Abstract
Introduction:
Venous ulceration is a multifactorial disease, and whether hyperhomocysteinemia (HHcy) promotes deterioration from primary varicose veins to venous ulcers remains unproven.
Methods:
This study retrospectively analyzed clinical data from 717 patients to investigate the potential correlation between HHcy and primary varicose veins ulcer formation, including 611 patients without ulcers (control group) and 106 with ulcers (case group).
Results:
In this study, 46.2% (49/106) of patients in the case group and 17.5% (107/611, p < 0.001) in the control group suffered from HHcy. Multivariate logistic analysis revealed that HHcy was closely associated with the incidence of venous ulceration in patients with primary varicose veins (p < 0.001). Propensity score matching created 101 matched pairs of patients with and without ulcers, and the analysis pointed to a potential link between HHcy and ulcer formation in the context of primary varicose veins (p < 0.001). Additional experiments showed that HHcy could induce endothelial dysfunction and phenotypic switching of vascular smooth muscle cells.
Conclusion:
Both clinical and experimental findings implicated HHcy as a key factor in the development of venous ulceration. Further research is needed to appraise the effectiveness of HHcy-lowering therapy in the prevention of venous ulcers in patients with varicose veins.
Background
Varicose veins are the most common manifestation of chronic venous diseases (CVDs). Although frequently considered a cosmetic issue, the presence of varicose veins has been closely related to venous thromboembolism and skin complications. 1 The Clinical-Etiology-Anatomy-Pathophysiology (CEAP) is an internationally accepted classification system of CVDs, with the clinical (C) class ranging from several types of varicosities (C1–2) to swelling (C3) and skin lesions (C4–6). 2
Venous hypertension and valve insufficiency were postulated to be the main risk factors for varicose veins; however, the natural history of CVDs remains elusive. 3 The Edinburgh Vein Study investigated the natural history of CVD and found that the varicose veins in nearly one-third of subjects deteriorated into skin lesions. 4 Venous ulcer is the most serious skin complication, with a complex etiology, long duration, and recurrent attacks. 5 Severe ulceration may lead to different degrees of disease and disability, seriously affecting the quality of life. 6 For patients aged over 65, nearly 4% in the United States and 3% in the UK suffer from active venous ulcers.3,7
Since not all patients with varicose veins develop venous ulcers, there might be some unknown risk factors implicated in the progression of CVDs. Homocysteine (Hcy) is an important intermediate product of the methionine cycle. Elevated Hcy concentrations are associated with multiple age-related diseases, such as cardiovascular diseases.8,9 Upcoming results from clinical trials of the effects of hyperhomocysteinemia (HHcy)-lowering treatment on cardiovascular diseases should contribute to more information about the relevance of Hcy levels to cardiovascular risks; however, the efficacy of HHcy-lowering interventions remains contested. 10 A previous study evaluated the efficacy of folic acid therapy on ulcer healing, and the results showed that HHcy-lowering treatment seems to expedite ulcer healing. 11 Although it was a small study, its findings suggest a potential link between HHcy and ulcer formation. Furthermore, the current literature has implicated Hcy in inducing cell dysfunction and inflammation, 12 key factors in ulcer formation. 13 However, whether elevated Hcy levels promote the deterioration of primary varicose veins to venous ulcers remains unproven. This study aimed to investigate the potential correlation between HHcy and ulcer formation and to explore its potential mechanisms.
Methods
This study was approved by the Ethics Committee of the Qingpu Branch of Zhongshan Hospital and conformed to the standards of the Declaration of Helsinki. A waiver of consent was granted due to the retrospective nature of this study.
Study population
Owing to the development of endovenous interventions, the treatment strategy for varicose veins has changed in recent years. Surgical indications for treatment include symptomatic superficial venous insufficiency, CEAP clinical class ⩾ 2, and duration of reversed venous flow exceeding 0.5 s. 14 From January 2017 to December 2020, 1204 patients hospitalized at the Division of Vascular Surgery, Qingpu Branch of Zhongshan Hospital with signs and symptoms of varicose veins (CEAP clinical class ⩾ 2) were enrolled. Patients with previous surgical intervention were not included.
Venous ulceration is a multifactorial disease. Accumulating evidence suggests that advanced age, higher body mass index (BMI), female sex, deep venous thrombosis, and multiple pregnancies are risk factors contributing to venous ulceration. 6 This study retrospectively collected demographic and clinical data from patients, including age, sex, duration, BMI, serum Hcy, medical history, CEAP clinical class, and venous clinical severity score (VCSS). CEAP clinical class and VCSS were used to evaluate the severity of CVDs. All patients received preoperative duplex ultrasonography to check the presence of reflux in deep and superficial veins and to exclude potential thrombosis. 15 The cut-off value of reflux in the superficial veins was defined as 500 ms. 14 The more severely affected limb was taken into consideration if patients had bilateral lesions. Patients meeting the following criteria were excluded: missing data, < 18 years of age, Klippel–Trenaunay syndrome, history of immune diseases, deep venous thrombosis, peripheral artery diseases (ankle–brachial index < 0.9), or iliac vein compression syndrome. The screening and diagnosis of iliac vein compression syndrome were as described previously. 16
Serum Homocysteine (Hcy)
Since Hcy is associated with multiple health risks (including atherosclerotic diseases and neurodegenerative processes), 10 Hcy measurement has been part of routine medical care. Blood samples were collected from patients after overnight fasting, and the serum was isolated and measured with an automated chemistry analyzer (Roche Group, Sweden). A serum concentration of Hcy > 15 μmol/L was defined as HHcy. 10
Cell culture
Human umbilical vein endothelial cells (HUVECs) and human umbilical vein smooth muscle cells (HUVSMCs) were purchased from CELL RESEARCH (Zhong Qiao Xin Zhou Biotechnology Co. Ltd, Shanghai, China). The cells were cultured in Dulbecco’s modified Eagle medium (KeyGEN Biotech Co. Ltd, Jiangsu, China) containing 10% fetal bovine serum. The cells were then incubated with Hcy (Sigma, St Louis, MO, USA).
Cell viability
Cell Counting Kit 8 (CCK-8) (Beyotime Biotech Co. Ltd, Haimen, China) was used to measure cell viability. Cells were seeded into 96-well plates (3 × 103/well). After administration of Hcy, CCK8 solution was added and incubated for another 2 h. Absorbance was determined at 450 nm wavelength by a microplate reader. Cell viability was calculated as: (ODassay – ODblank)/(ODcontrol – ODblank).
Cell migration rate
After the cells reached confluency greater than 90%, they were subjected to serum starvation. Then, a cell surface scratch test was performed with 200 µL pipette tips. The migration ability of cells treated with the different stimuli was determined as: (cell migration area)/(scratch area).
Cell apoptosis
An Annexin V-kFluor594 Apoptosis Detection Kit (KeyGEN Biotech Co. Ltd) was used to evaluate cell apoptosis. Cells were incubated with Annexin V-kFluor 594, and the apoptosis rate was then observed under a fluorescence microscope with an excitation wavelength of 590 nm. Flow cytometric analysis of apoptosis was also performed with a FITC Annexin V Apoptosis Detection Kit I (BD Biosciences, San Jose, CA, USA) to separately detect early apoptotic cells and late apoptotic or dead cells.
Western blotting
SDS–PAGE separation was performed on equivalent amounts of denatured protein samples, which were then transferred to polyvinylidene difluoride (PVDF) membranes and blocked in 5% bovine serum albumin (Sigma). The PVDF membranes were exposed to primary antibodies, followed by incubation with secondary antibodies.
Statistical analysis
Data from the clinical part of the study were processed with the statistical software IBM SPSS, Version 24.0 (IBM Corp., Armonk, NY, USA) using the χ2 test, Fisher’s exact test, or Student’s t-test. Univariate analyses were used to compare the risk factor levels between the control and case groups. Risk factors significantly associated with venous ulcers (p < 0.05) on univariate analyses were then included in the multivariate regression analysis. Propensity score-matching analysis was performed to create one-to-one matching pairs using the nearest neighbor matching method (IBM SPSS, Version 24.0). The significance of the differences was defined as p < 0.005 to reduce the false-positive rate.
Data from the experimental part were analyzed with one-way ANOVA for multiple statistical comparisons, followed by Tukey post hoc testing to compare the differences between groups. A statistically significant difference was taken as p < 0.05.
Results
Clinical characteristics
This study retrospectively collected 1204 patients with varicose veins, with 717 of these classified as having primary varicose veins. The patients with primary varicose veins were then divided into two groups based on their clinical manifestations: 611 patients without active ulcers (control group) and 106 patients with active ulcers (case group; online Supplementary Figure 1). The demographic data and clinical characteristics of the patients are described in Table 1.
Patient demographic and clinical data.
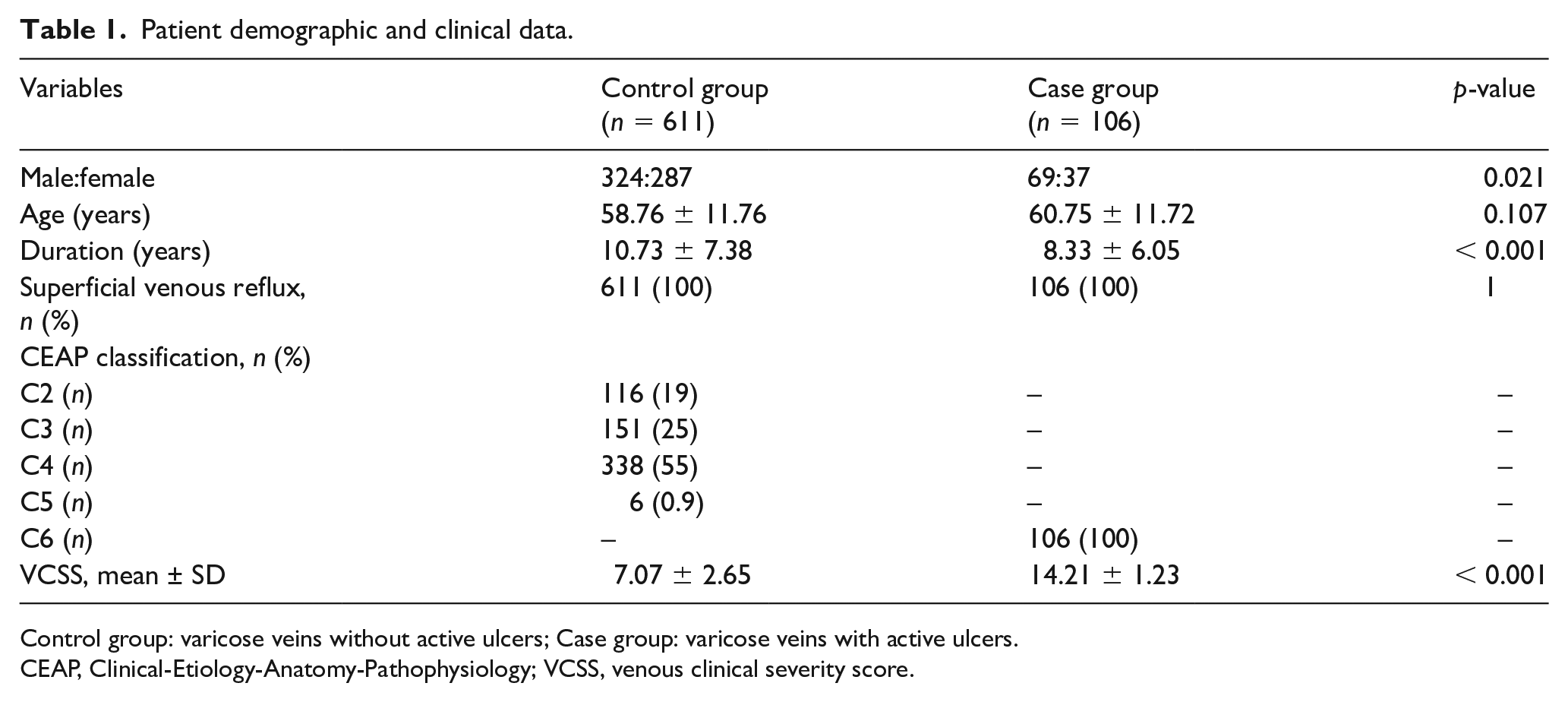
Control group: varicose veins without active ulcers; Case group: varicose veins with active ulcers.
CEAP, Clinical-Etiology-Anatomy-Pathophysiology; VCSS, venous clinical severity score.
Association of serum Hcy with venous ulceration
HHcy was found in 46.2% (49/106) of patients from the case group and 17.5% (107/611, p < 0.001) of patients from the control group. As described in online Supplementary Table 1, the univariate analysis demonstrated that diabetes, elevated serum Hcy and deep venous insufficiency were closely associated with venous ulceration. Multiple logistic regression analysis (Table 2) also demonstrated that diabetes and HHcy (OR 4.314; 95% CI 2.736–6.802, p < 0.001) were key risk factors. In addition, propensity score matching (Table 3) revealed that HHcy may serve as a potential predictor of venous leg ulcers (OR 2.906; 95% CI 1.591–5.305, p < 0.001).
Multiple logistic regression analysis.
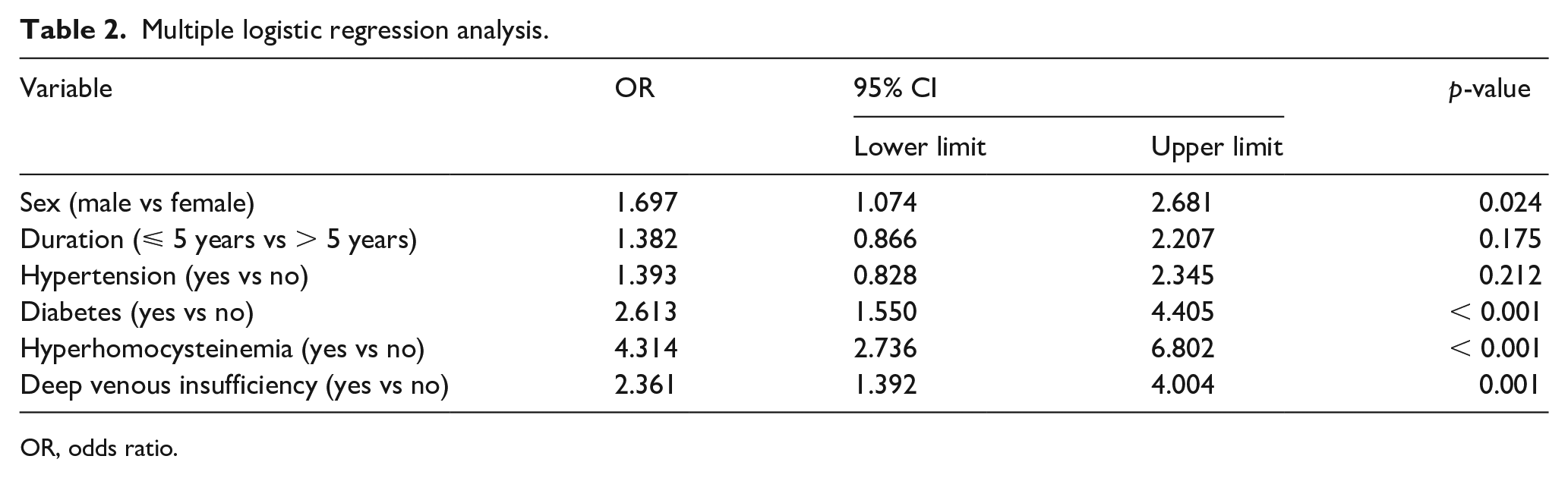
OR, odds ratio.
Propensity score matching.
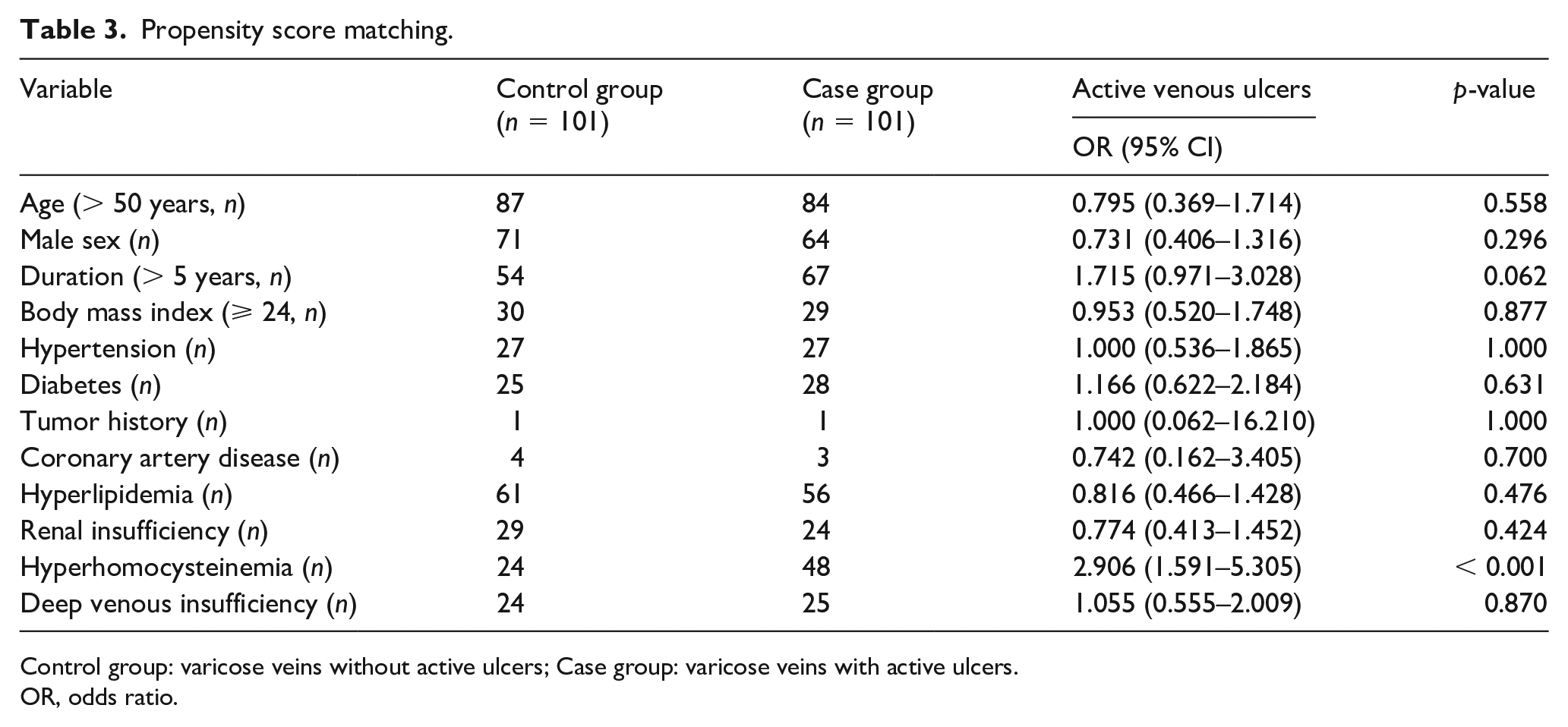
Control group: varicose veins without active ulcers; Case group: varicose veins with active ulcers.
OR, odds ratio.
Effect of Hcy on HUVECs
As shown in Figure 1A, cell viability decreased with increasing Hcy concentration after 24 hours of induction. Compared to the control group, the viability of HUVECs exposed to 1000 μmol/L Hcy decreased to 51.9% (p < 0.001). Based on this, we chose 500 μmol/L Hcy as a moderate cell treatment concentration for the following experiments. Similarly, CCK-8 assays revealed a trend of a gradual decrease in cell viability with prolonged Hcy (500 μmol/L) treatment duration (Figure 1B), as well as an increase in the total apoptotic cell percentage (Figure 1C and 1D). Likewise, flow cytometry showed an increase in the early apoptotic rate under HHcy treatment, accompanied by an increase in the late apoptotic or dead cell percentage (online Supplementary Figure 2). Western blot results also showed that the expression of proinflammatory cytokines (intercellular adhesion molecule 1 (ICAM-1), vascular cell adhesion molecule 1 (VCAM-1), phosphorylated-p65 (p-p65), and cyclooxygenase-2 (COX-2)) was markedly higher in the Hcy-treated HUVECs, accompanied by a decrease in the antiapoptotic protein Bcl-2 and an increase in the proapoptotic protein Bax (Figure 1E and 1F).
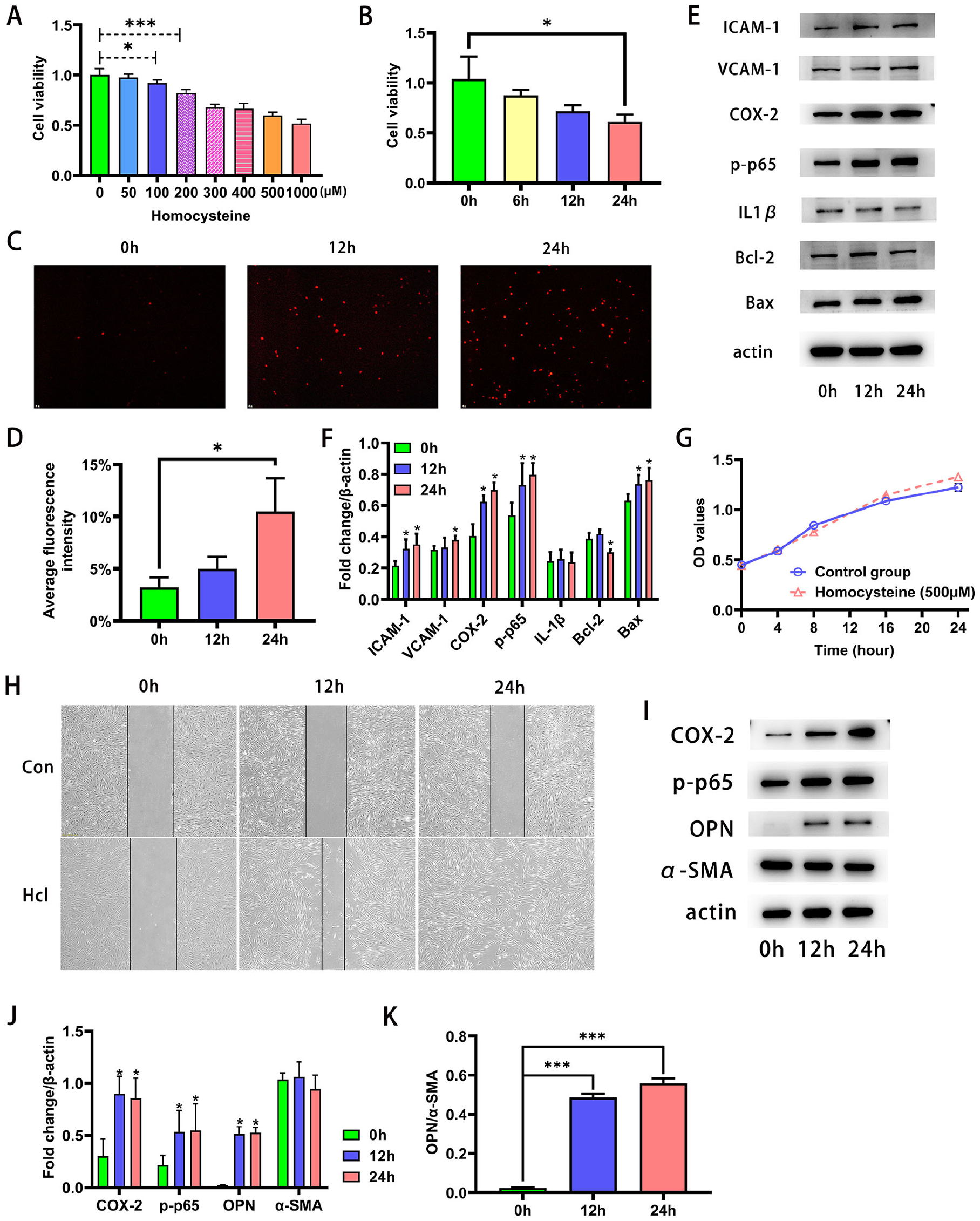
The adverse effects of Hcy.
Effect of Hcy on HUVSMCs
The viability of HUVSMCs incubated with an increased concentration of Hcy (from 0 to 1000 μmol/L) did not show significant changes. Then, the HUVSMCs were exposed to moderate treatment concentrations (500 μmol/L) to evaluate the effect of Hcy on the biological properties of proliferation and migration. Likewise, no significant changes were observed in cell proliferation between the two groups (Figure 1G). As shown in Figure 1H, however, HUVSMCs treated with Hcy displayed an augmentation in migratory capacity. In addition, western blotting analysis illustrated that exposure of HUVSMCs to Hcy promoted a significant increase in the protein expression of p-p65 and COX-2. However, we failed to detect the expression of interleukin (IL)-1β in the Hcy-treated group (Figure 1I and 1J). The western blot results demonstrated that HUVSMCs incubated with Hcy (500 μmol/L) induced an upregulation of OPN (secretory protein marker), and the ratio of OPN and α-SMA (contractile protein marker) increased with prolonged exposure duration (Figure 1I–K).
Discussion
This study retrospectively analyzed clinical data from patients with primary varicose veins, and we found higher concentrations of HHcy in patients with ulcers. Further cell experiments revealed that increased levels of HHcy were associated with cellular dysfunction. The main finding of our present research implied that HHcy was closely associated with the pathogenesis of venous ulceration.
Venous ulceration is the end result of a cascade of intricate cell events that are activated by hemodynamic abnormalities and amplified by genetic predisposition and other risk factors. 17 Inherited genetic susceptibility could cause perturbation of the venous system and affect the predisposition to venous disorders. In a large-scale vein study containing 2701 patients, genetic predisposition resulting in CVDs made up approximately 17%; however, no significant differences were found in genetic susceptibility among different clinical classes of CEAP. 18 Since not all patients with varicose veins would undergo progression to ulceration, there might be other factors involved in the development of skin complications. The role of Hcy in the pathogenesis of venous ulceration has gained increasing attention. 19 Several studies have revealed a positive correlation between serum Hcy and the severity of primary CVDs.20,21 Our study also identified HHcy as a significant risk factor for the development of venous ulceration. In the study population, a high BMI seemed to have no effect on developing ulcers. For patients aged over 50, however, the prevalence of BMI ⩾ 24 was 34.1% in the control group and 51.7% (p < 0.1) in the case group, suggesting that being overweight or obesity may play a greater role in the aging population.
The possible mechanisms of HHcy include nutritional deficiencies, genetic mutation, or secondary to other processes. 10 McCully first proposed that elevated Hcy levels might be associated with atherosclerosis. 22 Subsequently, multiple studies confirmed that a high level of Hcy adversely affects vascular structure and function. 23 However, the involvement of Hcy in the pathogenesis of venous disorders has rarely been reported. The transitional bridge linking hemodynamic abnormalities to ulcer development involves changes in microcirculation, among which microcirculation obstruction caused by thrombophilia is an important part. 24 Although the natural history of venous disorders is not completely understood, it is widely accepted that endothelial inflammation, thrombophilia and venous remodeling are implicated in the pathogenesis of venous ulceration. 6 Elevated Hcy is also clearly associated with an increase in thrombotic risk and cellular dysfunction, 11 which may play a role in the pathogenesis of venous ulceration.
Impaired endothelial cells (ECs) are critical in the progression of venous ulceration, characterized by enhanced proinflammatory activities and poor endothelium-dependent vasomotor activation when exposed to relaxing agents or stimulants of venous contraction. 13 During our study, treatment with Hcy led to a decrease in cell viability in a dose-dependent manner. In addition, incubation with Hcy promoted the expression of proinflammatory cytokines and induced cell apoptosis. These findings suggest that elevated Hcy levels may be responsible for endothelial dysfunction and may contribute to the progression of venous ulceration.
Our previous study revealed that phenotypic switching of vascular smooth muscle cells (VSMCs) from a contractile to a synthetic phenotype was the cytological basis for venous remodeling. 25 It has been previously described that Hcy could enhance VSMC proliferative and migratory capacities. 26 Our study failed to observe dramatic changes in cell proliferation; however, HUVSMCs exposed to Hcy displayed greater migration ability than normal controls. In addition, Hcy treatment induced an increased concentration of proinflammatory cytokines, accompanied by decreased contractile protein markers and increased secretory protein markers, indicating that HHcy may participate in the pathological process of venous ulceration.
Pathological alterations of venous ulceration involve varix formation, venous remodeling and cellular dysfunction,3,13 whereas ulcer healing depends on the rate of tissue remodeling, which involves fibroblast-mediated matrix contraction and keratinocyte epithelialization. 27 Previous studies have demonstrated dermal fibroblast dysfunction, abnormal responsiveness to growth factor stimulation, and fibroblast phenotypic alterations in ulceration-related skin damage. 13 Current investigations have also revealed that inflammatory conditions result in microcirculatory dysregulation and further promote the differentiation from secretory fibroblasts into myofibroblasts, affecting ulcer healing. 28 Chronic inflammation also contributes to leucocyte recruitment and extravasation into the dermis with the production of various proinflammatory cytokines. 13 In our study, we found that Hcy could induce cell dysfunction, accompanied by a significant upregulation of inflammation-related cytokines, including nuclear factor-κB (NF-κB, p65). It has been shown that inhibition of the NF-κB pathway alleviates Hcy-induced oxidative stress.29,30
Currently, the efficacy of HHcy-lowering therapy on cardiovascular events remains controversial. Prior randomized trials have failed to show a benefit from B-vitamin supplementation on cardiovascular events. 31 However, there is an anticipation that the effects of HHcy-lowering interventions might be dependent on the disease stage since most cardiovascular diseases develop slowly (typically taking more than 20 years from initial development to clinically associated events), whereas clinical trials of HHcy-lowering therapy on cardiovascular events are mostly based on exposure to HHcy for less than 5 years. 10 A meta-analysis on folic acid supplementation in stroke prevention demonstrated no significant effect in short-term studies but a greater beneficial effect in trials with a treatment duration of more than 36 months. 32 Therefore, HHcy-lowering therapy with a long duration may help prevent the deterioration from varicose veins into venous ulcers.
Study limitations
This study had several limitations. First, 102 patients were excluded due to incomplete data, which may underestimate the impact of other risk factors. Second, it is important to underline the mechanism of HHcy, as patients with genetic factors may have different cardiovascular outcomes than those with nutritional deficiency. During this study, we failed to collect relevant information about the mechanism of action of HHcy in ulcer formation. Third, another bias comes from limb infections, which may drive inflammation and affect Hcy levels. Finally, there may be some data bias in the experiment since Hcy of 500 μmol/L used in the cell experiments is higher than the common physiological levels of HHcy. These results, therefore, should be interpreted with caution.
Conclusion
This retrospective study revealed that HHcy was closely associated with the incidence of venous ulcers in patients with primary varicose veins and that exposure to HHcy contributed to cell functional disturbance. Further research is needed to appraise the effectiveness of HHcy-lowering therapy in the prevention of venous ulcers in the context of primary varicose veins.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221080486 – Supplemental material for Elevated serum homocysteine levels are associated with the development of chronic venous ulcers
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221080486 for Elevated serum homocysteine levels are associated with the development of chronic venous ulcers by Zhenyu Guo, Xu Li, Tao Wang, Xiaohu Yang and Longhua Fan in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X221080486 – Supplemental material for Elevated serum homocysteine levels are associated with the development of chronic venous ulcers
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X221080486 for Elevated serum homocysteine levels are associated with the development of chronic venous ulcers by Zhenyu Guo, Xu Li, Tao Wang, Xiaohu Yang and Longhua Fan in Vascular Medicine
Footnotes
Data availability statement
All data generated during this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Natural Science Foundation of Shanghai (grant 20ZR1411800).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
