Abstract
Background:
Controversy regarding the definition of the upper limit of normal (ULN) for dilated mid-ascending aorta (mAA) stems from variation in criteria, based on several small-sized studies with small datasets of normal subjects (DONS). The present study was carried out to demonstrate this variation in the prevalence of mAA dilation and to identify the optimal definition by creating the largest DONS.
Methods:
Echocardiographic studies of patients ≥ 15 years of age performed at a large tertiary care center over 4 years (n = 49,330) were retrospectively evaluated. The leading-edge-to-leading-edge technique was used to measure the mAA in diastole. The largest-to-date DONS (n = 2334) was created, including those who were normal on medical record review, did not have any of the 28 causes of dilated aorta, and had normal echocardiograms. Because age had the strongest correlation with mAA (multivariate adjusted R2 = 0.26), as compared with sex, height, and weight, we created a new ULN based on the DONS with narrow age stratification (10-year intervals).
Results:
The prevalence of dilated mAA varied between 17% and 23% when absolute criteria were used with sex stratification, and it varied between 6% and 11% when relative criteria (relative to age, body surface area, and sex) were used. Based on new criteria from the DONS, it was 7.6%, with a ULN of 3.07–3.64 cm in women and 3.3–3.91 cm in men.
Conclusions:
These data demonstrate the undesirable variation in the prevalence of dilated mAA based on prior criteria and propose a new ULN for dilated mAA.
Background
Thoracic aortic aneurysm and dissection (TAAD) still involves significantly high mortality rates despite significant diagnostic and therapeutic advances. The incidence of TAAD ranges from 2.4 cases to 10.4 cases per 100,000 people and has been increasing over the past three decades. 1 Overall mortality from ruptured thoracic aortic aneurysms is estimated to be up to 100%, with 59% of patients dying before reaching the hospital. 2 Mortality after elective repair of thoracic aortic aneurysm is about 4%, but it increases to 28.6% when the repair is performed emergently. 3 Since emergent repair is associated with such a high mortality rate, emphasis in recent decades has been placed on early diagnosis, leading to placement of patients on preventive measures. These measures, aimed at slowing the progression of disease, are beta-blockers (animal and human studies),4,5 angiotensin receptor blockers (animal and human studies),5,6 and doxycycline as a matrix metalloproteinase inhibitor (animal and human studies). 5
Detecting a dilated thoracic aorta allows early medical and surgical interventions in high-risk patients. Thus, the identification of the normal range of thoracic aortic measurement becomes imperative for screening and diagnosis. Many previous studies have attempted to define the reference range of aortic dimensions in healthy populations.7–10 These studies suffered from a small sample size for the mid-ascending aorta (mAA; Table 1), as well as lax criteria to define normal subjects. Some used clinical criteria for inclusion as a normal subject9,11 (Table 1), whereas others used echocardiographic criteria in addition to clinical criteria for inclusion.7,8,10 None of these studies systematically excluded all of the 28 causes of TAAD in adults as outlined by the American College of Cardiology (ACC)/American Heart Association (AHA) 2010 guidelines. 12
Summary of studies defining normal diameter of the mid-ascending aorta in individuals 15 years and older.
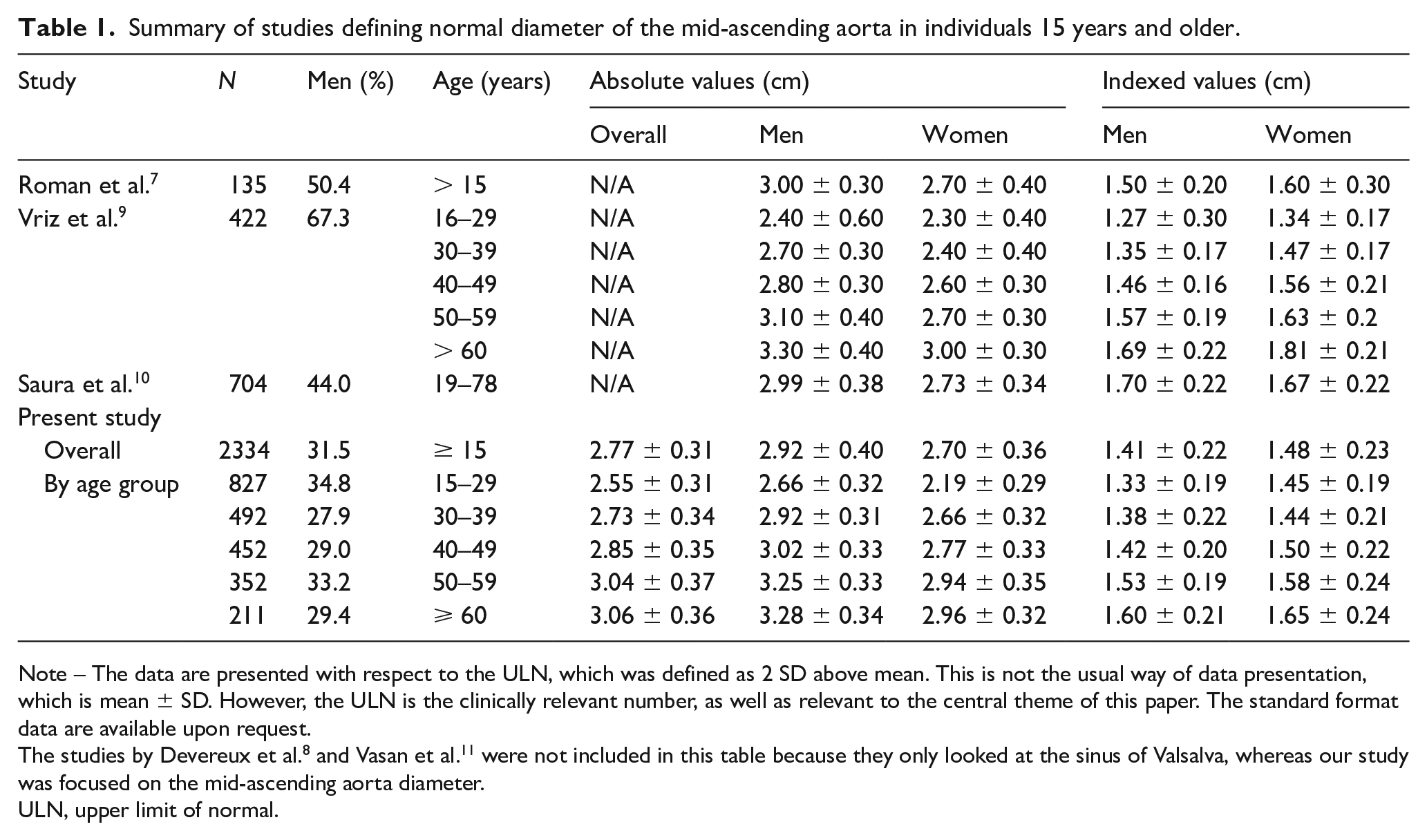
Note – The data are presented with respect to the ULN, which was defined as 2 SD above mean. This is not the usual way of data presentation, which is mean ± SD. However, the ULN is the clinically relevant number, as well as relevant to the central theme of this paper. The standard format data are available upon request.
The studies by Devereux et al. 8 and Vasan et al. 11 were not included in this table because they only looked at the sinus of Valsalva, whereas our study was focused on the mid-ascending aorta diameter.
ULN, upper limit of normal.
In view of these limitations, this study was carried out to determine variation in the prevalence of dilated mAA based on different published criteria. In addition, new criteria were created to define the upper limit of normal (ULN) by creation of a ‘normal subjects’ or ‘healthy population’ dataset that would not only be the largest dataset to-date, but also qualitatively a better dataset because subjects would be defined as normal both clinically, based on medical record review, and by echocardiography. We systematically excluded from the dataset of normal subjects (DONS) anyone who had one or more of the 28 causes of dilated mAA as outlined in the ACC/AHA 2010 guidelines. The ULN for mAA was defined as mean plus 2 SD. The prevalence of dilated mAA in our entire population was measured based on these new criteria and compared with prior published criteria.
Methods
This study was approved by the Aurora Health Care institutional review board (13-37E). The requirement for informed consent was waived due to the retrospective nature of this study.
Study population
This study included all patients aged 15 years or older who underwent echocardiography from November 2011 to December 2016 in our healthcare system based in Milwaukee, WI, and had aortic measurements available. We further queried the International Classification of Diseases, Ninth Revision (ICD-9)-based electronic medical record database for 28 risk factors of dilated aorta as described in the 2010 ACC/AHA guidelines. 12
Echocardiographic evaluation
Protocol at our institution dictates that mAA diameter be measured in the parasternal long-axis view in a plane perpendicular to that of the long axis of the aorta at end-diastole using the leading-edge-to-leading-edge technique as described in the American Society of Echocardiography guidelines 13 (Figure 1).
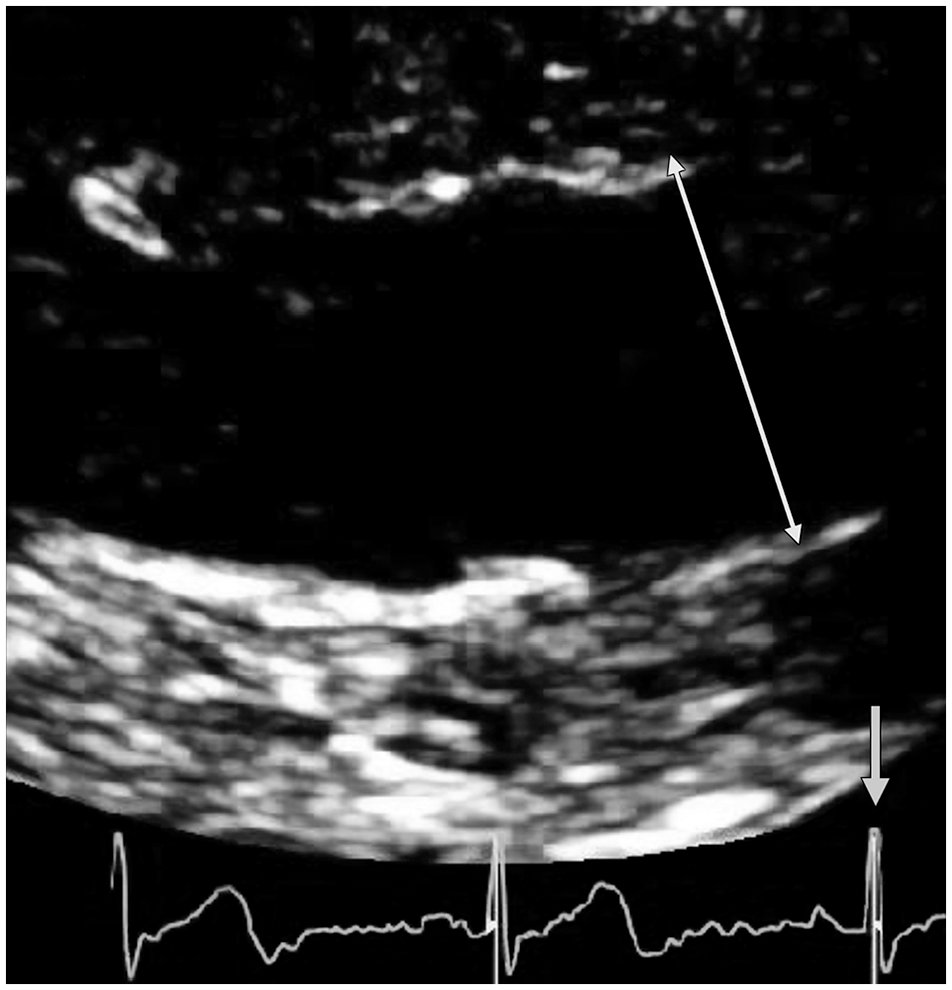
The double-headed arrow shows the leading-edge-to-leading-edge measurement of mid-ascending aorta diameter (3.42 cm), avoiding the ultrasonic blooming artifact created by the trailing edge. The single-headed arrow points toward the QRS, which indicates that the measurement was made in end-diastole.
Definition of normal subjects to create DONS
We used clinical and echocardiographic criteria based on the 2010 ACC/AHA guidelines 12 to exclude any subjects with the presence of risk factors for dilated aorta. The clinical and echocardiographic criteria are described in the following sections.
Clinical criteria
All patients with any of the 28 risk factors present were excluded from the DONS (Figure 2). These risk factors include hypertrophic obstructive cardiomyopathy, diabetes, hypertension, tobacco use, heart failure, Marfan syndrome, ischemic cardiomyopathy, bicuspid aortic valve, aortic valve stenosis or regurgitation, Turner syndrome, pheochromocytoma, cocaine use, coarctation of the aorta, history of valve replacement, Ehlers–Danlos syndrome, syphilis, Takayasu arteritis, giant cell arteritis, Behcet’s syndrome, polycystic kidney disease, and corticosteroid immunosuppression.
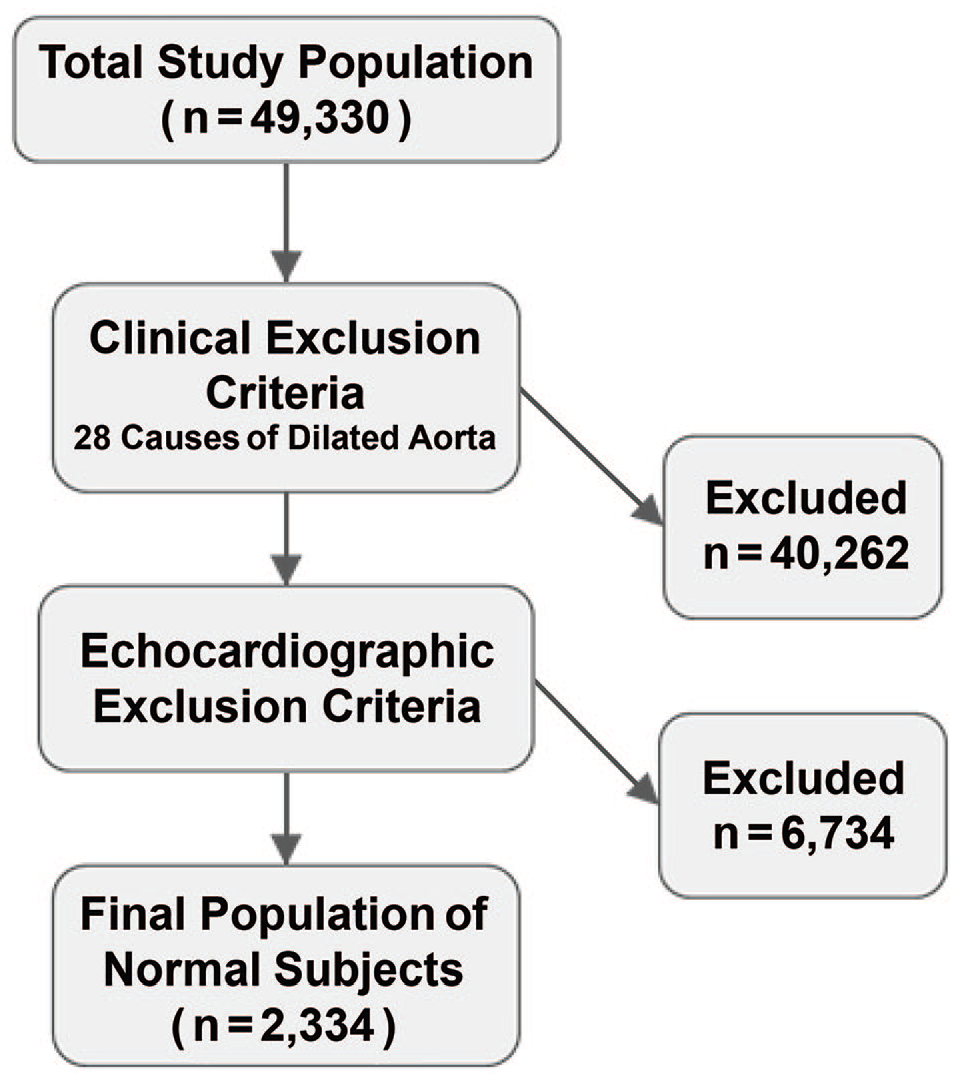
Study population flow diagram. Application of exclusion criteria decreased the study population from 49,330 to 2,334.
Echocardiographic criteria
Patients whose echocardiograms demonstrated abnormal findings also were excluded. These abnormal findings included overt aortic aneurysmal disease, coarctation of the aorta, aortic regurgitation (any degree of regurgitation), aortic stenosis (any degree of stenosis), bicuspid aortic valve, mitral regurgitation (moderate or worse), mitral stenosis (moderate or worse), diastolic dysfunction (any grade), regional wall motion abnormality, akinesis or hypokinesis, systolic dysfunction (left ventricular ejection fraction < 50%), mild or worse left ventricular hypertrophy, moderate or worse pulmonary hypertension, a severely enlarged left atrium, atrial septal defect or ventricular septal defect (repaired or unrepaired), and technically difficult studies.
Measurement of prevalence of dilated mAA based on different criteria
In order to compare the effect of different criteria on the prevalence of dilated mAA, the previously published criteria were applied to the entire study population. In addition, new criteria were created to define the ULN by creation of a ‘normal subjects’ or ‘healthy population’ dataset that would not only be the largest dataset to-date, but also qualitatively a better dataset, because subjects would be defined as normal both clinically, based on medical record review, and by echocardiography. The ULN for mAA was defined as mean plus 2 SD. The prevalence of dilated mAA in our entire population was then measured, based on these new criteria, and compared with prior published criteria.
Statistical analysis
Univariate analysis was performed; aortic diameters were presented as mean ± SD and prevalence was presented as a percentage of the overall population. To evaluate the strength of association between dilated mAA and different determinants such as age, sex, height, weight, etc., we performed bivariate linear regression followed by multivariate linear regression. The strength of association was defined by the slope (β) of association. Because the units of these parameters were very different, standardized β coefficients were used, which allows comparison between variables with markedly different ranges and units and their association with mAA.
In addition, to evaluate the impact of individual parameters such as age, sex, weight, blood pressure, etc., on mAA diameter, we utilized the adjusted R2, which is generated by multiple linear regression equations. For example, an R2 of 0.26 for age would mean that 26% of the variation in mAA diameter in normal subjects can be explained by age.
All analyses were carried out using JMP 12 software (SAS, Cary, NC, USA).
Results
We identified 49,330 people aged ≥ 15 years who had undergone echocardiography at our tertiary care center. The mean age of the study population was 64.2 ± 17.1 years, and 23,445 subjects (47.6%) were men. The mean height of our study population was 169.1 ± 10.9 cm, mean body surface area (BSA) was 2.01 ± 0.31 m2, and mean body mass index was 30.6 ± 7.7 kg/m2. Within the study population, 82.1% of participants were White, 12% Black, 4.8% Hispanic, 1.2% Asian, and 0.1% other races.
Application of exclusion criteria
As depicted in Figure 2, application of clinical exclusion criteria removed 40,262 patients. Subsequent review of echocardiographic reports led to the removal of an additional 6734 patients, leaving a final population of 2334 normal subjects.
Characteristics of final study population
Table 2 shows the baseline characteristics of our study population compared with the overall population. Out of the 2334 normal subjects, 735 (31.5%) were men. Normal subjects were significantly younger (37.5 ± 14.9 years vs 64.2 ± 17.1 years) and had lower weight (79.5 ± 20.7 kg vs 87.7 ± 24.4 kg), BSA (1.92 ± 0.27 m2 vs 2.01 ± 0.3 m2), and body mass index (27.8 ± 6.7 kg/m2 vs 30.6 ± 7.7 kg/m2) than the overall population (p < 0.0001). Mean height was similar between the two groups (168.9 ± 10.1 cm vs 169.1 ± 10.9 cm; p = 0.72).
Baseline clinical characteristics of the study population.
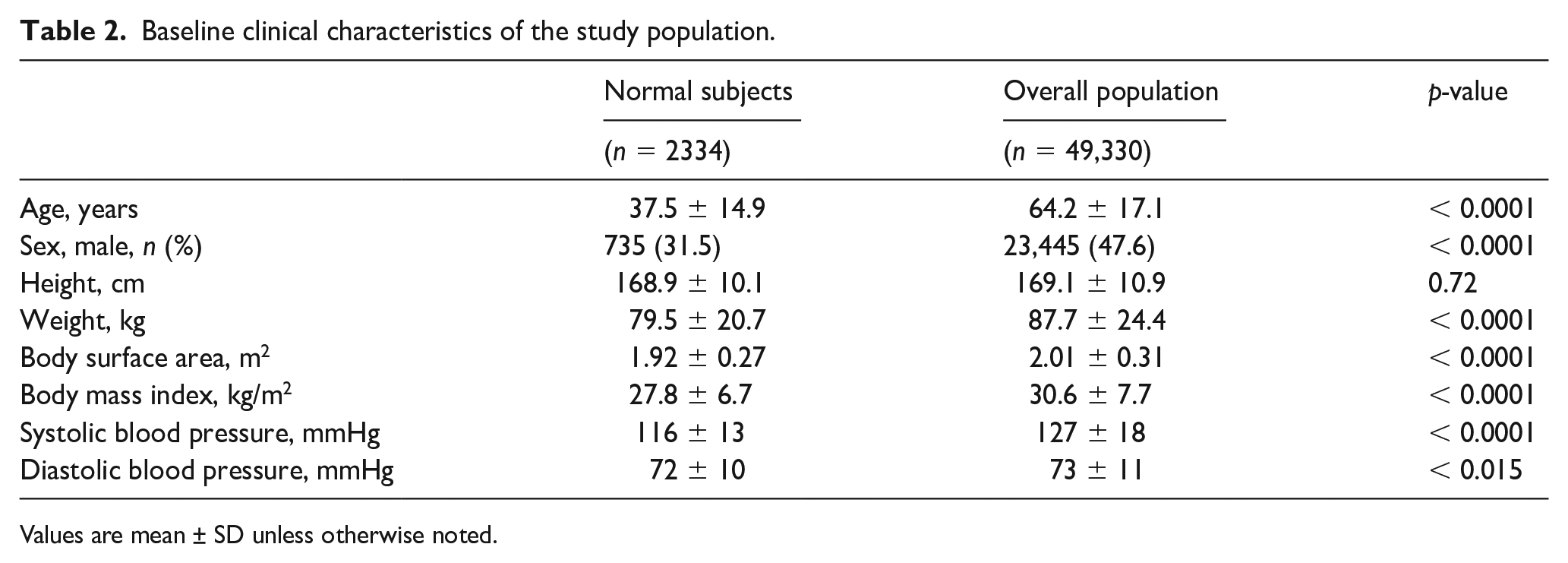
Values are mean ± SD unless otherwise noted.
Definition of normal aortic dimensions
The ULN mAA diameters are reported in Table 3. The results are depicted in absolute values as well as in ‘relative’ values (relative to BSA), and stratified by age and sex, defined by more than 2 SD above the mean among normal subjects. The absolute ULN stratified by age varied between 3.07 amd 3.64 cm in women and 3.3 and 3.91 cm in men. The BSA-indexed cutoff values varied between 1.79 and 2.07 cm/m2 in women and 1.85 and 1.92 cm/m2 in men, increasing with age. The absolute aortic dimensions were greater in men across all age groups, whereas BSA-indexed values for ULN were lower for men because they generally have a larger denominator, i.e., body surface area (height and weight). Aortic dimensions significantly increased with advancing age.
The upper limit of normal mid-ascending aortic diameter based on age and sex.
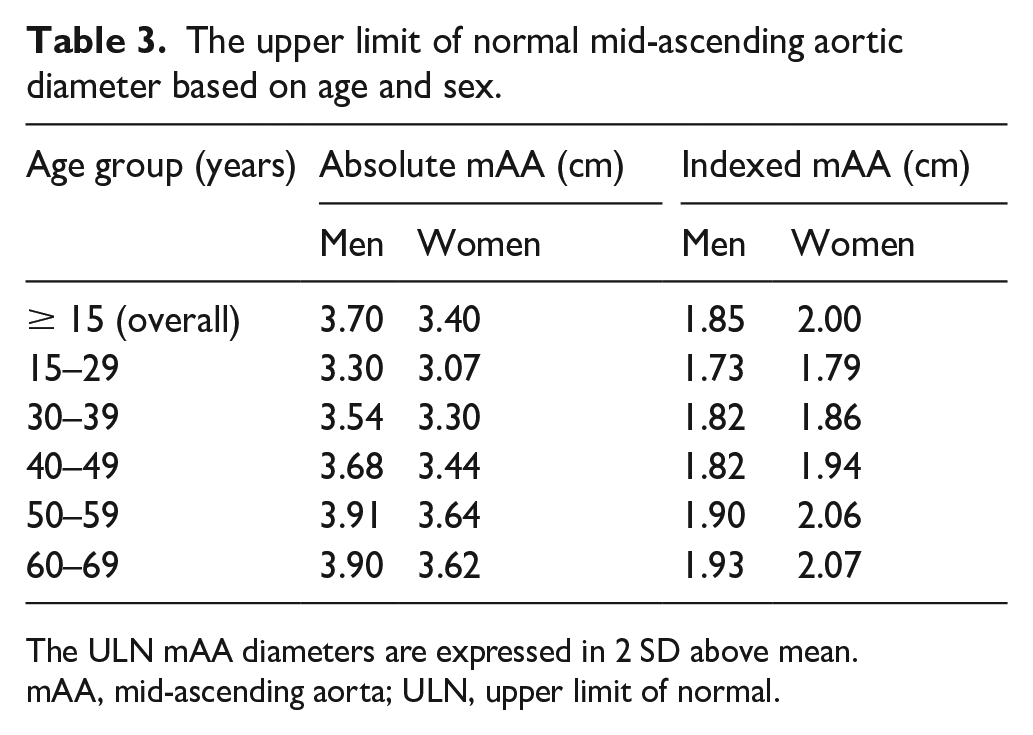
The ULN mAA diameters are expressed in 2 SD above mean.
mAA, mid-ascending aorta; ULN, upper limit of normal.
Prevalence of dilated aorta after application of new definition
The overall prevalence of dilated mAA was 23.2% (n = 11,444) when absolute cutoff values were utilized. Of these subjects, 5676 were men and 5768 were women. When BSA-indexed cutoff values were employed, the overall prevalence of dilated mAA decreased to 15.4% (n = 7571), with 3997 men and 3574 women.
Bivariate determinants of aortic dimensions
In our normal population, age had the highest standardized β (0.50, p < 0.0001), followed by weight (0.35, p < 0.0001), sex (0.27, p < 0.0001), and height (0.26, p < 0.0001). BSA had a much stronger association with mAA diameter than body mass index, with a higher standardized β (0.375 vs 0.247, p < 0.0001) and higher adjusted R2 as well (0.140 vs 0.061). Age had the highest R2 (0.260, p < 0.0001), followed by weight (0.12, p < 0.0001), sex (0.069, p < 0.0001), and height (0.065, p < 0.0001). Age accounted for 26% of mAA diameter variation. If age increases by 10 years, mAA diameter increases by 0.13 cm.
Multivariate determinants of aortic dimensions
The pattern remained in the multivariate linear regression model, with age having the highest adjusted standardized β (0.499, p < 0.0001), followed by weight (0.202, p < 0.0001), sex (0.150, p < 0.0001), and height (0.116, p < 0.0001). There was no meaningful difference in the adjusted R2 for the multivariate models, including age, sex, and height and weight vs age, sex, and BSA (39% vs 38.9%), indicating that height and weight can be replaced by BSA. Removal of sex increased R2 from 0.36 to 0.39. Removal of age reduced R2 from 0.39 to 0.14, indicating that age plays the biggest role.
Prevalence of dilated mAA by other definitions and their comparison with our criteria
The prevalence of dilated mAA measured by the present study was higher than the prevalence measured in prior studies, including those by Saura et al., 10 Lang et al., 14 and Campens et al. 15 (23.2% vs 20.2% vs 17.3% vs 14%, respectively) (Figure 3A). The prevalence found by Roman et al. 7 was similar to the prevalence in our study (23.3% vs 23.2%). The prevalence of dilated mAA was higher based on these criteria without age stratification and meaningfully dropped to 7.6% when criteria stratified by age group were used (Figures 3B, 3C). This was lower than the prevalence calculated using the age-stratified definition described by Vriz et al. (10.5% vs 7.6%). 9 The prevalence measured by the present study using the definition indexed by age, sex, and BSA was higher compared with the prevalence measured using definitions by Campens et al. and Vriz et al. using similar stratification (Figure 3D).
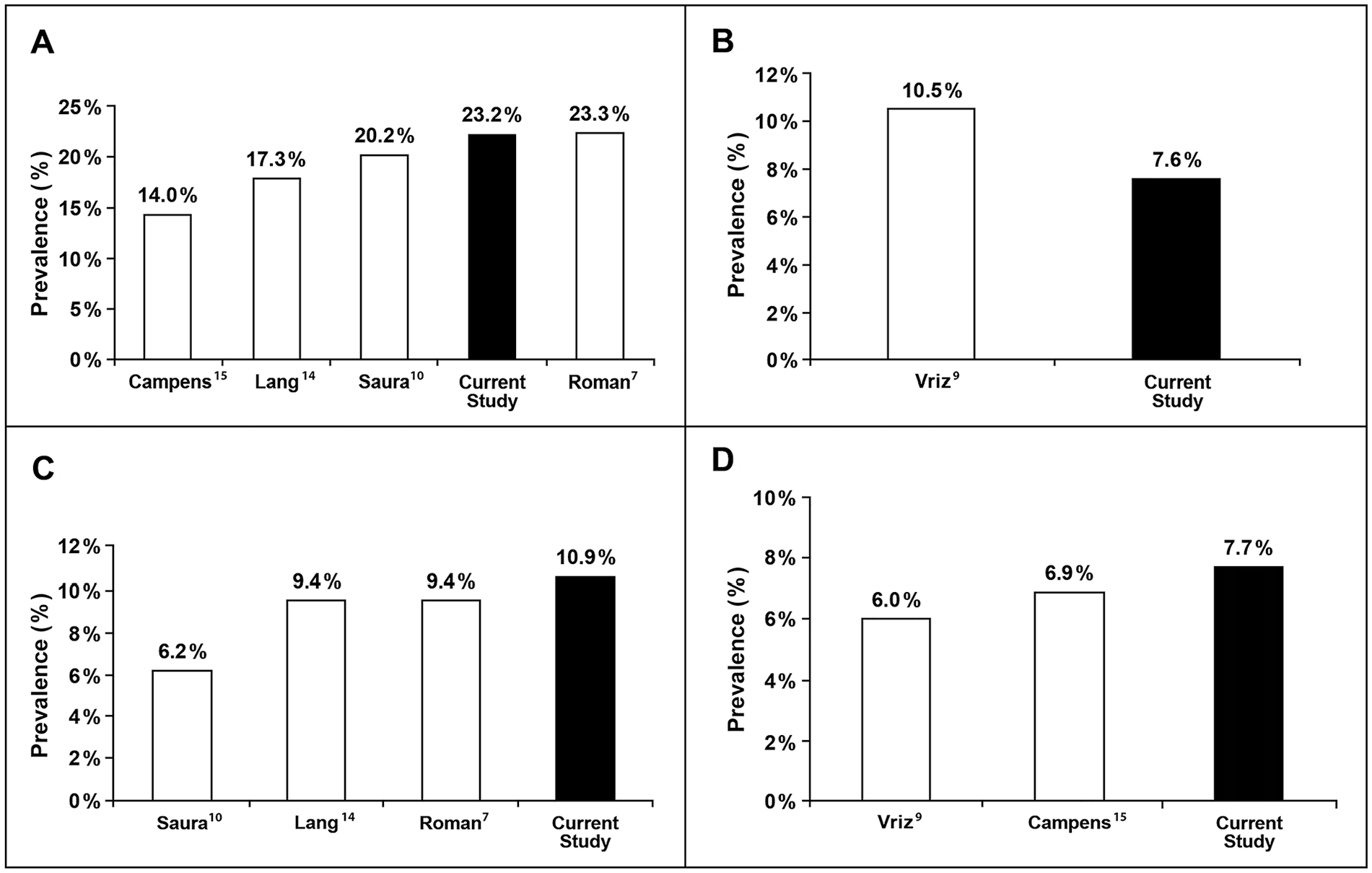
Prevalence of dilated mAA with increasingly sophisticated criteria used.
Discussion
The present study represents the largest dataset of mAA diameters in a normal population. It also represents the strictest definition of abnormal aorta in comparison to all prior studies.7,9,10 These data suggest lower cutoff points for dilated aorta in the general population, as well as when stratified by age and sex. The prevalence varies markedly depending on the criteria used and decreases from as much as 23% to 7.6% if age, sex, and BSA adjustment is used.
Because age and weight had the strongest impact on mAA diameter, definitions that incorporate age and weight (or BSA) should be preferred over others that predominantly use sex or height stratification.
The landmark study by Roman et al. had a much smaller sample size (135 vs 2334) than the present study, and it failed to account for sex-related differences. 7 The definition of normality in that study included unaffected family members of patients with mitral prolapse and Marfan syndrome, which is much weaker than ours because we excluded patients with 28 causes of dilated aorta based on clinical chart review (ICD-9) and echocardiographic reports. Studies subsequent to the Roman et al. study demonstrated that women had decreased aortic dimension compared with men across age groups. 8 In addition, only three age groups were used: < 20, 20–39, and > 40 years of age. Because age is the strongest determinant of mAA diameter, smaller intervals are preferable. This could be achieved in our study owing to a larger sample size. Similar to our study, the study carried out by Vriz et al. stratified the results by 15-year age intervals, 9 whereas previous studies, including those by Roman et al. and Saura et al., categorized study populations as less than or more than 40 years and 50 years of age, respectively.7,10 In addition, subjects aged 15–25 years were not included in the study by Saura et al., which is a limitation not faced by our data. The narrower age interval in our study is able to accommodate the age-specific increment of aortic dimensions, with Vriz et al. being the only other study to accomplish this goal. The present study minimized selection bias by including patients across different racial and ethnic groups, whereas some of the prior studies only included White patients.9,10
A major study by Campens et al. included a large sample of 849 subjects to determine reference values for sinus of Valsalva and mAA diameter in all age categories. 15 This study included all echocardiograms performed in the echocardiography laboratory at Ghent University after exclusion of bicuspid aortic valves, significant valvular disease, or connective tissue disorders like Marfan syndrome. All other causes of dilated aorta, including hypertension, were not excluded. Although that important study is clearly inferior to the current study in terms of sample size (Figure 4), racial diversity (100% White vs 82% White), and exclusion criteria (3 vs 28), it brings forth a similar message of a much lower prevalence of dilated mAA when sophisticated criteria utilizing not only age but sex and BSA as well are employed (Figure 2).
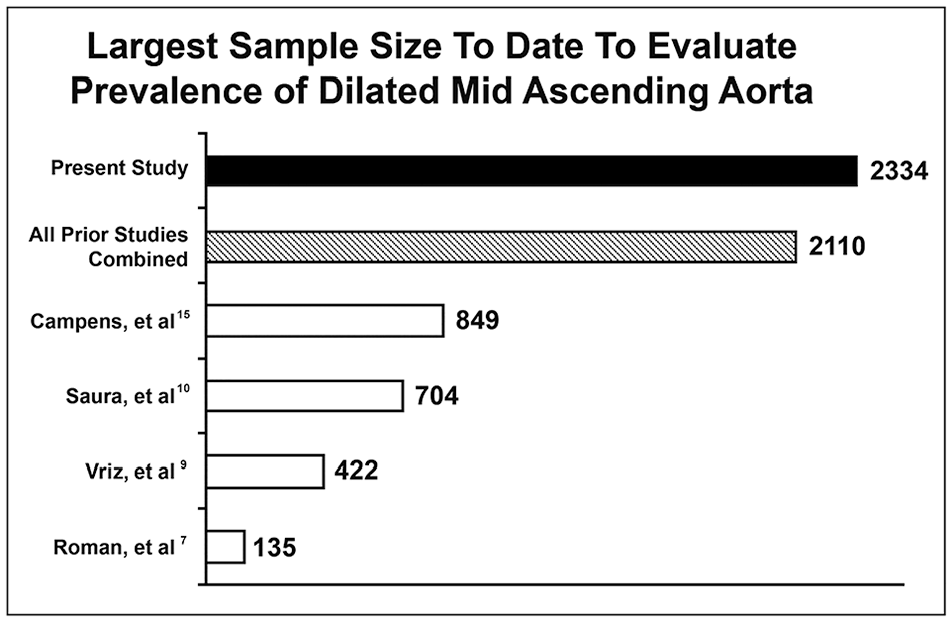
The present study has the largest sample size to date for the evaluation of the prevalence of dilated mid-ascending aorta.
Because age has a significant impact on mAA diameter (β = 0.50; p < 0.0001), the definitions that included 15- or 10-year age intervals should be preferred over those with larger age intervals. The present study compared the absolute cutoff values of normal mAA with the values stratified by age groups that were divided into 15–29 years old, 30–39 years old, 40–49 years old, 50–59 years old, and older than 59 years. The number of normal subjects decreased with advancing age. Our age-based definition of dilated mAA had the second lowest prevalence of dilated mAA (7.6%; Figure 3B) in the entire population (n = 49,330), second only to Vriz et al.’s definition (6.0%; Figure 3D) 9 and much lower than 23.2%, which is based on an absolute cutoff with sex stratification (Figure 3A). Similarly, the prevalence of mAA dilation with BSA indexed dropped from 10.9% without age stratification to 7.7% with age stratification.
When BSA-indexed criteria were used to define dilated mAA, the prevalence was highest by our definition and decreased with definitions given by Roman et al., Lang et al., and Saura et al. (10.9% vs 9.4% vs 9.4% vs 6.2%, respectively).7,10,14 The prevalence with age-stratified, BSA-indexed criteria in our study was also higher than the age-stratified prevalence with BSA indexed calculated by Vriz et al. (10.9% vs 7.7%). 9
Our data suggest the use of age stratification over sex stratification to define dilated mAA. BSA was the second most meaningful determinant of mAA; therefore, BSA should be the second factor taken into consideration when choosing a definition of dilated mAA. Age and sex are nonmodifiable risk factors, whereas weight is modifiable, indicating the importance of weight control, which is the single most important modifiable risk factor. Blood pressure was the least important modifiable risk factor for mAA dilation in normal subjects.
Our data pose the clinical and epidemiological question of whether we should use criteria that would render 23% of people (approximately one out of four) as abnormal or criteria that would declare only 7.6% of people (approximately one in 14) to have an abnormally dilated aorta. Though our data would favor the definition with lower prevalence, based on cross-sectional data of β and R2, a better way may be to employ prognostic data such as aneurysm rupture and death in future studies.
As mentioned in the 2022 AHA/ACC guidelines on aortic aneurysms, 16 the definition of arterial aneurysm has been an increase in diameter to 1.5 times the expected normal diameter of the artery. Although this classical and anatomical definition serves well the patients with descending thoracic aortic aneurysm and abdominal aortic aneurysm, it provides a major disservice to those with ascending aortic aneurysm. The risk of dissection in the ascending aorta, as compared with the risk of dissection in an aorta with a diameter < 3.5 cm, increased to 4.5 times normal in those with a diameter of 3.5–4 cm, to 89 times normal in those with a diameter of 4–4.4 cm, and to 6305 times normal for those with a diameter of ⩾ 4.5 cm. Considering that the conventional ULN of mAA is 3.5–3.8 cm, even a mAA with a 5 cm diameter would not be considered an arterial aneurysm under the current generic AHA definition of an arterial aneurysm, when we know that a mAA with a diameter of 5 cm is 6305 times more likely to develop a dissection. Therefore, the current study remains relevant, as instead of using the generic arterial aneurysm definition, it utilizes the philosophy of abnormal being 2 SD above the mean and stratifies it according to age, sex, and BSA. A follow-up to the current study is being planned to see if aortic dissection can be predicted based on the proposed definition.
Limitations and strengths
The present study suffers from a retrospective study design and its inherent limitations. The echocardiograms were performed as part of clinical care; therefore, they may not reach the excellence ensured in research echocardiograms. However, the studies were done in Intersocietal Commission for the Accreditation of Echocardiography Laboratories (ICAEL)-certified echocardiographic laboratories, which ensures a minimum standard of performance set by ICAEL.
No correlation of echocardiographic measurements was made with measurements in computed tomography or magnetic resonance imaging studies, which is a limitation of this study insofar as its results apply only to echocardiograms. There can be a variation of 2–4 mm in the measurement of aortic diameter, as the aortic wall is generally 2-mm thick and there are several methods of measurement in computed tomography and magnetic resonance imaging: aortic diameter can be 4 mm smaller when measured by inner-to-inner diameter versus outer-to-outer diameter.
This study suffers from selection bias, so it can be applied to a similar population only and cannot be used as a representative sample of the general community. However, although the exact prevalence may not turn out to be the same in a future study in a sample representative of a general population, we predict that the variation in prevalence of dilated mAA—the most important message that this paper aims to deliver—will turn out to be quite similar.
Another limitation is the lack of test-retest reproducibility performed in our cohort. However, we used a time-tested technique that has been shown in multiple publications to carry test-retest reproducibility. A study by Dai et al. in 1999 showed almost nil variation in measurements of the aorta whether done by a field technician or a research echocardiographer. 17 In a more recent study by Muraru et al., in 2014, reproducibility was reported in healthy volunteers. 18 Therefore, the investigators felt that testing the test-retest reproducibility in our sample was not essential.
The biggest strength of the current study is its sample size, which is larger than all prior studies. The second strength is its stricter criteria for normalcy. This study used ICD-9 codes as well as echocardiographic abnormalities to exclude 28 risk factors of dilated aorta as defined by 2010 ACC/AHA guidelines. 12 After exclusion, the present study still had more participants than in all three prior studies combined (Figure 4). Although women comprised more than two-thirds of the normal population, the unintentional bias this introduces is addressed by the much larger sample size of our study and sex stratification.
Conclusion
The prevalence of dilated mAA varies markedly depending on the criteria used and decreases from as much as 23% to 7.6% if age, sex, and BSA adjustment is used. Because age and weight had the strongest impact on mAA diameter, definitions that incorporate age and weight (or BSA) should be preferred over others that predominantly use sex or height stratification. This strategy yielded a lower prevalence of 7.6% for dilated mAA in our sample. As these definitions have wide public health implications for screening, detection, and therapy, these data suggest the adoption of age-, sex-, and BSA-dependent criteria and longitudinal follow-up of these patients in search of the criteria with optimal prognostic implications.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
