Abstract
Introduction:
Duplex ultrasound (DUS) is the modality of choice for surveillance of popliteal artery aneurysms (PAAs). However, noninvasive vascular laboratories have no standard guidelines for reporting results. This study assessed reports of PAA DUS for inclusion of information pertinent to operative decision-making and timing of surveillance.
Methods:
This study was a retrospective review of a multi-institutional repository that was queried for all patients with a PAA from 2008 to 2022 and confirmed via manual chart review. DUS reports were abstracted and images were individually annotated for features of interest including dimensions, flow abnormalities, and percent thrombus burden.
Results:
A total of 166 PAAs in 130 patients had at least one DUS available for viewing. Postoperative surveillance of PAAs was performed at several intervals: the first at 30 months (IQR 3.7–113, n = 44), the second at 64 months (IQR 20–172, n = 31), and the third at 152 months (IQR 46–217, n = 16) after the operation. The largest diameter of operative PAAs (median 27.5 mm, IQR 21.8–38.0) was significantly greater than nonoperative PAAs (median 20.9 mm, IQR 16.7–27.3); p < 0.01. Fewer than 33 (21%) reports commented on patency of distal runoff. We calculated an average percent thrombus of 60% (IQR 19–81) in nonoperative PAAs, which is significantly smaller than 75% (IQR 58–89) in operative PAAs; p < 0.01.
Conclusion:
In this multi-institutional retrospective study, PAAs are often not followed at intervals recommended by the Society for Vascular Surgery guidelines and do not include all measurements necessary for clinical decision-making in the multi-institutional repository studied. There should be standardization of PAA DUS protocols performed by all noninvasive vascular laboratories to ensure completeness of PAA DUS images and inclusion of characteristics pertinent to clinical decision-making in radiology reports.
Introduction
Duplex ultrasound (DUS) is the recommended modality for surveillance of vascular arterial aneurysms, including popliteal artery aneurysms (PAAs). 1 PAAs account for 70% of all peripheral arterial aneurysms, occurring at an incidence of 10.4 cases per million per year, 1 most commonly in 60- to 70-year-old men. 2 Although rupture of these aneurysms is uncommon at around 2%, 3 the more common and morbid complication of a PAA is a thromboembolic event resulting in acute limb ischemia requiring emergent repair and possible limb loss. 4
The Society for Vascular Surgery (SVS) has published a set of clinical practice guidelines based on limited available literature concerning PAAs, including recommendations for repair when the benefits outweigh the risks. 1 Currently, the grade 1 recommendation for a PAA with a diameter greater than 20 mm is surgical repair. However, the quality of evidence for this guideline is level B (moderate), 1 and size has not been shown to confer increased risk of thromboembolic complications or need for emergent surgery. 5 For PAAs with diameters smaller than 20 mm, the SVS guidelines provide a grade 2 recommendation for repair when the presence of thrombus or imaging of poor distal runoff are concerning for thromboembolic complications.
DUS can accurately diagnose PAAs and identify PAA features concerning for increased risk of a thromboembolic event, including mural thrombus and distal runoff. 6 Presence of a mural thrombus and poor distal runoff are associated with negative outcomes and therefore physicians usually proceed with operative intervention. Hence, early identification of high-risk features are essential for surgical planning. However, interpretation and result reporting of DUS for PAAs have often relied on institutional experience. 7 For example, noninvasive vascular laboratories that perform DUS for surveillance of PAAs may have internal criteria for reporting results. However, the SVS does not provide specific recommendations on different measurements to include in the DUS. There are no standard guidelines for reporting results apart from the PAA size itself, which is not the only feature used to dictate need for repair. 1 The purpose of this study was to assess reports of PAA DUS preoperatively and postoperatively for mention of characteristics that influence operative management per SVS guidelines and suggest new standards of reporting for PAAs.
Methods
The cohort of patients was derived from a multi-institutional repository from the Massachusetts General Brigham Research Patient Data Registry (RPDR). This centralized clinical data registry gathers demographics, diagnosis codes, encounter data, procedural codes, medications, and other patient clinical information. The RPDR database was queried for all patients with a diagnosis code for ‘Aneurysm of the artery of lower extremity’ (ICD-9: 442.3 / ICD-10: I72.4) and a preoperative DUS from inception of the database in 2008 to 2022 (Figure 1). The Partners Human Research Committee Institutional Review Board approved this study protocol for patients > 18 years of age and patient consent to participate was waived.
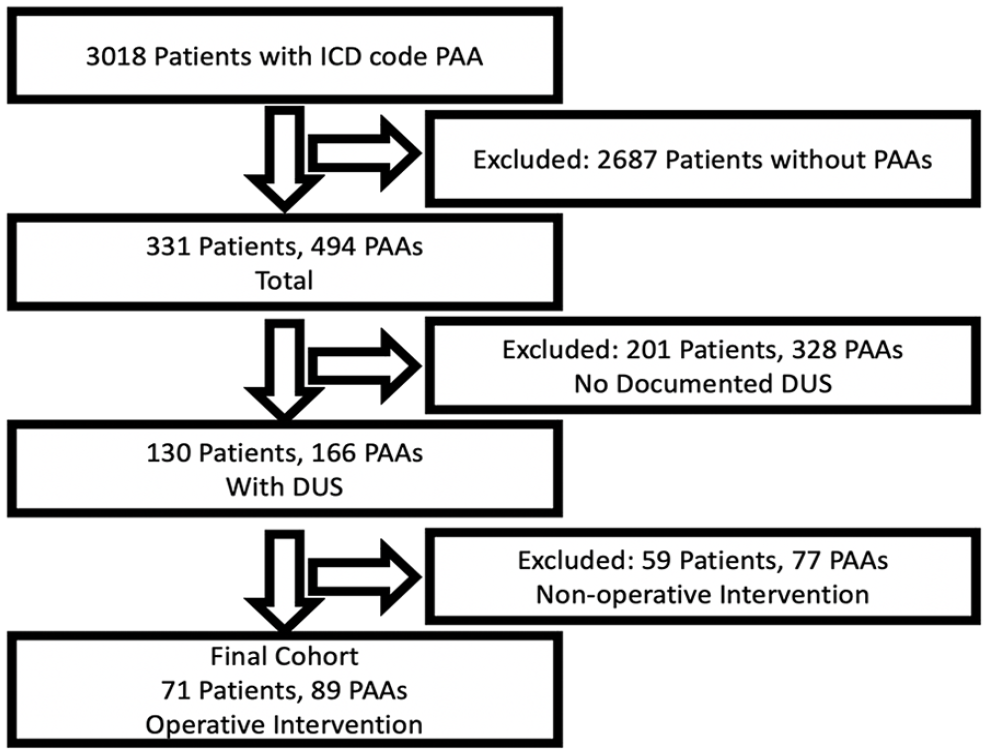
Flowchart of popliteal artery aneurysm identification and manual chart review to validate the presence or absence of a popliteal artery aneurysm.
Manual chart review was performed to confirm the presence of a PAA (Figure 1). PAAs that did not undergo operative intervention (i.e., nonoperative PAAs) were included if there was at least one DUS performed and the most recent DUS was used for analysis. Inclusion criteria for PAAs that underwent operative repair (i.e., operative PAAs) included the presence of at least one preoperative DUS and all measurements calculated were derived from the preoperative DUS performed closest to the time of the operation. All DUS were performed at an Intersocietal Accreditation Commission accredited vascular laboratory. Our institution currently has no standardized protocol for PAA DUS imaging. The reports of DUS for PAAs were abstracted to identify all information listed in the report for review. The operative criteria listed in SVS guidelines are a size > 20 mm, presence of a thrombus, and imaging evidence of poor distal runoff. A Registered Physician in Vascular Interpretation (RPVI) (GG) was blinded to ultrasound reports. GG annotated all DUS for operative criteria characteristics and additional characteristics that may be important for thromboembolic events, including the presence or absence of thrombus within the lumen (Figure 2). Index DUS was defined as the first DUS performed specifically for evaluation of a PAA. The definitions of each characteristic are as follows. Surface transversal area was defined as the surface of the popliteal aneurysm in cross section, with inclusion of the arterial wall. Internal surface area was defined as the surface of the popliteal aneurysm in cross section, without inclusion of the arterial wall. Patent channel area was defined as the surface of the circulating arterial lumen, free from thrombus. Thrombus area was defined as the area within the artery occupied by thrombus material. Anteroposterior diameter was defined as the largest diameter measured in the anterior to posterior dimension, outer wall to outer wall. Transverse diameter was defined as the largest diameter in any still image measured in the transverse dimension, outer wall to outer wall. Largest diameter was defined as the maximum diameter annotated in either anteroposterior or transverse dimension. Flow abnormalities were blood flow disturbances causing either occlusion or hemodynamically significant stenosis, defined as greater than two times the peak systolic velocity of the proximal segment. The percent thrombus burden calculated for each PAA was defined as the thrombus area divided by the total surface transversal area of the PAA.
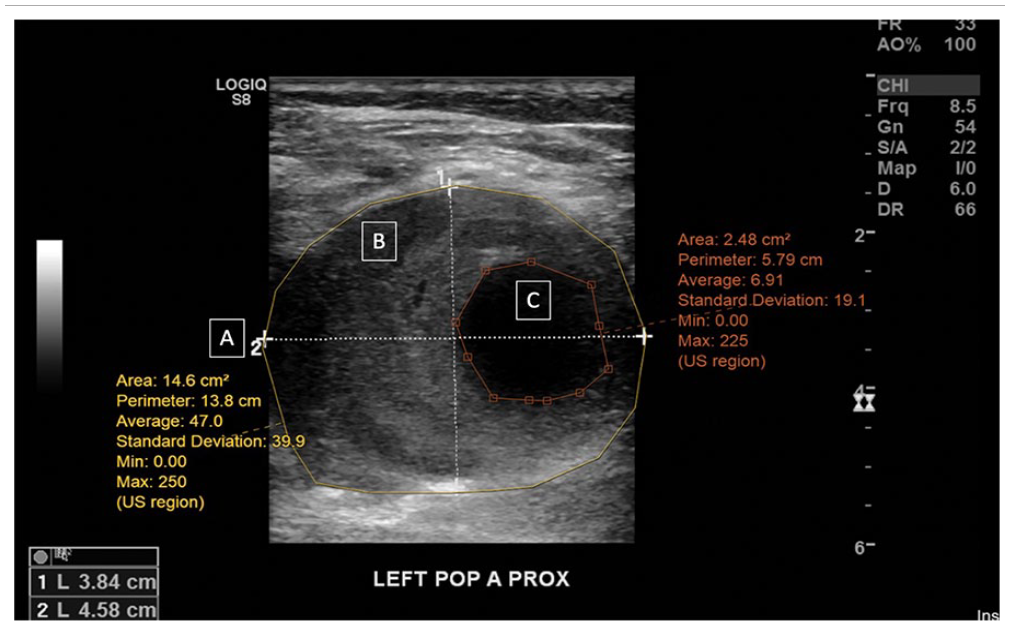
Duplex ultrasound measurements of left popliteal artery (LEFT POP A) aneurysm characteristics.
Statistical analysis
All results were expressed in terms of median and IQR given the nonnormality of the data. Each PAA was treated as a separate event, even in the case of a single patient with bilateral PAAs. Patient demographics were expressed for both operative and nonoperative PAAs using the descriptive statistics in Table 1. The interval of studies performed for surveillance of PAAs that underwent operative intervention were calculated for postoperative monitoring after both open and endovascular repair in Table 2. Differences between operative and nonoperative PAA characteristics on DUS were compared: continuous variables expressed as a median and IQR were compared using a Mann–Whitney test and categorical variables expressed as a percentage were compared using a chi-squared or Fisher’s exact test in Table 3.
Characteristics and demographics documented per individual patient (N = 130 patients).
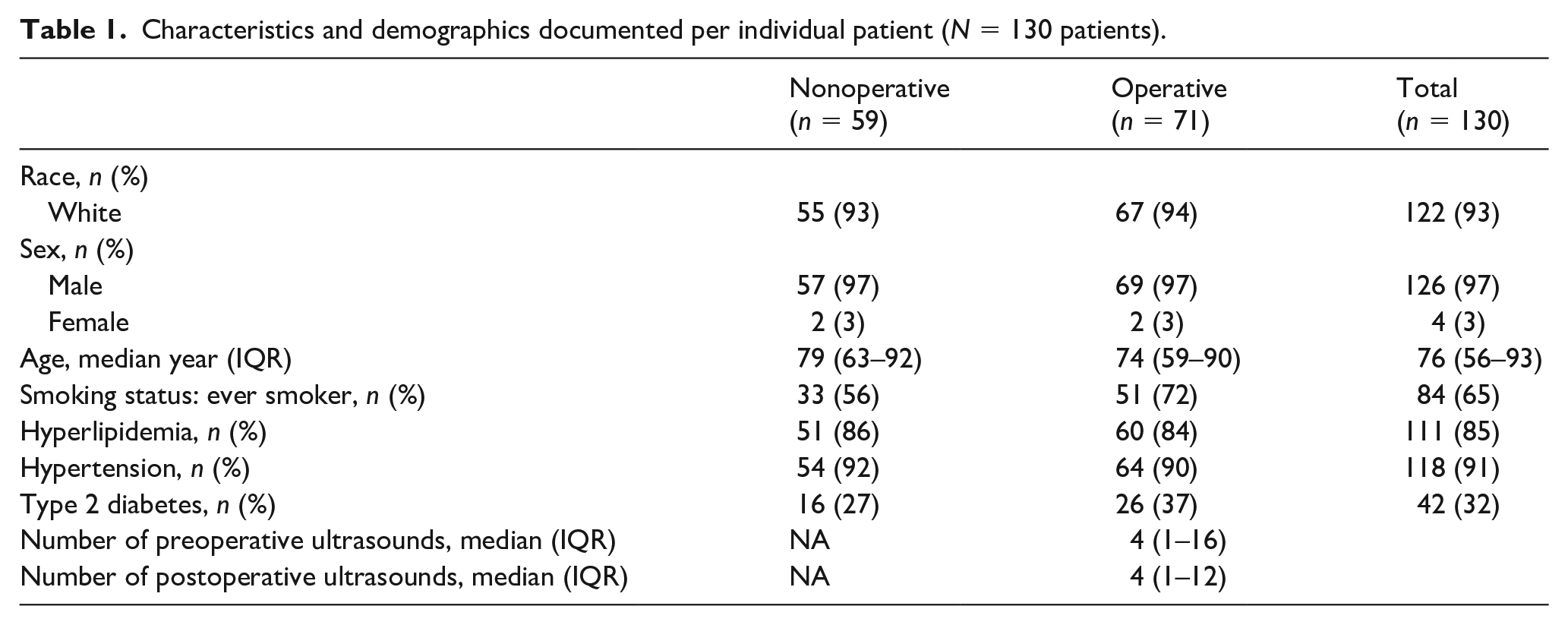
Duplex ultrasound surveillance frequency and intervals of popliteal artery aneurysms (n = 89) that required an operative intervention.
Index DUS was defined as the first DUS performed specifically for evaluation of a PAA.
DUS, duplex ultrasound; PAA, popliteal artery aneurysm.
Characteristics of 166 popliteal artery aneurysms measured directly from duplex ultrasound images.
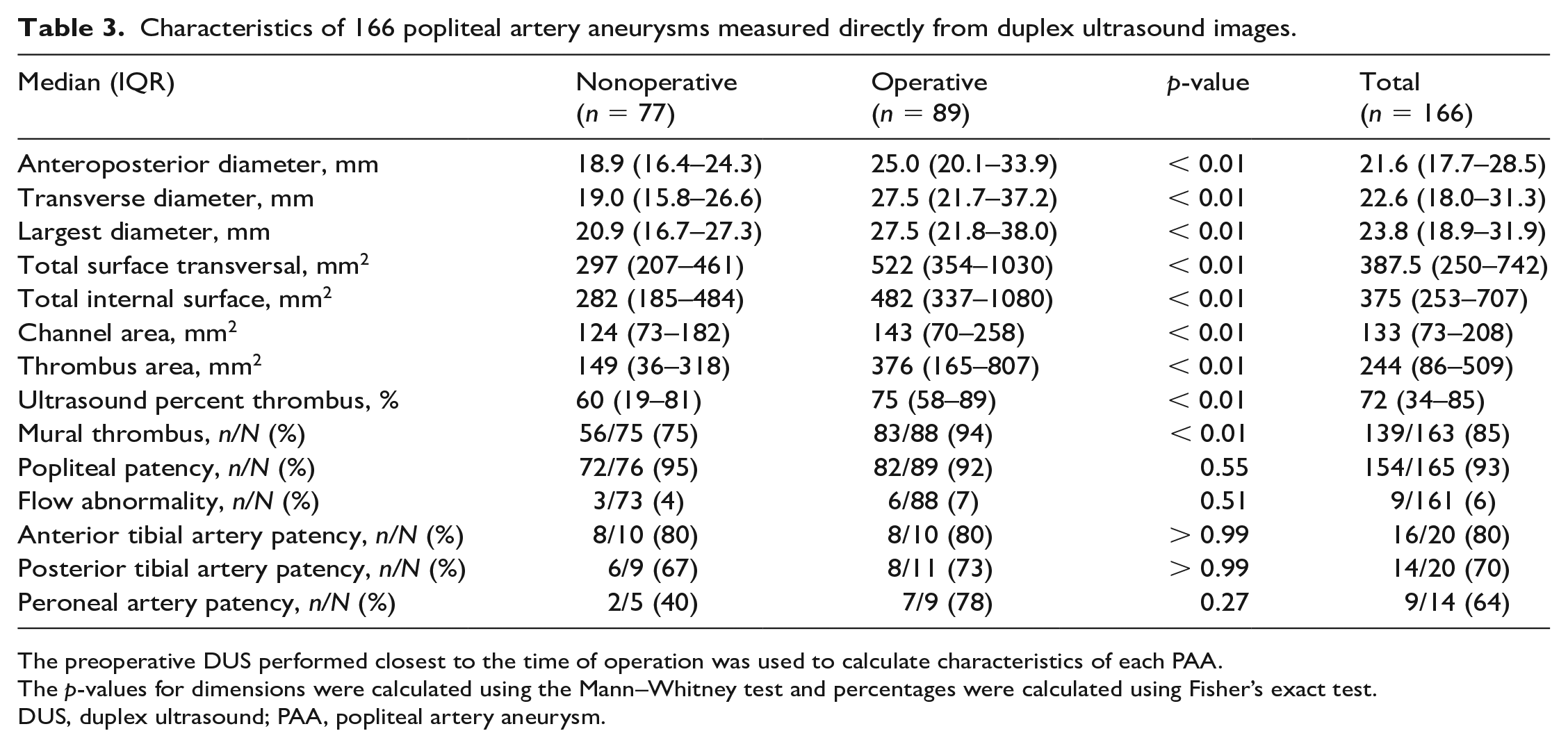
The preoperative DUS performed closest to the time of operation was used to calculate characteristics of each PAA.
The p-values for dimensions were calculated using the Mann–Whitney test and percentages were calculated using Fisher’s exact test.
DUS, duplex ultrasound; PAA, popliteal artery aneurysm.
Characteristics in DUS reports for operative and nonoperative PAAs appearing in the medical chart were compared to characteristics annotated by our RPVI certified MD (GG). Statistical significance was defined as p < 0.05. All analyses were performed using R version 4.2.1 (R Foundation for Statistical Computing).
Results
Demographics
There was a total of 166 PAAs studied in 130 patients who did not undergo operative intervention (n = 59 patients) or had at least one preoperative DUS and underwent operative intervention (n = 71 patients) (Figure 1). Of the 59 patients with nonoperative PAAs (n = 77 PAAs), 18 or 23% of patients had bilateral PAAs (n = 36 PAAs) and 41 or 53% of patients had unilateral PAAs. Of the 71 patients with operative PAAs (n = 89 PAAs), 18 or 25% of patients had bilateral PAAs (n = 36 PAAs) and 53 or 75% of patients had unilateral PAAs. A total of 130 patients had a median age of 76 (IQR 56–93) years, 126 (97%) were men, and 122 (93%) were White, with no significant difference between patients with nonoperative and operative PAAs (Table 1).
PAAs that did not undergo operative intervention had a median of five DUS (IQR 1–17) performed during the study period. PAAs that underwent operative repair had a median time from index DUS to operative repair of 10 months (Table 2). Each DUS surveillance occurred a median of four times (IQR 1–16) before and four times (IQR 1–12, n = 44) after the operation (Table 1). When conducted, postoperative DUS were performed at several intervals: the first at 30 months (IQR 3.7–113, n = 44), the second at 64 months (IQR 20–172, n = 31), and the third at 152 months (IQR 46–217, n = 16) after the operation (Table 2).
Popliteal aneurysm characteristics
The preoperative DUS performed closest to the time of operation was used to calculate characteristics of each PAA (Table 3). All aneurysm diameters calculated were significantly smaller in nonoperative PAAs compared to operative PAAs: the median anteroposterior diameter of nonoperative PAAs was 18.9 mm (IQR 16.4–24.3) compared to operative PAAs was 25.0 mm (IQR 20.1–33.9); p < 0.01. The median transverse diameter of nonoperative PAAs was 19.0 mm (IQR 15.8–26.6) compared to operative PAAs was 27.5 mm (IQR 21.7–37.2). The median largest diameter in nonoperative PAAs was 20.9 mm (IQR 16.7–27.3) compared to operative PAAs was 27.5 mm (IQR 21.8–38.0). Total areas were also calculated, including a median surface transversal area of 297 mm2 (IQR 185–484) in nonoperative PAAs and 522 mm2 (IQR 354–1030) in operative PAAs. The median internal surface area was significantly smaller in nonoperative PAAs at 282 mm2 (IQR 185–484) compared to operative PAAs at 482 mm2 (IQR 337–1080). The median patent channel area of 124 mm2 (IQR 73–182) for nonoperative and 143 mm2 (IQR 70–258) for operative PAAs with the median thrombus area of 149 mm2 (IQR 36–118) for nonoperative and 376 mm2 (IQR 165–807) for operative PAAs at the largest diameter were used to calculate a median percent thrombus burden for 166 aneurysms in 130 patients. The percent thrombus burden was significantly smaller in nonoperative PAAs at 60% (IQR 19–81) compared to operative PAAs at 75% (IQR 58–89); p = 0.001.
Mural thrombus was identified in 56 (75%) nonoperative PAAs and 83 (94%) operative PAAs (Table 3). Flow abnormalities within the PAA were identified in only three (4%) nonoperative PAAs and six (7%) operative PAAs. Patent distal runoff was identified in 16 (80%) of the operative and nonoperative anterior tibial arteries, six (67%) nonoperative and eight (73%) operative posterior tibial arteries, and two (40%) nonoperative and seven (78%) operative peroneal arteries when images were available.
Duplex ultrasound (DUS) reports
All preoperative DUS reports (n = 157) available for the authors to review were analyzed for the presence or absence of important PAA characteristics (Table 4). All DUS reports commented on the largest diameter in both operative and nonoperative PAAs. PAA patency was commented on in 73 (100%) nonoperative DUS reports and 82 (98%) operative DUS reports. Flow abnormalities were more frequently reported for nonoperative DUS in 71 (97%) reports than for operative DUS in 72 (86%) reports (p = 0.02) (Table 4). Out of all DUS reports, five (3%) calculated percent thrombus, 121 (77%) commented on mural thrombus, and fewer than 33 (21%) commented on patency of distal runoff: 16 (10%) anterior tibial, 33 (21%) posterior tibial, and three (2%) peroneal arteries (Table 4).
Reported popliteal artery aneurysm characteristics in 157 ultrasound reports available for review.
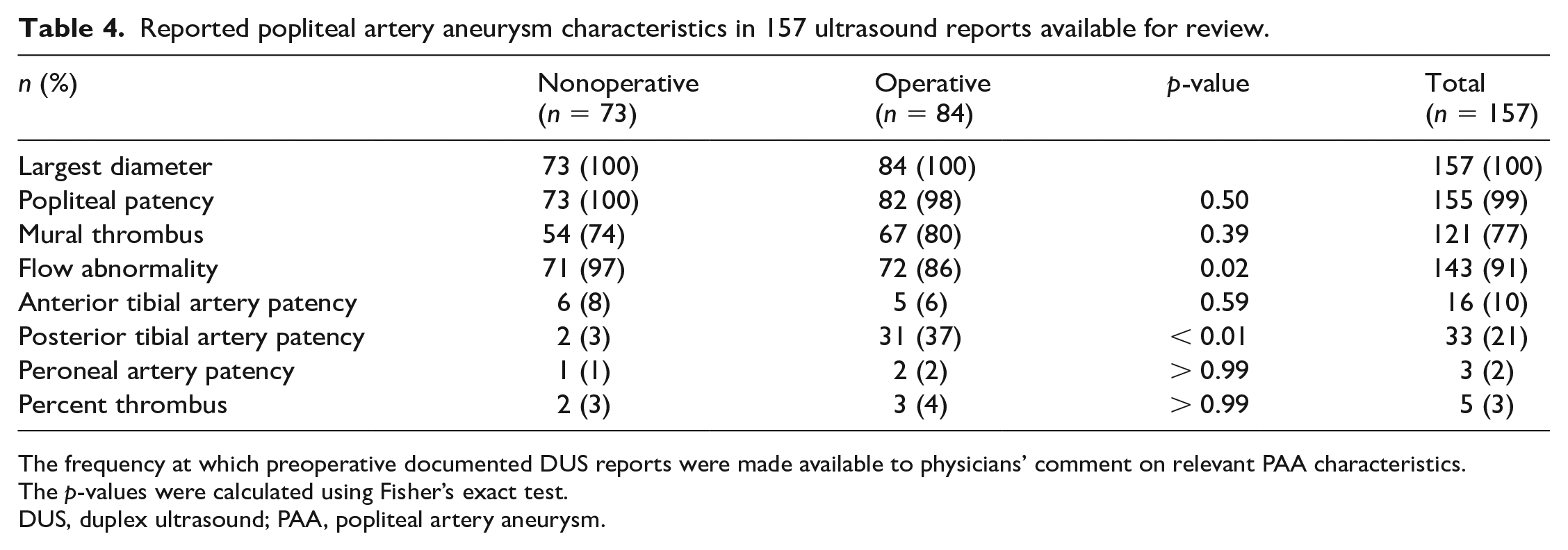
The frequency at which preoperative documented DUS reports were made available to physicians’ comment on relevant PAA characteristics.
The p-values were calculated using Fisher’s exact test.
DUS, duplex ultrasound; PAA, popliteal artery aneurysm.
Discussion
The current SVS guidelines concerning operative criteria for PAAs and postoperative DUS surveillance for PAAs are derived from a limited amount of literature. 1 Currently, DUS reports are not standardized and there are no societal recommendations on the standardization of PAA DUS protocols. In this dataset of PAAs, we show that PAAs are not monitored with DUS at intervals recommended by SVS guidelines and PAA characteristics that guide decision-making are performed and reported inconsistently. The operative criteria listed in the SVS guidelines are a size > 20 mm, presence of a thrombus, and imaging evidence of poor distal runoff. However, presence of a thrombus and imaging evidence of poor distal runoff are infrequently commented on in DUS reports in this study. Noninvasive vascular laboratories that perform DUS for surveillance of PAAs should include not only maximum diameter and flow abnormalities, but should also include presence of a mural thrombus and distal runoff.
Most patients were followed for 10.3 months before operative intervention and underwent a median of four postoperative DUS for surveillance at variable intervals. SVS guidelines recommend at least yearly DUS surveillance and physical examination for changes in symptoms once a PAA is diagnosed. 1 There are several clinical reasons for not performing yearly surveillance: different modalities of imaging were obtained, patients were lost to follow up, patients preferred less frequent monitoring, or multiple DUS demonstrated unchanged aneurysm size or slow growth.8,9 However, PAAs can undergo a period of slower growth before an accelerated growth phase in select patients with risk factors. 10 Accelerated growth has also been shown to occur once an aneurysm reaches a larger size. 11 Even still, small aneurysms can thrombose and cause distal limb ischemia. 11 There are specific SVS recommendations for PAA surveillance postoperatively, where DUS should be performed at 3, 6, and 12 months during the first postoperative year after endovascular or open repair. 1 However, this cohort of PAAs underwent their first postoperative DUS at 30 months (n = 44), second postoperative DUS at 64 months (n = 31), and third postoperative DUS at 152 months (n = 16) for those patients who did undergo a postoperative ultrasound. There are multiple explanations for the delay in DUS surveillance postoperatively: different modalities of imaging were obtained, patients were lost to follow up, or patients preferred less frequent monitoring. However, studies have shown most graft failures occur within 6 months of open intervention, 12 overall patency of endovascular intervention is only 50% at 12 months, 13 and thromboembolic complications develop within the first 18 months. 5 We would recommend institutions encourage adherence to the SVS guidelines of at least yearly DUS surveillance both preoperatively and incrementally postoperatively.
Nonoperative PAAs were found to have significantly smaller median diameters than operative PAAs at 18.9 mm versus 25.0 mm anteroposteriorly, 19.0 mm versus 27.5 mm transversely, and 20.9 mm versus 27.5 mm in the largest dimension. A diameter measurement was available in all DUS reports. However, the largest diameter in the nonoperative PAA group exceeded 20 mm, which is a criterion for repair, indicating other factors are being used to determine the need for operative repair. In our review of DUS, most of the operative and nonoperative PAAs had mural thrombus; distal runoff was not captured on a majority of DUS exams. Both mural thrombus and distal runoff abnormalities are strong indications for repair according to SVS guidelines. 1 However, a majority of DUS reports in the electronic health record did not comment on these specific criteria, even when mural thrombus was present or distal runoff abnormalities were present on available imaging. Mural thrombus is known to pose a risk of embolization in any aneurysm 14 and was identified as a significant predictor of sac expansion in a study of 87 PAAs. 12 The presence or absence of mural thrombus in a PAA has known predictive value of accelerated sac expansion and should be routinely identified in DUS reports. The occlusion of tibial vessels is evidence of distal embolization that can cause symptoms of limb ischemia, 13 and poor distal runoff in and of itself can cause thrombosis of PAA due to stagnant blood flow. 15 Distal runoff is a major consideration when determining the need for operative repair 1 and patency of all three tibial vessels should also be routinely assessed and reported in DUS.
The patent channel area and thrombus area are not well studied in the context of PAA but have been studied in the context of larger arteries, such as the aorta. Thrombus burden in the aortic wall of an abdominal aortic aneurysm, even after repair, has been associated with solid organ infarction from possible emboli. 16 One study by Ascher et al., focused on small PAAs, showed the presence and size of thrombus had more predictive value for thromboembolic events. 17 In this study, we found that thrombus area was significantly smaller in nonoperative PAAs, but we also found that the patent channel area was significantly smaller in nonoperative PAAs. When taking both values into context by calculating a ratio, we found that the overall percent thrombus burden calculated for 166 aneurysms in 130 patients was larger in operative PAAs at 75% versus nonoperative PAAs at 60%. This demonstrates that if an aneurysm is small but the thrombus burden is proportionally large, the patent channel area may remain unchanged. This indicates that both the patent channel area and the thrombus area must be considered. Reporting not only the presence or absence of thrombus, but the extent of thrombus burden is important in helping clinicians determine risk of embolization and need for repair.11,17 Standardizing DUS reports for PAAs to include this pertinent information may substantially aid clinician decision-making and management, just as standardization of carotid DUS has benefitted patient and provider management. 18
Study limitations
There are several limitations of the current study inherent to the population sampled. The Massachusetts General Brigham multi-institutional retrospective repository is comprised of tertiary hospitals from the Boston area, where referrals are received for complex cases, which limits the generalizability of the data. The retrospective nature of this study makes it difficult to obtain the exact technical details of the DUS and we cannot be sure the same techniques were used to yield consistent results across centers. Multiple different physicians have read the DUS studies in this retrospective review and therefore the result reporting may vary widely from provider to provider. DUS surveillance performed at other institutions outside of this healthcare system would not have been captured in this study, and therefore the median number of DUS could be underestimated. Follow up with computed tomography scans could have been performed in place of DUS surveillance. The quality of DUS varied by operator and therefore affected the diameters and areas calculated in this study. Institutional guidelines for DUS may have changed over the study period and precede publication of the SVS guidelines. Thus, information in current reports may include additional relevant information. Further studies should be conducted with more recent DUS data and a larger sample size to corroborate our results given our small cohort size. Similarly, a multicenter study could improve the generalizability of a larger sample size and aid in reforming the standardization of surveillance and surgical intervention guidelines. Certainly, a study identifying the relationship of these popliteal artery characteristics with choice of operative intervention and outcomes would be helpful in the management of such patients.
Conclusion
PAAs are often not followed at intervals recommended by SVS guidelines and do not include all measurements necessary for clinical decision-making in the multi-institutional repository studied. Interpretation and result reporting of DUS for PAAs are not standardized apart from a single measurement of the largest PAA diameter. To ensure completeness of DUS images as well as reports, there should be standardization of PAA DUS protocols. Standardizing DUS reports to include all characteristics of PAAs pertinent to clinical decision-making across noninvasive vascular laboratories could significantly improve patient care.
Footnotes
Data availability statement
Data used in this study are available on request from the corresponding author. Code to perform analyses in this manuscript are available from the authors upon request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
