Abstract
Introduction
The walls of large arteries require the presence of a fine network of microvessels within them to function correctly, as the transfer of oxygen and nutrients solely from the arterial lumen proves insufficient.1 –3 Under pathological conditions of the arterial wall, such as atherosclerosis or vasculitis, the increased metabolic activity and the influence of pro-angiogenic factors from inflammatory cells trigger this network to expand.4 –6 This microvascularization predominantly emerges from the adventitia, although the existence of microvessels originating from the intima, which directly interfaces with the arterial lumen, is a topic of ongoing debate. Few studies have identified this specific subset of microvessels referred to as vasa vasorum interna (VVI). 3 Takayasu arteritis (TA) involves all three layers of the arterial wall. Histologically, there is a thickened adventitia with cellular infiltration, destruction of the vascular smooth muscle cells of the media and the elastic fiber network by cellular infiltration, and ultimately intimal fibro-sis. These anomalies are associated with developing vasa vasorum, around which an accelerated inflammatory reaction is observed. 4 Imaging this complex dynamic network would allow a better assessment of inflammatory activity and early wall remodeling. Ultrasound imaging combined with intravenous microbubble (MB) injection can thus be used to observe the passage of bubbles through the arterial wall.7,8 Ultrasound localization microscopy (ULM), using MB tracking within tissues, has enabled in vivo visualization of the microvascular network.9,10 In the Takayasu Arteritis Activity Evaluation by Ultrafast Ultrasound Imaging (TAK-UF) study, we used this imaging technique on the common carotid arteries of patients with TA to image and evaluate the carotid vasa vasorum network. 11 ULM technology requires an ultrasound scanner with a conventional ultrasound probe, dedicated ultrafast ultrasound imaging sequences, and the intravenous injection of MBs, available in routine practice. ULM enabled visualization of microvessels within the thickened carotid wall in TA, with significantly higher MB density in active cases. ULM provided a precise in vivo visualization of the vasa vasorum and gives access to quantification of the arterial wall vas-cularization. 11
Based on the results of the TAK-UF study, we aim to report the description of VVI within the carotid wall of patients with an active form of TA and discuss their consequences.
Methods
Population
The TAK-UF study (ClinicalTrials.gov Identifier: NCT03-956394) methodology has been previously reported. 11 The study protocol was approved by an independent ethics committee. All the patients provided written informed consent before enrolment. Briefly, in a single-center observational study, we included 16 patients with TA, of whom five had an active form of TA according to the NIH activity index, 12 measuring biological inflammation, morphological evaluation by conventional color Doppler ultrasound, computed tomography (CT) angiography, and magnetic resonance imaging, and associated with 18-fluorodeoxyglucose positron emission tomography. With ULM, a substantial passage of bubbles allowing visualization of a microvascular network within the carotid wall was seen only in cases with an active form of TA, thus allowing a clear dissociation between the two groups of patients.
Ultrasound localization microscopy (ULM)
ULM is a novel imaging technique enabling the visualization of microvessels and measuring their hemodynamics with resolution past the diffraction limit. 7 It combines an ultrafast imaging loop 13 with the injection of an ultrasound contrast agent, gas MBs. Ultrafast ultrasound imaging was performed with a 6.4-MHz central frequency linear probe SuperLinear™ SL10-2 and Aixplorer ultrasound scanner (SuperSonic Imagine, Aix-en-Provence, France). We developed an ultrafast ultrasound cine loop sequence (8 plane waves ranging from −5° to 5° tilt, frame rate 500 Hz, voltage 8 V, duration of 8 seconds continuously) long enough to capture MBs in the microvasculature but suitable for handheld carotid imaging and with low voltage to ensure a nonsignificant destruction of MBs with a mechanical index (MI) < 0.2. The ultrafast ultrasound cine loop was coupled with the intravenous injection of sulfur hexafluoride MBs (2.4 mL bolus of Sonovue; Bracco, Milano, Italy). Raw data were saved and processed offline.
ULM processing is presented in Figure 1. Preprocessing included the reconstruction of the ultrafast ultrasound cine loop images with delay-and-sum beamforming and coherent compounding to obtain in-phase and quadrature (IQ) demodulated images. Diastolic blocks were selected, based on axial wall displacements from Kasai autocorrelation of IQ data, 14 to prevent pulsatility motion in the walls. MB signals were extracted from the surrounding tissue based on singular value decomposition (SVD) discarding the first 20% singular value contributions. SVD filtering takes advantage of ultrafast high-spatiotemporal-resolution imaging and of the higher spatiotemporal coherence of tissue than MB or flow signals. 15 We can retrieve an ultrafast Doppler cine loop from the root mean squared norm of filtered IQ data over a temporal window. Finally, motion from the handheld probe was estimated based on the rigid registration of 200-ms lumen blocks. Each lumen block resulted from thresholded 200-ms ultrafast Doppler images.
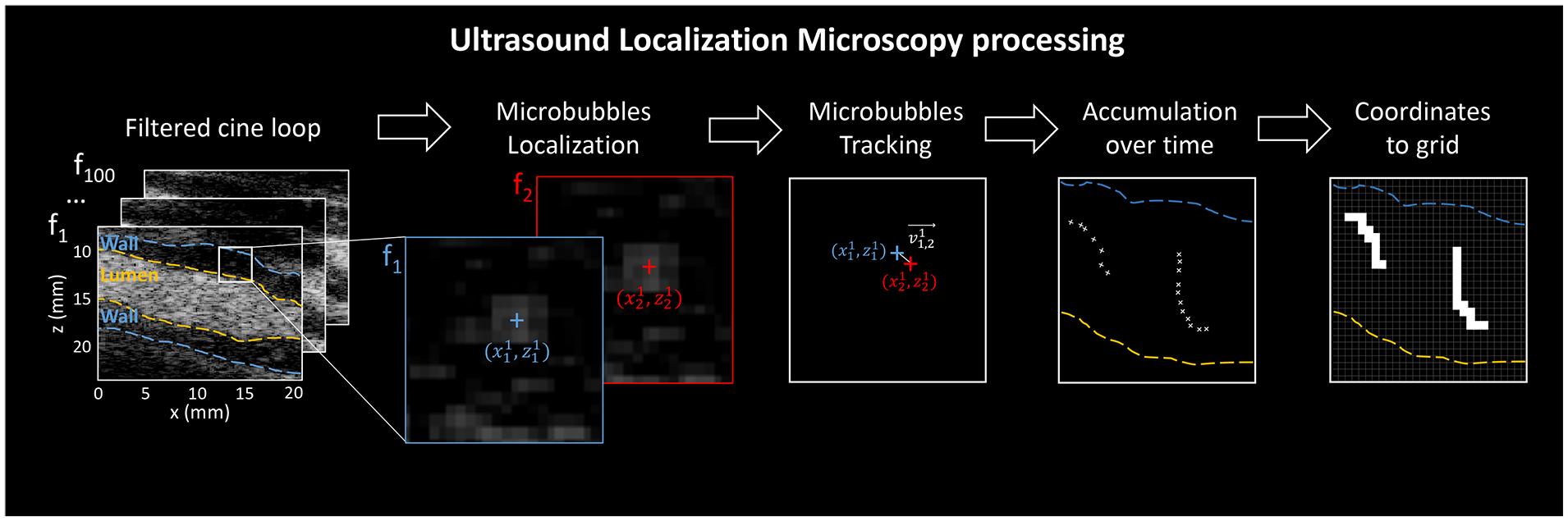
Ultrasound localization microscopy processing. After preprocessing, we retrieved a filtered ultrafast ultrasound cine loop. Microbubbles are localized at the sub-pixel resolution; microbubble positions are paired frame by frame for tracking, also giving instantaneous microbubble velocity; microbubble tracks are accumulated over time, and maps are reconstructed from the coordinates to sub-wavelength grid.
We performed ULM on the norm of the IQ images previously selected for diastole and SVD filtering. 16
First, we localized MBs in each frame using local maxima plus intensity above −40 dB and correlation above 0.6 with an ideal backscattered response of an isolated MB, as 2D gaussian (σx, σz) = (pitch, λsystem/2) = (200, 110) μm2. Then, we performed sub-wavelength localization, looking for the center of the MB spot. We detected the maxima in the second-order polynomial fitted 5 × 5 (550 × 550 μm2) MB neighborhood.
Next, we tracked the paired MB positions frame by frame, using a particle tracking algorithm 17 with a maximum distance corresponding to 100 m·s–1 and no gap filling. We only kept the MB tracks linked over three successive frames. Then, we corrected for handheld probe motion estimated previously. We also retrieved MB velocities from MB positions tracking and lag between frames. Finally, all MB tracks were accumulated over the acquisition.
We reconstructed ULM on a grid with pixel size divided by four, as (dx/4, dz/4) = (50.0, 27.5) μm2. Maps were reconstructed – for density counting all the MBs passing through a pixel and for velocity averaging the MB velocities passing through a pixel.
The arterial wall and lumen were manually segmented based on B-mode and ultrafast Doppler images.
Single microvessel identification obtained from microbubble (MB) tracking
Microvessels were visualized by tracking single MB using ULM within the arterial wall. We aimed to identify single vasa vasorum microvessel for further quantification. But a single microvessel can correspond to several MB tracks, preventing direct microvessel identification. The number of passages of MBs are proportional to the blood flow. In the arterial wall, the vasa vasorum flow rate is very low compared to the lumen. As a matter of comparison, the vasa vasorum flow rate was estimated at 3 ± 1 mL/min per 100 g of tissue in the coronary arteries of monkeys 18 and the carotid flow rate in humans is around 395 ± 79 mL/min. 19 In our acquisitions, we obtained a mean of 1.3 [1.2–1.4] tracks per ULM pixel in the arterial wall during the 8-second ultrafast ultrasound cine loop (i.e., almost a single passage of MBs in each vessel’s portion visualized). Therefore, we can consider that a single microvessel corresponded to a single MB track in the arterial wall.
Vasa vasorum interna (VVI) identification
From the microvascular network obtained within the arterial wall, we automatically identified some vasa vasorum directly interfacing with the arterial lumen. We previously retrieved vasa vasorum of the carotid wall as the MB tracks from ULM. Then VVI is roughly selected with MB tracks starting close to the lumen (defined as a close distance of < 0.2 mm from the lumen). We refined the selection by choosing MB tracks whether orthogonal to the lumen (45° < αtrack < 135°) or going deep within the wall (> 0.5 mm from the lumen), to exclude any possibility of artefactual tracks such as lumen flow MBs. All the postprocessing steps were performed using MATLAB software (MathWorks, Natick, MA, USA).
Results
Observation of vasa vasorum in communication with the arterial lumen
ULM provides an overview of the vasa vasorum by visualizing the MB tracks within the arterial wall (Figure 2). Among the MB tracks we found 2.2 [1.1–3.0] tracks per second in communication with the carotid lumen in a 2D acquisition. Figure 2 presents the postprocessing dedicated to VVI identification, and Figure 3 shows the result of the selected tracks corresponding with the VVI in a case. The measured flow velocity for tracks communicating with the carotid lumen was 35 [31–42] mm·s–1, slightly lower, even though not significant, than all MBs’ velocities: 40.5 [39.0–42.9] mm·s–1 (p = 0.081). The online video of the MBs’ passage is even more noticeable. This subset of vasa vasorum was found in all five cases of an active form of TA (see Video in the online supplementary material).
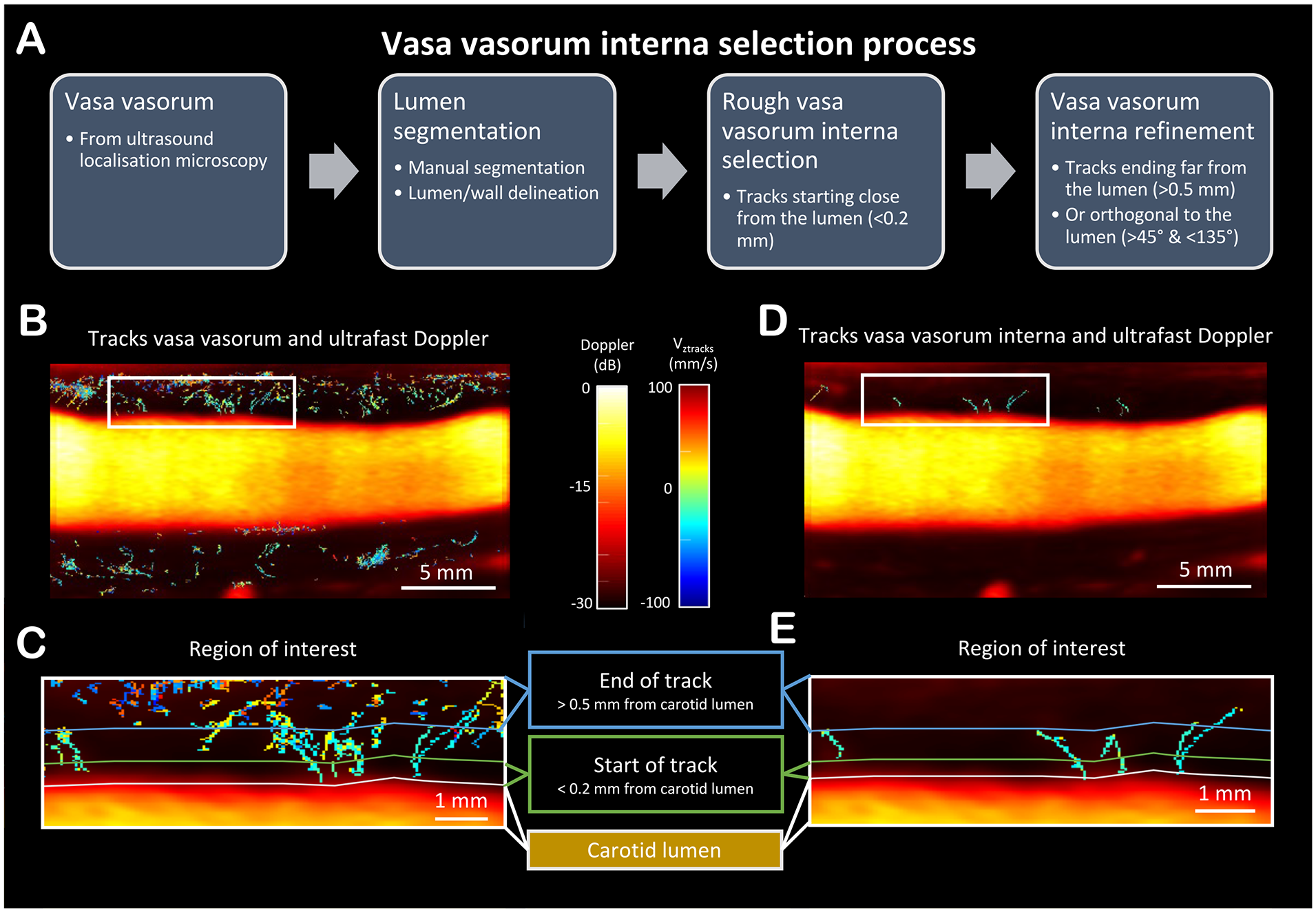
The vasa vasorum interna selection process with the different steps are described in
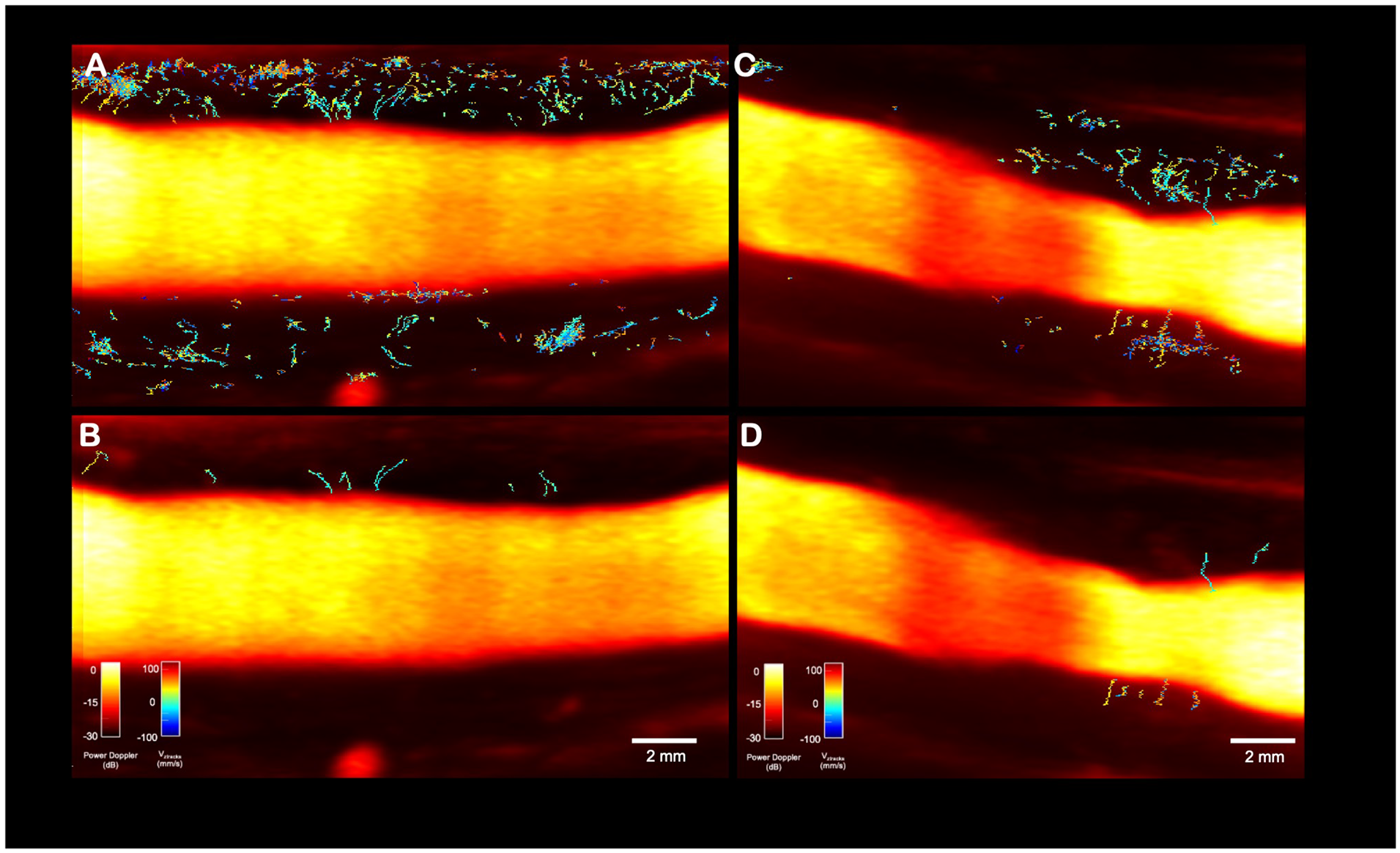
Ultrasound localization microscopy images with power Doppler background of two common carotid arteries
Vessels observed in the inner part of the wall cannot be vascular loops from the adventitia
The main confusion with a VVI would be the recording of a vascular loop entering the intima and then rising into the adventitia. This is a possibility linked to 2D imaging only. However, if the vessels observed in the vicinity of the lumen originated from the adventitia, then the following equation should be obtained:
Number of microvessels heading toward the carotid lumen = Number of microvessels heading toward the adventitia.
Based on the orientation of MB tracks, we obtained a more important rate of MB tracks heading toward the adventitia than MB tracks heading toward the lumen. Of all acquisitions of active TA patients, 88% [54–100] of MB tracks were moving toward the adventitia. The directional asymmetry of MB direction in the vicinity of the lumen was repeatedly observed on the acquisitions among the five patients with an active form of TA (Figure 4).
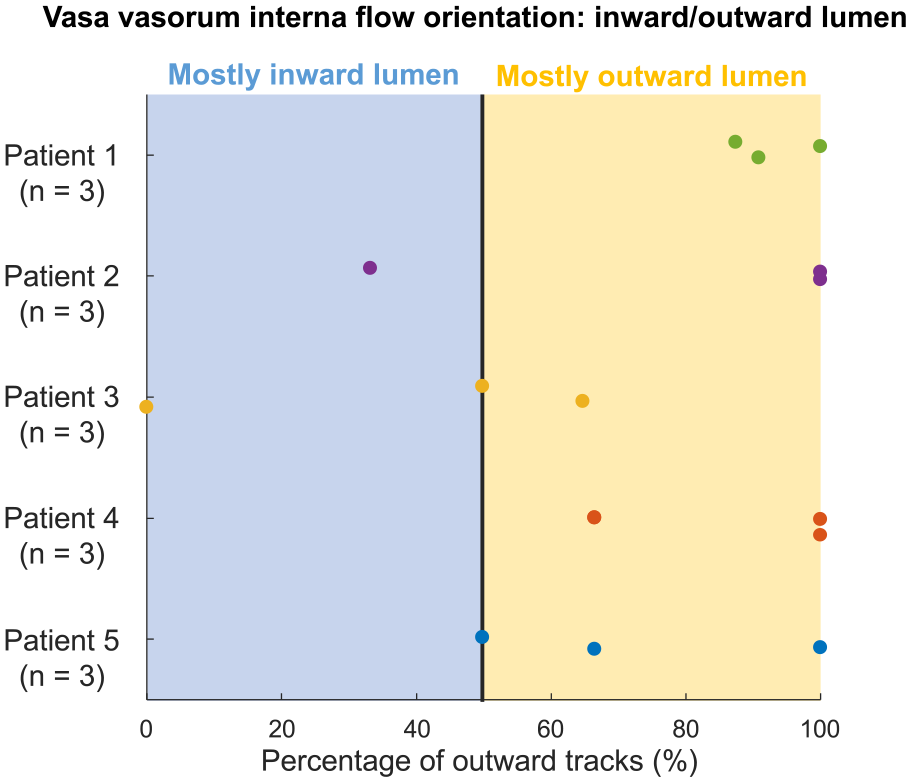
Vasa vasorum interna: inward/outward lumen.
Discussion
From the ULM data of patients with an active form of TA, our study documents the presence of VVI within the carotid wall; that is, vasa vasorum interfaced with the carotid lumen and extending deep into the wall. VVI have been demonstrated in several circumstances but more rarely described compared to the classic and predominant subset of vasa vasorum externa arising from the periarterial vascular networks and connected to the wall through the adventitia. The literature is weak, with different types of arteries investigated, and in various animal species. VVI were first identified by micro-CT analysis of pig coronary arteries,3,20 and then by electron microscopy of vascular corrosion cast on ex vivo arteries. 21 VVI have been reported in the ascending aorta of hooded seal, a species with a particularly thick aortic wall from 3 to 5 mm. 22 In this species, the VVI of the aorta appeared numerous, with their origin visible on the inner side of the aorta with the naked eye and imaged ex vivo using micro-CT. In humans, the presence of VVI has been suggested in vulnerable atherosclerotic carotid plaques by intraoperative indocyanine green video angiography. Some vasa vasorum appeared immediately after intracarotid injection of indocyanine green due to the direct passage from the lumen through vasa vasorum in direct communication with the lumen, whereas most vasa vasorum appeared later. 23 Lastly, VVI have been observed in the wall of saphenous grafts, where the vessel is detached from its physiological vascularization during coronary bypass surgery. 24
We cannot state whether VVI are present in a physiological state because the carotid wall is too thin and does not allow sufficient visualization by ULM. In inflammatory arteritis, the thickening of the wall and the increase of vasa vasorum density and diameter favors their visualization. In cases of non active arteritis (11 patients included in the TAK-UF study), these VVI were not observed, and the visualized microvascular network was sparse. The VVI observation emphasizes the involvement of the intima layer in TA. Endothelial cells appear to be critical in disease formation and progression.25,26 The hypotheses of the involvement of the intima in large vessel vasculitis or directly of the vasa vasorum in the pathogenesis of the disease remain to be explored. Beyond the technical challenge and the unusual nature of these observations, which nonetheless confirm histological data, the impact on clinical practice is not yet demonstrable, as these VVIs have received little or no attention to date.
Study limitations
A 3D proof would allow us to verify our hypothesis. However, there is no 3D probe available yet in clinical practice with a sufficient resolution in ultrafast ultrasound imaging that would allow us to perform this confirmation. Data here are limited to TA with carotid wall thickening. The TAK-UF study did not include patients without Takayasu arteritis. ULM on the arterial wall is difficult to perform without carotid wall thickening.
Conclusion
In Takayasu arteritis, the thickening of the carotid wall associated with inflammatory activity leads to the development of an increased vascularization within the carotid wall. Not only the vasa vasorum externa, the classical subset, but also the vasa vasorum interna, more rarely described up to now, were documented using the ULM. These microvessels, only observed in the active form of TA, show the involvement of the intimal layer in the disease’s inflammatory process, with microvascular development reaching the three tunics of the arterial wall.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241228262 – Supplemental material for Vasa vasorum interna in the carotid wall of active forms of Takayasu arteritis evidenced by ultrasound localization microscopy
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241228262 for Vasa vasorum interna in the carotid wall of active forms of Takayasu arteritis evidenced by ultrasound localization microscopy by Guillaume Goudot, Anatole Jimenez, Nassim Mohamedi, Jonas Sitruk, Louise Z Wang, Lina Khider, Patrick Bruneval, Emmanuel Messas, Mathieu Pernot and Tristan Mirault in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
French Society of Cardiology. ART (Technological Research Accelerator) biomedical ultrasound program of INSERM, France.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
