Abstract

Claudication in peripheral artery disease (PAD) results from abnormalities in skeletal muscle oxygen delivery, mitochondrial oxygen utilization, or both. 1 Supervised exercise training improves claudication, although the mechanisms of benefit remain incompletely defined. Near-infrared spectroscopy (NIRS) provides an index of oxygen delivery and utilization in skeletal muscle.2–4 We previously explored the role of NIRS in a pilot study of supervised exercise in patients with PAD, finding that NIRS-directed exercise had a similar benefit to a traditional pain-based training strategy. 5 A knowledge gap remains regarding whether adaptations from exercise rehabilitation are the result of increases in oxygen delivery, enhanced oxygen use, or both. Accordingly, we revisited training data from these reported cohorts to explore the possible ‘dose–response’ relationship between mitochondrial adaptations in skeletal muscle and improvements in claudication symptoms after supervised treadmill exercise. 7
From local clinics, we screened 71 patients with mild to moderate PAD, among whom 36 were enrolled and 18 completed the 12-week study. (Most enrolled patients who discontinued the study before completion cited leg pain as the major factor.) The study was conducted between June 2013 and May 2016 with the approval of the institutional review board at the Augusta University and was conducted on the campuses of the University of Georgia and Athens Regional Medical Center. Informed consent was obtained from all participants. Patients were assessed on a 12-minute walk test. Treadmill speed was kept constant at 2 miles per hour (mph) while the grade increased by 2% every 2 minutes. Muscle mitochondrial capacity (i.e., rate of oxygen use, or Tc) and microcirculation perfusion index (i.e., rate of oxygen delivery, or T1/2) were measured using NIRS. 4 Muscle work was calculated based on training performance using the summed product of subject mass in kg multiplied by the total vertical distance traveled (derived from pace, walking time, and slope). NIRS-based determination of mitochondrial capacity and microvascular flow were measured at baseline and after 12 weeks. A Pearson correlation was also performed to determine the association between oxyhemoglobin (HbO2) and muscle work.
Eighteen patients completed the training protocol; exercise data were available for 17 patients (22% women). Mean age was 71 years and mean body mass index was 27.1 kg/m2. Baseline mean ankle-brachial index (ABI) was 0.85 ± 0.28. In the baseline progressive walk test, the mean time to first pain symptoms was 3.2 ± 2.3 min and the mean total walking time was 8.6 ± 3.7 min; baseline pain-free walking time was similar in each group (2.5 ± 0.9 vs 3.6 ± 1.0 min, p = 0.5). The mean NIRS-derived microcirculation perfusion index (T1/2) was 46.4 seconds. Patients trained for an average of 32.9 ± 5.9 sessions, walking at 54.4 ± 13.4 m/min per session. Patients achieved a mean of average workload of 4767 (± 5112) kilogram-force meters (kpm) per session and an aggregate of 148,536 (± 148,020) kpm for the training program. A baseline pain-free walking time of 2.95 (± 2.1) minutes increased to a mean of 3.74 (± 2.47) minutes. An increase in walking time was proportional to the mean work level achieved per session, with a correlating trend to the overall work level performed over 12 weeks (r = 0.543, p = 0.024). There was no increase in blood flow measured by ABI (p = 0.49) or limb microcirculation perfusion index (T1/2, p = 0.55) over the training period. Skeletal muscle mitochondrial capacity increased over the training period and correlated with the overall work performed (r = 0.573, p = 0.016) (Figure 1).
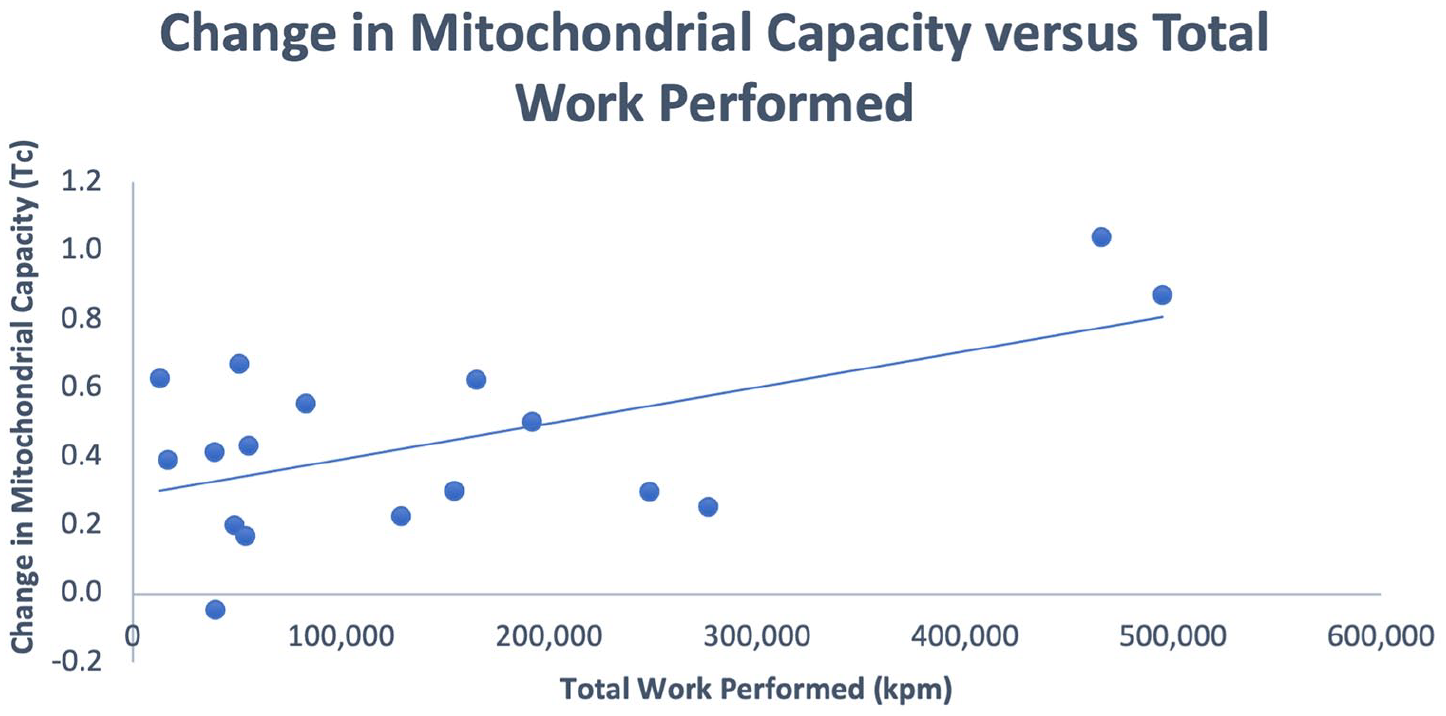
Correlation between change in pain-free walking time and total work performed.
The correlation between mitochondrial adaptation and exercise exposure suggests a ‘dose–effect’. Exercise rehabilitation in PAD has long been held as an effective means of improving clinical performance, and in healthy populations exercise is associated with adaptive changes in mitochondrial size and number. 6 The mechanism of benefit has been debated, but skeletal myopathy has been noted in PAD as measured in rodent models, human muscle biopsy, and 31P magnetic resonance spectroscopy phosphocreatine recovery time. 7 Improvements in claudication from treadmill exercise training may result more so from adaptations in skeletal muscle mitochondrial oxidative capacity than improvements in blood flow. Van Schaardenburgh et al. found that in patients with claudication, respondents to exercise training demonstrated improvements in mitochondrial respiration by muscle biopsy compared to negative responders. 8 Baker et al. observed an improvement of oxygen extraction in patients with PAD after exercise training, attributed by these authors to enhanced muscle oxidative metabolism. 9
A limitation in this study is the heterogeneous exposure to exercise by participants reflected in the work performed, despite the 12-week, thrice weekly treadmill prescription. In addition, there was no control group of patients with claudication that did not undergo exercise training. This group would have confirmed that usual care does not lead to adaptations in walking performance, and therefore changes in mitochondrial oxidative capacity as seen in other studies.
Our findings suggest that the clinical focus on improving ischemia in claudication by revascularization strategies may need to be matched with new approaches to improve oxygen utilization capacity in muscle. This study was meant to explore whether mitochondrial adaptation corresponded to exercise exposure. Transferability of the findings might include considering a novel intermediate marker of rehabilitation response.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jonathan Murrow, Kent Nilsson Jr, and Kevin McCully have ownership interest in Infrared Rx, Inc. The remaining authors have no conflicts to disclose.
Funding
This study was supported by a Grant in Aid (13GRNT1693006) from the American Heart Association.
