Abstract
Background:
Inflammation and immune dysregulation have been associated with adverse outcomes in cardiovascular disease. There is limited understanding of the association of different profiles of white blood cell (WBC) subsets and red cell distribution width (RDW) in patients with chronic limb-threatening ischemia (CLTI).
Methods:
Patients with CLTI undergoing endovascular revascularization in our single-center, tertiary care hospital from 2017 to 2019, who had a preceding complete blood count (CBC) with WBC differentials (n =213), were included in the analysis. Patient characteristics, laboratory values, and clinical outcomes were collected. Cox proportional hazards regression models were used to assess for associations between all-cause mortality and leukocyte subset; multivariate analysis was used to account for confounders. Kaplan–Meier curves were generated to depict survival censored at 1 year postrevascularization using baseline CBC indices.
Results:
Adjusting for confounders, elevated RDW was associated with increased mortality (continuous per % increase, adjusted hazard ratio [HR] 1.33, p < 0.001). Baseline lymphopenia was associated with mortality in univariate analysis. Other leukocyte subtypes were not associated with mortality outcomes in our population. Exploratory analysis showed negative deflections in ∆WBC from pre- to postprocedure day 1 were affiliated with increased mortality when adjusted for age, sex, race, chronic kidney disease, and baseline hemoglobin (∆WBC HR 1.16, p = 0.004). Further exploratory analysis showed an association between RDW and all-comers readmission.
Conclusions:
The utilization of a periprocedural WBC subset differential can be a useful adjunct to risk-stratify patients with CLTI undergoing endovascular revascularization. Further studies are needed to understand potential ways to modulate immune dysregulation so as to improve mortality outcomes.
Background
Inflammation and immune dysregulation are prevalent among patients with cardiovascular disease (CVD).1–4 Furthermore, markers of immune function can be useful predictors of overall and cardiovascular survival.5–7 Among markers of immune dysregulation, anemia and lymphopenia are associated with worse outcomes in patients with CVD.1,8–10 Pharmacologic agents that target immune regulation have shown promise in cardiovascular outcomes, but improved methods to identify those at greatest risk are needed.7,11,12
Patients with chronic limb-threatening ischemia (CLTI) are the most vulnerable within the peripheral artery disease (PAD) spectrum, carrying the highest risk of major adverse limb events, including major amputation and mortality. 13 Certain markers of inflammation, such as C-reactive protein (CRP), have been associated with adverse outcomes in patients with PAD.14,15 However, the effects of white blood cell (WBC) counts, WBC subsets (neutrophils, lymphocytes, and monocytes), and red cell distribution width (RDW) on clinical outcomes of patients with CLTI have not yet been fully evaluated. We hypothesize that levels of WBC subsets and RDW, as markers of immune and hematologic homeostasis, will be associated with all-cause survival of patients with CLTI undergoing endovascular revascularization for limb salvage.
Methods
Patient population
This was a single-center, observational study of patients ⩾ 18 years of age, who underwent endovascular revascularization at our tertiary care center for CLTI from August 2017 to April 2019. Patients presented with either rest pain or ischemic wounds consistent with Rutherford classification IV–VI, with evidence of perfusion deficiency detected on noninvasive studies (e.g., ankle–brachial index [ABI], toe–brachial index [TBI], pulse volume recording, Duplex ultrasound, or computed tomography angiography). We included in this analysis the patients with complete blood count (CBC) with leukocyte subset differentials prior to revascularization.
Clinical and laboratory data
Patient data, including baseline characteristics, medical history, laboratory results, procedural details, and postprocedural outcomes were obtained from the electronic medical record.
For all patients, baseline CBC was obtained within 30 days preceding the intervention. WBC with leukocyte subset differentials (absolute neutrophil counts [ANC], absolute lymphocyte counts [ALC], and absolute monocyte counts [AMC]) as well as RDW were recorded for analysis. The primary endpoint was all-cause mortality at 1 year postrevascularization. For exploratory purposes, a follow-up CBC was analyzed on postintervention day 1 on the patients with available data. Other outcomes, including wound healing, target vessel revascularization (TVR), major amputation, and readmission were assessed using Kaplan–Meier curves. Time-to-event data were obtained via extraction from the electronic medical record. If the event did not occur, the date of the last follow-up was used.
This study was approved by the institutional review board at University Hospitals Cleveland Medical Center with a waiver of consent. Clinical data were collected from the electronic medical record and entered into the REDCap (Research Electronic Data Capture) tool 16 hosted at University Hospitals Cleveland Medical Center.
Statistical analysis
Kruskal–Wallis and Pearson chi-squared tests were used to assess for differences in baseline characteristics in continuous and categorical variables, respectively. To understand whether there was an interaction between ALC and RDW, we ran a Pearson’s correlation between the variables. Cox proportional hazards regression models were used to assess for associations between all-cause mortality and leukocyte subset levels in preprocedure and postrevascularization day 1 labs. The change in each biomarker was expressed as the difference between the postprocedural value minus the preprocedural value. Kaplan–Meier curves were generated to depict survival censored at 1-year postrevascularization using baseline CBC indices. Kaplan–Meier curves were also generated for other outcomes, including time-to-wound healing, time-to-readmission, and time-to-major amputation censored at the date of last follow-up. Tertiles of leukocyte counts and baseline characteristics were compared using analysis of variance and multiple comparisons were made using post hoc Tukey tests. All p-values < 0.05 were considered statistically significant. Statistical analysis was performed using IBM SPSS Statistics, Version 26 (IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics and patient demographics are reported in Tables 1 and 2 based on RDW and ALC tertiles, respectively. Baseline characteristics according to ANC and AMC tertiles are reported in Supplemental Tables S1 and S2, respectively (available online). Of a total of 297 patients who underwent endovascular revascularization for CLTI during the study period, 213 patients had available CBC with differential preprocedure and were included in this analysis. Overall, the patients in the study had an average age of 70 ± 13.6 years, 61% male, and 69% White. Prior surgery or endovascular intervention for PAD, unrelated to the current CLTI presentation, was common (56%).
Baseline characteristics according to red cell distribution width (RDW) tertiles.
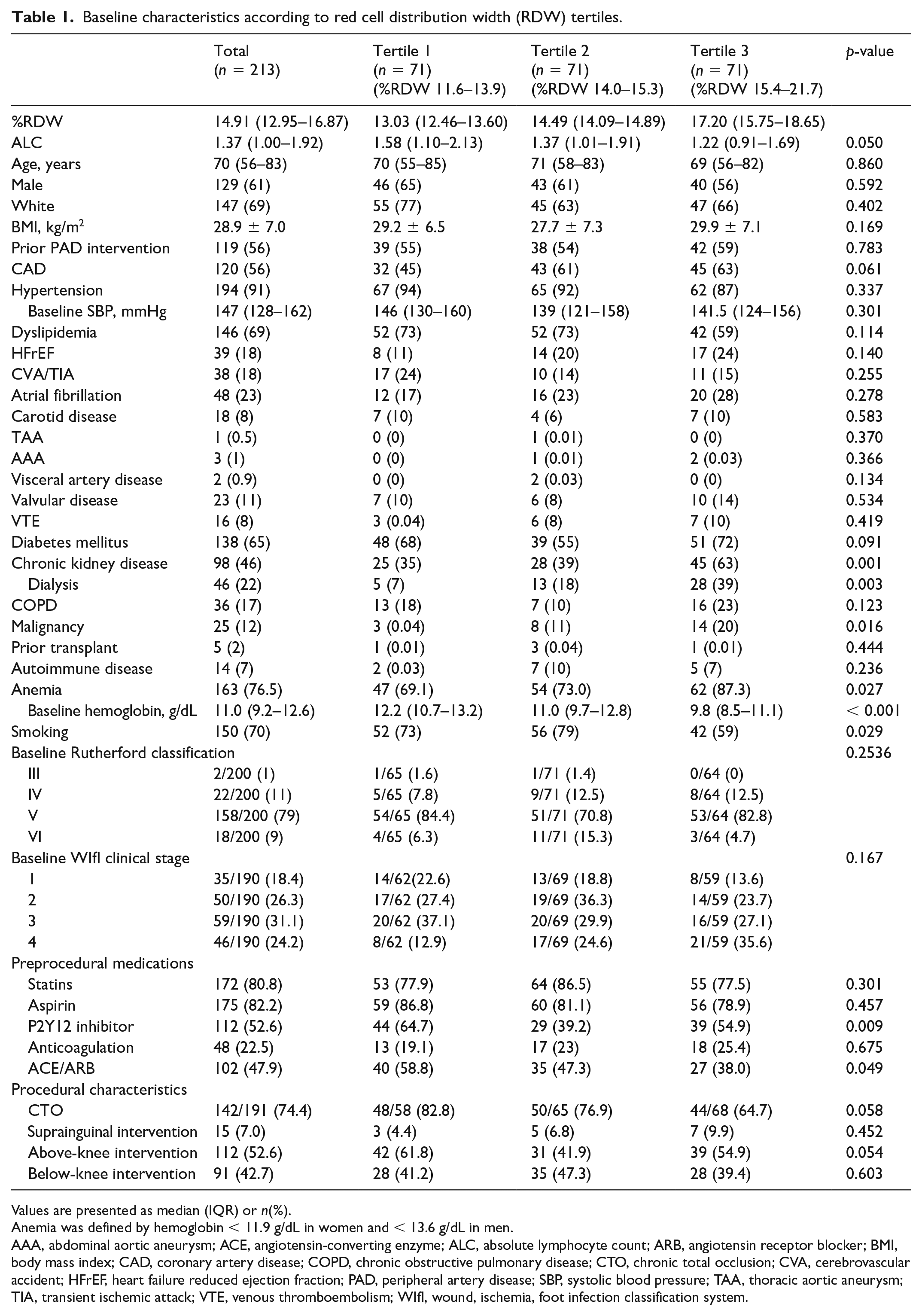
Values are presented as median (IQR) or n(%).
Anemia was defined by hemoglobin < 11.9 g/dL in women and < 13.6 g/dL in men.
AAA, abdominal aortic aneurysm; ACE, angiotensin-converting enzyme; ALC, absolute lymphocyte count; ARB, angiotensin receptor blocker; BMI, body mass index; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CTO, chronic total occlusion; CVA, cerebrovascular accident; HFrEF, heart failure reduced ejection fraction; PAD, peripheral artery disease; SBP, systolic blood pressure; TAA, thoracic aortic aneurysm; TIA, transient ischemic attack; VTE, venous thromboembolism; WIfI, wound, ischemia, foot infection classification system.
Baseline characteristics according to absolute lymphocyte count (ALC) tertiles.
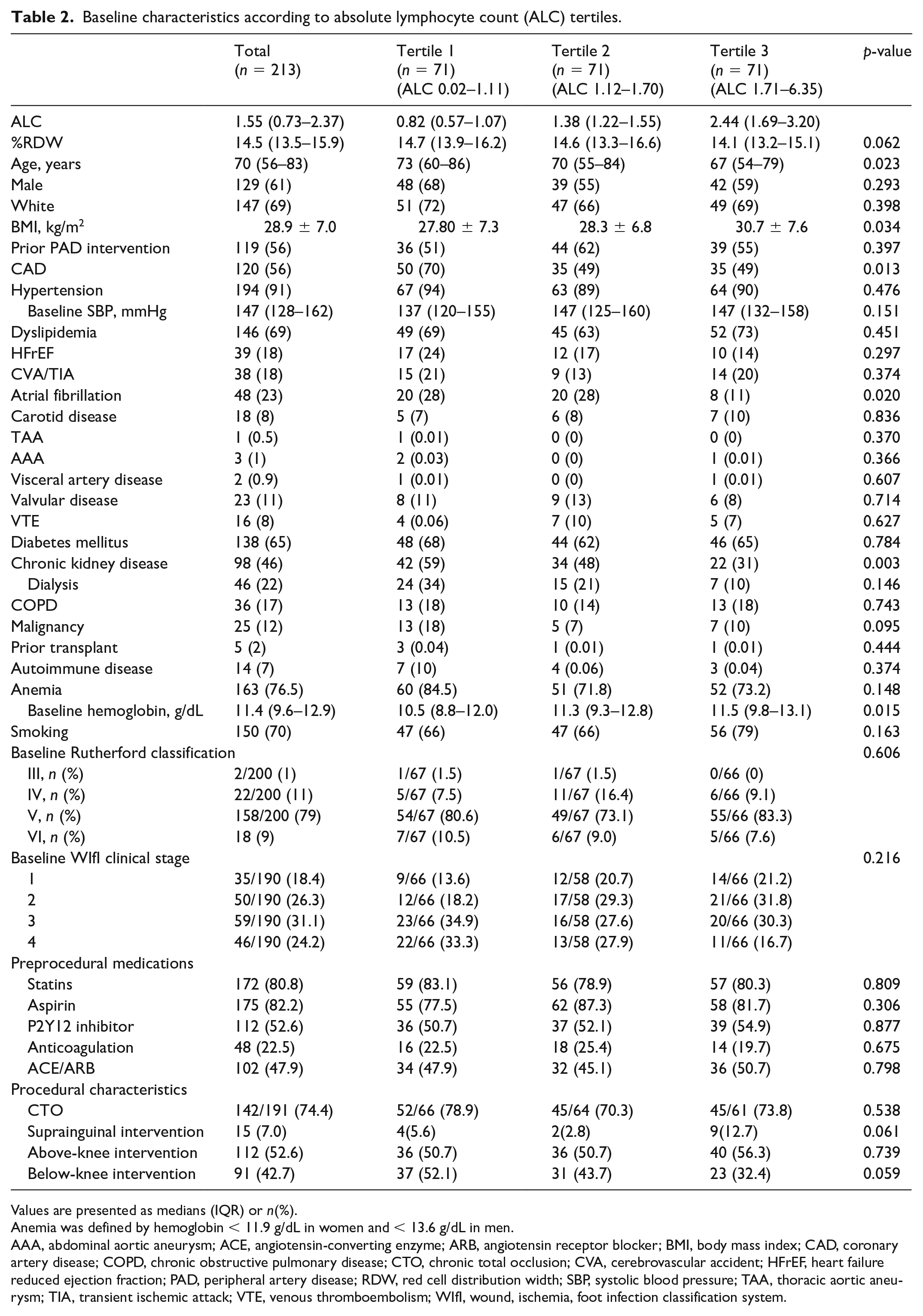
Values are presented as medians (IQR) or n(%).
Anemia was defined by hemoglobin < 11.9 g/dL in women and < 13.6 g/dL in men.
AAA, abdominal aortic aneurysm; ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; BMI, body mass index; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CTO, chronic total occlusion; CVA, cerebrovascular accident; HFrEF, heart failure reduced ejection fraction; PAD, peripheral artery disease; RDW, red cell distribution width; SBP, systolic blood pressure; TAA, thoracic aortic aneurysm; TIA, transient ischemic attack; VTE, venous thromboembolism; WIfI, wound, ischemia, foot infection classification system.
A total of 212 patients had a CBC on postrevascularization day 1. Of these, 70 patients had a leukocyte differential on postrevascularization day 1 (Figure 1).
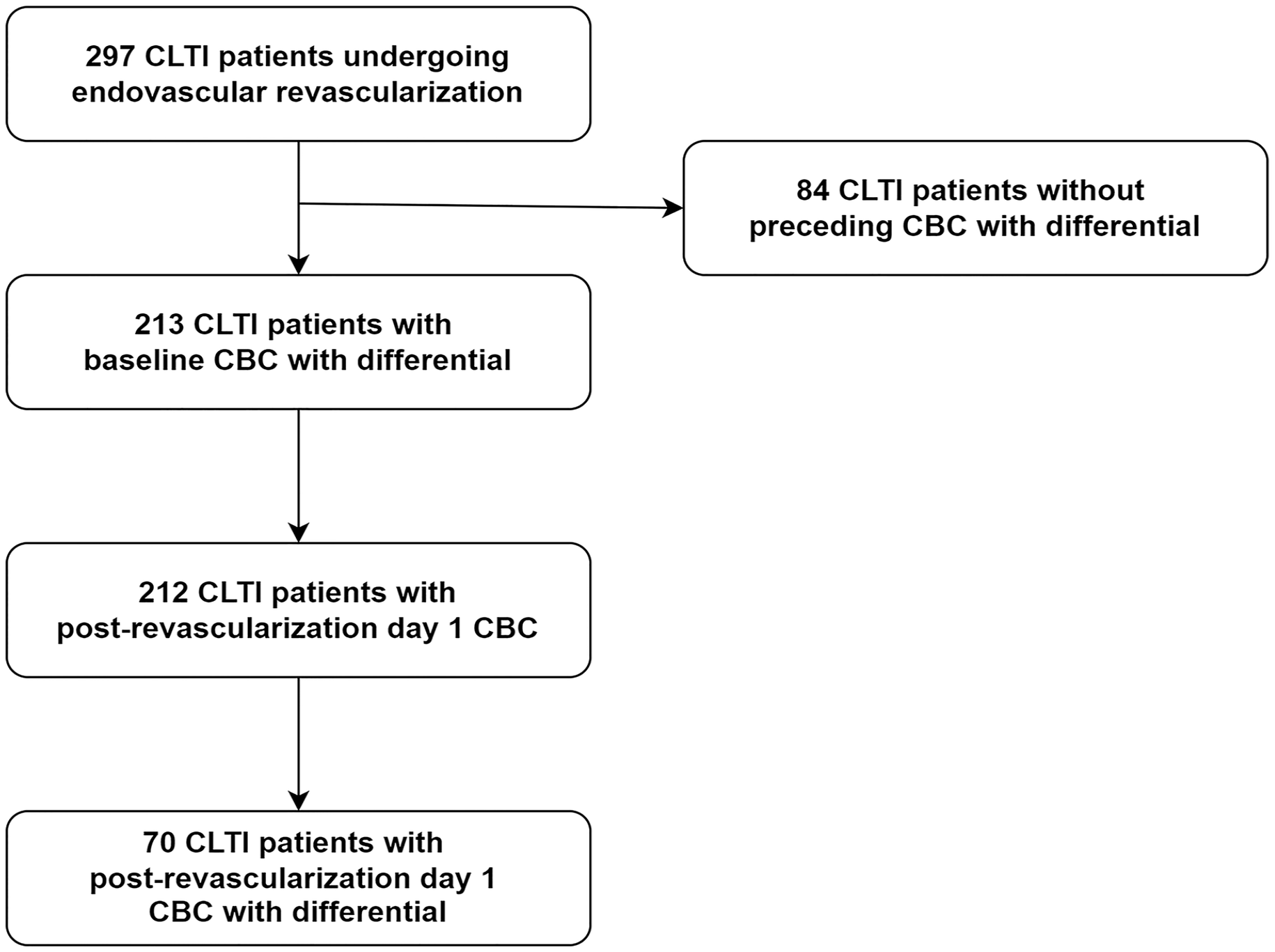
Flowchart of our study population.
Baseline CBC and mortality – univariate analysis
Patients with CLTI undergoing revascularization demonstrated marked differences in mortality associated with preprocedural ALC and RDW tertiles (Figure 2; p < 0.001 for both), whereas heterogeneity in antecedent ANC or AMC were not prognostic.
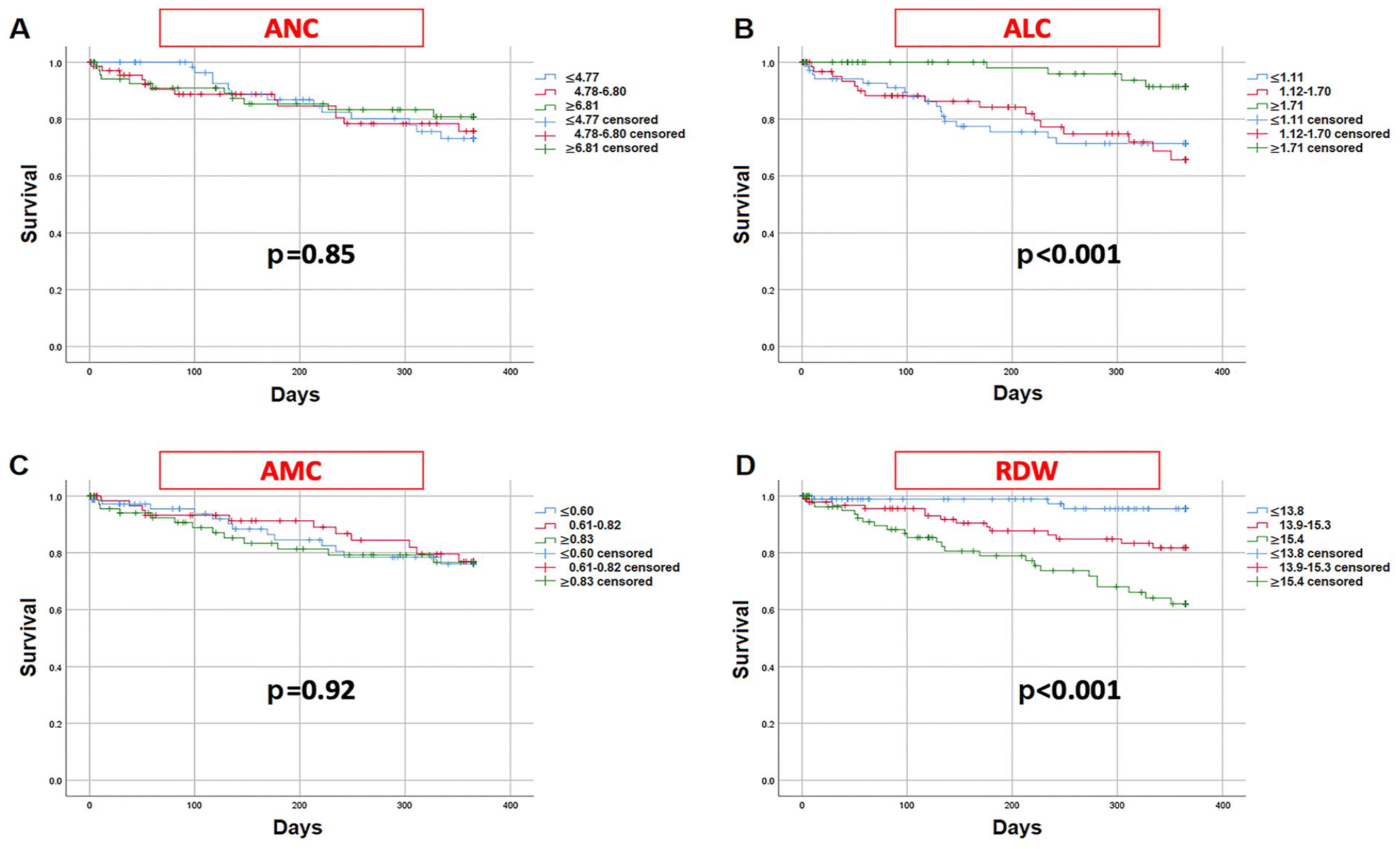
Kaplan–Meier survival curves against baseline leukocyte subsets (A-C) and RDW (D), each divided into tertiles.
Higher RDW tertiles were incrementally associated with increased mortality (Tertile 3 vs 1, hazard ratio [HR] 5.65, CI 2.13–14.99, p < 0.001; Table 3A). Similarly, as RDW continuously increased, the risk of mortality increased (HR 1.31 per each percentage of RDW elevation, CI 1.15–1.49, p < 0.001). The presence of anisocytosis, as defined by RDW > 14.5%, carried an unadjusted HR of 3.12 (CI 1.57–6.20, p = 0.001; Figure 3) for mortality.
Hazard ratios (HR) for mortality adjusted for potential confounders for red cell distribution width (RDW) and absolute lymphocyte count (ALC).
Table 3A.
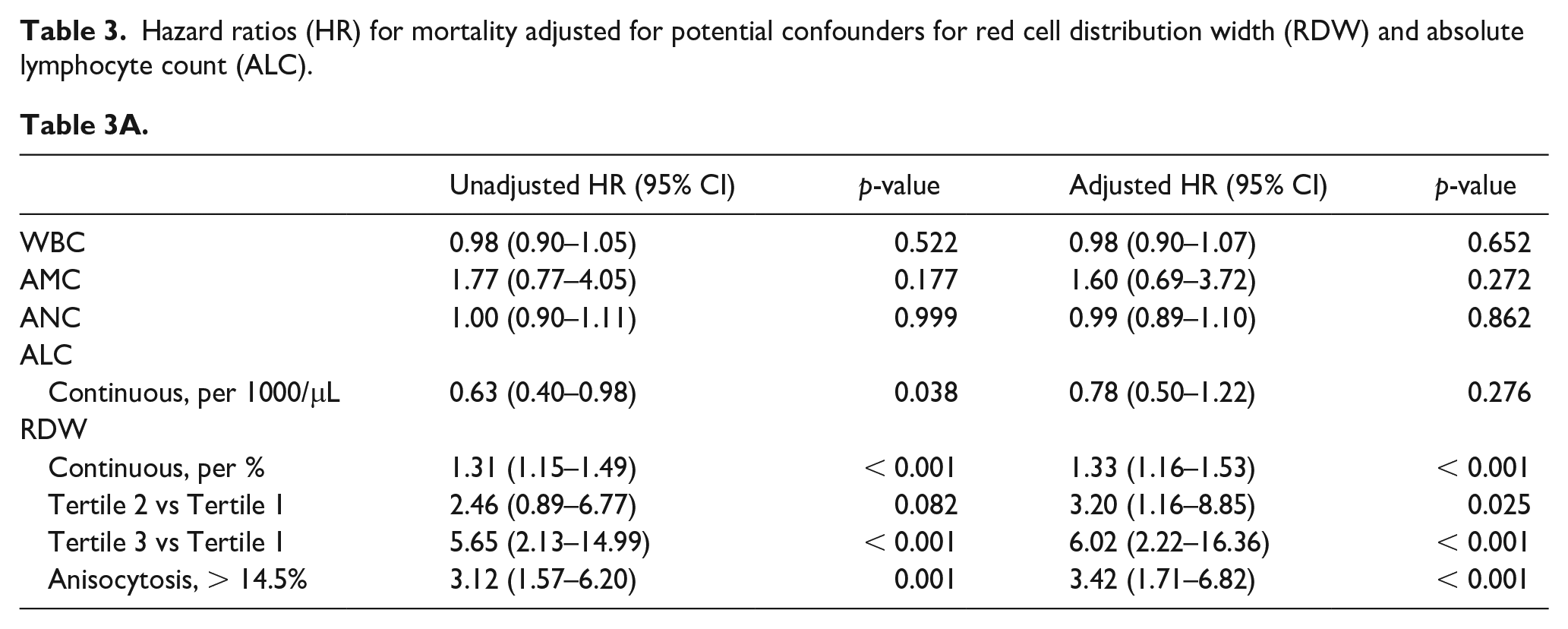
Table 3B.
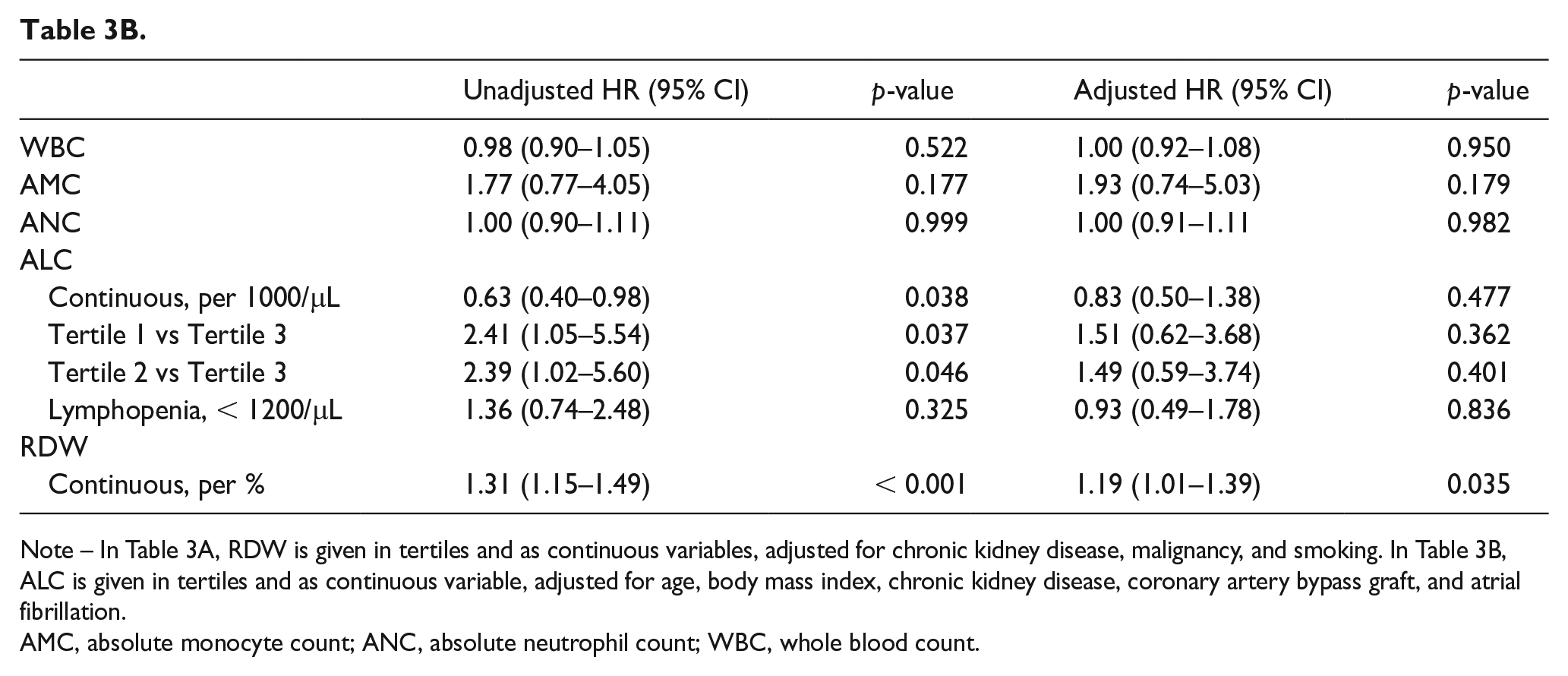
Note – In Table 3A, RDW is given in tertiles and as continuous variables, adjusted for chronic kidney disease, malignancy, and smoking. In Table 3B, ALC is given in tertiles and as continuous variable, adjusted for age, body mass index, chronic kidney disease, coronary artery bypass graft, and atrial fibrillation.AMC, absolute monocyte count; ANC, absolute neutrophil count; WBC, whole blood count.
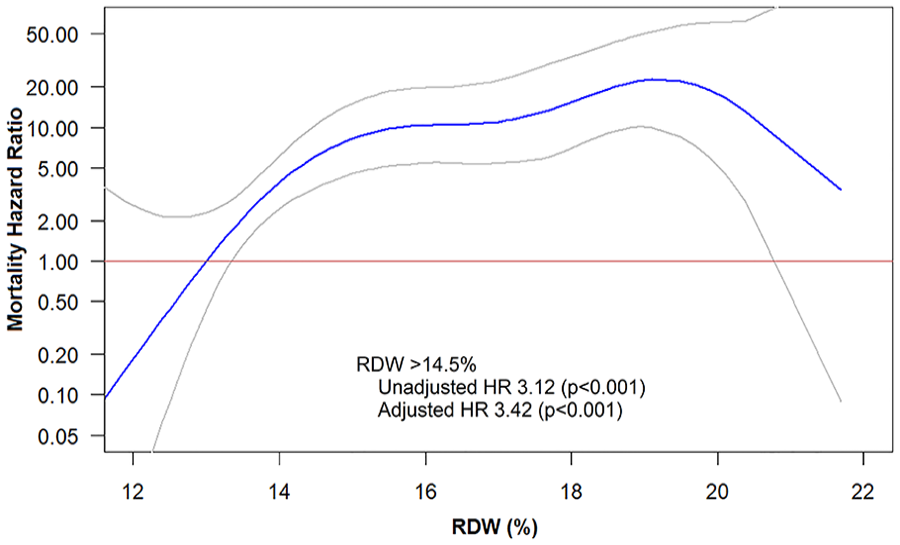
Spline analysis of RDW on a continuous scale against mortality.
Patients in lower ALC tertiles (Tertile 1 and 2) had increased risk of mortality compared to those with the highest ALC in Tertile 3 (Tertile 1 vs 3, HR 2.41, CI 1.05–5.54, p = 0.037; Tertile 2 vs 3, HR 2.39, CI 1.02–5.60, p = 0.046; Table 3B). Lymphopenia, defined by ALC < 1200/µL, carried an unadjusted HR of 1.36, but this was not statistically significant (online Supplemental Figure 1).
Overall, at the univariate level, these data suggest that those in the lowest tertile for RDW and/or the highest tertile of ALC carry strikingly low 1-year mortality (< 10%) compared to their counterparts (approximately 30%). In an exploratory fashion, evaluation of patients based on their ALC and RDW profiles combined (Figure 4) shows generally worse outcomes for those with RDW Tertile 2 and 3 in combination with ALC Tertile 1 or 2, with early mortality drop off as rapidly as at 100 days postprocedure. On Pearson’s correlation, there was a weak but nonsignificant correlation between ALC and RDW (online Supplemental Figure 2), suggesting that each component independently affects the outcome.
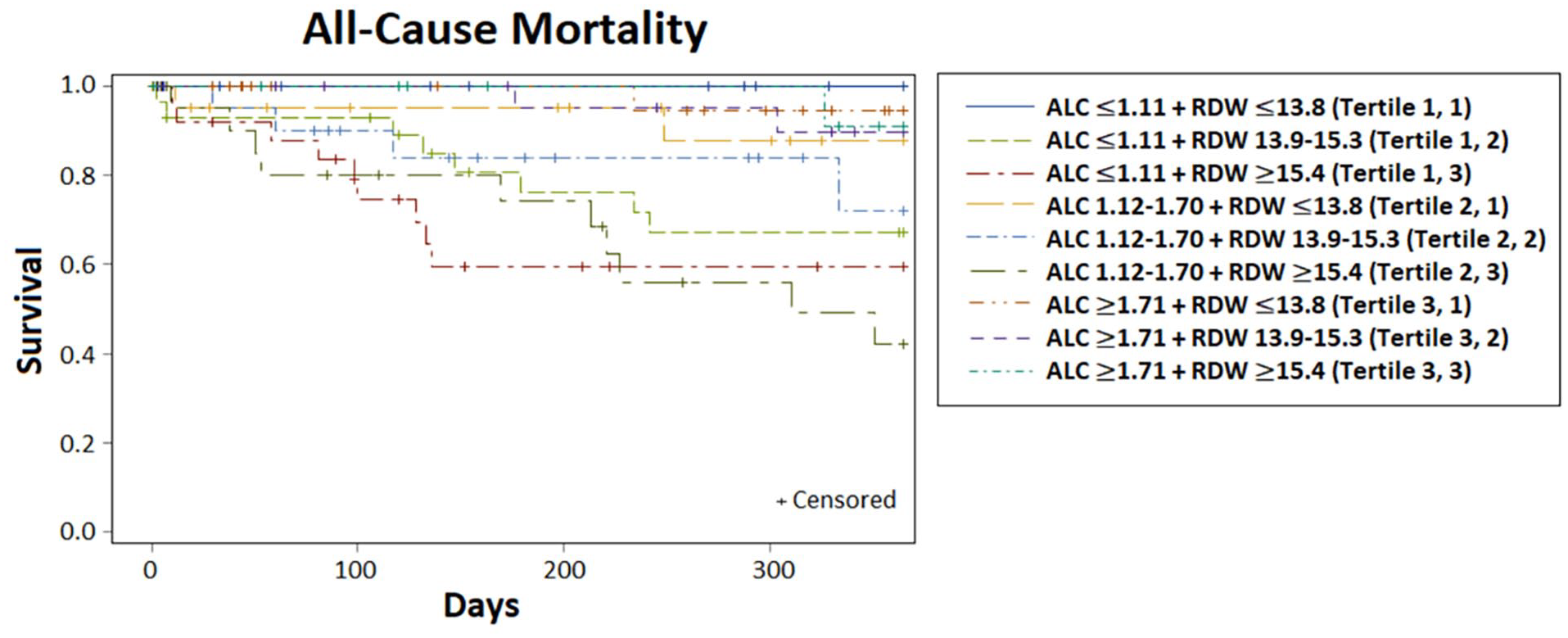
Kaplan–Meier survival curves based on various combinations of tertiles of ALC and RDW. Tertile 1 is the lowest, and Tertile 3 is the highest.
Baseline CBC and mortality – multivariate analysis
Some underlying characteristics were noted to vary based on RDW and ALC tertiles (Table 1 and 2). Multivariate analysis was performed with adjustments for the associated variables (Table 3A and 3B). With multivariate analysis, we detected persistently elevated all-cause mortality in association with higher RDW (Tertile 2 vs 1, HR 3.20, CI 1.16–8.85, p = 0.025; Tertile 3 vs 1, HR 6.02, CI 2.22–16.36, p < 0.001). Continuous variable analysis showed adjusted HR of 1.33 per each percentage of RDW elevation (p < 0.001). However, our multivariate analysis showed no persistent relationship between all-cause mortality and ALC in either tertiles or on a continuous scale.
Changes in CBC postrevascularization
In an exploratory fashion, we sought to analyze whether postprocedural trajectories in immunohematologic factors can predict mortality. On postprocedure day 1, as compared to baseline, there was a significant decrease in ALC level (∆ALC, p = 0.01) and increase in RDW (∆RDW, p = 0.004; Table 4). Changes in overall WBC, ANC, and AMC were not significant.
Paired analysis for change (Δ) in CBC values after revascularization.
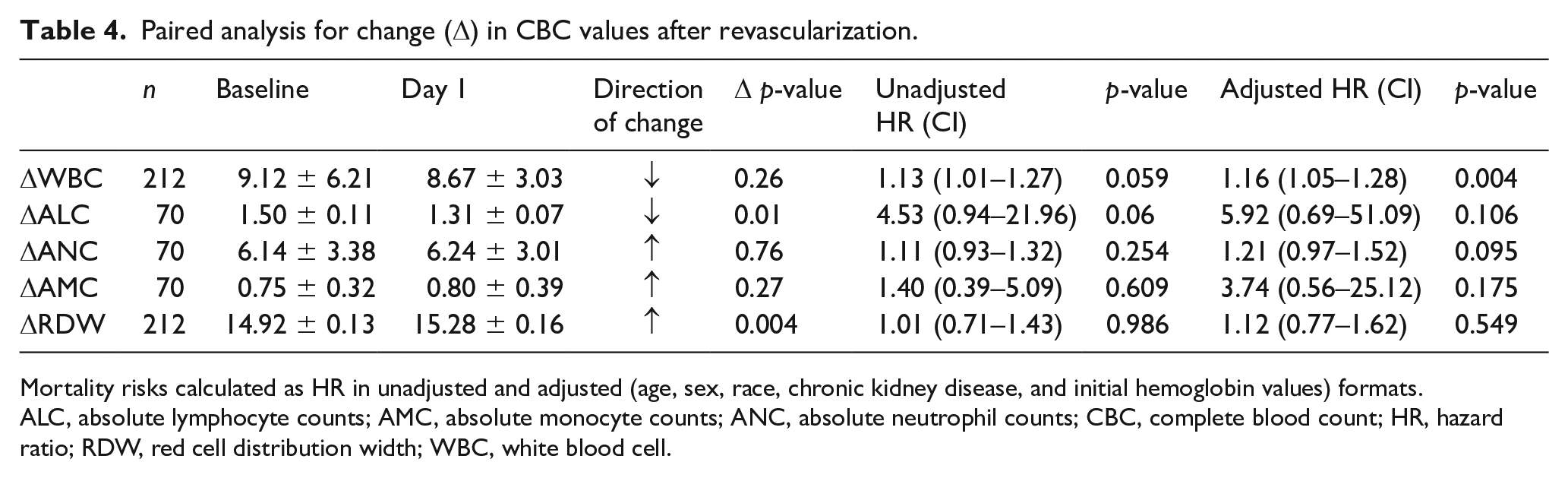
Mortality risks calculated as HR in unadjusted and adjusted (age, sex, race, chronic kidney disease, and initial hemoglobin values) formats.
ALC, absolute lymphocyte counts; AMC, absolute monocyte counts; ANC, absolute neutrophil counts; CBC, complete blood count; HR, hazard ratio; RDW, red cell distribution width; WBC, white blood cell.
Unadjusted analysis showed the negative ∆WBC and ∆ALC deflections to trend towards increasing mortality (p = 0.059 and 0.060, respectively). Adjusting for age, sex, race, chronic kidney disease (CKD), and baseline hemoglobin, we noted a statistically significant increase in mortality associated with ∆WBC (HR 1.16, CI 1.05–1.28, p = 0.004) but no statistically significant difference in mortality risk based on ∆ALC and ∆RDW (Table 4).
Other outcomes data
We further evaluated wound healing, TVR, major amputation, and all-comers readmissions against the tertiles of ALC and RDW (online Supplemental Table S3) in an exploratory fashion. The highest RDW tertile conveyed an increased risk of readmission in comparison to the lowest tertile (HR 1.83, p = 0.001), but no other statistically significant findings were noted.
Discussion
To our knowledge, this is the largest study to date assessing clinical outcomes in patients with CLTI undergoing endovascular intervention for limb salvage using WBC subsets and RDW as biomarkers. Preprocedural ALC and RDW were statistically significant markers of mortality in an unadjusted analysis, and multivariate analysis accounting for confounders showed persistent difference in mortality signal for higher tertiles of RDW. Acute postprocedural drop in leukocytes also portends increased risk of mortality. Further analyses showed RDW tertiles to be associated with readmission rates, but no other differences were noted in wound healing, TVR, or major amputation.
PAD is characteristically a chronic inflammatory state, with disease propagation rooted in endothelial dysfunction, plaque disruption, and oxidative stress. 17 Inflammation associated with CLTI, the most extreme of PAD presentations, is expected to be even more robust in comparison to patients with stable PAD. 18 Leukocytosis has been widely associated with poor outcomes in patients with coronary artery disease,19–21 and certain markers of inflammation (including CRP, erythrocyte sedimentation rate, and procalcitonin) are increasingly being used as cardiovascular prognostic indicators.14,15,22 However, associations between inflammation and mortality in patients with CLTI have not been as well studied.23–25
Others have previously shown an elevated neutrophil–lymphocyte ratio (NLR) to be an important prognosticator of mortality in patients with CVD.21,26–29 Neutrophils are activated by acute ischemia and associated with atherosclerotic plaque progression, and lymphopenia can also be a response to proinflammatory stimuli and/or neurohormonal excess.30–32 Whether the reactionary lymphopenic response is indicative of an appropriate or a pathologic response has yet to be determined. In our cohort, baseline lymphopenia, but not reactionary lymphopenia, appears to be associated with increased mortality. We were surprised to find that lymphopenia did not appear to predict wound healing or major amputation in our cohort, suggesting that more detailed immunophenotypic studies are needed to assess immune function in this setting.
RDW has previously been found to be associated with increased PAD severity and complexity. 33 We and others have shown that RDW is associated with elevation in certain proinflammatory conditions, as well as imbalances in multiple inflammatory and regulatory cytokines, and thus may serve as a pragmatic indicator of chronic immune dysregulation.34–38 Anisocytosis is known to be present in many forms of anemia, as is seen in our population. To avoid over-adjustment of confounders, which would increase the variance inflation factor, anemia and baseline hemoglobin were not adjusted for within our RDW multivariate model. Additionally, RDW is known to be associated with CKD, as reflected in our findings. However, even after our multivariate adjustment (Table 3A) for risk factors including CKD, mortality remained significantly higher in those with elevated RDW. Our current data confirm that within the tenuous CLTI population, anisocytosis heralds increased mortality along a linear continuum.
Based on our findings, those at the highest risk of mortality in our CLTI cohort are those with (i) elevated RDW at baseline and/or (ii) more dramatic negative deflections in ∆WBC on postprocedure day 1. Preintervention immunohematologic profiles and postintervention changes may be useful prognostic markers in the CLTI population undergoing endovascular interventions. These laboratory findings may help us identify the most vulnerable patients with CLTI requiring closer posthospitalization follow-up and perhaps more aggressive secondary prevention.
Study limitations
This study has several limitations, including its single-site, retrospective design. Given a relatively small number of patients, propensity matching between the tertiles of ALC and RDW could not be accomplished. The comparisons between the WBC subgroups and mortality were not defined prior to obtaining such data, so multiplicity and chance findings may contribute to the associations observed in this study. Moreover, the cause of mortality was not identified in subjects, which may limit the conclusions drawn. Finally, the dataset was obtained before the onset of the COVID-19 pandemic, and we do not yet know the interplay of COVID-19 with inflammatory markers and CLTI outcomes.
Future directions
Identification of the most vulnerable patients with CLTI can help guide comprehensive discussions between patients and providers, define treatment goals, and ultimately affect patient outcomes. Based on our findings, elevated RDW at baseline and negative changes in ∆WBC postprocedurally may help predict those at highest risk of longer-term mortality, even with successful endovascular revascularization. This may serve as an adjunctive tool to currently available CLTI risk scores39–42 and may help us identify those who can derive the most benefit from aggressive risk-factor modification. Future studies are needed to identify WBC subset profiles of the most vulnerable CLTI population, and to ascertain methods to alter their immune dysregulation and mitigate these risks. Lastly, the implications of WBC subsets on the patient with CLTI undergoing surgical revascularization needs to be better understood in comparison to those undergoing endovascular revascularization.
Conclusions
Baseline blood counts and trends in leukocytes following revascularization in patients presenting with CLTI may be useful prognostic variables in determining overall mortality following intervention. Such information can easily be obtained with a CBC and WBC subset differential upon admission and with close follow-up. More studies are needed to confirm the association of RDW and WBC subsets with mortality in CLTI, as well as determining potential methods of intervention to lower associated mortality.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X231169323 – Supplemental material for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization
Supplemental material, sj-docx-1-vmj-10.1177_1358863X231169323 for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization by Jun Li, Shilpkumar Arora, Heather Wheat, Siddhartha Dash, Stephen Kimura, Justin Smith, Yulanka Castro-Dominguez, Clint Oommen, Tarek A Hammad, Mehdi H Shishehbor, Sadeer Al-Kindi and David A Zidar in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X231169323 – Supplemental material for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X231169323 for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization by Jun Li, Shilpkumar Arora, Heather Wheat, Siddhartha Dash, Stephen Kimura, Justin Smith, Yulanka Castro-Dominguez, Clint Oommen, Tarek A Hammad, Mehdi H Shishehbor, Sadeer Al-Kindi and David A Zidar in Vascular Medicine
Supplemental Material
sj-pdf-3-vmj-10.1177_1358863X231169323 – Supplemental material for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization
Supplemental material, sj-pdf-3-vmj-10.1177_1358863X231169323 for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization by Jun Li, Shilpkumar Arora, Heather Wheat, Siddhartha Dash, Stephen Kimura, Justin Smith, Yulanka Castro-Dominguez, Clint Oommen, Tarek A Hammad, Mehdi H Shishehbor, Sadeer Al-Kindi and David A Zidar in Vascular Medicine
Supplemental Material
sj-pdf-4-vmj-10.1177_1358863X231169323 – Supplemental material for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization
Supplemental material, sj-pdf-4-vmj-10.1177_1358863X231169323 for Immunologic profiles in patients with chronic limb-threatening ischemia undergoing endovascular revascularization by Jun Li, Shilpkumar Arora, Heather Wheat, Siddhartha Dash, Stephen Kimura, Justin Smith, Yulanka Castro-Dominguez, Clint Oommen, Tarek A Hammad, Mehdi H Shishehbor, Sadeer Al-Kindi and David A Zidar in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Li: advisory board for Boston Scientific, Inari Medical, and Medtronic; consultant for Abbott Vascular, Endovascular Engineering, Inquis Medical, and Philips. Dr Shishehbor: global advisory board for Abbott Vascular, Medtronic, Terumo, Philips, Boston Scientific, ANT, and Inquis Medical. Drs Castro-Dominguez, Oommen, and Hammad: consultants for Medtronic. The remaining authors have no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
