Abstract
Background:
Vascular Ehlers–Danlos syndrome (vEDS) is an inherited connective tissue disorder characterized by arterial fragility. Celiprolol has been suggested to significantly reduce rates of vascular events in this setting, though real-world evidence is limited. The aim of this study was to report our experience with celiprolol therapy in vEDS management.
Methods:
Patients with a genetically confirmed diagnosis of vEDS who were referred for outpatient consultation at the Brescia University Hospital between January 2011 and July 2023 were included. At each visit, patients’ medical history, results of vascular imaging, and office blood pressure measurements were recorded. Celiprolol therapy was progressively titrated to the maximum tolerated dose of up to 400 mg daily, according to the patients’ tolerance.
Results:
Overall, 26 patients were included. Female sex was prevalent (62%). Mean (SD) age was 37 (16) years. Follow-up duration was 72 (41) months. At the last follow-up visit, all patients were on celiprolol therapy, 80% of whom were taking the maximum recommended dose. The yearly risk of symptomatic vascular events was 8.8%, the majority of which occurred after reaching the maximum recommended dose of celiprolol. No significant predictor of symptomatic vascular events was identified among patients’ clinical characteristics.
Conclusion:
In our cohort, rates of celiprolol use were high and the drug was well tolerated overall. Nonetheless, the risk of symptomatic vascular events remained nonnegligible. Future studies should identify reliable predictors of major adverse events and explore additional therapeutic strategies that could further lower the risk of life-threatening events in this population.
Introduction
Vascular Ehlers–Danlos syndrome (vEDS) is a rare autosomal dominant connective tissue disorder resulting from pathogenetic variants in COL3A1, which encodes type III collagen that provides strength and flexibility to blood vessels and membranes lining body cavities. The main feature of vEDS is severe tissue fragility leading to aneurysm, arterial dissection, or rupture, as well as spontaneous bowel perforation, especially of the sigmoid colon, and uterine rupture during pregnancy. 1 As a result, patients with vEDS experience a significantly reduced lifespan, with a median age of about 50 years. 2 The majority of deaths result from arterial rupture, 3 and only one in five patients remains free from vascular complications at the age of 60. 4 The diagnosis of vEDS is based on the 2017 international classification of EDS, which lists major and minor criteria that are suggestive of vEDS and should lead to diagnostic testing.1,5 It is widely recognized that even for skilled clinicians, confirming a diagnosis of vEDS requires molecular confirmation to differentiate from conditions with a similar presentation. A vEDS diagnosis is established upon the identification of a pathogenic variant on one allele of the COL3A1 gene. The majority of disease-causing variants are missense or splice site mutations that introduce a glycine substitution or result in an in-frame exon skip within the triple helical domain of the protein. A small proportion of affected individuals have null-mutations leading to COL3A1 haploinsufficiency, which is associated with a delayed onset of complications by almost two decades and an overall milder phenotype.1,2,4,6,7
In 2010, a randomized controlled trial (RCT) including 53 patients with vEDS, the Beta-Blocker in Ehlers–Danlos Syndrome Treatment (BBEST) trial, demonstrated that celiprolol therapy significantly reduced the risk of major vascular events by more than 50%. 8 These findings were further supported by a recent cohort study involving 40 Swedish patients, which revealed a similar yearly risk of major vascular events (~5%), despite a lower proportion of patients achieving the target dose of 400 mg per day (65%, compared with 88% in the BBEST study). 9 Interestingly, neither study was able to identify predictors for life-threatening events among patients’ clinical characteristics, although both low diastolic blood pressure (DBP) (< 62 mmHg) and high pulse pressure (PP) (> 50 mmHg) at baseline have been suggested to predict a poor response to celiprolol therapy.8,9
Owing to the rarity of the disease, real-world data on vEDS natural history after the publication of the BBEST trial remain limited.6,9,10 Accordingly, the aim of our study is to describe the clinical characteristics of a cohort of patients with vEDS followed at our center and to report our experience with celiprolol therapy in managing vEDS.
Materials and methods
Study population
We included patients with a molecularly confirmed diagnosis of vEDS who were referred for outpatient consultation at the Center for Rare Vascular Diseases at the Brescia University Hospital (‘Spedali Civili di Brescia’) between January 1, 2011 and July 31, 2023. Patients were initially evaluated at the Center for Hereditary Connective Tissue Disorders and Ehlers–Danlos Syndromes (‘Centro Connettivopatie Ereditarie e Sindromi di Ehlers–Danlos’) at the Department of Dermatology of the same institution. After obtaining written informed consent in compliance with the Italian legislation on genetic diagnostic tests, each patient underwent blood sampling to detect COL3A1 gene mutations. Specifically, at the Division of Biology and Genetics, Department of Molecular and Translational Medicine of the University of Brescia, bidirectional Sanger sequencing of COL3A1 coding sequences and exon–intron junctions was performed, as previously reported. 7 Once the presence of a causal mutation was confirmed, patients were referred to the aforementioned outpatient consultation to start celiprolol therapy, according to our local clinical protocols (‘Percorsi Diagnostico Terapeutici Assistenziali’). Patients were then offered annual follow-ups, except in cases of intercurrent clinical conditions necessitating closer monitoring. During follow-ups, patients underwent regular assessment of their vascular status by total body computed tomography angiography (CTA) or magnetic resonance imaging (MRI) before proceeding to medical examination. At each visit, all patients underwent a complete clinical assessment, which included medical history review, physical examination, and office BP measurement. Celiprolol therapy was progressively titrated to the highest tolerated dose (up to 400 mg daily) based on individual tolerance, as well as BP and heart rate (HR) values.
This study was carried out from routine clinical activity, and formal ethics review was not sought according to ethical standards in our institution during the study period. All patients included in the present study gave written informed consent allowing the reuse of their anonymized clinical data. The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice.
Clinical parameters
For each participant, the following data were collected at baseline: age, sex, body mass index (BMI; calculated as body weight [kg] divided by the square of the height [m]), age at vEDS diagnosis, presence of major and minor diagnostic criteria according to the 2017 EDS nosology, 5 the Beighton score (BS) for joint hypermobility, 11 reason for undergoing genetic testing, type of COL3A1 variant, concomitant drug treatment, and presence of relevant comorbidities.
Major vEDS-related events included vascular events (arterial rupture, thrombosis, dissection, or aneurysm), gastro-intestinal events (bowel rupture), development of spontaneous carotid-cavernous sinus fistula (sCCF) or other arteriovenous fistula, pulmonary events (pneumo- or hemothorax), other organ rupture, and complications during pregnancy (such as uterine rupture). During follow-up, in addition to the events mentioned above, instances of all-cause mortality were also recorded. Vascular events were classified as either asymptomatic (i.e., arterial lesions discovered incidentally during follow-up imaging) or symptomatic. The latter cases were further categorized based on their potential life-threatening nature, determined after careful reassessment of the patient’s record by two authors with expertise in vEDS management (MLM and GB). For each patient, we recorded the maximum achieved dose of celiprolol and any side effects. During initial evaluation and subsequent follow-up visits, the patients’ resting office BP was measured by the same physician using a semiautomatic sphygmomanometer. Three measurements were taken in a sitting position after 10 minutes of rest. Systolic, diastolic, and mean BP (SBP, DBP, and MBP, respectively) (mmHg), PP (mmHg), as well as HR (bpm) were recorded. For each parameter, the average of the three values was then calculated.
Statistical analysis
The Shapiro–Wilk test was used to test the normality of the data. Continuous variables are presented as mean and SD, regardless of the distribution’s normality, to ensure simplicity. Categorical variables were represented using proportions and frequencies. First, descriptive analyses of the cohort at baseline were performed. Additionally, we compared the evolution of office BP parameters in patients who started celiprolol therapy either during or after their first visit using a paired t-test. The occurrence of symptomatic vascular events during follow-up (such as arterial rupture, thrombosis, or dissection) was assessed through survival curves with the Kaplan–Meier method, splitting the overall cohort according to sex, genotype (glycine substitution vs others), and previous symptomatic vascular events to assess their effect on event rates with a two-tailed log rank test. Patients were censored at the time of their last follow-up visit. Baseline clinical characteristics were compared in patients with versus without symptomatic vascular events during follow-up by using the Student’s t-test. Then, the impact of potential predictors on such events was assessed by Cox regression analysis. Variables that achieved p < 0.10 in unadjusted analysis were included in a multivariable regression model. All statistical tests were two-tailed and carried out at a significance level of 0.05. Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA).
Results
Patient characteristics at baseline
Overall, the study cohort included 26 patients from 19 different families. Among the participants, 20 patients have been previously published, 7 whereas six additional patients were novel. Patients’ clinical and molecular features are summarized in Tables 1 and 2. Most of the cohort consisted of female patients (16; 62%). At baseline, the mean (SD) age was 37 (16) years, and mean BMI was 21.6 (3.1) kg/m2. The mean age at molecular diagnosis was 35 (16) years. In 21 patients (81%), genetic testing was performed after a major vEDS-related event, with an average time of 4.2 years (SD 6.7) between the event and diagnosis. Overall, the reason for genetic testing included positive family history in nine patients (35%), vascular events in 13 patients (50%), and spontaneous colonic perforation in two cases (8%). A family history of vEDS with a documented pathogenic COL3A1 variant was present in 46% of the cases (12 patients). Additionally, in five more subjects, a positive family history was assumed as well since they reported sudden death due to vascular and/or intestinal rupture in a close or first-degree relative.
Patients’ clinical characteristics.
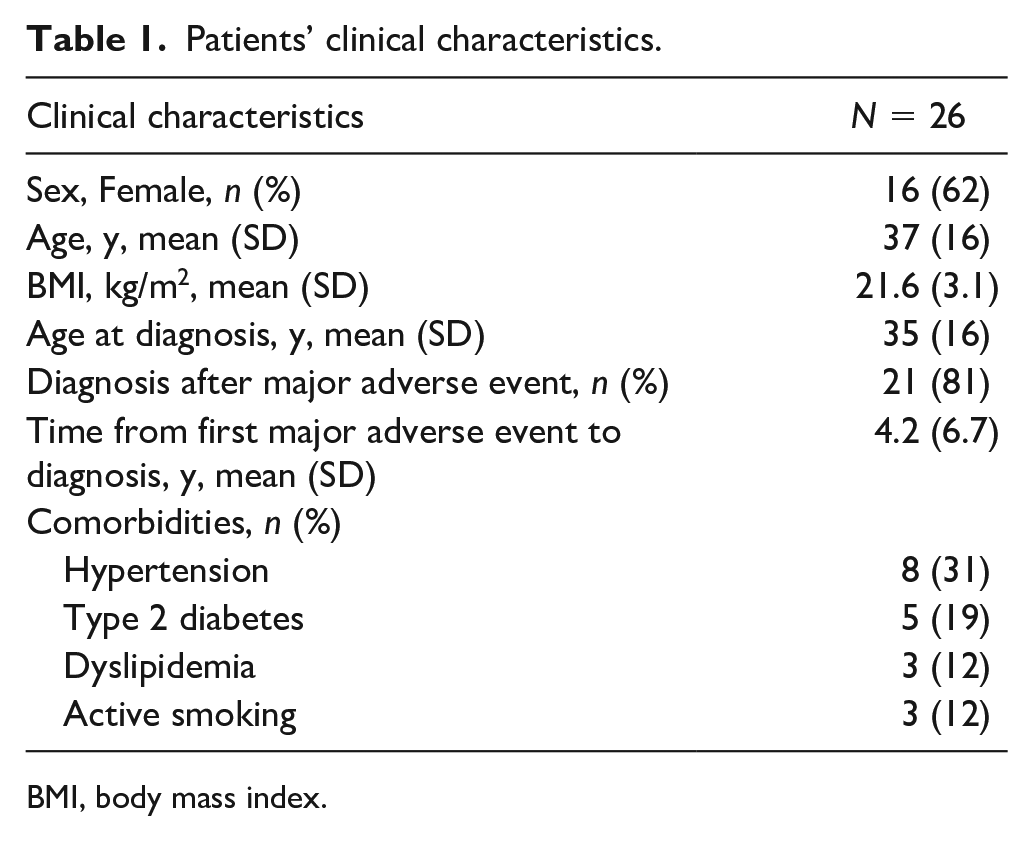
BMI, body mass index.
Patients’ medical history.
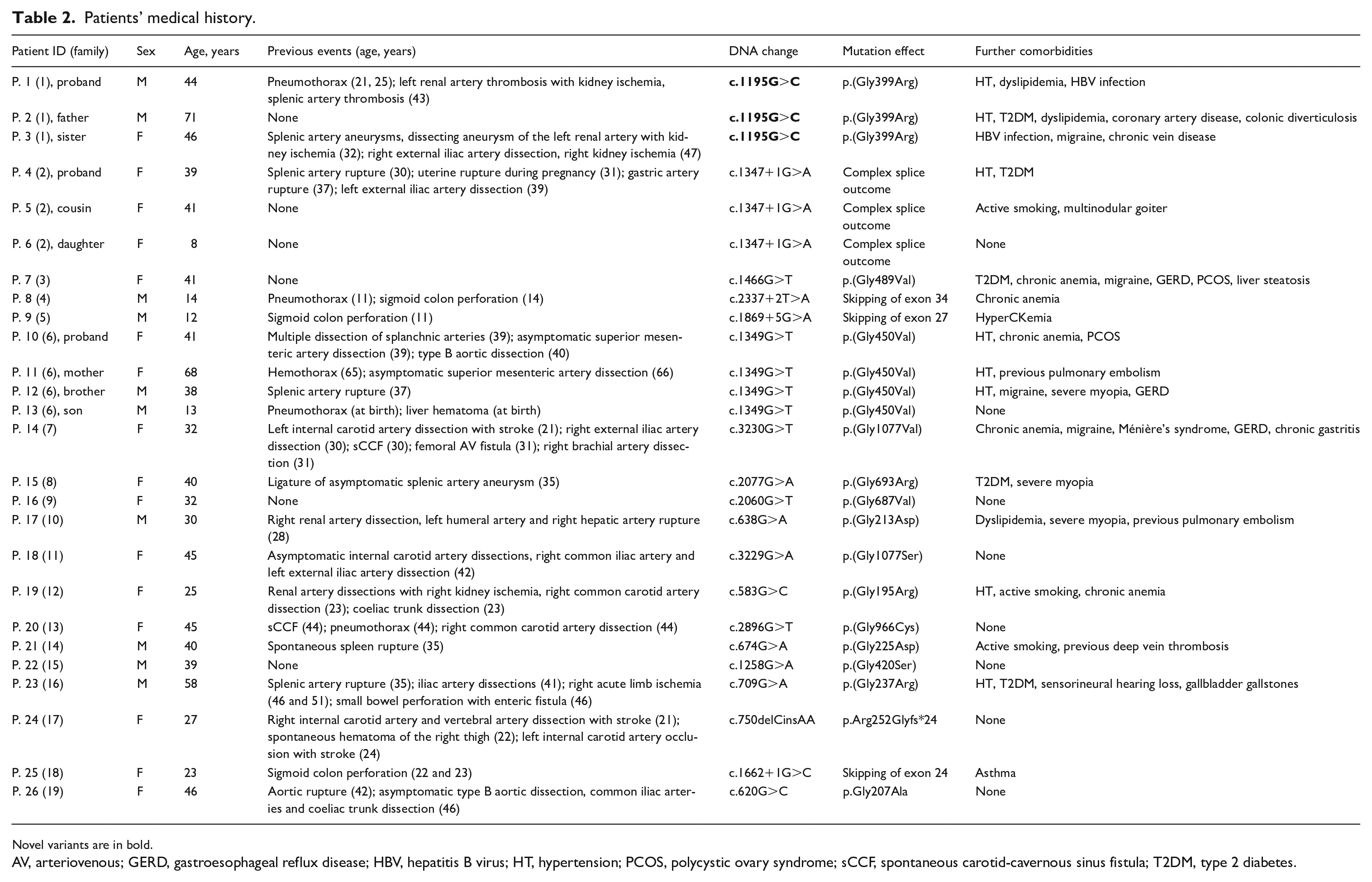
Novel variants are in bold.
AV, arteriovenous; GERD, gastroesophageal reflux disease; HBV, hepatitis B virus; HT, hypertension; PCOS, polycystic ovary syndrome; sCCF, spontaneous carotid-cavernous sinus fistula; T2DM, type 2 diabetes.
In all patients, a pathogenetic COL3A1 variant was identified. In particular, 18 variants were previously documented (i.e., 13 glycine substitutions, four in-frame exon skipping splice variants, all within the collagenous domain of the protein with a predicted dominant negative effect, and one small deletion/insertion likely representing a null-allele leading to haploinsufficiency). One variant was novel (a glycine substitution). Out of 19 probands, 14 were sporadic cases, with a verified de novo mutational event. Details of the pathogenic variants identified in the cohort are summarized in Table 2.
Comorbidities included hypertension in eight subjects (31%), type 2 diabetes in five patients (19%), and dyslipidemia in three patients (12%), whereas three subjects (12%) were active smokers. Further relevant comorbidities are listed in Table 2.
Concerning the major 2017 diagnostic criteria, seven patients (27%) reported previous arterial rupture at young age, four (15%) had spontaneous sigmoid colon perforation, and two (8%) had sCCF, whereas one out of eight women who experienced pregnancies (4%) had uterine rupture during the third trimester. In the entire cohort, 20 patients (77%) showed at least one major criterion; however, when considering index-patients only, this percentage decreased to 50% (13/26) due to the high prevalence of family history with documented pathogenic variants. In terms of minor criteria, thin, translucent skin (85%), extensive bruising (73%), hypermobility of small joints (54%), and a characteristic facial appearance (46%) were most frequently observed, whereas gingival recession/fragility (27%), early-onset varicose veins (23%), talipes equinovarus (19%), spontaneous pneumothorax (12%), and acrogeria (4%) were less common. No patient had a history of congenital hip dislocation, keratoconus, or tendon and muscle rupture (Table 3). The mean (SD) BS was 3.3 (2.4), with eight patients (29%) demonstrating generalized joint hypermobility scoring ⩾ 5.
Diagnostic criteria according to the 2017 Ehlers–Danlos syndrome nosology.
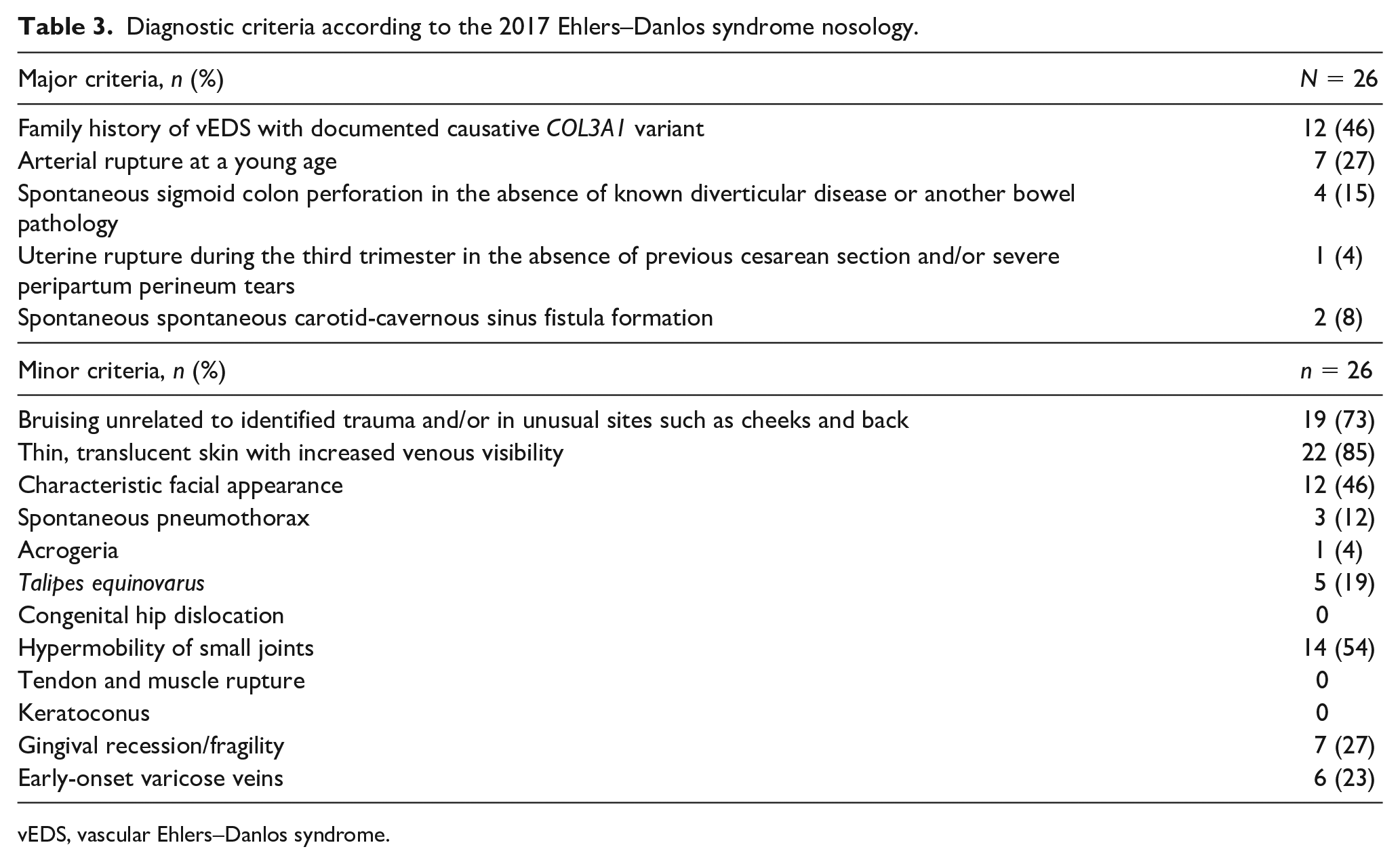
vEDS, vascular Ehlers–Danlos syndrome.
Clinical events at baseline
Overall, 21 patients (81% of the cohort) had one or more major vEDS-related events at the first outpatient visit. Vascular events occurred in 62% of the cases (16/26), for a total of 29 events (ranging from one to four per patient), including 24 symptomatic events and five arterial lesions detected incidentally on vascular imaging (three arterial dissections and one splenic artery aneurysm treated with ligation). Symptomatic events included aortic rupture or type B aortic dissection (two cases), renal artery thrombosis or dissection with renal infarction (three cases), splenic artery thrombosis with spleen infarction (one case), splenic or gastric artery rupture (one cases), coeliac trunk dissection with pancreatic infarction (one case), liver hematoma or hepatic artery rupture (two cases), carotid artery thrombosis or dissection with ischemic stroke (two cases), iliac artery thrombosis or dissection (three cases), and brachial artery rupture or dissection (two cases). Four patients experienced gastro-intestinal events for a total of six events, including five spontaneous sigmoid colon perforations and one small bowel tearing with enteric fistula development. Pulmonary events occurred in five patients for a total of six events, including five pneumothorax and one hemothorax. Three patients presented with arteriovenous fistula for a total of four events, including two sCCFs, and one splenic and one femoral arteriovenous fistula, whereas one patient developed spontaneous spleen rupture.
Overall, the mean (SD) age at first major adverse event was 30 (14) years: 36 (11) years for vascular events, 23 (16) years for gastro-intestinal events, 46 (15) years for sCCF or other arteriovenous fistula, and 27 (24) years for pulmonary events. Spontaneous spleen rupture occurred in one patient at age 36, whereas uterine rupture occurred in another woman at age 31.
Treatment with celiprolol and events during follow-up
Overall, 19 patients from the cohort had at least one follow-up visit, and one patient died after the first visit. Mean (SD) follow-up duration was 72 (41) months, ranging from 10 to 140 months, with a total follow-up of 125 patient-years. At baseline, six patients were already on celiprolol therapy. In three subjects, treatment was not initiated at the first visit: one pediatric patient (the only subject initially judged ineligible for celiprolol therapy) and two patients who initially refused treatment. At the end of the follow-up period, however, all patients were under celiprolol therapy, 16 of whom (80%) were on the recommended dose of 400 mg daily. The mean (SD) time from baseline visit to maximum recommended dose was 31 (30) months. One patient reached a maximum dose of 200 mg due to intolerance to higher doses (experiencing fatigue). Another patient was still undergoing drug titration at the last follow-up, reporting no adverse events. Two patients died during celiprolol titration. Overall, celiprolol therapy was started before the development of vascular pathology in eight out of 20 subjects. Four of these patients had been diagnosed with vEDS as first-degree relatives of affected individuals, three following gastro-intestinal events, and one due to the presence of other clinical features suggestive of vEDS. Some patients received other medications, either as single therapy or in combination: angiotensin II receptor blockers (11 patients), calcium channel blockers (three patients), diuretics (one patient), statins (one patient), and antithrombotic drugs (six patients).
Among the 20 patients with at least one follow-up visit, 14 (70%) had one or more major vEDS-related events during their follow-up period. The mean (SD) time from the baseline visit to the occurrence of the first event was 34 (24) months. Vascular events occurred in 60% of the cases (12/20), for a total of 18 events (ranging from one to two per patient), including 11 symptomatic events in eight patients. Additionally, seven arterial lesions were incidentally detected on vascular imaging, including one iliac artery pseudoaneurysm, four iliac artery dissections, one carotid artery aneurysm, and one carotid artery dissection. Symptomatic events included type B aortic dissection (two cases), renal artery thrombosis or dissection with renal infarction (three cases), hepatic artery rupture (two cases), splenic artery rupture (one case), iliac artery thrombosis or dissection (three cases), and spontaneous hematoma of the iliopsoas or thigh (two cases). Two patients experienced gastro-intestinal events: one with a spontaneous sigmoid colon perforation and the other with an appendicular perforation. One patient experienced spontaneous hemothorax. Two patients developed spontaneous spleen rupture, one of whom also presented with gallbladder rupture. No cases of arteriovenous fistula or uterine rupture during pregnancy were observed. Three patients died: a 15-year-old boy and a 37-year-old woman, both due to type B aortic dissection and rupture of hepatic arteries during splenectomy for spontaneous spleen rupture, and a 42-year-old man from splenic artery rupture. All these patients were on celiprolol therapy. Ten out of 18 vascular events (six symptomatic events including one fatal event, and four arterial lesions detected incidentally on vascular imaging) occurred after reaching the recommended dose of 400 mg daily of celiprolol. In the three patients with delayed initiation of celiprolol therapy, no adverse events were reported in the period between the first visit and the start of treatment (range 8–43 months). Overall symptomatic vascular events occurring after the initiation of celiprolol therapy are depicted in Figure 1.
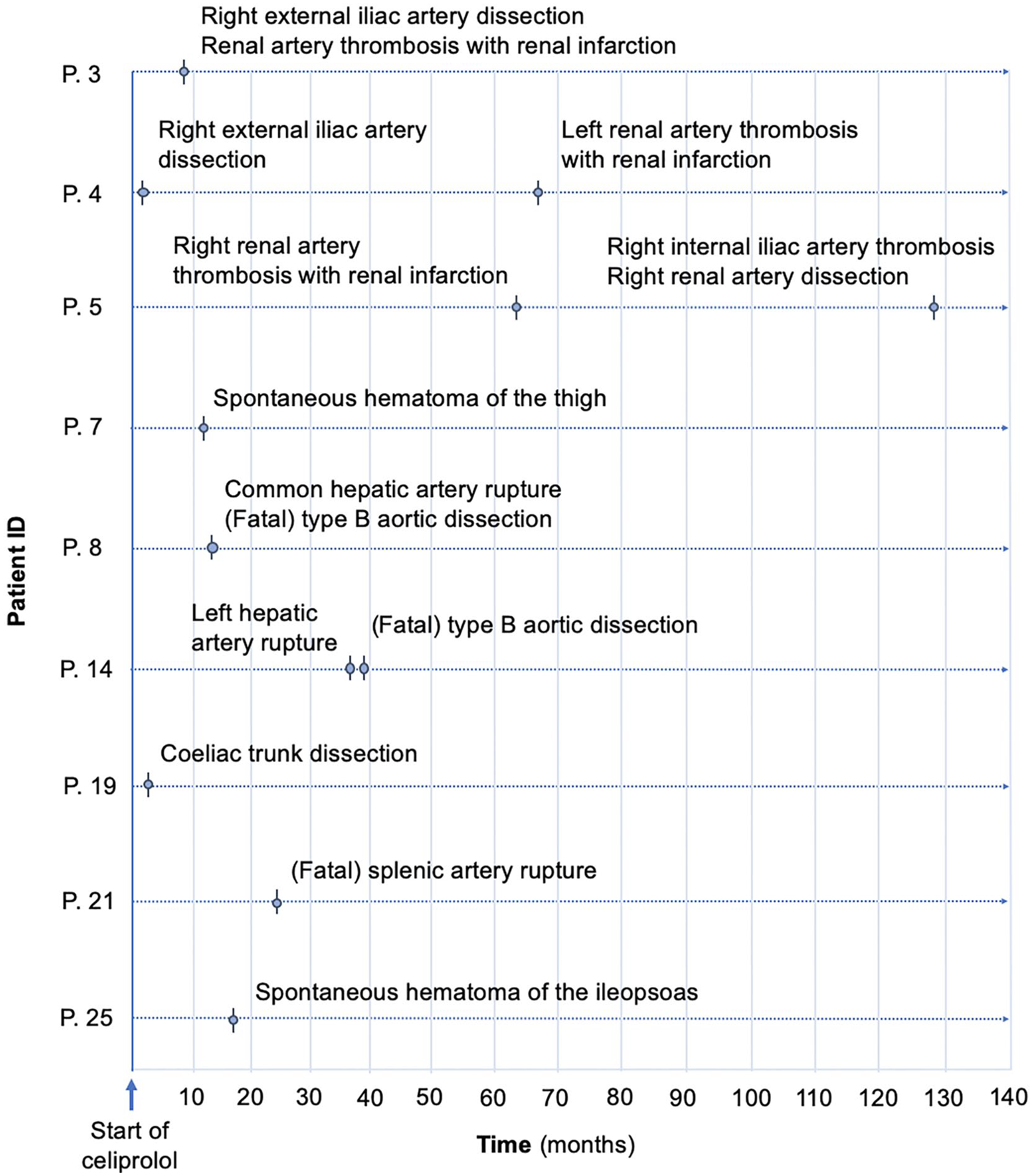
Symptomatic vascular events occurring after the initiation of celiprolol therapy.
The yearly risk of symptomatic vascular events in this cohort was 8.8% (11/125), which decreased to 6.4% (8/125) when only considering life-threatening incidents and increased to 14.4% (18/125) when asymptomatic new lesions were included. The survival analysis for symptomatic vascular events is illustrated in Figure 2. There were no significant differences in terms of baseline clinical characteristics between patients with versus without events during follow-up (online Supplementary Table 1), nor did Cox regression analysis identify any predictor of these events among the baseline clinical characteristics, including glycine substitution as DNA change (hazard ratio [HR] 2.18; 95% CI 0.54–8.73; p = 0.27), age on celiprolol introduction (HR 1.02; 95% CI 0.97–1.08; p = 0.50), DBP (HR 1.04; 95% CI 0.97–1.10; p = 0.28), and PP (HR 0.92; 95% CI 0.82–1.02; p = 0.10) (online Supplementary Table 2).
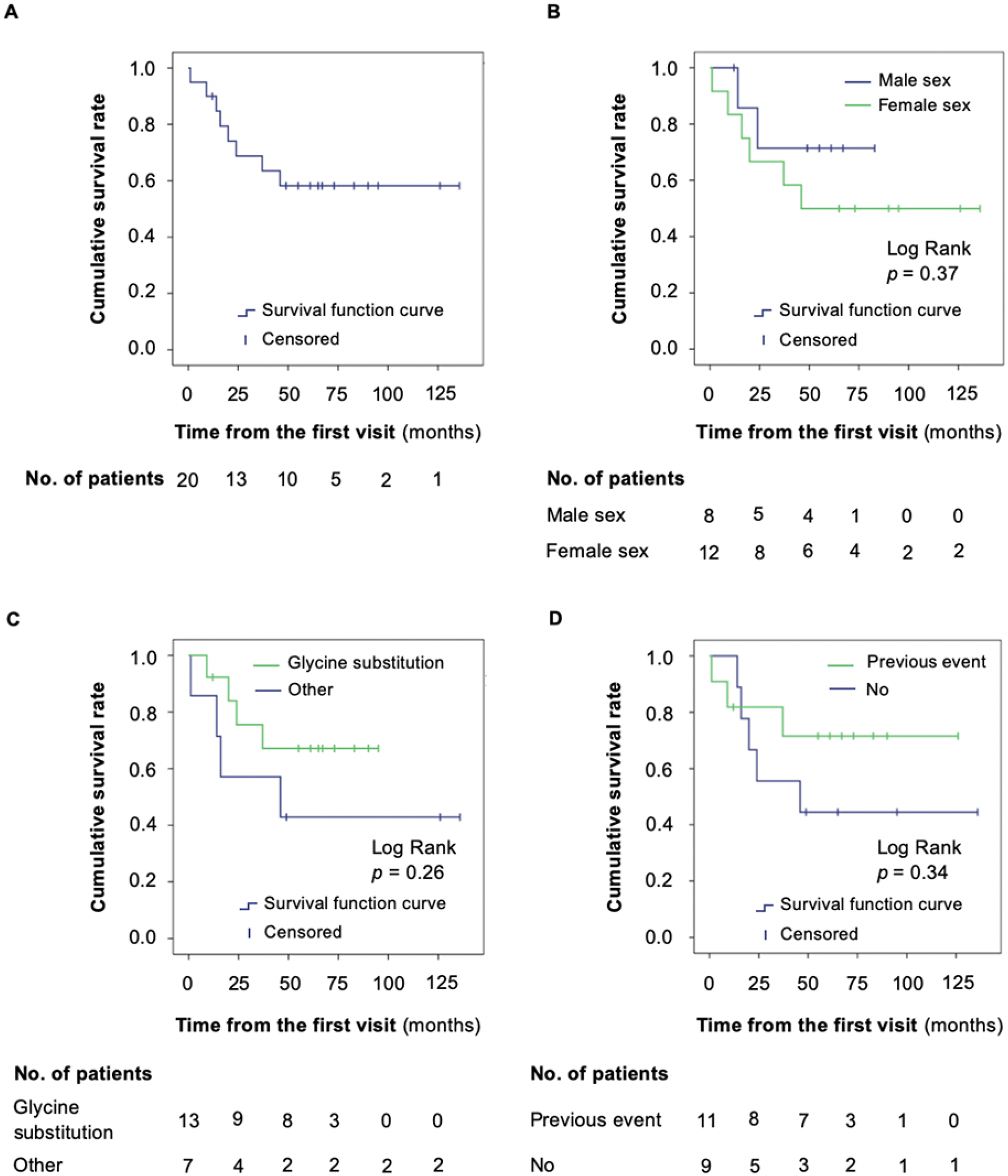
Survival function (Kaplan–Meier) of symptomatic vascular events from the first visit: altogether
Among the 13 patients who started celiprolol therapy either at or after the first visit and had at least one follow-up visit, no significant changes were observed over time in terms of mean SBP (116 vs 114 mmHg, first vs last visits, respectively; p = 0.66), DBP (71 vs 72 mmHg; p = 0.80), MBP (86 vs 86 mmHg; p = 0.99), PP (45 vs 42 mmHg; p = 0.37), and HR (69 vs 71 mmHg; p = 0.71). Among these, three patients had baseline DBP < 62 mmHg, with one experiencing a fatal vascular event during follow-up. Moreover, two patients had baseline PP > 50 mmHg, and neither of them subsequently encountered any major vEDS-related events.
Discussion
In the present study, we share our experience with a cohort of patients with vEDS followed in an Italian tertiary hospital after the publication of the BBEST trial. Overall, celiprolol therapy was generally well tolerated, with only one patient reporting fatigue with doses above 200 mg daily. During a total follow-up of 125 patient-years, we documented 11 symptomatic vascular events across eight patients, of which three were fatal. Importantly, over half of such events occurred after reaching the maximum recommended dose of celiprolol. The yearly risk of symptomatic, life-threatening vascular events in our cohort was 6.4%, which is lower than the risk in untreated patients in the BBEST trial (12%), 8 and similar to the rates observed in treated subjects within both the BBEST trial (5%) and the study by Baderkhan et al. (4.7%).8,9 The percentages were notably higher when considering all vascular events, including asymptomatic ones (up to 14.4%). Remarkably, these results were observed despite the fact that treatment was started before the development of vascular lesions in almost half of the cases and the high rates of patients being on celiprolol at the maximum recommended dose at the last follow-up visit (80%), a percentage similar to that observed in the BBEST study (88%) 8 and higher than that of the Swedish cohort (65%). 9 Interestingly, a retrospective cohort study by Frank et al. demonstrated that the highest degree of protection against major vascular events was achieved with a daily dose of celiprolol at 400 mg. 10 Although this treatment has certainly reduced the burden of disease over the past decade, our results rather suggest that additional efforts should be made to further diminish the risk of vascular events in this specific population.
Mechanisms by which celiprolol provides protection to patients with vEDS against vascular events are still unclear and require further elucidation. Celiprolol functions as a β1 cardioselective blocker with a β2 agonist vasodilator effect. 12 Celiprolol may thus reduce total peripheral resistance by dilating skeletal muscle resistance vessels through β2-adrenergic receptors and might decrease arterial wall shear stress by lowering HR through β1-adrenergic receptor antagonism. In the aforementioned Swedish cohort, SBP, DBP, and PP were all significantly reduced by the end of the follow-up period. 9 In our study, on the other hand, among subjects who started celiprolol therapy at or after the first visit, no significant change was noted in any BP parameter or HR over time. These outcomes are partially in line with those of Ong et al., though in the latter study, PP values showed a significant increase throughout celiprolol therapy. 8 Importantly, no patients in our cohort had ascending aortic aneurysm or clinically relevant aortic valve insufficiency on echocardiography (data not shown), conditions that could have influenced our results, particularly in terms of PP.
Beyond these aspects, it is also possible that celiprolol provides more stable hemodynamic conditions by preventing excessive BP spikes throughout the day, especially during physical exertion and emotional stress. 13 Such aspects may not be revealed by office BP measurement that does not allow the capture of variations in BP over time.
The link between β-adrenergic stimulation and transforming growth factor beta (TGFβ) expression is another potential mechanism to mention in this regard. 14 In fact, Brooke et al. 15 highlighted the role of β1 antagonism in the inhibition of renin secretion and TGFβ-mediated pro-fibrotic mechanisms, whereas Ong et al. 8 stressed the importance of β2 stimulation in the activation of TGFβ and promotion of collagen synthesis. 16 Celiprolol therapy could therefore have a protective effect by increasing arterial stiffness. To support such hypotheses, Boutouyrie et al. demonstrated significantly higher carotid distensibility and circumferential and pulsatile wall stress in patients with vEDS compared with healthy controls, 17 whereas celiprolol selectively increased Young’s elastic modulus by reducing carotid distensibility in the former. 8 Further research must elucidate these aspects.
Consistent with previous findings,8,9 our study did not identify any predictor for symptomatic vascular events among baseline clinical characteristics, which is likely a type II error because of the small simple size. In the BBEST trial, low DBP and high PP have been shown to predict poor response to celiprolol therapy in patients with vEDS, 8 given their association with reduced mechanical stress on the vessel. In our cohort, only one out of three patients with baseline DBP < 62 mmHg experienced a fatal vascular event, whereas no patient with baseline PP > 50 mmHg developed any type of vascular event during follow-up.
Study limitations and strengths
The main limitation of our study is the small sample size, which is related to the rarity of the disease. Accordingly, as already mentioned, the study is clearly underpowered to identify predictors of adverse events in this clinical setting and further studies on larger cohorts are needed to clarify these aspects. Another limitation is clinical heterogeneity of the included patients, mainly in terms of age, frequency of visits, and duration of follow-up. Moreover, due to the lack of a control arm, our study cannot provide any insight about celiprolol treatment in terms of efficacy and safety in patients with vEDS.
Nevertheless, our study has several strengths. To the best of our knowledge, this is the first Italian study on the natural history of vEDS after the BBEST trial publication, as well as one of the largest cohorts detailing the experience with celiprolol in this clinical setting. Furthermore, all included patients had a confirmed molecular diagnosis of the disease, with one variant being novel. Lastly, the substantial follow-up duration in some cases (extending up to 12 years) and the high rates of patients on celiprolol at the maximum recommended dose at the last follow-up visit constitute additional points of strength.
Conclusion
In conclusion, within our cohort of patients with vEDS, rates of celiprolol use at the maximum recommended dose were high and the drug was well tolerated overall, but the risk of vascular events remains remarkable in this population. Future studies should aim to identify reliable predictors of major adverse events during celiprolol therapy, elucidate the mechanisms underlying the treatment’s efficacy, and provide additional therapeutic strategies for further reducing the risk of life-threatening events among affected individuals.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231215330 – Supplemental material for Despite celiprolol therapy, patients with vascular Ehlers–Danlos syndrome remain at risk of vascular events: A 12-year experience in an Italian referral center
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231215330 for Despite celiprolol therapy, patients with vascular Ehlers–Danlos syndrome remain at risk of vascular events: A 12-year experience in an Italian referral center by Giacomo Buso, Anna Paini, Claudia Agabiti-Rosei, Carolina De Ciuceis, Fabio Bertacchini, Deborah Stassaldi, Massimo Salvetti, Marco Ritelli, Marina Venturini, Marina Colombi and Maria Lorenza Muiesan in Vascular Medicine
Footnotes
Data availability statement
The participants of this study did not give written consent for their data to be shared publicly, so due to the sensitive nature of the research, data are not publicly available.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
