Abstract
Spectral Doppler and plethysmographic waveforms vary significantly in the presence of circulatory assist devices. Understanding the effect these devices have on the waveforms and flow characteristics is necessary for the appropriate interpretation of duplex ultrasound and other noninvasive vascular studies. We review the different arterial waveforms that can be seen with circulatory assist devices.
Keywords
Introduction
Circulatory assist devices such as left ventricular assist devices (LVADs), intra-aortic balloon pump (IABP), and extracorporeal membrane oxygenation (ECMO) are used in cardiac or pulmonary failure refractory to medical management.
Duplex ultrasound is frequently used in critically ill patients treated with these devices to evaluate for vascular access site complications, vascular thrombosis, and distal limb malperfusion. The effect of the assist devices on hemodynamics produces sonographic flow patterns, which are important to recognize for accurate interpretation of the vascular studies.
In this pictorial review, we discuss normal Doppler and pulse volume recording waveforms, and changes observed in the presence of circulatory assist devices.
Normal peripheral arterial waveforms
In a resting upper or lower extremity, spectral Doppler waveforms are typically multiphasic in nature (Figure 1). The first phase is characterized by a brisk systolic upstroke, which is followed by the second phase – reversal of flow in early diastole. The final phase is a low, or absent, end-diastolic flow in an upward deflection, reflecting forward flow due to elastic recoil of the vessel wall. 1
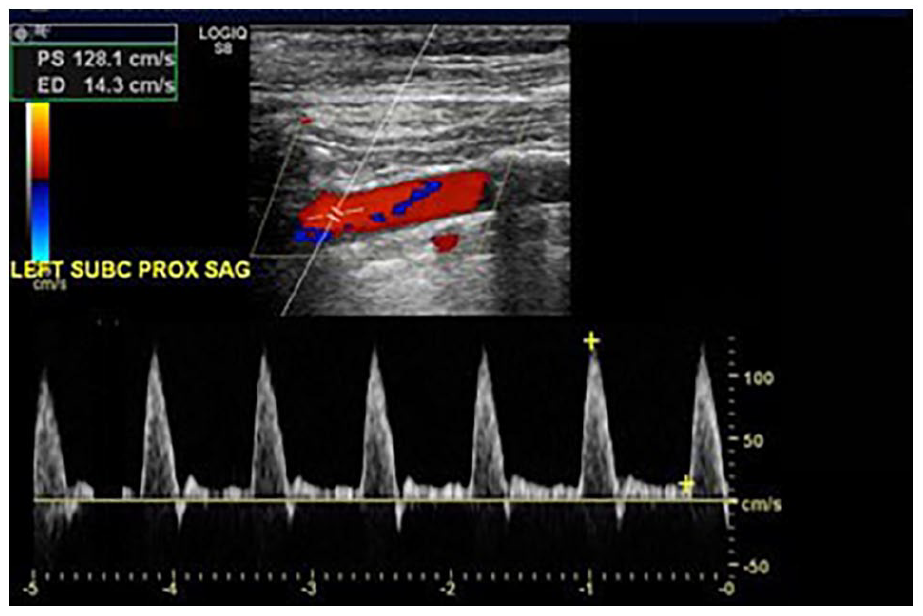
Normal peripheral arterial spectral Doppler waveform.
Normal carotid artery spectral Doppler waveforms
The spectral Doppler waveforms of the carotid arteries are reflective of the vascular bed being supplied. 1 Common carotid artery (CCA) spectral Doppler waveform has characteristics of the waveforms seen in the internal carotid (ICA) and external carotid arteries (Figure 2A). Owing to a predominance of CCA flow through the ICA, the waveform in CCA is mostly that of a low-resistive pattern, which is characterized by forward flow throughout diastole.
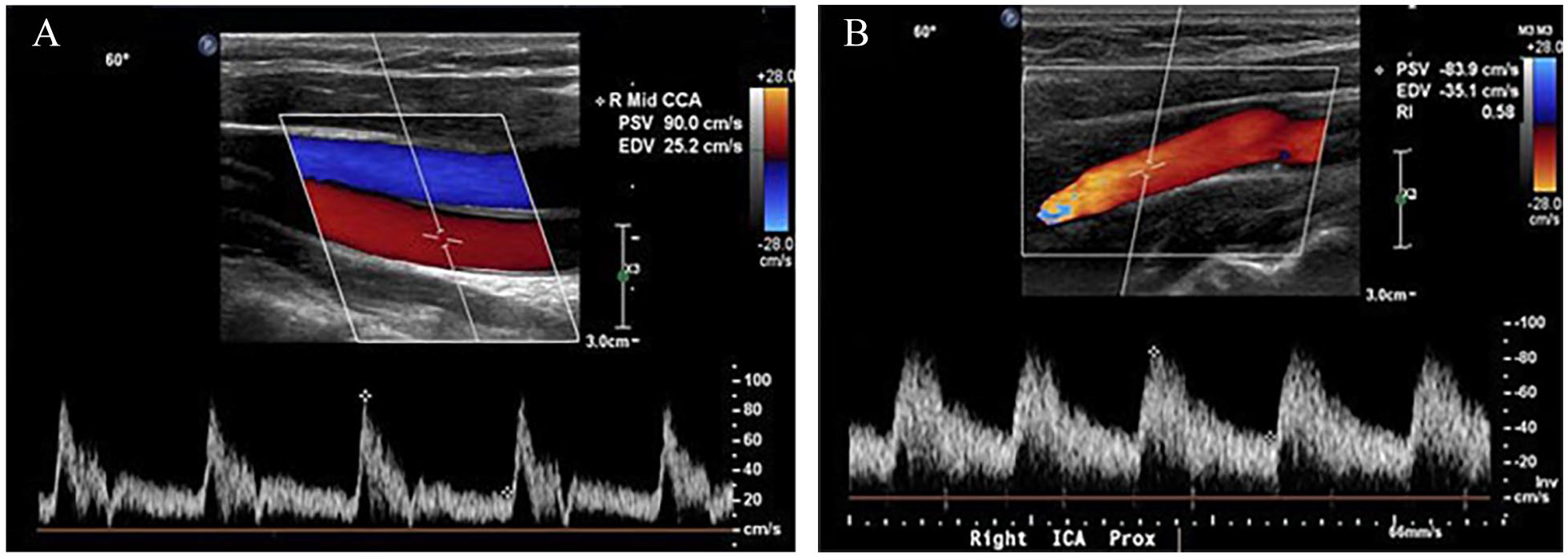
ICA spectral Doppler waveform has a monophasic low-resistive flow pattern. It is characterized by a sharp upstroke during systole followed by a continuous antegrade flow throughout diastole (Figure 2B).
Normal peripheral arterial physiologic study
A normal plethysmographic waveform has a sharp systolic rise, a narrow peak, and a dicrotic notch during downslope. 2 An ankle–brachial index (ABI) ⩾ 1.0 is considered normal. An ABI of ⩽ 0.9 is diagnostic of peripheral artery disease (PAD), whereas an ABI of 0.91–0.99 is considered borderline. In cases of noncompressible vessels and an ABI of > 1.40, the use of a toe–brachial index is recommended. A value of < 0.7 is abnormal and diagnostic of PAD. Figure 3 shows an example of a normal pulse volume recording with no segmental pressure gradients and bilaterally normal ankle–brachial indices.
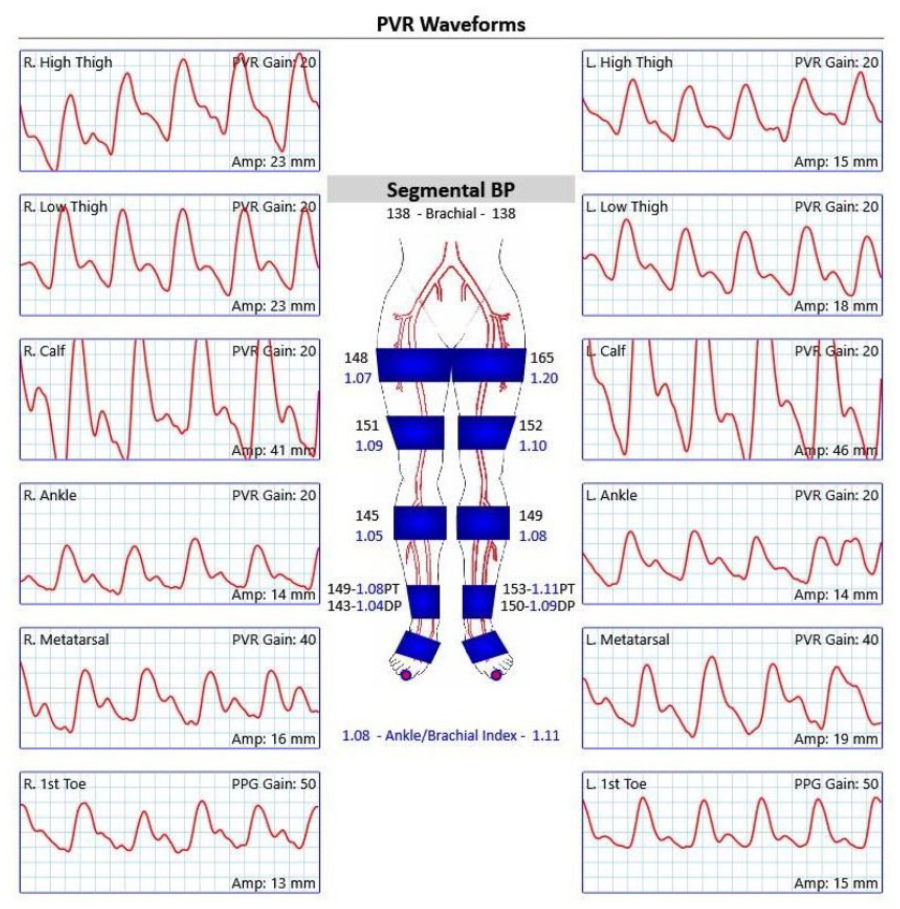
Normal PVR and ankle–brachial indices.
Intra-aortic balloon pump
IABP is a device used to increase cardiac output in cases of cardiogenic shock, and/or as a support device in patients at risk for hemodynamic compensation during high-risk coronary interventions. 3 The device consists of a helium-filled balloon that is inserted into the aorta, and it operates by deflating during systole and inflating during diastole.
Typical spectral Doppler waveform in the setting of an IABP at 1:1 setting demonstrates two separate systolic peaks. The first peak corresponds to the unassisted systole and the second peak corresponds to the inflation of the balloon during diastole. The second peak is followed by the reversal of flow due to the deflation of the balloon (Figures 4A and 4B). Plethysmographic waveform in a patient undergoing treatment with IABP retains the characteristic two peaks in 1:1 setting (Figure 4C).
The level of assistance provided by an IABP can be adjusted to assist every cardiac cycle (1:1 setting) as in Figures 4A and 4B, or less frequently (1:2 setting) as in Figure 4D. Changing the level of assistance will change the frequency of the augmented systolic peak in relation to the unassisted beat. Velocities cannot be measured accurately unless the IABP is turned off. If this is not possible, the peak systolic velocity is measured from the unassisted peak as in Figures 4A and 4B.
Left ventricular assist device
A LVAD is a device used in cases of end-stage heart failure. The device consists of a pump that has its inlet in the apex of the left ventricle and its output in the ascending or descending aorta. 4 The device works by continuously pumping blood from its inlet to the outlet, augmenting cardiac outflow.
Contemporary LVADs are continuous flow (CF) devices with either axial (e.g., HeartMate II, Abbott, Abbott Park, IL, USA; Impella, ABIOMED, Danvers, MA, USA) or centrifugal pump (e.g., HeartMate III, Abbott) designs. In CF-LVADs, blood flow is inversely related to the pressure differential between the left ventricle and aorta. 5 Centrifugal pumps are more sensitive to changes in preload and afterload; consequently, for the same changes in pressure, centrifugal pumps generate greater changes in flow and this results in greater pulsatility to waveforms. Therefore, LVADs with centrifugal pump designs produce a more pulsatile waveform relative to LVADs with axial pump designs.5,6 In addition to pump design, the systolic pulsatility of the Doppler waveforms of LVAD patients depends on the device settings, and the patient’s myocardial reserve. 6 Patients with poor myocardial reserve will have a decreased pulsatility as compared to patients with a preserved myocardial reserve. Furthermore, the HeartMate III can be programmed using pump speed modulation to generate more pulsatility from the LVAD and to intermittently increase flow from the left ventricle during systole and diastole. 5
Doppler waveform in the patients with a poor myocardial reserve treated with a CF-LVAD with an axial pump design demonstrates a monophasic and low-resistive waveform with a prolonged systolic upstroke and minimal pulsatility, as in Figure 5A. Monophasic and low-resistive spectral Doppler waveforms with preserved pulsatility is seen in patients with a CF device with a centrifugal pump design, as in Figure 5B.
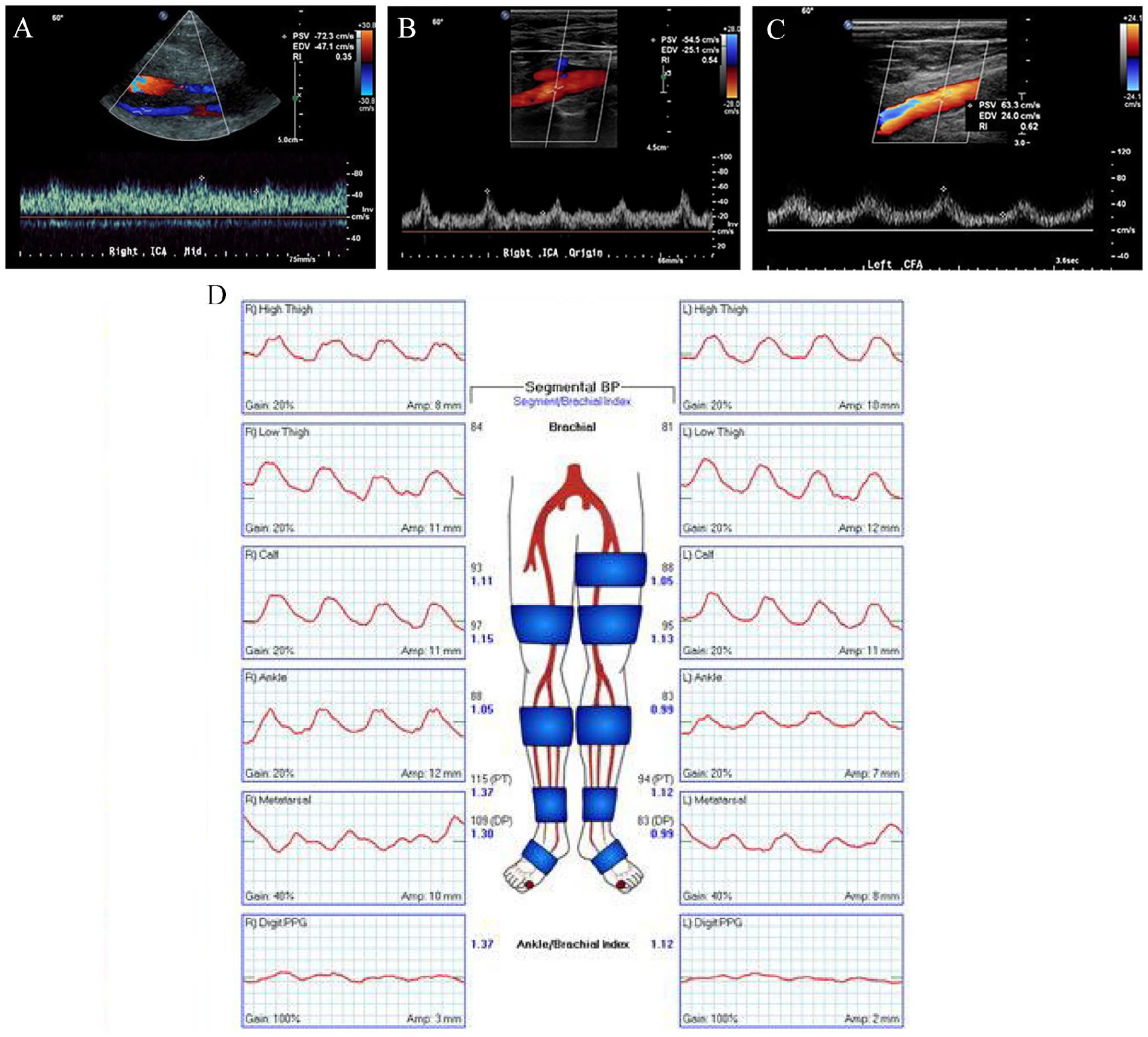
Peripheral arterial spectral Doppler waveform demonstrates a monophasic, low-resistive pattern in the presence of an LVAD (Figure 5C). LVAD-associated qualitative and quantitative hemodynamic changes limit accuracy of the duplex ultrasound for the estimation of stenosis.7,8 Further investigation on the use of additional parameters such as velocity ratios and plaque characterization is needed in such cases.
Plethysmographic waveforms in a patient with LVAD are dampened throughout with loss of dicrotic notch bilaterally (Figure 5D). Morphology of the plethysmographic waveforms may be variable, dependent on the LVAD pump settings. The localization and severity of the disease cannot be accurately determined based on the waveforms in the presence of the LVAD.
Extracorporeal membrane oxygenation
ECMO is a method of oxygenating the blood outside the body. There are two types of ECMO – veno-venous (VV) and veno-arterial (VA). VA ECMO runs parallel to circulation, where blood is obtained from the right atrium or vena cava and reintroduced through peripheral cannulations or central cannulation to the ascending aorta. 9
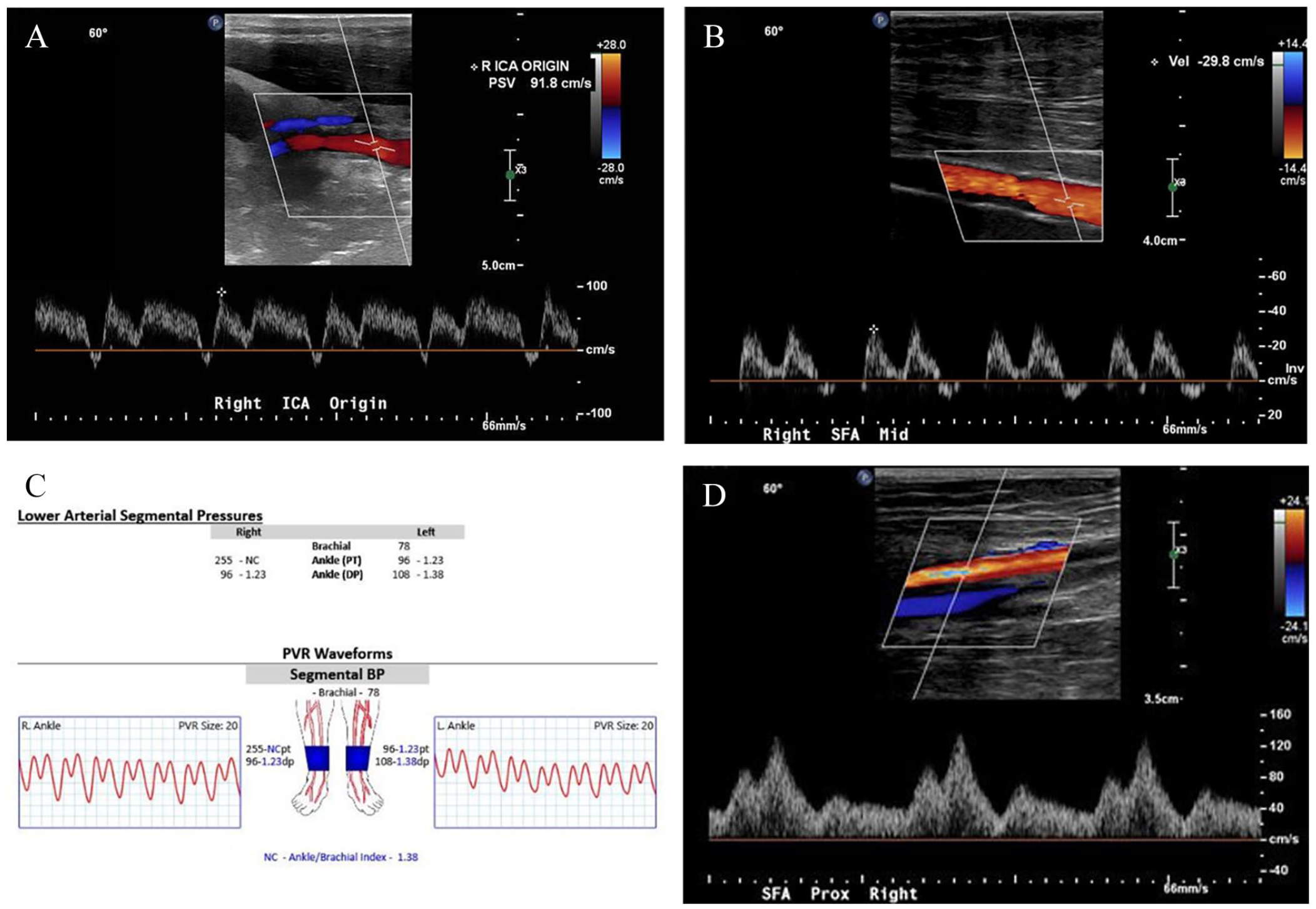
Doppler waveform in the presence of ECMO demonstrates a high-flow, low-resistive pattern with variable systolic pulsatility, depending on the myocardial reserve of a patient and degree of ECMO support. 6 Monophasic low-resistive spectral Doppler waveform with diminished systolic pulsatility suggests poor myocardial reserve but may also be seen with increased VA ECMO support (Figure 6A). High-flow, monophasic, low-resistive spectral Doppler waveform with systolic pulsatility suggests preserved myocardial reserve (Figure 6B).
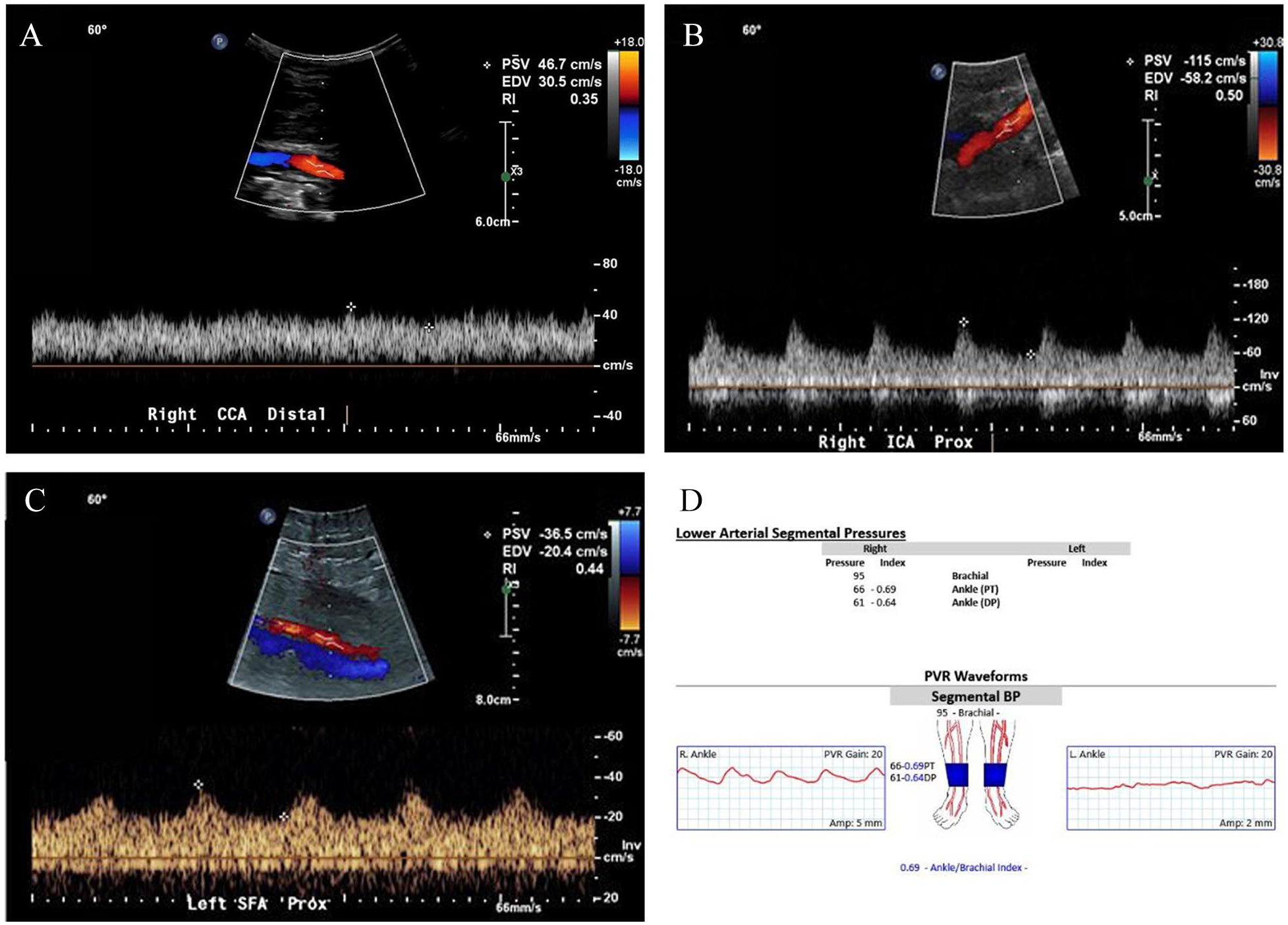
Peripheral arterial spectral Doppler waveforms obtained in the presence of ECMO show monophasic, low-resistive pattern and delayed systolic upstroke (Figure 6C). Change in the waveform morphology and limited visualization due to presence of cannulas present challenges in evaluation of stenosis in patients treated with ECMO. There are no established criteria to accurately assess stenosis in the presence of LVAD or ECMO devices. Further research into the role of additional hemodynamic parameters is necessary.
Vascular ultrasound has a role in the diagnosis of complications caused by circulatory devices. Severely dampened left ankle plethysmographic waveforms and nonaudible pedal Doppler signals were demonstrated in a patient with VA ECMO with an arterial cannula in the left common femoral artery, which indicate severely impaired limb perfusion (Figure 6D).
Conclusion
Presence of the cardiac assist devices has a distinct effect on the morphology of the spectral Doppler and plethysmographic waveforms, flow velocities, and pulsatility. Recognition of these changes and limitations of the diagnostic criteria in the presence of mechanical assist devices are essential for the accurate interpretation of the arterial studies.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
