Abstract

Keywords
The lipid hypothesis proposes that lowering serum cholesterol can prevent major adverse cardiovascular events (MACE) and is supported by evidence from randomized controlled trials (RCTs). The magnitude of clinical benefit associated with lipid lowering appears to be proportional to the absolute reduction in low-density lipoprotein cholesterol (LDL-C) concentrations. 1 Contemporary clinical practice guidelines recommend LDL-C lowering with statins as first-line therapy in patients with or at risk for clinically evident atherosclerotic cardiovascular disease. 2 Treatment to goal LDL-C concentrations below conventional targets (< 70–100 mg/dL) may be beneficial. Although Mendelian randomization studies demonstrate that lifelong modest reductions of LDL-C are associated with robust reduction in MACE, current guidelines provide limited recommendations for the initiation of pharmacology to lower LDL-C in adults < 40 years of age.2,3 We hypothesize that the clinical benefit of lipid-lowering therapy is related to both the absolute reduction in LDL-C and the duration of time over which LDL-C is reduced. The objective of the present analysis was to evaluate the relationship between the clinical benefit of LDL-C lowering with the magnitude of LDL-C reduction and the duration of time LDL-C was reduced.
Cardiovascular outcome RCTs of guideline-recommended LDL-C-lowering therapies (including statins, ezetimibe, and PCSK9 inhibitors), with > 1000 participants and > 2-year follow-up, were identified as described by the Cholesterol Treatment Trialists’ Collaboration in accordance with PRISMA guidelines (see online Supplemental Material).1,2 Intervention and comparator therapies, baseline and postintervention LDL-C levels, and median length of follow-up were extracted. The cross product of LDL-C reduction and the duration of follow-up were calculated for each trial. Clinical outcomes from each trial were selected to most closely approximate the composite endpoint of cardiovascular death, myocardial infarction or other acute coronary syndrome, revascularization, and stroke (see online Supplemental Material). 1 Sensitivity analyses were performed in secondary prevention trials, trials with greater than 3-year follow-up, and after excluding trials that exclusively enrolled participants with heart failure or end-stage renal disease on dialysis, since US and European guidelines do not recommend lipid-lowering treatment in the absence of another indication for therapy. 2 Statistical analyses were performed with Stata (StataCorp LLC, College Station, TX, USA) and R (R Foundation for Statistical Computing, Vienna, Austria) using the metareg package. Random effects meta-regression analysis was carried out to assess the impact of trial-specific LDL-C lowering and duration of therapy on the effect size for MACE. A two-tailed p-value less than 0.05 was considered statistically significant.
A total of 33 RCTs enrolling 249,887 participants with a median follow-up of 48 months (IQR: 38–60) were included in the analysis. Thirty-one trials met criteria for low risk of bias per Cochrane’s assessment tool. 1 These trials tested statins (n = 29), ezetimibe (n = 2), and PCSK9 inhibitors (n = 2). Eight trials evaluated lipid-lowering therapy for primary prevention; the remainder evaluated lipid lowering for secondary prevention. The cross product of LDL-C reduction and the duration of therapy correlated with the relative risk reduction of MACE in meta-regression analyses (r2 = 0.66; p < 0.001; Figure 1A). This association was most robust in secondary prevention trials (r2 = 0.79; p < 0.001) and in trials with greater than 3-year follow-up (r2 = 0.82; p < 0.001). In a sensitivity analysis that excluded four trials focused on patients with heart failure or renal failure requiring dialysis, the cross-product of LDL-C reduction and time was associated with reductions in MACE (r2 = 0.77; p < 0.001). In contrast, the association between absolute LDL-C lowering and MACE, without the dimension of time, was less robust in meta-regression analyses (r2 = 0.31; p = 0.014; Figure 1B).
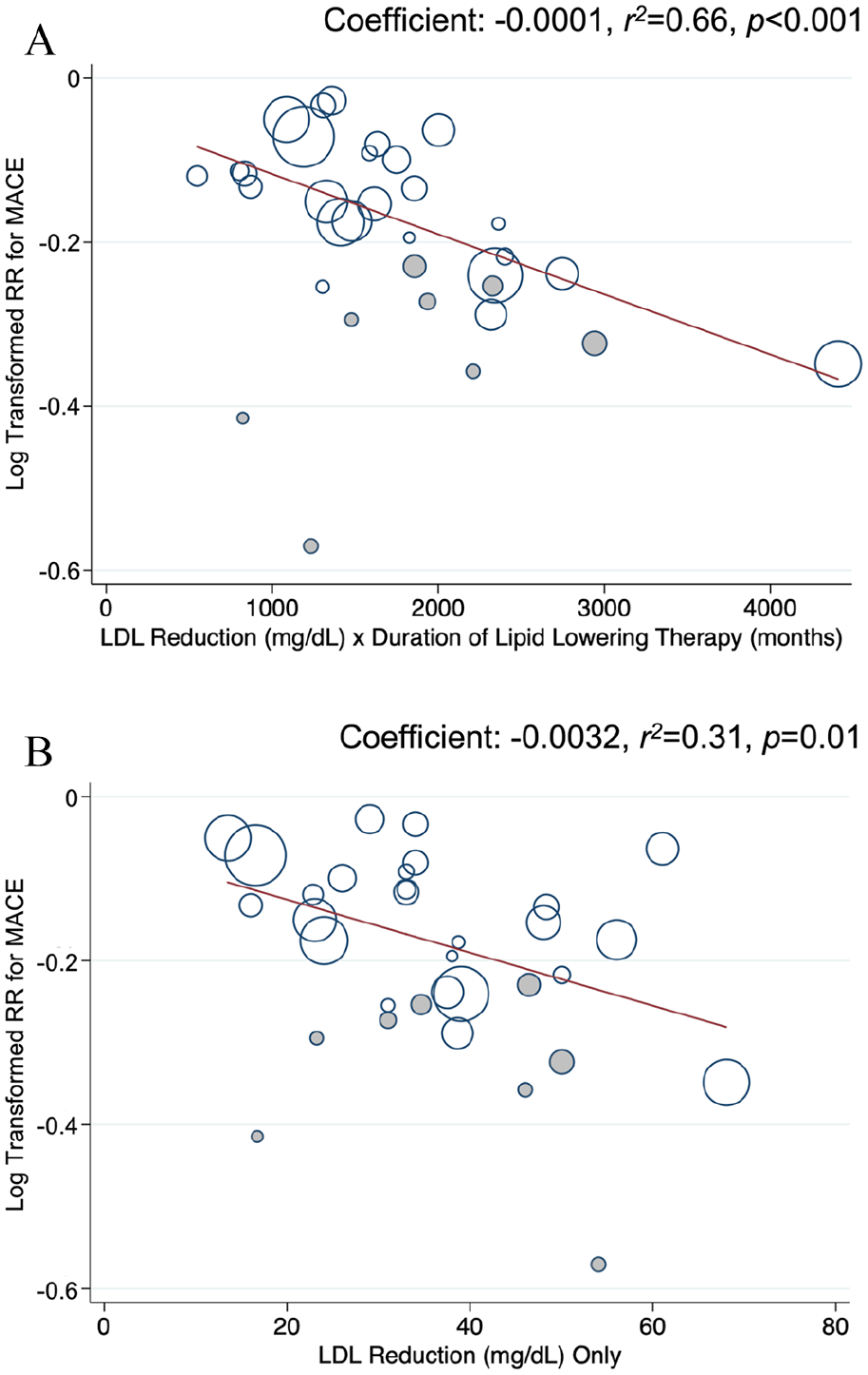
The clinical benefit of lipid lowering is clear. These data suggest a stronger association between the cross product of absolute LDL-C reduction and duration of therapy and the relative risk of MACE compared with absolute LDL-C lowering alone. Our findings support the notion that cardiovascular risks are related to exposure of elevated LDL-C over time, and that intensity and duration of LDL-C lowering affect clinical outcomes. The median study duration was 4 years; extended follow-up might demonstrate even more robust associations. Based on the current findings, prolonged duration of lipid lowering would confer greater clinical benefits. This is supported by Mendelian randomization studies which show that prolonged exposure to lower LDL-C beginning early in life is associated with a threefold greater reduction in the risk of MACE for each unit lower LDL-C than treatment with an LDL-C lowering agent started later in life. 3 These observations together may support a potential benefit of early initiation of intensive lipid-lowering therapy in patients at risk for clinical events associated with atherosclerotic cardiovascular disease.4,5
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221098459 – Supplemental material for Systematic review and meta-regression on the duration of LDL-C lowering and major adverse cardiovascular events
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221098459 for Systematic review and meta-regression on the duration of LDL-C lowering and major adverse cardiovascular events by Gabriel Redel-Traub, Nathaniel R Smilowitz, Yuhe Xia and Jeffrey S Berger in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
