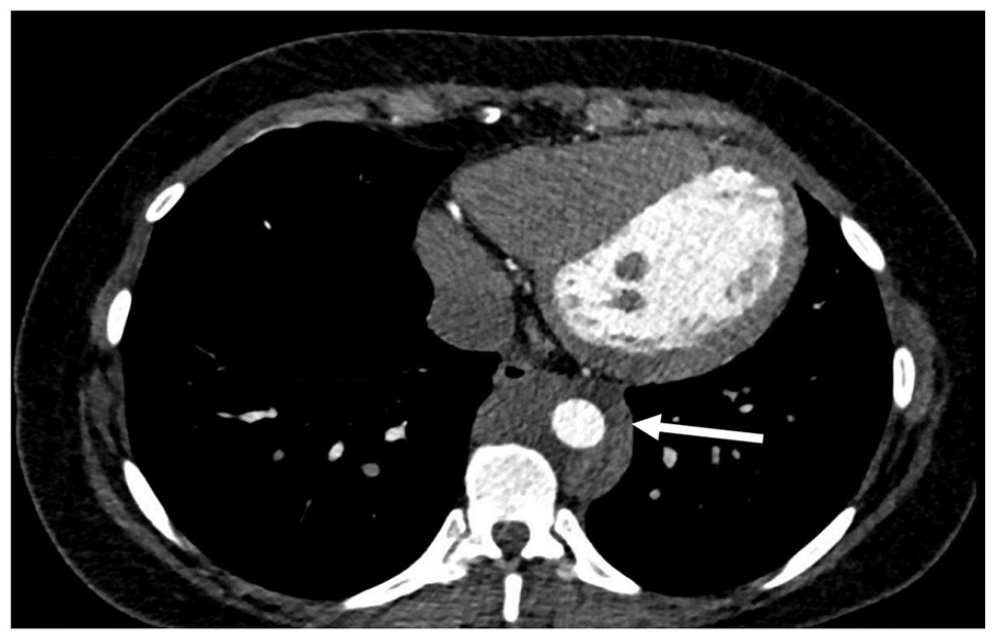
Computed tomography angiogram showing the classic finding of a ‘coated’ aorta (arrow) in an Erdheim–Chester disease patient.
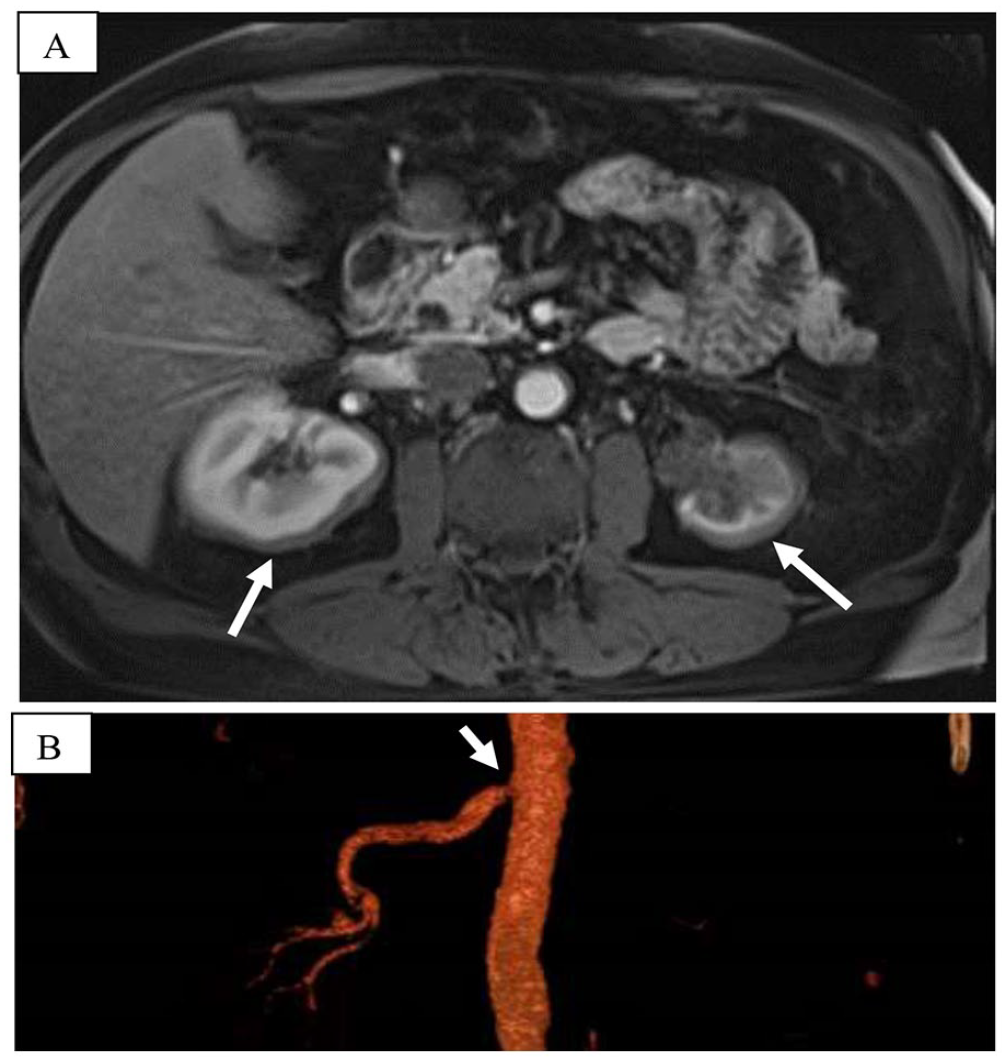
Case two is a 68-year-old man referred to vascular medicine for renal artery stenosis (RAS) and resistant hypertension (HTN). During his early 50s, he developed new-onset HTN up to 200/100 mmHg and was found to have bilateral RAS and a right renal mass. His HTN was medically managed. He developed progressive renal dysfunction and an atrophic left kidney secondary to left renal artery occlusion. During this time, he was undergoing evaluation for right hip replacement and was noted to have abnormal lower-extremity X-rays. He subsequently underwent bone marrow and right renal biopsies. The initial bone biopsy showed sclerotic bone with marrow replaced by fibrosis and lymphocytes. Occasional histiocytes were seen, but aggregates of foamy histiocytes typical of ECD were not present. He then underwent right renal biopsy, which showed a predominance of lipid-laden foamy histiocytes, and immunohistochemical staining, which was negative for CD1a and S100 proteins, suggesting a diagnosis of Langerhans cell histiocytosis. Immunohistochemical staining for BRAF-V600E mutation was not performed. Imaging at that time had revealed enhancing soft tissue around both kidneys (Figure 2A), renal sinuses, ureteropelvic junction, and proximal ureters; inflammatory involvement of the left adrenal gland; occluded left renal artery and inferior mesenteric artery; severe stenosis of the celiac trunk, superior mesenteric artery (SMA), and right renal artery due to retroperitoneal fibrosis – all of which suggested ECD (Figure 2B). A diagnosis of ECD was made based on renal and bone involvement, imaging, as well as histology. In both cases, close collaboration between the pathologist, radiologist, and clinician eventually led to the diagnosis of ECD.
Epidemiology
ECD is a rare condition that was previously classified as a non-Langerhans histiocytosis. In 2016, it was reclassified as an ‘L’ (Langerhans) group histiocytic neoplasm that affects a multitude of organ systems ranging from skeletal, cardiovascular, renal, nervous, and integumentary. 1 The incidence of ECD is unknown due to scarcity of data and lack of national registries. Based on data from the National Institutes of Health (NIH) and a French cohort, ECD primarily affects middle-aged adults, with a mean age of 40s to 50s, though patients much younger and older have been afflicted. It predominantly affects men, who make up 70–75% of the ECD population. Pediatric cases of ECD are rare, mostly affect boys, often carry the BRAF-V600E mutation, and usually present with a juvenile xanthogranuloma mass lesion in the CNS, though it spares the heart.1,2
Pathophysiology
ECD is classically characterized by accumulation and infiltration of CD68-positive and CD1a-negative foamy histiocytes in multiple organ systems. 1 Histiocytes are derived from bone marrow precursor cells. They belong in the family of mononuclear phagocytic cells which include macrophages. 4 In histiocytoses, these cells fail to apoptose and accumulate. They have historically been classified as Lan-gerhans cell or non-Langerhans cell subtypes. 4 ECD was classified as a non-Langerhans cell histiocytosis. A new classification for histiocytic disorders puts Langerhans cell and non-Langerhans cell histiocytoses into the group L (Langerhans) histiocytoses because of their clinical and molecular similarities.1,4
The majority of patients with ECD have activating mut-ations in genes of the MAPK (RAS-RAF-MEK-ERK) or PI3K-AKT pathways which drives histiocyte proliferation. Pathologic mutation in the BRAF-V600E gene is capable of inducing multiple cell functions (angiogenesis, apoptosis, migration, proliferation, and survival), ultimately leading to RAS pathway activation. 2 Up to 60% of patients with ECD have the BRAF-V600E mutation with some data showing rates as high as 80% in those with cardiac involvement.1,5
In 2018, Cohen-Aubart et al. determined that the presence of the BRAF-V600E mutation was significantly associated with low plasma high-density lipoprotein (HDL), decreased monocyte cholesterol efflux, with increased risk of both cardiac and aortic infiltration with odds ratios of 5.75 and 18.28, respectively. 6 Azoulay et al. also found a statistically significant association between BRAF-V600E mutation and cardiac involvement in their ECD cohort, with an odds ratio of 5.95 (CI = 3.5–16.8, p < 0.001). 5
Mutations in RAS genes, specifically KRAS and NRAS, are known to cause ECD and are associated with upregulation of vascular endothelial growth factor (VEGF) expression. 7 Roeser et al. found that high serum VEGF levels (> 500 pg/mL) were associated with cardiovascular involvement. 8 They noted 68.8% of their patients with high VEGF had vascular involvement (‘coated’ aorta, mesenteric arteries, renal arteries) compared to 48.6% with low VEGF (p = 0.001). There was a statistically significant difference in the presence of ‘coated’ aorta, which was found in 47.5% of patients with ECD with high VEGF compared to 33% with low VEGF (p = 0.025). Cardiac involvement was also more prevalent in the high VEGF cohort (59% vs 41.2%, p = 0.007), with coronary artery involvement being almost twice as prevalent in the high VEGF group (28.7% vs 14.7%, p = 0.01). BRAF-V600E status was not significantly different between patients with ECD with high or low serum VEGF in their study.
Classic histopathologic features include foamy histiocytes with small nuclei and surrounding fibrosis, multinucleated giant cells, and Touton giant cells, although the absence of these findings do not exclude the diagnosis of ECD. 1 Atypical features include florid lymphohistiocytic infiltrates or fibrotic lamellae with scattered foamy histiocytes and rare Touton giant cells. ECD can share features within the xanthrogranuloma family, such as a prominence of reactive lymphocytes, plasma cells, and rarely neutrophils. 1
Immunohistochemically, histiocytes in ECD were classically believed to be negative for S100, but weak or focally positive staining has been seen in up to 30% of cases. 1 Histiocytes in ECD can test negative for CD1a and CD207, and can be positive for CD68, CD163, factor XIIIa, and fascin. 1
Clinical cardiovascular manifestations
Cardiovascular involvement of ECD is common and can have wide variability in presentation ranging from asymptomatic to life-threatening. 2 Pericardial involvement can present as pericarditis, pericardial effusions, or even fatal cardiac tamponade. 2 Coronary artery involvement can lead to stenosis and patients can present with myocardial infarction. 2 Conduction system abnormalities and heart failure have also been seen in patients with ECD.5,9–11 Periaortic sheathing is considered a classic feature of ECD but is not associated with dilation, dissection, or aneurysm (Figure 1). 2 Involvement of the renal arteries can lead to RAS and resistant HTN (Figure 2). 1 Patients can also develop mesenteric ischemia and claudication if ECD involves the visceral and peripheral arterial systems.4,12 Careful attention should be paid to patients with ECD and lower-extremity long-bone involvement complaining of lower-extremity pain, as complaints may often be misattributed to bone pain and a diagnosis of peripheral artery disease could be missed. 12 The superior and inferior vena cava can also be affected, even portal HTN secondary to portocaval involvement has been documented.4,13
Cardiac and coronary involvement
A retrospective cohort study by Villatoro-Villar et al. sought to determine the frequency and distribution of cardiovascular involvement in 63 patients with ECD who underwent baseline imaging within 6 months of diagnosis. 12 Of those with cardiac imaging, 61% had cardiac involvement, 45% had pericardial effusion, 38% had myocardial thickening, and 31% had pericardial thickening. A study by Azoulay et al. with a cohort of 200 patients with ECD who underwent cardiac imaging reported pericardial effusion in 24% of patients, 4% with pericarditis, and 5% with cardiac tamponade. 5 A systematic review by Sánchez-Nadales et al. of 52 articles on patients with ECD and pericardial effusions found that approximately 60% required a pericardial procedure, 30.3% of patients received at least pericardiocentesis, four patients received a pleuro-pericardial window, three received a pericardial window, and two received a pericardiectomy. 14
Right atrial pseudotumors are often seen, with multiple cohorts reporting its presence in 13–63% of patients with ECD.4,12,13 The left heart is not affected as frequently, with left atrial involvement ranging from 4% to 11% among cohorts.5,13 Cardiac imaging has also detected ECD infiltration around the heart, with cohorts reporting 37–76% with atrioventricular groove involvement.5,14 Palmisano et al. 13 found superior vena cava infiltration in 38% of their patients, and thoracic inferior vena cava infiltration in 24%, although this is likely a consequence of atriocaval compression by right atrial pseudomass rather than extension of myocardial infiltration to surrounding vasculature. Only two of their patients had superior vena cava stenosis in the absence of right atrium involvement.
Coronary artery disease (CAD) is known to occur in ECD, due to both traditional and disease-related factors. In a cohort of 200 patients, 23% had CAD, 3% had three-vessel CAD, 16% underwent percutaneous angioplasty, and 4% underwent surgical coronary artery bypass. 5 Left anterior descending artery stenosis due to extrinsic compression from pseudomass was noted in one patient; however, they found no statistically significant association between CAD and cardiac involvement, or between right atrioventricular sulcus infiltration and right coronary involvement. A cohort of 63 patients with ECD with coronary imaging found 5% had periarterial thickening and 10% had stenosis due to ECD which was unrelated to atherosclerosis seen in 42%. 12 On histology, foamy histiocytes can be seen surrounding coronary atheromatous lesions. 5 Azoulay et al. speculate CAD occurred frequently in ECD due to a high frequency of clonal hematopoiesis, chronic inflammatory syndrome, and burden of classical cardiovascular risk factors in predominantly male patients. 5
Cardiac magnetic resonance (CMR) findings have shown that patients with ECD with cardiac involvement had mild diffuse myocardial edema compared to those without cardiac involvement. 13 Almost all patients with ECD and cardiac involvement had nondense late gadolinium enhancement (LGE) following coronary distribution. In most cases, nondense fibrosis following a transmural pattern. Palmisano et al. suggest that, based on the CMR information, cardiac ECD can lead to fibrosis by causing ischemia, either by extrinsic compression of coronary arteries, by direct infiltration by histiocytes, or by promoting a chronic inflammatory state in downstream coronary circulation. 13
Vascular involvement
ECD notably affects large and medium-sized arteries, often seen as circumferential thickening around the artery, giving a coated appearance that mimics other conditions such as vasculitis15,16 and retroperitoneal fibrosis. 17
In a large cohort of 200 patients, 64% had aortic involvement on imaging. 5 Smaller cohorts of 63 and 54 patients found similar rates of aortic infiltration at 56% and 59%, respectively.12,13 Villatoro-Villar et al. saw equal distribution among thoracic and abdominal aorta at 48% each. 12 Further analysis revealed the frequency of individual aortic segments to be infra-renal abdominal aorta (46%), then descending thoracic aorta (41%), supra-renal abdominal aorta (40%), aortic arch (38%), and ascending aorta (35%). The most common abnormality of the thoracic aorta was periarterial thickening followed by atherosclerosis, 43% and 38%, respectively. The opposite presentation was seen in the abdominal aorta, with atherosclerosis occurring in 48% and periarterial thickening in 44%. Visceral artery branches (40%) were more often affected by ECD compared to aortic arch (26%) and pelvic/lower-extremity vessels (31%). Visceral arteries also displayed increased frequency of periarterial thickening compared to stenosis/occlusion, but higher rates of both than other branch arteries. Of the visceral branches, renal involvement was more frequent than mesenteric. Periarterial thickening also occurred more frequently than atherosclerosis in the visceral arteries, though the opposite was seen in arch branches and pelvic/lower-extremity arteries. The authors of this study note the presence of calcified atherosclerotic changes in abdominal and pelvic arteries was greater than expected based on lipid profiles and cardiovascular risk in their patients. 12
Only eight patients in total required arterial intervention: three required renal artery stenting for severe renovascular HTN, five required stenting in the lower extremities for claudication, and one patient required femoral-popliteal artery bypass. 12 Azoulay et al. report 16% of their cohort had renal artery involvement, with 8% of the cohort having undergone renal artery stenting. 5
Aortic ectasia and aneurysm have not been commonly reported in the literature. Of 63 patients with ECD, thoracic aortic ectasia was seen in eight individuals, more than double the rate of ectasia seen in branch arteries. 12 Aneurysms in ECD are less frequent but when reported are associated with rapid growth and rupture.18,19
Thrombosis
Venous and arterial thrombotic events are very rare in ECD with very few case reports in the literature. In 2014, there was a published case of a 50-year-old man with cerebral venous sinus thrombosis and multiple brain lesions who was later diagnosed with ECD. 20 A recent case report published in 2022 reported a 36-year-old woman with ECD initially presenting with multiple arterial thromboses involving the aortic arch, left subclavian artery, right common femoral artery, and bilateral internal iliac arteries who required stenting of the thoracic aorta and left common carotid artery, and embolectomy of the lower-extremity arteries. 21
Diagnosis and genetics
Diagnosis of ECD is difficult owing to its rarity, multisystem organ involvement, and variable presentation. In addition, ECD is not exclusively a pathologic diagnosis as classic histopathologic features may be absent, and requires consideration of clinical and radiographic findings with histopathology.
Consensus recommendations favor fluorodeoxyglucose (FDG) PET-CT from vertex to toes as part of diagnostic workup and to evaluate the extent of osseous lesions and other organ involvement. 1 Contrast-enhanced CT is second in line when PET-CT is not available. If diagnosed, further imaging including MRI of the brain with gadolinium and cardiac MRI is recommended. 1 Contrast and noncontrast CT shows concentric hypodense perivascular soft tissue, due to adventitial infiltration, coating the aorta.18,22 These areas show increased FDG uptake on PET-CT and appear isointense compared to muscle on both T1 and T2 on MRI.22,23 Large-vessel vasculitis (LVV) can mimic ECD, so it is important to differentiate the two. The adventitial sheath in ECD can be quite asymmetric in contrast to the more symmetric intimal/medial or transmural thickening seen in LVV.23,24 This difference can be seen on PET-CT, where LVV tends to have linear, diffuse, and circumferential uptake. 25 LVV most often involves the aortic arch and its branches, although Takayasu arteritis can also involve the mesenteric and renal arteries. 25 Involvement of the cranial arteries, specifically temporal arteries, is considered the hallmark of giant cell arteritis.25,26 This is contrary to ECD where temporal artery biopsy would be negative for vasculitic changes. Arterial involvement in ECD can also be segmental, similar to retroperitoneal fibrosis, though it is usually limited to anterior and lateral sides of the infra-renal and aorto-iliac regions of the abdominal aorta, and does not present with skeletal lesions.15,17,23,27 A summary of the key imaging features on CT, PET-CT, and MRI in ECD, retroperitoneal fibrosis, and LVV is provided in Table 1.
Key imaging features in Erdheim–Chester disease, retroperitoneal fibrosis, and large-vessel vasculitis.
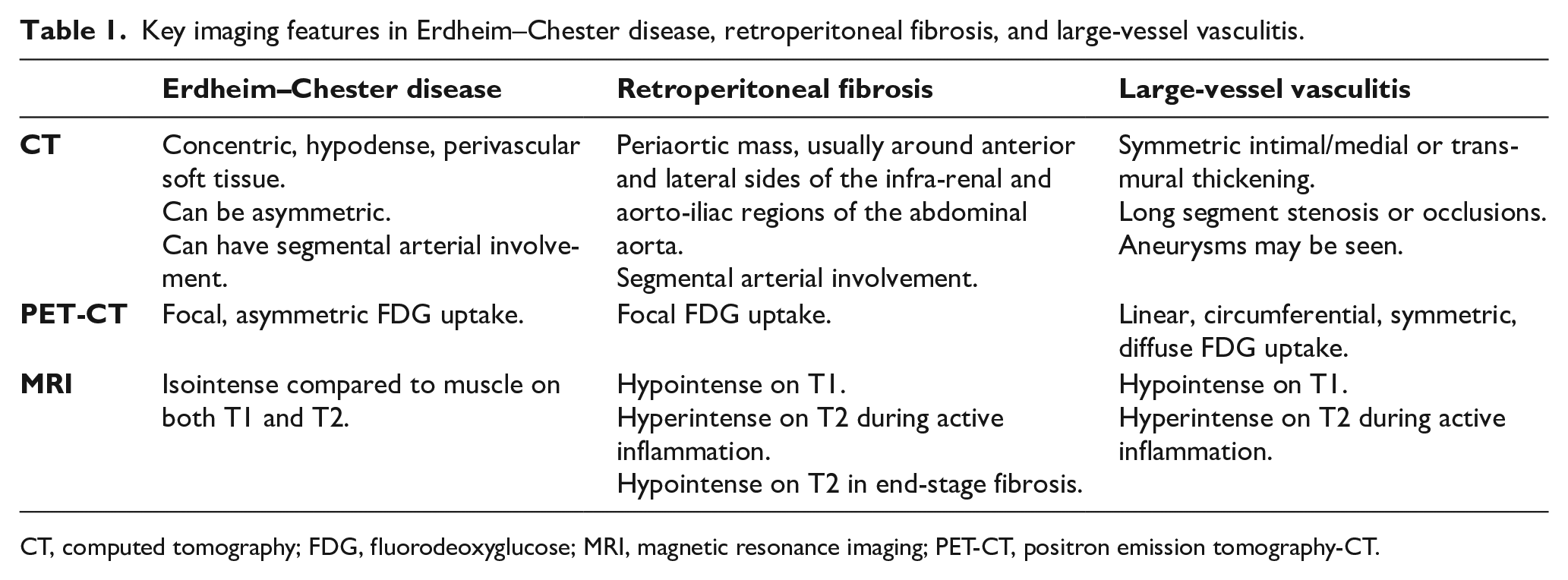
CT, computed tomography; FDG, fluorodeoxyglucose; MRI, magnetic resonance imaging; PET-CT, positron emission tomography-CT.
Biopsy is recommended for histopathologic assessment and immunohistochemical testing. Sites for biopsy include skin, bone, or soft tissue infiltrates, or the safest, most FDG-avid site. Classic histopathologic features include foamy histiocytes with small nuclei and surrounding fibrosis, multinucleated giant cells and Touton giant cells, although absence of these findings do not exclude the diagnosis of ECD. 1 Atypical features include florid lymphohistiocytic infiltrates or fibrotic lamellae with scattered foamy histiocytes and rare Touton giant cells. ECD can share features within the xanthogranuloma family, such as prominence of reactive lymphocytes, plasma cells, and rarely neutrophils. 1
Immunohistochemically, histiocytes in ECD were classically believed to be negative for S100, but weak or focally positive staining has been seen in up to 30% of cases. 1 Histiocytes in ECD can test negative for CD1a and CD207, and can be positive for CD68, CD163, factor XIIIa, and fascin. 1 All patients should be tested for the BRAF-V600E mutation as this can be positive in up to 60% of those with ECD. 1 When this is negative, consensus guidelines recommend testing for KRAS, NRAS, ARAF, RAF1, MAP2K1, MAP2K2, BRAF indels, and PI3KCA genes. 1
Treatment
Treatment of ECD continues to evolve but includes targeted therapies. Vemurafenib is first-line treatment for those with the BRAF-V600E mutation and life-threatening cardiac or neurologic involvement; dabrafenib and encorafenib can also be used. In addition to restoring plasma HDL levels, vemurafenib is noted to produce marked improvement at disease sites, including aortic infiltration, in patients with the BRAF-V600E mutation.1,6 For those without this mutation, targeted inhibition of MEK, mTOR, along with other tyrosine kinase inhibitors is recommended. Cobimetinib is a MEK inhibitor approved for the treatment of patients with ECD. Prior to the advent of targeted therapies, conventional therapy consisted of immunosuppressive and chemotherapeutic regimens. When mutation-directed therapy is not possible, then immune suppression with interferon-α-2a, cytokine-directed therapies (anakinra, canakinumab, tocilizumab, infliximab, etanercept), and cytotoxic chemotherapies (cladribine, cyclophosphamide, vinblastine) is used. Though the cornerstone of treatment of LVV is glucocorticoids, ECD monotherapy with glucocorticoids is not effective, though may be used as adjunctive therapy.1,26
Though vemurafenib leads to marked improvement in aortic infiltration, cardiac infiltration, and pseudotumors, such marked improvement was not seen in ECD cohorts that received other treatment regimens.4,5,12 In a cohort of 63 patients, complete resolution of arterial involvement was seen in less than 10%. Partial improvement was more common in the infra-renal aorta at 11% compared to 6% in both thoracic and supra-renal abdominal aorta. 12 Progression was also uncommon, but reported with decreasing frequency in the aortic arch, descending thoracic aorta, supra and infra-abdominal aorta, and iliac arteries. 12 A smaller cohort of 60 patients, of which 49 had follow-up imaging, also reported on disease response to therapy. 4 Stable disease was seen in 78% treated with interferon (IFN)-α, 57% treated with anakinra, and 20% with vemurafenib. 4 Progression was noted in 17% treated with IFN-α and 43% treated with anakinra. Partial response was seen in 5% treated with IFN-α and 60% treated with vemurafenib. 4 Recent data show that VEGF serum concentrations correlate with the level of improvement in cardiovascular manifestations, irrespective of the treatment regimen. 8 Surgery is not curative, and ECD is not radiosensitive, but these options may be considered for palliation. 1
Prognosis
The mortality rate of ECD can be high, estimated to be 60% at 3 years from the time of diagnosis, but varies depending on the sites of disease. 4 Cardiovascular involvement tends to have a poorer prognosis. Haroche et al. highlight that among their cohort of patients with ECD with follow-up data, 60% died and 31% of these deaths were due to cardiovascular involvement. 10 Infiltration of the central nervous system, lungs, and retroperitoneum have also been associated with poor prognosis, as well as advanced age. 1 Recent advances in diagnosis and treatment have led to improvements in 5-year survival rates, from 43% in 1996 to 83% in 2016. 1
Discussion
Cardiovascular involvement is frequent and can vary from asymptomatic to life-threatening with rapid deterioration requiring intervention.5,14 Cardiac involvement is significantly associated with extra-cardiovascular manifestations: renal, ocular, pulmonary, elevated inflammatory markers, and aortic involvement including coated aorta. 5 Clinical cardiac presentation can include pericarditis, arrhythmias, myocardial infarction, and symptomatic tamponade.2,5,9–11 In fact, 60% with pericardial effusions required a procedure. 14 Though atypical or severe myocardial involvement (symptomatic heart failure, decrease in left ventricular ejection fraction (LVEF), LGE lesions, or conduction disorders) can occur in ECD, CAD should be ruled-out first as nearly a quarter of patients with ECD also have CAD. 5
Aortic infiltration (‘coating’) is commonly reported; however, dilation is quite uncommon and the presence of aneurysm, dissection, or rupture is rarely seen in the literature.1,2,5,12,14 The few published cases involve men beyond their 8th decade of life with multiple comorbidities that could have also contributed to the aggressive nature of their aneurysmal disease.18,19 Perirenal artery thickening leads to extrinsic compression, stenosis/occlusion, resulting in resistant HTN and decreased renal function.2,5,12,14 ECD-induced RAS is amenable to angioplasty and stenting, often with marked improvement in HTN, though long-term outcomes are not known.3,5,12,28 Data on lower-extremity involvement are lacking but available literature suggest patients are prone to disease in pelvic and lower-extremity arteries. 12 Those complaining of claudication or leg pain may have their symptoms incorrectly attributed to osteolytic bone pain thereby delaying diagnosis and treatment. Revascularization with either stenting or bypass has been performed successfully. Coronary and peripheral arterial atherosclerotic disease appear to be prevalent among ECD, and are likely multifactorial. Villatoro-Villar et al. note that the severity and frequency of abdominal and pelvic atherosclerotic disease were more advanced than expected based on lipid and cardiovascular profiles. 12 They postulate this may be due to ECD upregulation of cytokines that also play a role in atherosclerosis formation. 12 Azoulay et al. note the high burden of traditional atherosclerotic risk factors in a largely male population in addition to ECD-specific factors and recommend that patients with cardiac symptoms undergo CAD evaluation prior to attributing symptoms to ECD. 5 Another possible process in both cohorts is the BRAF-V600E-mediated decrease in HDL with elevations in both triglycerides and lipoprotein(a). Though current consensus recommendations do not address atherosclerotic risk nor its management, there may be a role for lipid-lowering therapies such as statins and PCSK9 inhibitors, particularly in those who cannot tolerate targeted therapy.
VEGF has also shown itself to be a marker of interest in ECD. High levels are significantly associated with cardiovascular involvement and improvement in VEGF mirrors cardiovascular response to treatment and survival. 7 VEGF may serve as a method of detection and monitoring of cardiovascular manifestations in ECD.
Targeted therapy is important due to its ability to produce robust multisystem improvement, especially for those with life-threatening neurologic or cardiac complications. Compared to LVV and retroperitoneal fibrosis, which can have a similar radiologic appearance to ECD, ECD does not improve with glucocorticoid monotherapy, though it can be used as adjuncts to targeted therapies. 1 Surgery is not curative, and ECD is not ra-diosensitive, but these options may be considered for palliation. 1
Revisiting the two cases from our institution, the 24-year-old patient underwent mitral valve repair for her severe mitral regurgitation after multidisciplinary discussion between structural cardiology and cardiothoracic surgery. She was started on trametinib in 2019 and had complete response. Later she developed end-stage renal disease secondary to focal segmental glomerulosclerosis requiring a kidney transplant in 2022. Trametinib was stopped due to her immune suppression for her transplant, but she continued to have stable PET/CT imaging and is being monitored with routine imaging. The 68-year-old patient was without vascular imaging for many years and therefore MRA was performed, which confirmed previous findings in addition to worsening right RAS, ascending aortic ectasia, infra-renal abdominal aortic aneurysm (AAA), and left renal mass. He is currently being evaluated for the new left renal mass. In the setting of chronic kidney disease (CKD) with resistant HTN and a solitary functioning right kidney, he is also under consideration for right renal artery stenting.
Through reviewing the available literature and specific cases, it becomes clear that cardiovascular manifestations are common though quite variable in presentation and potentially lethal. Cardiovascular imaging is necessary in all patients with a diagnosis of ECD to assess and monitor cardiovascular involvement. Although studies continue to be published, more data are needed on patients with ECD, especially in terms of genetic and molecular markers which may lead to new therapeutic targets and allow for monitoring response to treatment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
