Abstract
Purpose:
To determine the diagnostic accuracy of preinterventional imaging modalities in patients being evaluated for iliocaval venous recanalization and stent placement.
Methods:
Consecutive patients with iliocaval postthrombotic obstructions or nonthrombotic iliac vein lesions (NIVL), who were scheduled for recanalization, underwent duplex ultrasound (DUS), magnetic resonance venography (MRV), multiplanar venography (MPV), and intravascular ultrasound (IVUS). The diagnostic accuracies of DUS, MRV, and MPV were analyzed using IVUS as reference.
Results:
A total of 216 limbs in 108 patients (80 patients with postthrombotic obstructions, 28 patients with NIVL) were examined. In patients with postthrombotic obstructions, the diagnostic sensitivities for the detection of lesions of the common femoral vein were 81% (95% CI 71–89%) for DUS, 76% (95% CI 65–85%) for MRV, and 86% (95% CI 76–93%) for MPV. The sensitivities for detecting lesions of the iliac veins were 96% (95% CI 89–99%) for DUS, 99% (95% CI 92–100%) for MRV, and 100% (95% CI 94–100%) for MPV. Regarding the inferior vena cava, the sensitivities were 44% (95% CI 24–65%) for DUS, 52% (95% CI 31–73%) for MRV, and 70% (95% CI 47–86%) for MPV. The sensitivities for detecting NIVL were 58% (95% CI 34–79%) for DUS, 90% (95% CI 68–97%) for MRV, and 95% (95% CI 73–99%) for MPV.
Conclusion:
In patients scheduled for recanalization of iliocaval postthrombotic obstructions, the sensitivities of DUS, MRV, and MPV were similar. In patients with suspected inferior vena cava involvement and in patients with NIVL, additional imaging with MR or conventional venography is required.
Introduction
Iliocaval venous obstructions are a common cause of chronic venous hypertension of the lower limbs. 1 Flow-limiting venous lesions can either be caused by postthrombotic obstructions or by nonthrombotic iliac vein lesions (NIVL).
Current guidelines recommend endovascular recanalization and stent placement in patients with clinically relevant chronic postthrombotic iliocaval or iliofemoral obstructions and for patients with symptomatic NIVL.2,3
In postthrombotic obstructions and NIVL, endovascular recanalization with stent placement effectively reduces symptoms of venous hypertension and – in case of skin ulcerations – facilitates ulcer healing.4–7 Success and durability of stent placement substantially depends on inflow and outflow of the recanalized venous segments as well as on the appropriate determination of stent landing zones. Therefore, optimal preinterventional imaging is crucial for estimating the extent of affected venous segments.
Duplex ultrasonography (DUS) is commonly used as the first imaging modality in patients with chronic venous disease. In patients who are scheduled for endovascular procedures because of suspected iliocaval venous obstructions, DUS is often complemented by cross-sectional imaging, such as magnetic resonance venography (MRV).3,8–10 Especially in young patients, MRV is preferred over computed tomography venography to minimize radiation exposure. 8 Regarding contrast-enhanced imaging methods, catheter-based multiplane venography (MPV) provides direct visualization of affected venous segments. In the scope of invasive procedures, the highest diagnostic accuracy in detecting iliac vein obstructions was found for intravascular ultrasound (IVUS).11,12
However, data on the diagnostic accuracy deriving from direct head-to-head comparisons of different imaging modalities in patients with iliocaval venous obstructions are limited.2,10
We therefore sought to directly compare the diagnostic accuracies of noncontrast and contrast-enhanced imaging modalities using IVUS as the standard of reference in patients with iliocaval venous obstructions.
Methods
Patient selection
Consecutive patients who were referred to our institution, a tertiary care university hospital, for further assessment of suspected chronic venous disease of the lower limbs were screened. Of these patients, subjects with suspicion of postthrombotic obstructions of the common femoral vein, the iliac veins (external and/or common iliac vein), and/or of the inferior vena cava, which were further scheduled for endovascular imaging and treatment, were eligible for this study. In addition, patients without a history of deep vein thrombosis but suspicion of NIVL, who were scheduled for endovascular diagnostic work-up and stent placement, if appropriate, were included.
Patients with active cancer, tumor compression or recurrent iliocaval thrombosis after previous recanalization and stent placement were excluded from this study.
All included patients systematically underwent DUS, MRV, MPV, and IVUS of both (right and left) common femoral veins, both (right and left) iliac veins, and of the inferior vena cava. IVUS served as the reference method for the comparison of the diagnostic accuracy of individual imaging techniques.11,12
The study was performed in accordance with good clinical practice guidelines and was approved by the ethical committee of the Medical University of Vienna (1685/2018). As this study constitutes a retrospective data analysis, no informed consent was required.
Clinical evaluation
Prior to imaging studies, all patients underwent a careful clinical evaluation. The severity of clinical symptoms was routinely categorized according to the CEAP (Clinical Etiology Anatomy Pathophysiology) classification, and the Villalta score was obtained. Clinically manifest comorbidities and history of previous deep vein thrombosis were recorded.
Definition of venous lesions
The presence of endoluminal fibrosis, strands, septae, scarring, or total chronic obstruction of the common femoral veins, iliac veins, or inferior vena cava resulting in a > 50% lumen reduction were classified as presence of postthrombotic obstructions. In patients with no history of deep vein thrombosis and without evidence of postthrombotic obstructions, a > 50% narrowing of the common iliac vein between the crossing artery and the spine was defined as presence of NIVL. 13
Duplex ultrasound (DUS)
All duplex scans were conducted by two trained medical technicians in the presence of one investigator. Representative duplex images of both (right and left) common femoral veins, both (right and left) iliac veins, and of the inferior vena cava were stored, and each examination was additionally reviewed offline by an experienced vascular sonographer.
For all examinations the patients were placed in a supine position in a quiet room with a constant room temperature after an overnight fasting period of 8 hours. High-resolution B-mode and duplex investigations were performed by using two ultrasound machines (Siemens Sequoia 512, Germany, and Mindray Resona 7, P.R. China) at the institutional duplex laboratory. Appropriate transducer probes were used for longitudinal ultrasound scans along the individual venous segments. As an initial approach, a 9-MHz linear transducer was used. In addition, especially for the visualization of the iliocaval confluence and of the inferior vena cava, a 5-MHz curvilinear transducer was additionally applied. B-mode was used to identify narrowing of the respective venous segments, endoluminal fibrosis, formation of intraluminal septae, and/or strands or a complete obstructive scarring of the respective venous lumen. In addition, duplex was used to confirm the presence of venous obstructions (turbulent flow, flow acceleration, loss of flow modulation, or absent flow). An integrative approach based on these qualitative criteria was used for identification of relevant venous stenosis. Lack of spontaneous respiratory variation in the Doppler waveform and during Valsalva maneuver upon Doppler interrogation of the common femoral vein was considered indicative of relevant proximal/central venous obstruction. No routine measurements of lesion length were obtained by DUS. The presence or absence of postthrombotic obstructions was finally rated for each venous segment separately (right and left common femoral vein, right and left iliac veins, and inferior vena cava). In patients without a history of deep vein thrombosis, the presence or absence of NIVL was documented in included patients.
Magnetic resonance venography (MRV)
MRV was performed using a 1.5 T MR System (Siemens Avanto Fit, Siemens Healthineer, Erlangen, Germany) using body surface coils. All examinations were performed under spontaneous breathing of patients. MRV included a native time-of-flight venography of the iliac veins as well as contrast-enhanced, time-resolved magnetic resonance angiography (MRA)/MRV of the whole abdominal region after the application of 1 mmol/kg gadobutrol (Gadovist, Bayer AG, Germany). This dynamic 3D MRA technique allows for visualization of the contrast dynamics within the abdomen including the venous structures, and especially allows for detection of collateral pathways. 14 By creating 3D datasets at different time points during contrast application, the sequence also allows for multiplanar reconstructions. Additionally, T1-weighted sequences with fat saturation after the application of contrast were available for the evaluation of the abdominal veins. This sequence allows visualization of thrombotic/postthrombotic remnants within the abdominal veins. The respective MRV protocol is can be accesssed in the Supplemental material. All MRV studies were reviewed independently by two experienced investigators. Excellent agreement between both investigators was observed (common femoral veins: κ = 0.91, iliac veins: κ = 0.88, inferior vena cava: κ = 1). In cases of disagreement regarding the rating on presence or absence of venous lesions, a third investigator was consulted for final judgement. A reduction in venous cross-sectional area in the time-of-flight sequence of ⩾ 50% was considered significant.
Multiplane venography (MPV)
Endovascular procedures were performed at the institutional catheterization laboratory by two experienced interventionists. For all examinations the same angiography system (Siemens Artis Q with PURE, Siemens Healthineer) was used.
For each endovascular procedure, lower limb venous access was obtained under local anesthesia and ultrasound guidance. Depending on the infrainguinal extent of disease, the superficial femoral vein or the popliteal vein were punctured, and a 10-French sheath was inserted. Aiming to achieve comprehensive MPV images of both (right and left) common femoral veins (involving the confluence of the respective femoral veins and deep femoral veins), both iliac veins, and of the inferior vena cava (up to the right atrium), bilateral access was routinely used allowing simultaneous bilateral injections of contrast media. For contrast injections, 20 mL of Iopamiro 300 mg/mL (iopamidol; Bracco Imaging, Milan, Italy), diluted 1:1 with saline, were applied manually via the inserted sheaths or via diagnostic catheters, and lesion imaging by fluoroscopy was obtained in two different projections. Regarding fluoroscopy, a standardized protocol (7.5 pulses per second, four pictures per second) was used. All MPV images were evaluated before balloon dilation or stent placement. In patients with postthrombotic obstructions, a reduction in venous diameter of ⩾ 50% in at least two planes was considered significant, whereas the angle between the two planes had to be at least 30°. In suspected NIVL, local contrast translucency at the left common iliac vein at the crossing point of the right common iliac artery accompanied by collateral flow was considered an indicator of significant venous stenosis.
Intravascular ultrasound (IVUS)
For IVUS imaging, iliocaval lesions were crossed by a 0.035" hydrophilic guidewire and support catheters. Before IVUS imaging the correct position of the guidewire was confirmed by multiplane imaging. Along this guidewire, a Visions PV .035 IVUS catheter (Philips Volcano, the Netherlands) was inserted and the transducer probe at the tip of this catheter was placed in the inferior vena cava proximal of the central end of the lesion. Then, the IVUS catheter was pulled back manually from the inferior vena cava to the respective introducer sheath. This allows continuous IVUS evaluation of the inferior vena cava, the iliac veins, and the common femoral veins. Continuous cross-sectional IVUS imaging, as well as the longitudinal reconstruction of the assessed venous segments, were stored on the attached console for offline analysis. IVUS assessment was obtained before balloon dilation or stent placement. A reduction in cross-sectional area of ⩾ 50% compared to the adjacent healthy venous segment was considered significant.
Comparison of imaging methods
The presence or absence of postthrombotic obstructions were analyzed separately for each venous segment (right and left common femoral veins, right and left iliac veins, and inferior vena cava) and by each imaging modality (DUS, MRV, MPV, IVUS) to determine diagnostic accuracy of the respective modalities. By analogy, in nonthrombotic patients, the presence of NIVL was studied by DUS, MRV, MPV, and IVUS (right and left iliac veins only). The findings of DUS, MRV, and MPV were compared with IVUS, which was used as reference. In all included patients, the target limb, which was the clinically symptomatic limb, as well as the contralateral limb, were assessed for the presence of obstructive venous lesions. Finally, the respective lesion lengths were assessed by MRV, MPV, and IVUS.
Statistical analysis
Discrete data are given as counts and percentages, continuous data are given as mean and SD. For the assessment of interrater agreement (of MRV), Cohen’s kappa was calculated based on the binary ratings (presence or absence of venous obstructions, defined as stenotic lesions (> 50%) or total occlusions) of both examiners at each venous segment (common femoral veins, iliac veins, inferior vena cava). To assess the relation between the ordinal CEAP C class and the respective lesion length as determined by IVUS, Spearman’s correlation coefficient was calculated.
Sensitivity, specificity, positive predictive values (PPV), and negative predictive values (NPV) of DUS, MRV, and MPV for the detection of obstructive venous lesions of the right and left common femoral veins, the right and left iliac veins, and the inferior vena cava were calculated using IVUS as reference. The analysis of the respective nontarget limbs was used additionally to include negative controls in this selected group of patients.
To take into account correlated observations within patients (two legs), sensitivity, specificity, NPV, and PPV, as well as the corresponding 95% CIs, were estimated using generalized estimating equations (R-package mmmgee; R Foundation for Statistical Computing, Vienna, Austria ). 15 In case only independent observations were considered, the 95% CIs were calculated using the Wilson score method with continuity correction. In patients with postthrombotic obstructions, differences in lesion lengths as assessed by MRV and IVUS, as well as MPV and IVUS, respectively, were examined and plotted as a function of their respective averages of measured lesion lengths using Bland–Altman plots.16,17 The respective Bland–Altman plots depict the deviations of lesion length measurements of MRV and MPV, both in comparison with IVUS. In both plots, the average bias is indicated by bold dashed lines. The 95% CIs for the average bias are depicted by the two thin dashed lines. The 1.96-fold SD of the average bias is shown by the upper and lower dashed bold lines. All statistical calculations were done using the software package R version 3.6.1 (R Foundation for Statistical Computing) and the R-package ‘mmmgee’ was used for simultaneous inference for multiple marginal GEE-models. 15
Results
Imaging studies in 111 patients with suspected iliocaval venous obstructions, who had been assigned to DUS, MRV, MPV, and IVUS between July 2016 and February 2022, were examined. After exclusion of three patients with incomplete IVUS imaging, 108 patients (80 patients with postthrombotic obstructions and 28 patients with NIVL) were included in the final analysis. In each patient, the presence or absence of obstructive venous lesions of both (right and left) common femoral veins, both (right and left) iliac veins, and of the inferior vena cava was assessed by DUS, MRV, MPV, and IVUS. Demographic and clinical characteristics of included patients with postthrombotic obstructions and NIVL are shown in Table 1.
Demographic and clinical characteristics of patients with iliocaval venous outflow obstructions.
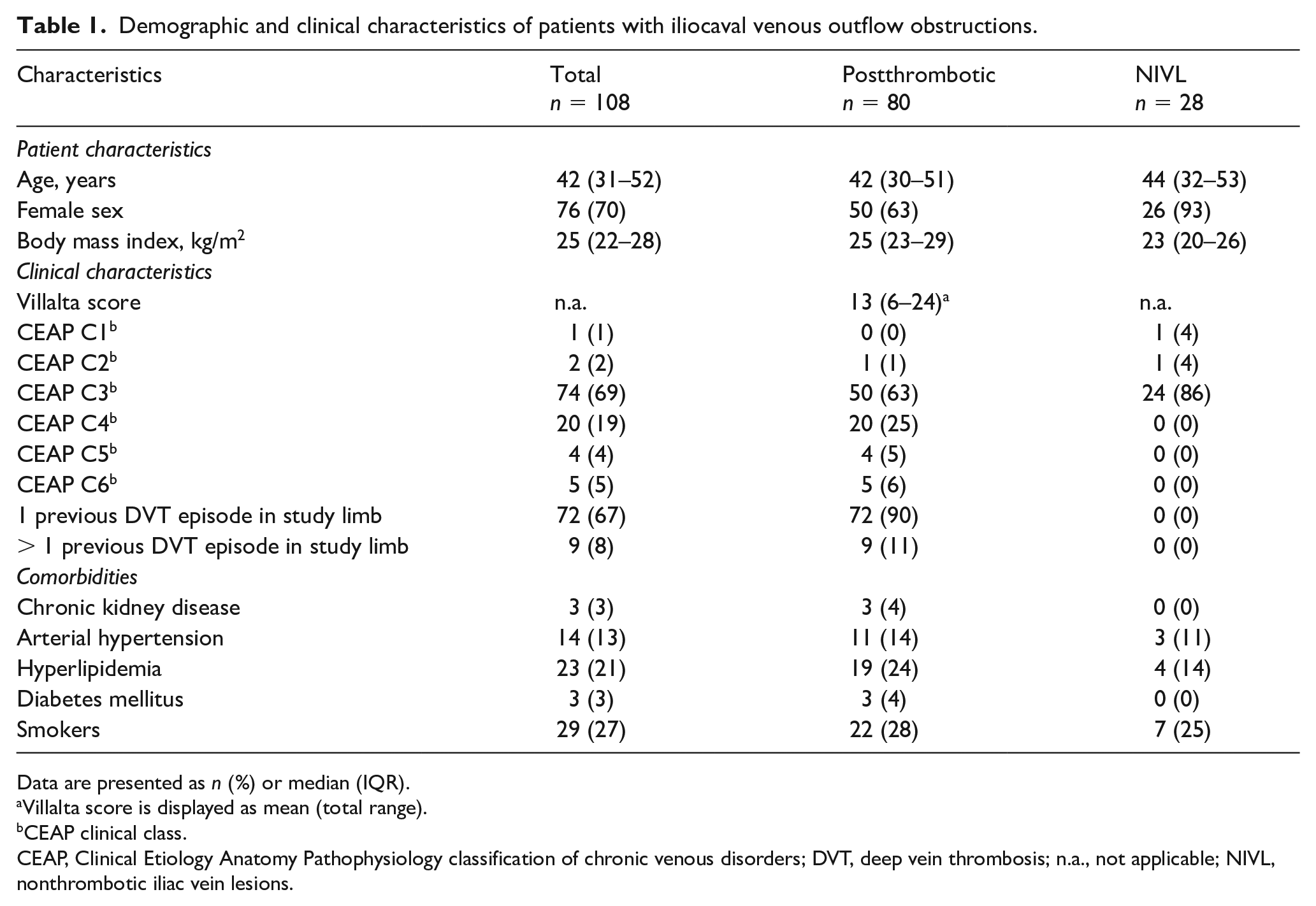
Data are presented as n (%) or median (IQR).
Villalta score is displayed as mean (total range).
CEAP clinical class.
CEAP, Clinical Etiology Anatomy Pathophysiology classification of chronic venous disorders; DVT, deep vein thrombosis; n.a., not applicable; NIVL, nonthrombotic iliac vein lesions.
Anatomic lesion distribution
Focusing on the symptomatic limb, the percentage distribution of obstructive venous lesions in patients with postthrombotic obstructions and NIVL as detected by the individual imaging methods is shown in Figure 1. In patients with postthrombotic obstructions, the proportion of venous occlusions versus stenotic lesions (> 50%) within the total number of obstructive lesions is shown in Figure 2. In patients with NIVL, only the iliac veins were affected.
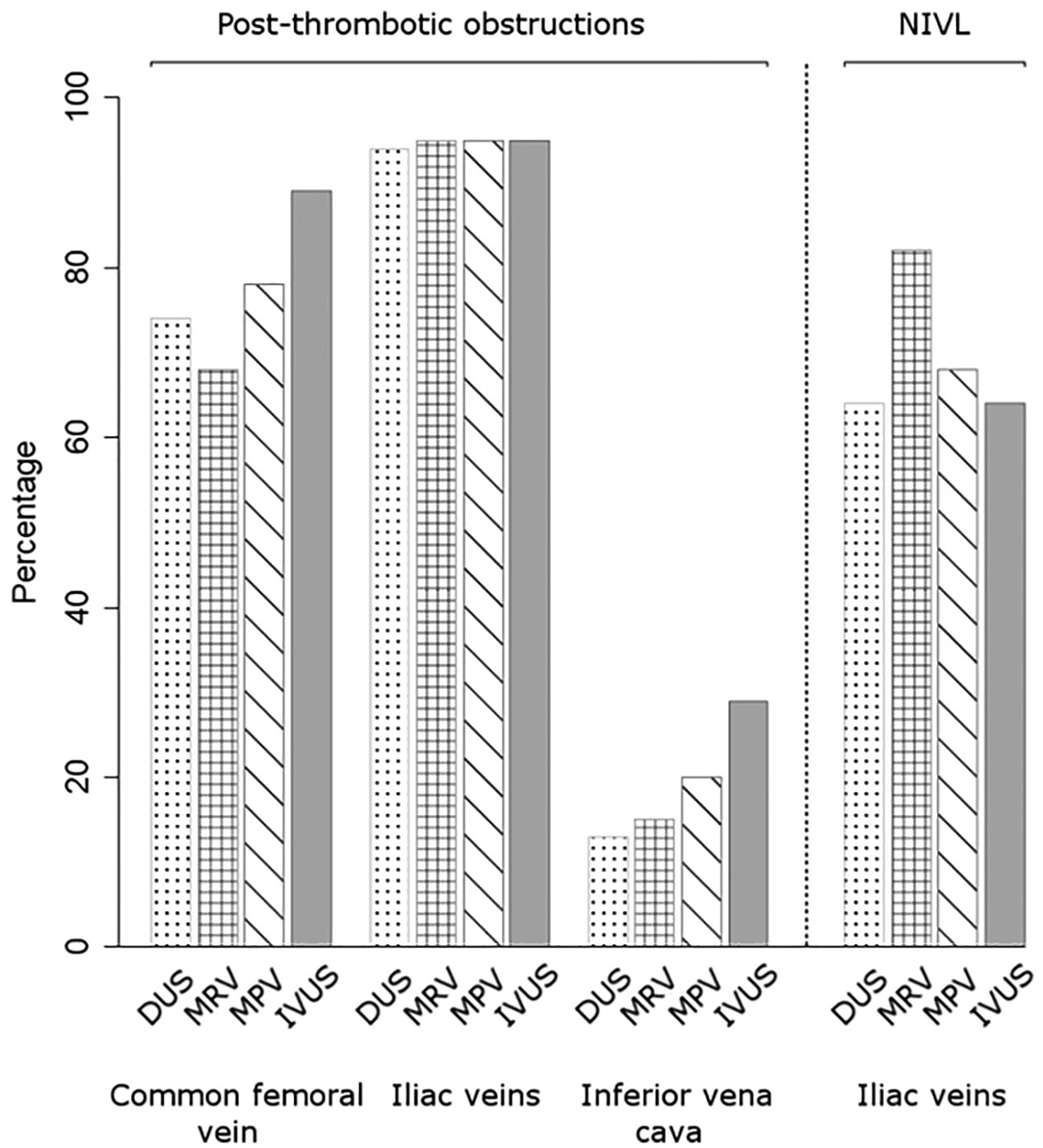
Frequency of lesion detection (stenotic lesions [> 50%] and total occlusions) according to individual venous segments and imaging modality (DUS, MRV, MPV, IVUS) in the target limbs of 80 patients with iliocaval postthrombotic obstructions and 28 patients with NIVL.
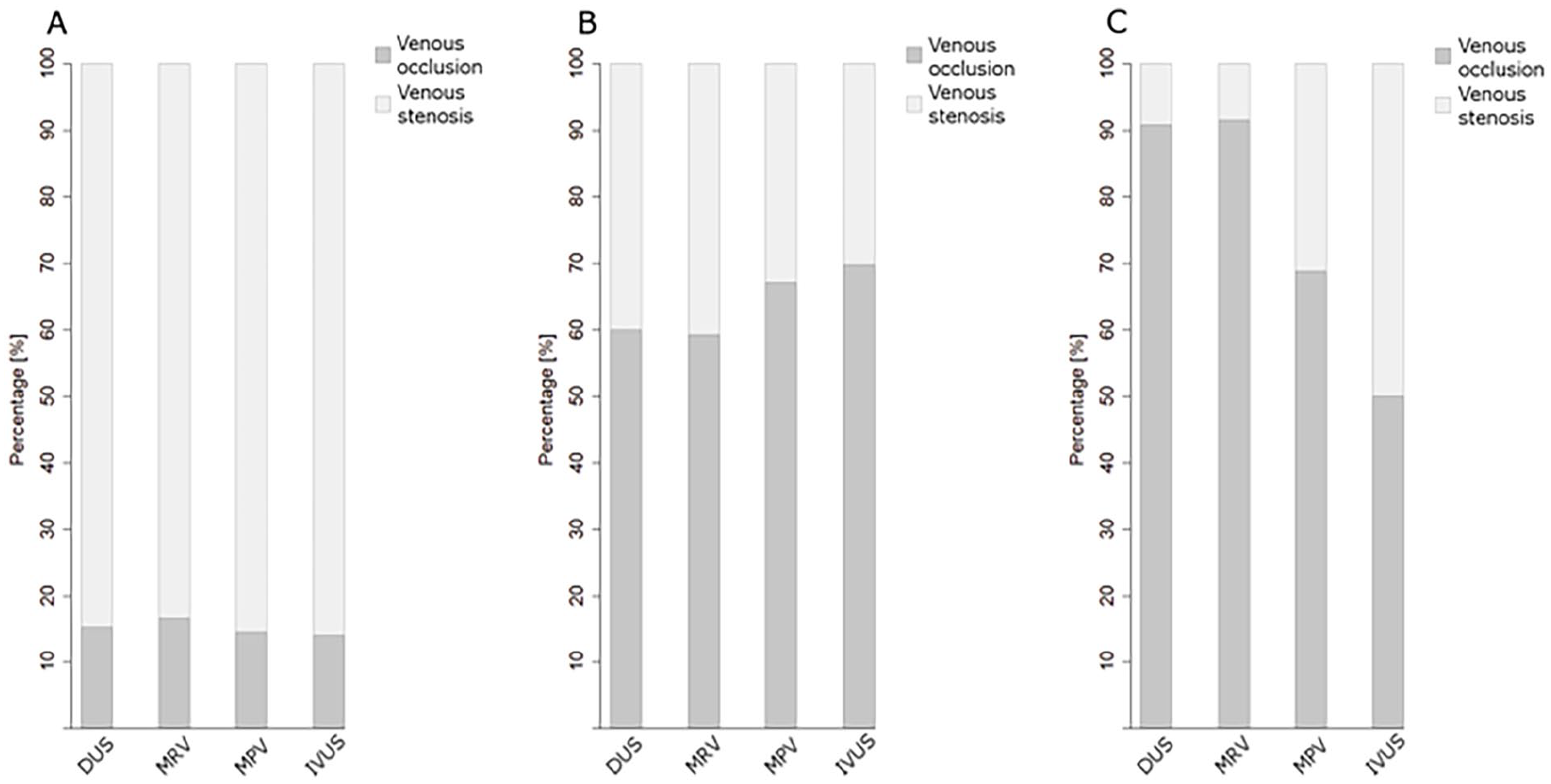
Percentage proportions of total occlusions and stenotic lesions (> 50%) of all lesions, as assessed by DUS, MRV, MPV, and IVUS within
Diagnostic sensitivity
In patients with postthrombotic obstructions, the diagnostic sensitivities of DUS, MRV, and MPV in detecting venous obstructions of the common femoral veins and iliac veins were similar (using IVUS as reference; Table 2). Regarding the inferior vena cava, the diagnostic sensitivities of DUS, MRV, and MPV were lower; however, DUS, MRV, and MPV comparably conferred high specificities and NPV (Table 2). In patients with NIVL, the diagnostic sensitivity and specificity of DUS were low in comparison with MRV and MPV (Table 3).
Sensitivities, PPVs, specificities, and NPVs of duplex ultrasound, magnetic resonance venography, and multiplane venography for the detection of postthrombotic obstructions.
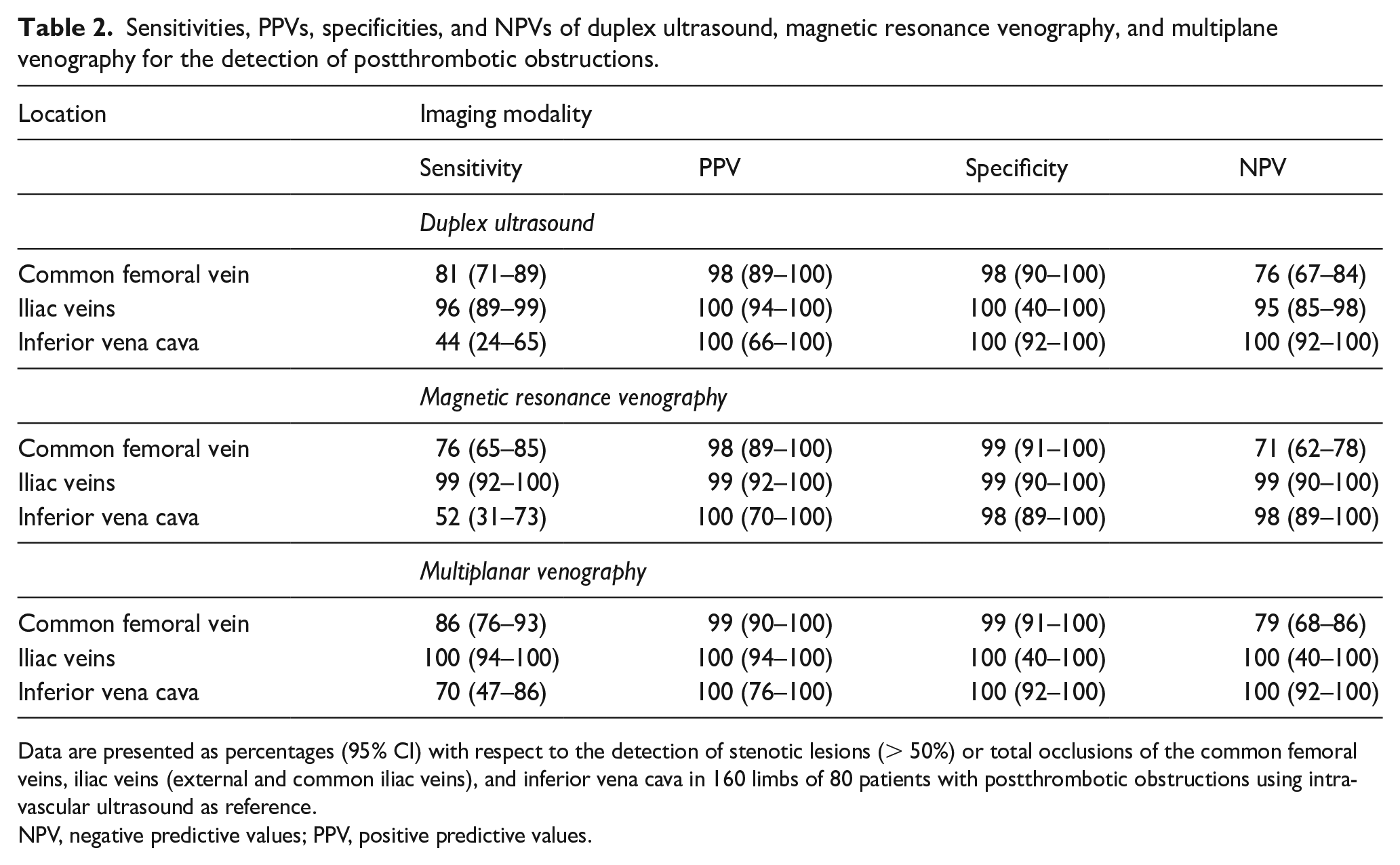
Data are presented as percentages (95% CI) with respect to the detection of stenotic lesions (> 50%) or total occlusions of the common femoral veins, iliac veins (external and common iliac veins), and inferior vena cava in 160 limbs of 80 patients with postthrombotic obstructions using intravascular ultrasound as reference.
NPV, negative predictive values; PPV, positive predictive values.
Sensitivities, PPVs, specificities, and NPVs of duplex ultrasound, magnetic resonance venography, and multiplane venography for the detection of nonthrombotic iliac vein lesions.
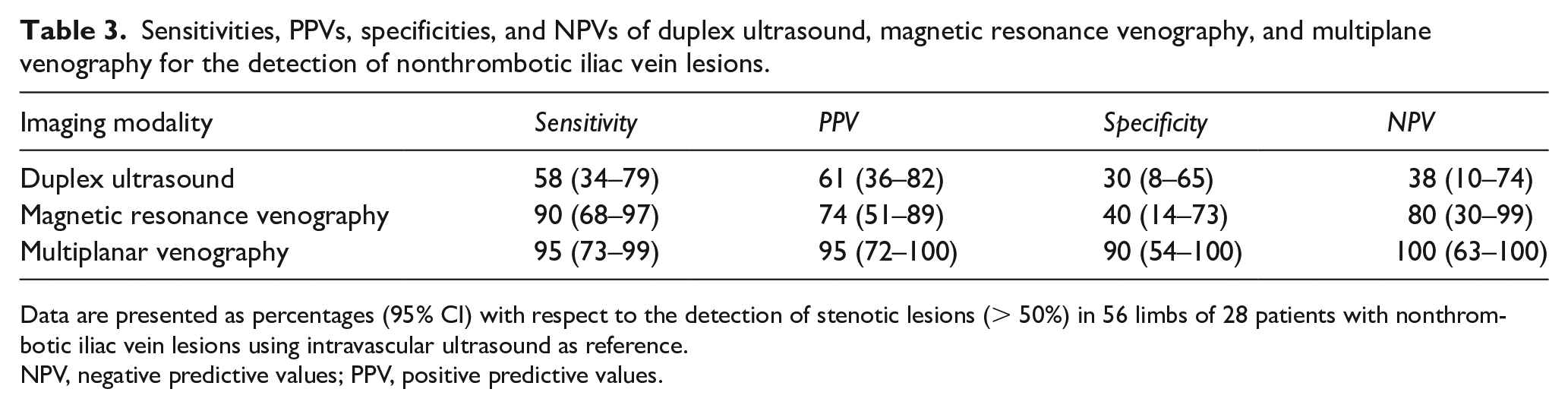
Data are presented as percentages (95% CI) with respect to the detection of stenotic lesions (> 50%) in 56 limbs of 28 patients with nonthrombotic iliac vein lesions using intravascular ultrasound as reference.
NPV, negative predictive values; PPV, positive predictive values.
Lesion length
The mean lesion length of postthrombotic obstructions was 237
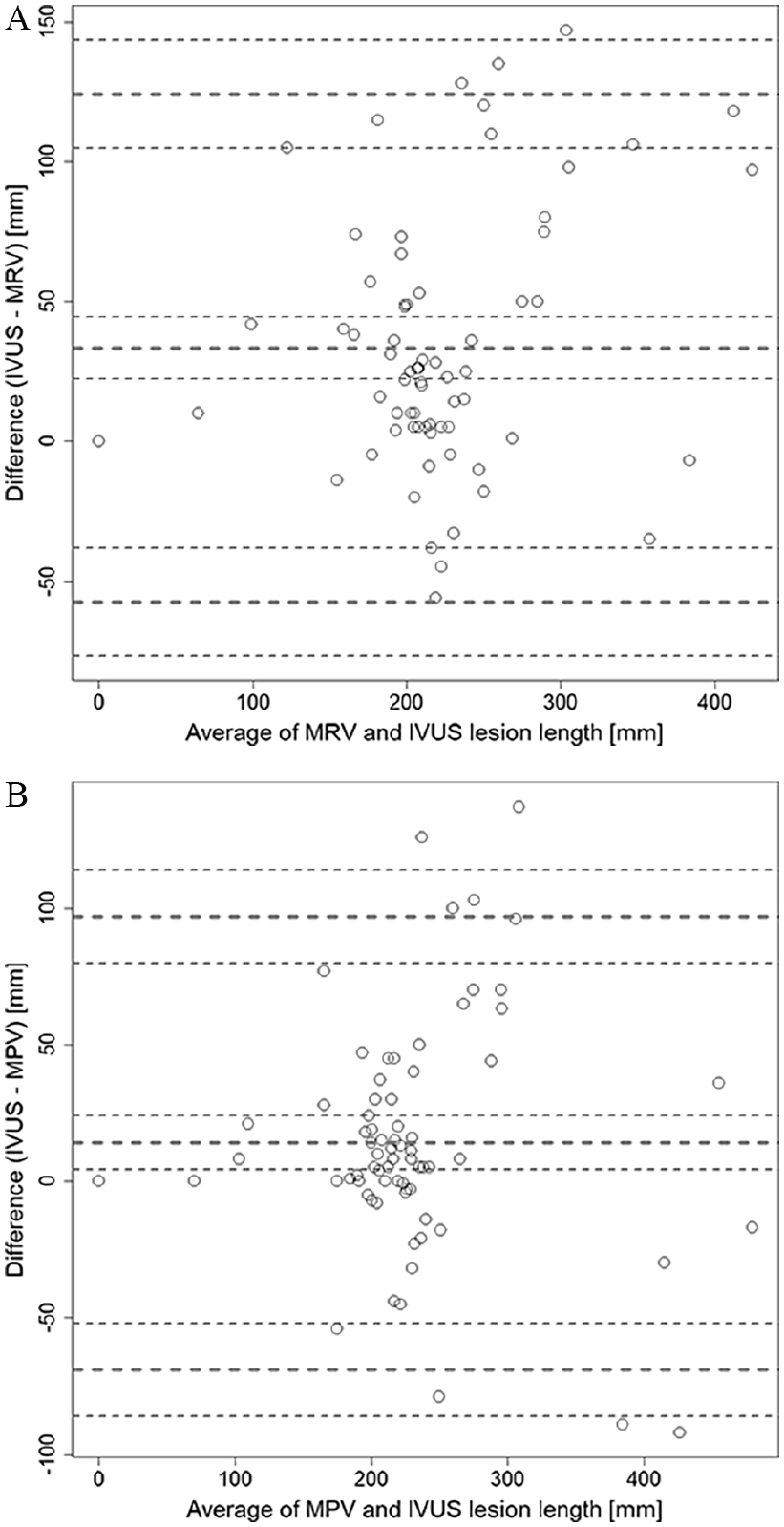
Bland–Altman plots showing the deviations of lesion length measurements of
In patients with NIVL, the mean lesion length was 64
Regarding patients’ clinical presentation, no correlation was observed between the lesion length (assessed by IVUS) and the clinical CEAP class (Spearman’s correlation ρ = −0.05, p = 0.67).
Discussion
As a main finding, the findings of the present study suggest comparable sensitivities of DUS, MRV, and MPV for the detection of postthrombotic obstructions of the iliofemoral segment. Regarding postthrombotic lesions of the inferior vena cava, however, the diagnostic sensitivity of DUS was low in comparison with other imaging modalities.
Within the spectrum of the applied imaging methods, DUS is most commonly used as the initial approach in the clinical work-up of patients with chronic venous disease to identify venous valvular reflux and proximal venous outflow obstructions, as well. 18 DUS provides excellent information on the inflow of the superficial, deep, and common femoral veins, which is required for iliac venous stent patency.
The diagnostic value of DUS in the detection of iliac vein obstructions, however, is still a matter of discussion. Previous studies on the accuracy of DUS-guided planimetry measurements of chronic iliac venous lesions are inconsistent.19,20 Apart from planimetric measurements, sonographers commonly refer to indirect measures, such as venous flow phasicity in the common femoral vein. However, especially in chronic venous outflow obstructions, indirect flow evaluation of femoral veins might not be reliable as a single criterion. 21 In the present study, we therefore additionally focused on longitudinal B-mode scans of the common femoral veins, the iliac veins, and the inferior vena cava to identify venous lumen narrowing, intraluminal septae, or obstructive scarring, and to complement this we integrated color-coded DUS imaging information. When discussing DUS, one must keep in mind that DUS depends on the investigators’ experience as well as on the resolution quality of the respective ultrasound machine. To minimize the variability of DUS, all examinations were performed by the same trained technicians and proven qualitative standard.
More than in other imaging modalities, the required penetration depth of ultrasound potentially has an impact on imaging accuracy. This explains the gap of the diagnostic sensitivity of DUS regarding the iliofemoral veins and the inferior vena cava. Additionally, the interpretation of DUS images may be severely impaired in patients with obesity or clinical conditions causing significant abdominal distension, and alternative imaging modalities may be sought in these cases. In the given study population, the median BMI was 25 (IQR 22–28), facilitating DUS image acquisition in most participants.
From a clinical point of view, however, the key question in patients with a history of unilateral deep vein thrombosis rather would be whether DUS allows the exclusion of inferior vena cava involvement. Based on the observed NPV of DUS for the detection of inferior vena cava lesions in this study, a negative DUS scan of the inferior vena cava could be used to rule out inferior vena cava involvement.
Importantly, it should be noted that the choice of the most appropriate imaging method always should be made upon each patient’s individual clinical context. Especially in patients with clinical suspicion of inferior vena cava involvement, one should consider additional imaging on top of DUS to evaluate inferior vena cava involvement.
When estimating lesion length, clinicians and interventionalists should be aware of a potential underestimation when referring to MRV or MPV as additional imaging. According to the extensive length of postthrombotic obstructions and the curved course of the iliocaval venous axis, we refrained from DUS-guided length measurements. Regarding the observed underestimation of lesion length by MRV and MPV, one explanation could be the presence of intraluminal postthrombotic septae extending over the central or peripheral lesion, which were more reliably captured by IVUS than by MRV or MPV. This observation confirms that final stent length fitting should be obtained during the procedure itself by IVUS length measurements. To avoid unnecessary stent elongation or extensive stent overlaps, an appropriate selection of different stent lengths should be available on stock.
Regarding preprocedural imaging in patients with NIVL, the current study demonstrated a lower diagnostic accuracy of DUS in comparison with MRV or MPV. This finding contrasts with the diagnostic sensitivity of DUS in the detection of postthrombotic obstructions, which was similar with MRV and MPV. The discrepancy of the diagnostic accuracy of DUS between postthrombotic obstructions and NIVL regarding the detection of iliac vein obstructions might be attributed to different lesion lengths and properties of postthrombotic obstructions and NIVL. First, iliocaval postthrombotic obstructions are characterized by intraluminal changes, such as endoluminal fibrosis, scarring, and intraluminal septae. In contrast, NIVL is caused by extraluminal compression of iliac veins between crossing arteries and – at its typical location – the lumbar spine. 13 Although NIVL can be associated with occasional formation of intraluminal webs or spurs, which increase the NIVL length over the pure diameter of the crossing iliac artery, these structural changes are rarely captured by DUS. Second, NIVL typically involve the iliocaval confluence beyond the crossing arteries, which potentially results in a lower imaging resolution of DUS than of other imaging modalities. Third, the mean lesion length of NIVL is naturally shorter than the mean length of postthrombotic obstructions. Postthrombotic obstructions commonly affect the entire venous iliocaval segment including more superficial parts of the iliac veins and sometimes the common femoral veins, which both can easily be visualized by DUS. In contrast to patients with postthrombotic obstructions, patients with suspected NIVL should therefore generously be referred to further imaging modalities in addition to DUS.
Study limitations and strengths
In summary, our findings must be viewed in the light of the study’s strengths and limitations. First, it should be acknowledged that this study was conceptualized as a descriptive diagnostic study exploratively analyzing the diagnostic sensitivity of various imaging methods. Regarding the choice of the preinterventional cross-sectional imaging method, regional variations in the applicability and deployment of MRV warrant mention. According to the young age of many patients undergoing iliocaval recanalization and stent placement, our aim was to minimize radiation exposure of these patients. Therefore, we integrated MRV as preferred cross-sectional imaging of iliocaval veins in the diagnostic work-up of patients with venous diseases. However, we are aware that computed tomography venography is commonly used in the evaluation of iliocaval venous obstructions. Since we did not obtain MRV and computed tomography venography in parallel, we cannot additionally provide data on the agreement of DUS, MPV, and MRV with computed tomography venography. Future studies need to elucidate the diagnostic accuracy of computed tomography venography in comparison with DUS, MRV, and MPV.
To justify the use of IVUS as the reference method in deep vein imaging, it needs to be stated that this is based on a greatly appreciated, but still limited amount of data.11,12 In turn, this aspect underlines the need for further studies on imaging accuracy in the deep venous system. Moreover, according to the invasive character of IVUS, additional data on the diagnostic reliability of noninvasive modalities in the evaluation of the deep venous system are required.
Finally, the characterization of venous lesions warrants mention. We defined endoluminal fibrosis, formation of intraluminal septae or strands, as well as a complete obstructive scarring resulting in a > 50% lumen reduction as a relevant venous lesion. Nevertheless, it needs to be acknowledged that there is a lack of standardized definitions, which underlines the need for further research in this field.
Nevertheless, the major strength of this study is the availability of all distinct imaging data in patients with postthrombotic obstructions and NIVL, allowing a direct head-to-head comparison of the diagnostic accuracies of three imaging modalities using IVUS as the standard of reference. A recent review on venous imaging modalities summarized previous studies comparing individual imaging techniques with IVUS. 10 However, none of these studies directly compared the three distinct imaging techniques with IVUS.
A small retrospective study reported a lower specificity and a lower PPV of MRV in the diagnosis of postthrombotic obstructions than we observed in our investigation. 22 In contrast to our study, however, MRV images were imported from various radiological facilities, implementing different protocols. Aiming to optimize MRV reliability and to reduce the risk of variation, all MRV examinations were obtained at the same institutional Department of Radiology.
Most importantly, iliocaval imaging comparisons including DUS are scarce.
In the future, prospective multicentric studies are warranted to validate the findings of this investigation. In addition to the diagnostic sensitivity, future studies might further include hemodynamic data that could noninvasively be obtained by DUS and might help to additionally specify the hemodynamic and clinical relevance of deep vein lesions.
Conclusion
The diagnostic sensitivities of DUS, MRV, and MPV in the detection of postthrombotic obstructions of the iliofemoral veins are similar. In patients with suspected inferior vena cava involvement and in NIVL, additional imaging methods with MRV or MPV are required. Regarding the extent of postthrombotic obstruction lesions, MRV and MPV could lead to an underestimation of total lesion length. In patients with NIVL, DUS needs to be complemented by additional imaging modalities.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231161938 – Supplemental material for Preprocedural imaging modalities in patients undergoing iliocaval venous recanalization and stent placement
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231161938 for Preprocedural imaging modalities in patients undergoing iliocaval venous recanalization and stent placement by Markus Müller, Florian Wolf, Christian Loewe, Dietrich Beitzke, Sonja Zehetmayer, Michael E Gschwandtner, Andrea Willfort-Ehringer, Renate Koppensteiner and Oliver Schlager in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
