Abstract

What is giant cell arteritis (GCA)?
Giant cell arteritis (GCA; also called temporal arteritis) is an inflammatory condition of the large arteries of the body, including those which supply blood to the head and neck, which can occur in older patients (50+ years of age). It is the most common systemic vasculitis, or inflammatory vascular condition. The clinical manifestations of GCA depend on the type and location of the affected arteries. It is important to diagnose and treat GCA without delay because serious complications including blindness and nerve disorders can occur if left untreated. 1
Physicians in internal medicine, rheumatology, vascular medicine, vascular surgery, dermatology, and/or ophthalmology may care for patients with GCA. Typical evaluation for GCA includes a patient’s history and symptoms, physical examination, blood tests for inflammation, appropriate imaging, biopsy of the affected artery or arteries, and referral to a specialist if warranted. Treatment of GCA is aimed at suppressing inflammation. Despite proper diagnosis and treatment, long-term cardiovascular complications can sometimes occur; however, most cases of GCA can be well-controlled and managed over a patient’s lifetime.
What causes GCA?
There is no single known, identifiable cause of GCA. The disease process of GCA begins with an inflammatory cascade. An often-unknown trigger can cause large ‘giant’ cells to form in the walls of large arteries, including the aorta and the carotid, subclavian, and iliac arteries. 2 The most commonly affected arteries are those that start in the neck and travel into the head and scalp, including the temporal arteries which course up each side of the forehead near the ears. 3 The temporal arteries enlarge, swell, or thicken in many patients with GCA. Although GCA and temporal arteritis are synonymous, GCA can also simultaneously affect multiple arteries, even smaller arteries in the head and neck, including the ophthalmic, occipital, vertebral, posterior ciliary, or proximal vertebral arteries. Blindness, the most serious complication of GCA, is caused by damage to the posterior ciliary branch of the ophthalmic artery. Inflammation in larger arteries can cause both aneurysm (dilation) or stenosis (narrowing) of the vessel.
Who is at risk for GCA?
The risk of developing GCA depends on several factors, including age, sex, geography, genetics, and other medical problems. Age is the most important factor. GCA almost never occurs before the age of 50, and it becomes more common with increasing age. In western countries, women are approximately twice as likely to develop GCA as men. 4 GCA is also associated with an inflammatory condition affecting the muscles called ‘polymyalgia rheumatica’ (PMR). 4 Other risk factors include ethnicity, with the highest rates in Scandinavia; smoking; genetically determined HLA-DRB1*04 alleles (common in Northern Europeans); and other genetic mutations.
What are the symptoms of GCA?
As shown in Figure 1, the symptoms of GCA occur both at the site of the affected artery or arteries and throughout the body due to inflammation. Vision loss remains the most serious symptom, but it typically happens after the more general symptoms appear. These may include intermittent headaches; visual changes; pain or tiredness in the jaw when chewing (also called jaw claudication); tenderness of the scalp; pain in the throat, tongue (related to tissue death or ‘necrosis’ due to lack of blood flow), neck, or temples; muscle aches; and flu-like symptoms including cough, fever, anemia, weight loss, loss of appetite, or fatigue.
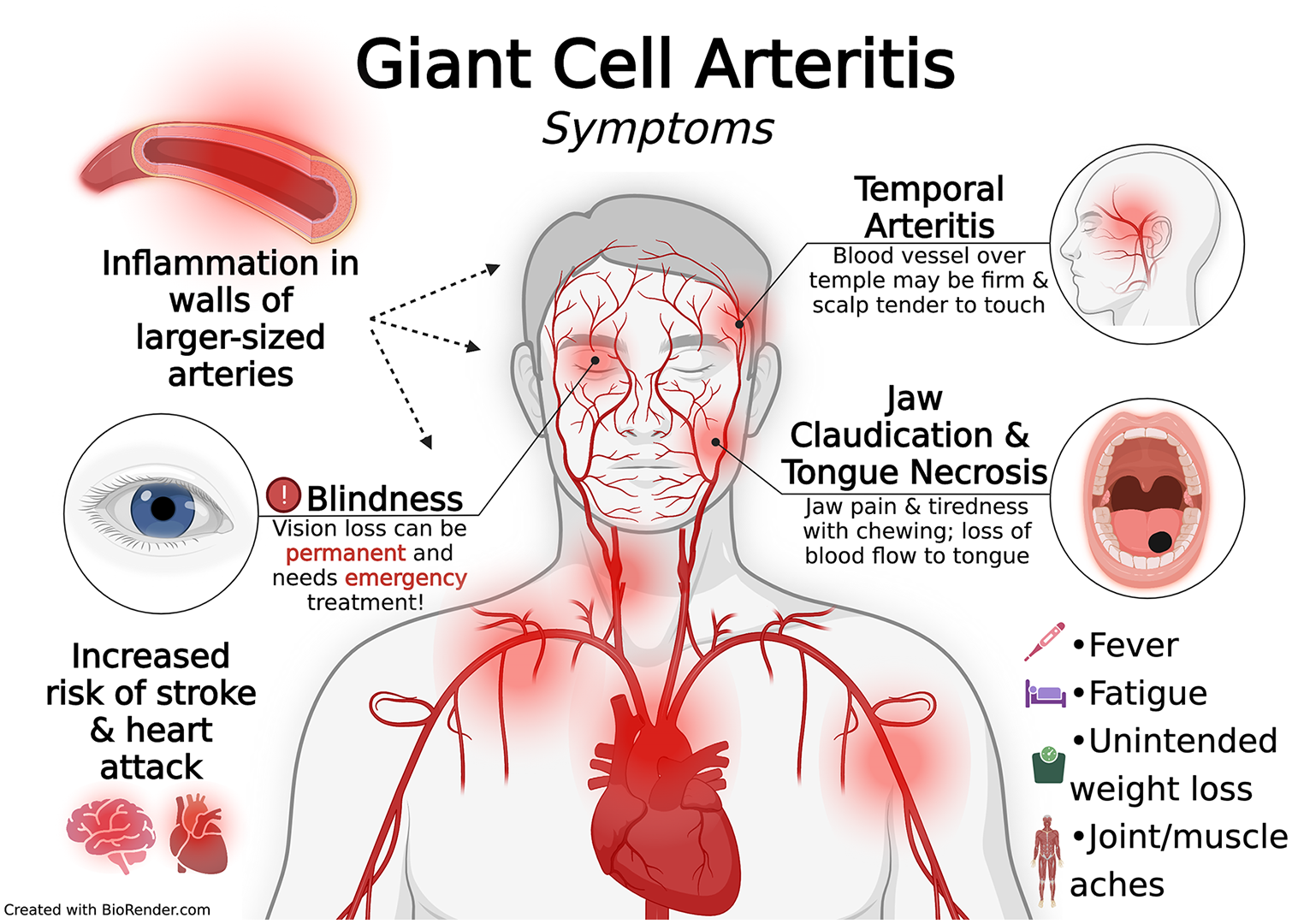
Signs and symptoms of giant cell (temporal) arteritis.
How is GCA diagnosed?
If a patient has the typical symptoms and risk factors for GCA, then a clinical diagnosis can be made immediately; however, some tests can be helpful. Figure 2 illustrates the general approach to diagnose GCA. Diagnosis of GCA requires a stepwise process, including review of symptoms, blood testing, and imaging studies. 5 The most important blood tests are measures of inflammation: the erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP). These inflammatory markers are often elevated with GCA, but they do not rule out GCA if normal, and they can be elevated due to a number of other conditions. Other less important blood tests, such as serum alkaline phosphatase and leukocyte counts, can also help determine the diagnosis.
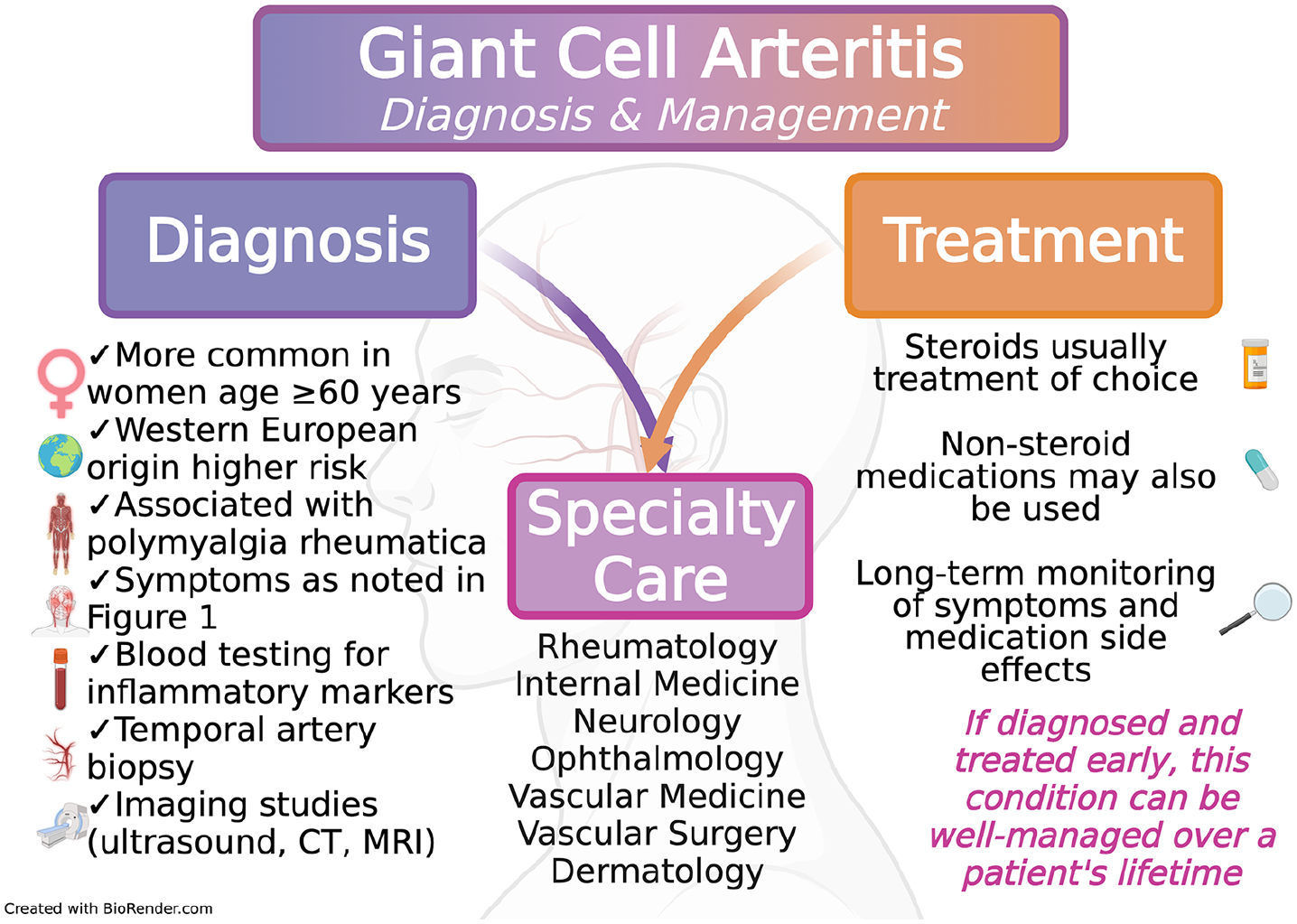
Overview of diagnosis and management of giant cell arteritis.
For diagnosis, some clinicians rely on the American College of Rheumatology, which sets five criteria for diagnosis: age ⩾ 50 years; localized pain in the head; temporal artery abnormality; ESR ⩾ 50 mm/h; and abnormal artery biopsy. Three of five criteria are needed to make the diagnosis. 6
A temporal artery biopsy or temporal artery color Doppler ultrasound can help to confirm a suspected diagnosis of GCA. The temporal artery will appear thickened and fragmented on biopsy approximately 85% of the time in patients with GCA. In some cases, a normal biopsy result on one side of the head may require another biopsy on the other side because GCA may not affect all parts of the artery. On ultrasound, a ‘halo’ sign can be seen in the temporal artery, which indicates thickening or swelling of the blood vessel wall.
In some cases, GCA may affect larger vessels such as the aorta and its major branches rather than (or in addition to) the temporal artery.7,8 Large blood vessel involvement does not always cause symptoms initially; therefore, advanced imaging is needed to help exclude it. The larger vessels may be imaged by computed tomography (CT), CT angiography (CTA), magnetic resonance imaging (MRI), MR angiography (MRA), positron emission tomography (PET), and/or PET with CT.
How is GCA related to PMR?
GCA and PMR are both inflammatory disorders with an unknown trigger. 3 The symptoms of PMR, which include morning stiffness at the shoulders, neck, torso, and hips, distinguish it from those of GCA. However, in patients with GCA, PMR occurs 40–50% of the time. 8 Conversely, in patients with PMR, GCA occurs 10% of the time. Though symptoms of the two conditions may or may not occur at the same time, the initial treatment is the same (i.e., steroids), as noted below.
What other conditions have symptoms similar to GCA?
Some of the symptoms of GCA can seem like a viral or flu-like illness. Other vascular conditions can have symptoms similar to those of GCA, but they are all less common than GCA and can be distinguished with proper testing. For example, other types of vasculitis (Takayasu arteritis, medium-vessel vasculitis, and small-vessel vasculitis) are inflammatory conditions that may cause symptoms of fever, fatigue, and anemia, just like GCA. Similarly, they may also have elevated inflammatory biomarkers including ESR and CRP. However, Takayasu arteritis generally occurs in a younger age group (< 50 years). Medium-vessel and small-vessel vasculitides, including microscopic polyangiitis, granulomatosis with polyangiitis, and polyarteritis nodosa, rarely affect the temporal artery. Stroke is another diagnosis that must be considered in patients with GCA symptoms, especially when vision changes occur. Trigeminal neuralgia and temporomandibular joint (TMJ) disorders may also cause pain in the face and jaw, and these are other diagnoses to consider in making the diagnosis of GCA.
How is GCA treated?
To reduce inflammation in GCA, steroids are the first-line treatment and are generally given even
Other antiinflammatory medications such as methotrexate, azathioprine, or cyclophosphamide may also be prescribed. These steroid-sparing therapies are used in patients who have GCA that does not improve with steroids, in relapsing–remitting GCA, or when side effects from steroids are a concern. Relapsing–remitting GCA occurs when symptoms of GCA return during tapering of steroids. This can be common within the first year after diagnosis, and headache is the most common symptom.
The newest medications used to treat GCA are called biological therapies (or ‘biologics’). One biologic medication, tocilizumab, targets a small protein in the blood called interleukin-6 (IL-6) to reduce inflammation. 9 Recently, it has been shown that combining tocilizumab with steroids may be a better treatment regimen compared with taking steroids alone. New biologic therapies that target specific inflammatory pathways are an active area of research and development in ongoing clinical trials (see ClinicalTrials.gov).
What are the side effects of treatment for GCA?
Once a patient starts steroids, regular monitoring is required to identify treatment side effects, the severity of which is typically increased with higher doses and longer durations of therapy. Common side effects of steroid medications include weight gain, increased blood sugar, high blood pressure, changes in mood, muscle weakness, blurred vision, glaucoma (increased pressure in the eyes), increased body hair, and easy bruising. Serious side effects include life-threatening infection and early cataracts, which require immediate re-evaluation of treatment. Long-term side effects of steroid use can also include an increased risk of cardiovascular events (such as heart attack or stroke). 8
In patients who are at higher risk for problems with steroids, such as those with osteoporosis or diabetes, side effects must be closely monitored. Steroids can affect bone density and lead to osteoporosis and compression fractures. On the other hand, steroid-sparing therapies, including tocilizumab, typically have fewer side effects but can compromise the immune response, increasing risk of infection.
What are the long-term problems after diagnosis of GCA?
In addition to the potential side effects of the treatment itself, ongoing inflammation in GCA leads to increased cardiovascular risk. Inflammation plays a role in the development of atherosclerosis, which is a condition that leads to plaque build-up within the arteries. Ongoing inflammation, such as that seen with vasculitis or other conditions like rheumatoid arthritis, accelerates the development and progression of plaque in the arteries. Other potential long-term problems associated with GCA include thoracic aortic aneurysm (i.e., bulging of the aorta) with associated risk of aortic valve regurgitation (leakage), heart failure, and aortic dissection (tear of the aorta). Periodic vascular imaging to assess for vessel stability can be helpful to monitor for complications.
What can be done to prevent blood vessel problems?
Maintaining a healthy lifestyle is vital to blood vessel health. Following a healthy diet, avoiding tobacco, and getting regular exercise are important in preventing the progression of blood vessel problems. These interventions can also help to reduce blood vessel inflammation. Exercise is particularly beneficial, and all adults should aim to achieve at least 30 minutes per day of moderate-intensity exercise. 10 For more information on the benefits of exercise, please see the Patient Information Page on ‘Exercise as medicine’. 11 Maintaining adequate control of blood pressure, blood sugar, and cholesterol are also important strategies to improve cardiovascular health and prevent blood vessel problems, especially among those on steroids for management of GCA.
Summary
GCA, or temporal arteritis, is an inflammatory condition (vasculitis) of the large blood vessels that is more common in older patients. Symptoms of GCA may include fever, headache, pain over the temples, vision changes, or jaw pain with chewing. Treatment should start immediately to prevent blindness, even without confirmation of the diagnosis by imaging or biopsy of the temporal artery. Blood tests are helpful to identify and monitor inflammation. High-dose steroids are considered first-line treatment of GCA with eventual slow tapering of the steroid dose as symptoms and inflammatory markers improve. Other medications can be added or substituted to prevent the side effects of steroids. With appropriate treatment and long-term monitoring, most patients improve and experience good control of symptoms.
Footnotes
Declaration of conflicting interests
Dr Elizabeth Ratchford reports that her spouse is employed by Genentech, Inc. (manufacturer of tocilizumab). The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Dr Elizabeth Ratchford’s work was supported in part by the generosity of David Kotick (1926–2021). Dr Alexandra Solomon was an Anticoagulation Forum Ansell Fellow at Boston Medical Center. Boston Medical Center received an Anticoagulation Forum Ansell Fellowship Award, which was supported by an unrestricted educational grant from Janssen Biotech, Inc., administered by Janssen Scientific Affairs, LLC.
