Abstract
The Wells score had shown weak performance to determine pre-test probability of deep vein thrombosis (DVT) for inpatients. So, we evaluated the impact of thromboprophylaxis on the utility of the Wells score for risk stratification of inpatients with suspected DVT. This bicentric cross-sectional study from February 1, 2018 to January 31, 2019 included consecutive medical and surgical inpatients who underwent lower limb ultrasound study for suspected DVT. Wells score clinical predictors were assessed by both ordering and vascular physicians within 24 h after clinical suspicion of DVT. Primary outcome was the Wells score’s accuracy for pre-test risk stratification of suspected DVT, accounting for anticoagulation (AC) treatment (thromboprophylaxis for ⩾ 72 hours or long-term anticoagulation). We compared prevalence of proximal DVT among the low, moderate and high pre-test probability groups. The discrimination accuracy was defined as area under the receiver operating characteristics (ROC) curve. Of the 415 included patients, 30 (7.2%) had proximal DVT. Prevalence of proximal DVT was lower than expected in all pre-test probability groups. The prevalence in low, moderate and high pre-test probability groups was 0.0%, 3.1% and 8.2% (p = 0.22) and 1.7%, 4.2% and 25.8% (p < 0.001) for inpatients with or without AC, respectively. Area under ROC curves for discriminatory accuracy of the Wells score, for risk of proximal DVT with or without AC, was 0.72 and 0.88, respectively. The Wells score performed poorly for discrimination of risk for proximal DVT in hospitalized patients with AC but performed reasonably well among patients without AC; and showed low inter-rater reliability between physicians.
Keywords
Introduction
Venous thromboembolism (VTE) is a major global burden with a prevalence of approximately 10 million cases yearly, thereby representing the third leading vascular disease after myocardial disease and stroke. 1 Thrombo-embolism is a frequent and well-known complication in hospitalized patients. Several million inpatients are considered at high risk of deep vein thrombosis (DVT) every year worldwide with life-threatening complications such as pulmonary embolism. 2 Inpatients with hospital stays of 7 days or more have a 5.4% risk of presenting DVT during hospitalization, and a cumulative risk of almost 10% during the following 6 months. 3 As DVT presents a wide panel of nonspecific signs, lower extremity ultrasound study (LEUS) is essential to rule out DVT. To reduce multiple and repeated imaging, Wells et al. developed a 10-item score to assess the clinical pre-test probability of DVT. 4 The Wells score has been validated for outpatients and emergency patients for whom the clinical pre-test probability for DVT was established by a medical specialist of venous thromboembolism.5–8
Currently, the Wells score is used in tertiary and quaternary hospitals worldwide to help predict the pretest probability for DVT in inpatients. Yet hospitalized patients have very distinct risk profiles of DVT, with frequent use of thromboprophylaxis9,10 and high prevalence of many thromboembolic risk factors such as cancer, immobilization, recent major surgery, acute infection, and cardiac and/or respiratory insufficiency. 11 We also believe that DVT signs can be reduced in inpatients owing to less walking or bed rest and wearing compression stockings, which can improve venous return and reduces edema in comparison with a sitting or standing position. Thromboprophylaxis during hospitalization and after discharge is effective to prevent DVT 12 and inpatients receiving thromboprophylaxis develop DVT far less frequently than those without. 10
Some studies have suggested that the Wells score should not be applied to inpatients,13,14 whereas others have proved a strong performance of this score in predicting proximal DVT in hospitalized patients.15–17 To our knowledge, no study has assessed whether thromboprophylaxis influences the performance of the Wells probability score. Therefore, the aim of this study was to evaluate the impact of thromboprophylaxis (or full-dose anticoagulation) on Wells pre-test clinical score efficiency and validate its use in a real-life hospital context. Our secondary objective was to analyze inter-observer correlation of the Wells score assessed by ordering physician and by a study physician specialized in thromboembolism.
Methods
This study was a bicentric, cross-sectional study of inpatients with suspected symptomatic DVT, referred to the vascular medicine unit of two quaternary university hospitals (University Hospital of Nîmes, France and University Hospital of Caen-Normandy, France). The R-WITT study protocol (RCB 2017-A02316-47) received ethical review committee approval (Comité de Protection des Personnes: #180101) and was registered on clinicaltrials.gov (NCT03784937). Informed patient consent was required before enrollment.
Patient demographics
From February 1, 2018 to January 31, 2019, we included consecutive adult medical or surgical hospitalized patients, with suspected DVT, addressed to our unit for LEUS. Included patients had to be admitted to hospital at least 72 hours before clinical suspicion of DVT. Exclusion criteria were: prior diagnosis of DVT in the last 3 months, LEUS performed by a physician not participating in the study (private practice or junior resident), clinical suspicion of current pulmonary embolism, less than 18 years old, incomplete predictors of the Wells score by ordering physicians, and patients in whom death was imminent. For patients who underwent multiple LEUS during the recruiting period, only the first was considered in our study. LEUS had to be performed within 24 hours after clinical suspicion.
Measures
Prior to inclusion, ordering physicians had to evaluate the pre-test clinical probability of DVT of each patient with a modified Wells clinical score. Clinical items were recorded using a data capture screen into our computerized healthcare software. A response to each item was not required to complete the ordering process because the ethics committee considered it a potential harm in patient care, but patients with incomplete predictors of the Wells score by ordering physicians were excluded.
Upon admission to the vascular medicine unit for LEUS, one of the study physicians blindly evaluated the pre-test clinical probability of DVT with a modified Wells score, blinded to the result of the ordering physician, and before LEUS was performed. The Wells score pre-test probability of proximal DVT is a 10-item tool, with a total score ranging from −2 to 9, classifying the patient as low (−2 to 0), moderate (1 to 2) or high (> 2) probability.7,15 Patient clinical and treatment status was recorded, then LEUS was performed to identify DVT. Study practitioners followed the same protocol for LEUS with systematic exploration of the deep venous system from the inferior vena cava to the distal calf veins, on both lower extremities. A diagnosis of DVT was made if a noncompressible segment of deep vein was observed with a diameter under compression of 3 mm or more. For inferior vena cava and iliac veins, absence of color Doppler filling (with appropriate settings) for spontaneous and provoked venous flow (compression of the muscle compartments of the homolateral thigh) indicated venous thrombosis. Full compression of all the investigated veins was considered a normal test result. LEUS was classified as positive or negative for proximal DVT. Thrombosis of the popliteal vein and/or more proximal segments indicated proximal DVT. Distal DVT was explored during LEUS as recommended by the French Society of Vascular Medicine. Distal DVT was considered to be present if thrombosis of the calf veins alone was demonstrated. Cases where patients presented both proximal and distal DVT were classified as proximal.
Patients were split into two groups according to thromboprophylaxis. Group 1 was defined by the presence of effective thromboprophylaxis for at least 72 hours (n = 195), and Group 2 was defined by the absence, or presence for less than 72 hours, of effective thromboprophylaxis (n = 220). We considered that patients were under effective thromboprophylaxis in the following situations: (1) thromboprophylaxis treatment for at least 72 hours prior to DVT suspicion and defined as subcutaneous use of enoxaparin 40 mg/day or subcutaneous heparin sodium 5000 UI twice daily; (2) long-term, full-dose anticoagulation therapy either by direct oral anticoagulant therapy (DOAC) or by vitamin K anticoagulant (VKA) with a recent INR in the target 2.0–3.0 range, or by subcutaneous low molecular heparin therapy or parenteral unfractionated heparin at a dosage enabling effective and consistent anticoagulation. Henceforth, ‘thromboprophylaxis for at least 72 hours or long-term, full-dose anticoagulation’ is summarized as ‘thromboprophylaxis’. We used a 72-hour cut-off of thromboprophylaxis prior to DVT clinical suspicion to simulate diagnosis delay inherent to daily practice and in consideration that the VTE hazard peak occurs at day 8, with 1.05 events per 1000 person-days in medically ill hospitalized patients. 18 The choice of thromboprophylaxis was left to the attending physician.
Ordering physicians were all practitioners employed by the referral hospitals. They were physicians or residents from different specialties (including general medicine) working in different medicine or surgery departments except from vascular medicine (considered as vascular specialists) and pediatric departments (exclusion of patients aged less than 18 years old). A Wells score assessment by residents was considered only if supervised by the senior physician. All ordering physicians were previously informed, by electronic message, of the study and its objectives. Fourteen study physicians participated in the study in two centers. All had at least 2 years of experience (after residency) in vascular medicine and thromboembolism.
Outcomes
The primary outcome was the Wells score’s utility for risk stratification for proximal DVT, depending on presence or absence of thromboprophylaxis. This was evaluated by the prevalence of proximal DVT among the three different risk categories (i.e. high, moderate, and low, and as the area under the receiver operating characteristics (ROC) curve (AUC)). A Wells score assessed by the study physician (considered as the thrombosis specialist) was used to evaluate the primary outcome, as originally validated by Wells et al.4,6 Secondary outcomes were the Wells score’s concordance and usefulness in clinical decision-making between ordering and study physicians by AUC, and evaluation of the prevalence of proximal DVT among the three different risk categories.
For both primary and secondary outcomes, only proximal DVT was used to evaluate Wells score accuracy as it has been validated in this clinical situation. 6
Statistical analysis
Statistical analysis was performed using commercially available software (IBM SPSS 22.0, Armonk, NY, USA and XLSTAT, Addinsoft, NY, USA). The AUC representing sensitivity versus 1 – specificity (i.e. true positive results plotted against false positive results) was used to measure the accuracy of the Wells prediction score for risk of proximal DVT according to the presence or absence of thromboprophylaxis and for evaluation of the prevalence of proximal DVT according to risk category. For Wells score concordance and usefulness in clinical decision-making between ordering and study physicians, we compared AUC and prevalence of proximal DVT among the three different risk categories. Efficiency rate was defined as the proportion of patients in the low probability group. Failure rate (estimated as mean predicted probabilities of DVT for patients with a low Wells score with a negative D-dimer test) could not be calculated as, in both recruiting centers, D-dimer testing is not recommended for hospitalized patients. Inter-rater reliability for the Wells score was evaluated by intraclass correlation coefficient (ICC) between ordering and specialist physicians, and each Wells score predictor was evaluated by weighted Cohen’s kappa coefficient, with a score ⩽ 0.4 considered a poor inter-rater agreement. The differences in rates of thromboembolic events between the three pre-test probability groups, as well as comparisons of percentages, were done using a χ2 test, with a cut-off for significance of < 0.05.
Based on preliminary data, 19 we calculated that the sample size should be 400 patients in order to prove a difference of at least 10% in DVT prevalence in the three risk categories depending on thromboprophylaxis status for an alpha risk of 5% and a statistical power of 80%.
Results
A total of 425 patients were initially recruited. Ten patients with incomplete DVT probability scoring by ordering physicians were excluded. All were classified as low risk and no DVT was found. Characteristics of the 415 patients included during the study period are summarized in Table 1. As expected in an inpatient setting, most patients (69.2%; 95% CI: 64.7–73.6%) were recently bedridden for at least 3 days or had undergone a major surgical procedure within the past 4 weeks. Almost one-fifth (19.3%; 95% CI: 15.5–23.1%) had active cancer. More than two-thirds (67.7%; 95% CI: 63.2–73.2%) were hospitalized in a medicine hospital service. A total of 195 patients (47.0%) received ‘thromboprophylaxis’, either by thromboprophylaxis for at least 72 hours in 147 patients (35.4%) or by long-term, full-dose anticoagulation in 50 patients (12.1%). Two patients received both thromboprophylaxis (subcutaneous enoxaparine 40 mg/day) and full-dose anticoagulation (VKA therapy with INR in the therapeutic range) due to prescription error from attending physicians.
Characteristics of the total population (n = 415).

Data presented as numbers (percentages) unless indicated otherwise.
DVT, deep vein thrombosis; LEUS, lower extremities ultrasound study.
D-dimers were assessed only for 14 inpatients. Mean D-dimer rates in patients with or without proximal DVT were 3031.8 (95% CI: 0–7000) and 1439.6 (95% CI: 0–3875) (p < 0.001), respectively.
DVT risk assessment could only be calculated for a small number of patients. For 55 medical inpatients, the Padua score (20) with or without thromboprophylaxis was 4.36 (95% CI: 1.29–7.43) and 3.03 (95% CI: 0.31–5.75) (p < 0.001), respectively. In the Padua score, VTE risk is categorized as being low (< 4 points) and high (⩾ 4 points). 20 For 25 surgical inpatients, the Caprini score, 21 with or without thromboprophylaxis, was 10.4 (95% CI: 0.4–20.4) and 6.4 (95% CI: 2.25–10.55) (p < 0.001). In the Caprini score, VTE risk is categorized as being very low (0–1 point), low (2 points), moderate (3–4 points), and high (⩾ 5 points). 22
Proximal DVT was diagnosed in 30 of 415 patients (7.2%; 95% CI: 4.8–9.7%) and isolated distal DVT in 53 of 415 patients (12.8%; 95% CI: 9.6–16.0%).
Effectiveness of the Wells score in predicting proximal DVT in inpatients depending on anticoagulation therapy
Patients under current thromboprophylaxis were older (mean age: Group 1: 73 ± 16 vs Group 2: 68 ± 18, p = 0.002) with a lower prevalence of proximal DVT (4.1% vs 10.0%, p = 0.03); whereas there was no significant difference between groups for Wells total or single-item score (Table 2).
Characteristics of inpatients with suspected DVT classified by thromboprophylaxis status.
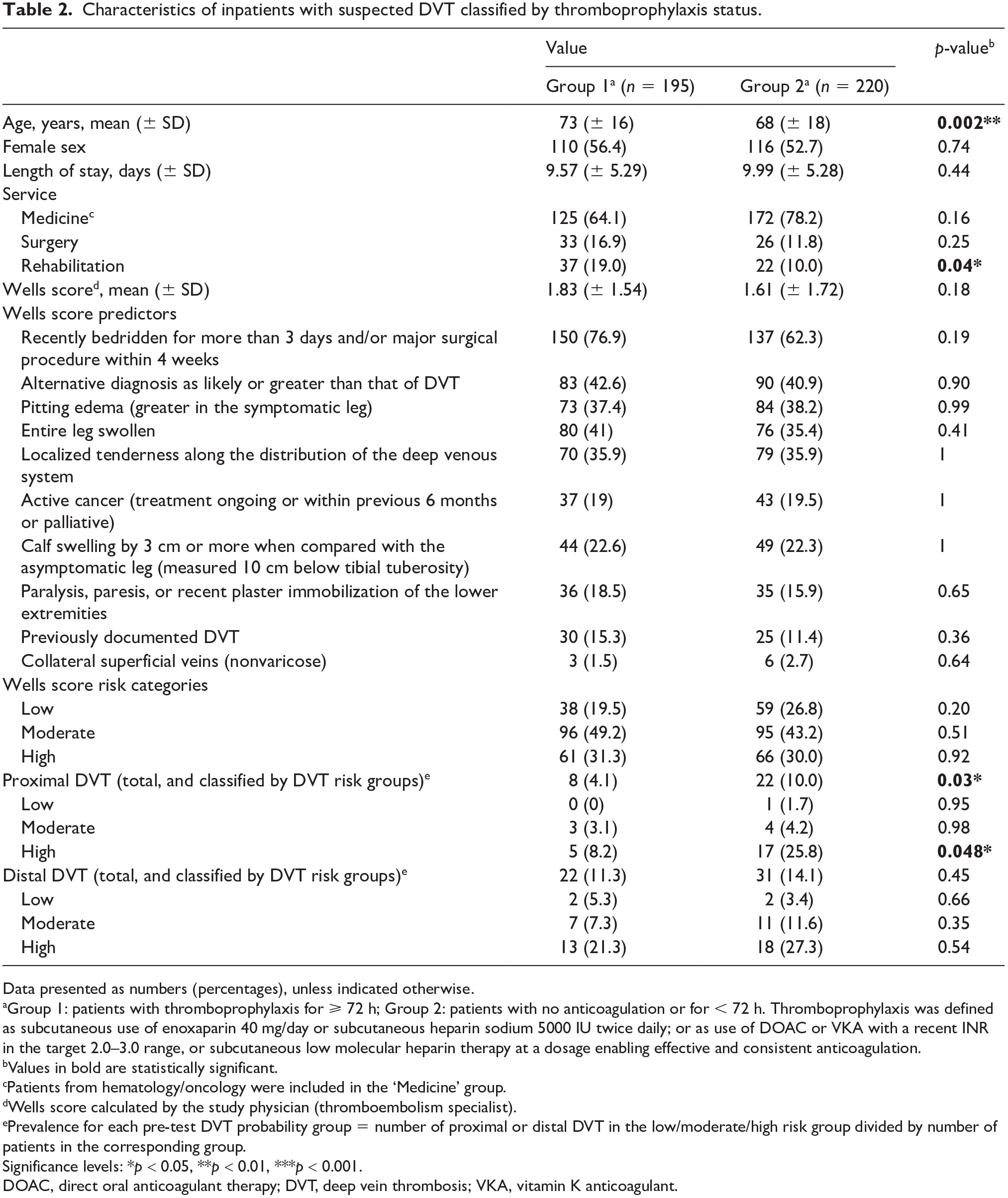
Data presented as numbers (percentages), unless indicated otherwise.
Group 1: patients with thromboprophylaxis for ⩾ 72 h; Group 2: patients with no anticoagulation or for < 72 h. Thromboprophylaxis was defined as subcutaneous use of enoxaparin 40 mg/day or subcutaneous heparin sodium 5000 IU twice daily; or as use of DOAC or VKA with a recent INR in the target 2.0–3.0 range, or subcutaneous low molecular heparin therapy at a dosage enabling effective and consistent anticoagulation.
Values in bold are statistically significant.
Patients from hematology/oncology were included in the ‘Medicine’ group.
Wells score calculated by the study physician (thromboembolism specialist).
Prevalence for each pre-test DVT probability group = number of proximal or distal DVT in the low/moderate/high risk group divided by number of patients in the corresponding group.
Significance levels: * p < 0.05, ** p < 0.01, *** p < 0.001.
DOAC, direct oral anticoagulant therapy; DVT, deep vein thrombosis; VKA, vitamin K anticoagulant.
Prevalence of proximal DVT in the low, moderate, and high pre-test probability groups was 0.0%, 3.1%, and 8.2% (p = 0.14) for Group 1; and 1.7%, 4.2%, and 25.8% (p < 0.001) for Group 2. Difference of prevalence was significant between groups in the high pre-test probability group: 8.2% in Group 1 vs 25.8% in Group 2 (p = 0.048), but not in the low (0.0% vs 1.7%, p = 0.95) or moderate (3.1% vs 4.2%, p = 0.98) pre-test probability groups.
The AUC for discriminatory accuracy of the Wells clinical score for risk of proximal DVT in inpatients with or without thromboprophylaxis was 0.72 (95% CI: 0.57–0.87) versus 0.88 (95% CI: 0.82–0.94), respectively (Figure 1). The efficiency of the Wells score was 23.4% (95% CI: 19.3–27.4%) for the entire inpatient population. As explained in the Method section, the failure rate could not be calculated.
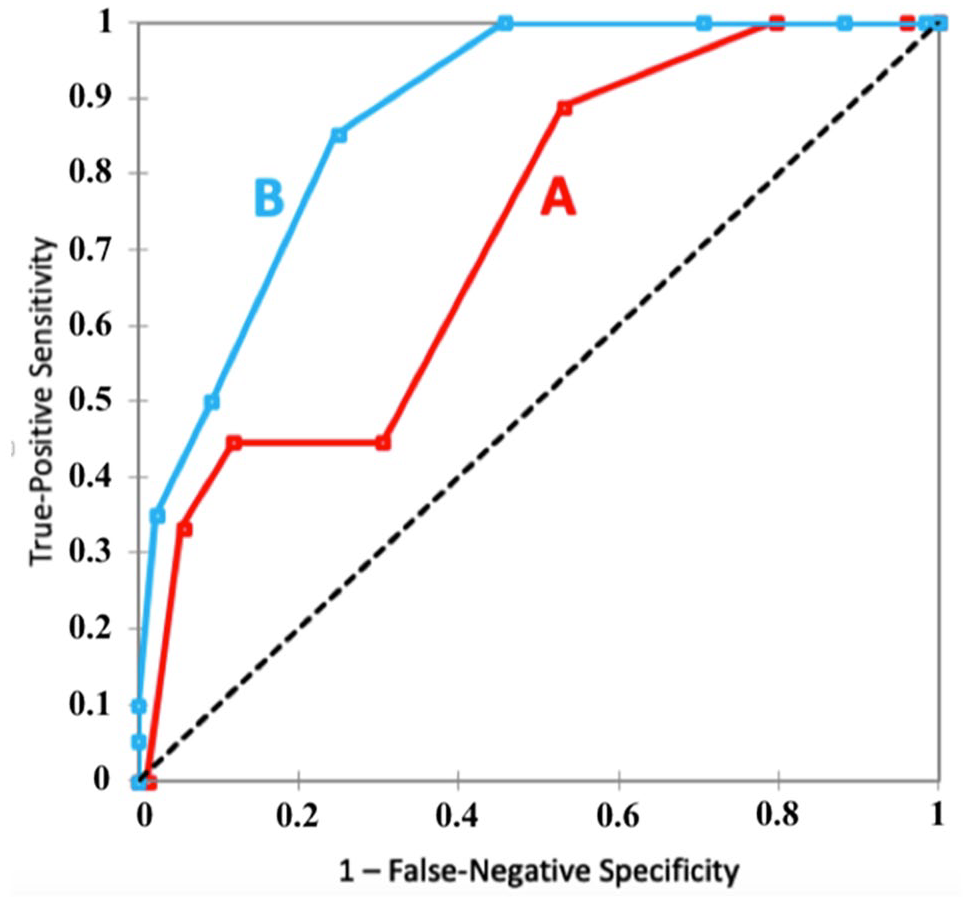
AUC of ROC curves comparing the discriminatory accuracy of the Wells score for predicting proximal DVT in two distinct inpatient populations: those with preventive or full-dose AC for ⩾ 72 h (A) and those with no (or for < 72 h) thromboprophylaxis (B).
For information, regarding Wells scores assessed by a nonthrombosis specialist, prevalence of proximal DVT in the low, moderate, and high pre-test probability groups was 0.0%, 4.1%, and 5.8% (p = 0.84) for Group 1; and 12.5%, 8.1%, and 9.6% (p = 0.89) for Group 2.
Concordance of the Wells score between ordering and study physicians
The number of patients classified as low, moderate, and high proximal-DVT risk was 10 (2.4%), 160 (38.6%), and 245 (59.0%) for ordering physicians, and 97 (23.4%), 191 (46.0%), and 127 (30.6%) for study physicians (p < 0.001), respectively.
Global prevalence of proximal DVT in the low, moderate, and high pre-test probability groups based on the Wells probability score established by ordering physicians or by study vascular physicians was 10.0%, 6.3%, and 7.8% (p = 0.82), and 0.0%, 4.2%, and 17.3% (p < 0.001) respectively. Difference of prevalence was significant only in the high pre-test probability group: 7.8% vs 17.3% (p = 0.021), but not in the low (p = 0.19) or moderate (p = 0.56) pre-test probability groups. The AUC for discriminatory accuracy of the Wells prediction score for risk of proximal DVT (confirmed by LEUS) was 0.83 (95% CI: 0.76–0.89) for the study vascular physician-assessed score and 0.57 (95% CI: 0.45–0.68) for the ordering physician-assessed score (Figure 2). Comparison of AUC 23 showed a z-score ⩾ 5 (where 1.96 is the z-score corresponding to a 95% two-sided confidence level, and 2.58 corresponding to a 99% two-sided confidence level) showing a high level of significance. The efficiency rate of the Wells score was 0.5% for the ordering physician and 23.4% for the study physician (p < 0.001).
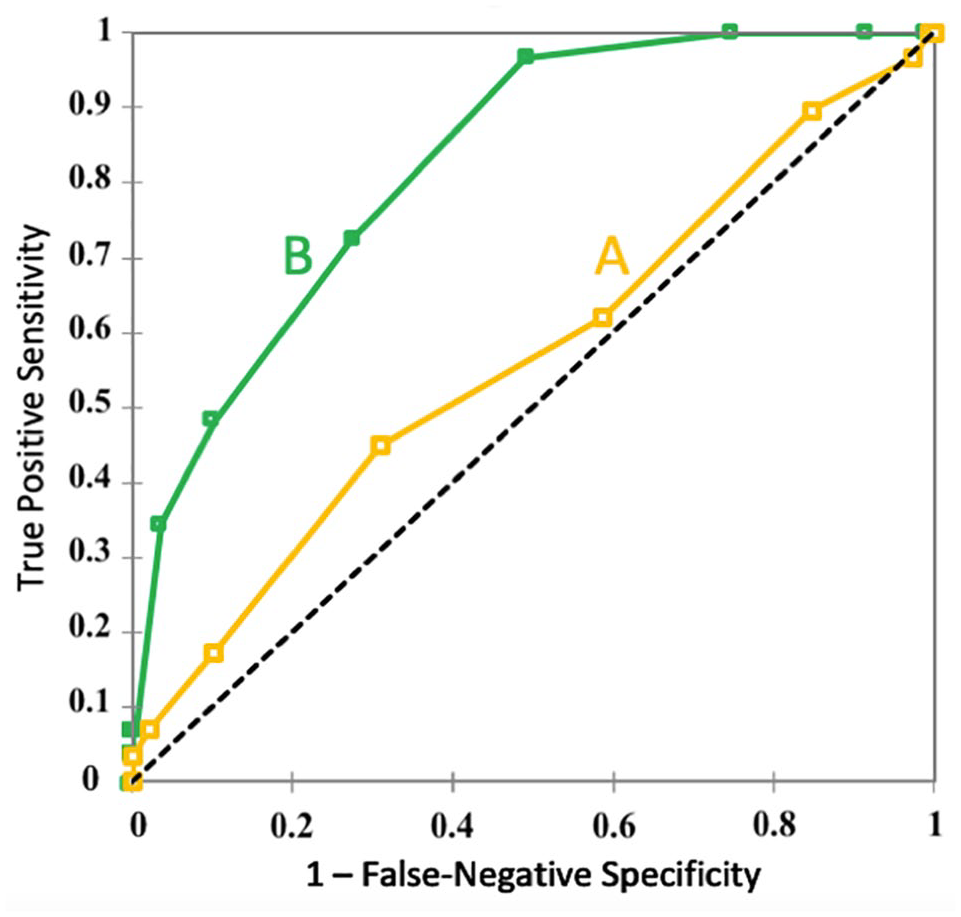
AUC of ROC curves comparing the discriminatory accuracy of the Wells score for predicting proximal DVT in inpatients between ordering physicians (A) and thromboembolism specialist physicians (B).
Inter-rater reliability between ordering and study vascular physicians for each Wells score predictor is summarized in Table 3. Wells score predictors based on interrogation of the patient or their medical file showed good to excellent inter-rater agreement, whereas those based on patient clinical examination showed moderate, poor or very poor inter-rater agreement. The ICC coefficient was 0.37 for the total score, representing a poor inter-rater agreement.
Inter-rater reliability for the Wells score and each Wells score predictor.
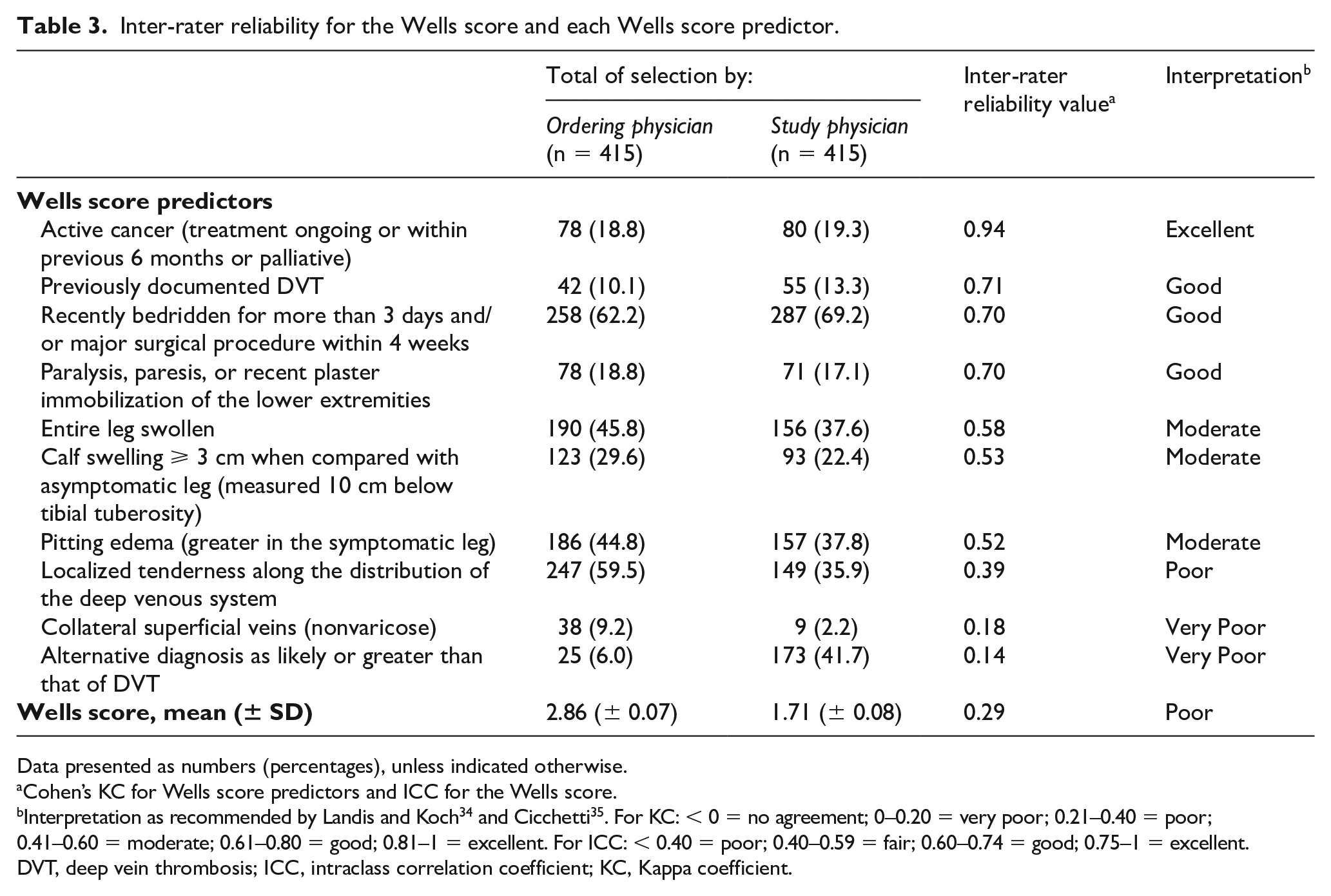
Data presented as numbers (percentages), unless indicated otherwise.
Cohen’s KC for Wells score predictors and ICC for the Wells score.
Interpretation as recommended by Landis and Koch 34 and Cicchetti 35 . For KC: < 0 = no agreement; 0–0.20 = very poor; 0.21–0.40 = poor; 0.41–0.60 = moderate; 0.61–0.80 = good; 0.81–1 = excellent. For ICC: < 0.40 = poor; 0.40–0.59 = fair; 0.60–0.74 = good; 0.75–1 = excellent.
DVT, deep vein thrombosis; ICC, intraclass correlation coefficient; KC, Kappa coefficient.
Discussion
Our study focused on the difference in performance of the Wells score for inpatients according to thromboprophylaxis, showing a marked decrease of performance in the inpatient group under thromboprophylaxis. We found that the difference of proximal DVT prevalence in inpatients across the low, moderate, and high pre-test probability groups was much narrower (0.0%, 4.2%, and 17.3%, respectively) compared with the outpatient ranges reported by Wells et al. (3%, 17%, and 75%, respectively). 5 The diagnostic accuracy of the Wells score was moderate (AUC = 0.72) for discriminating the risk of proximal DVT for inpatients with thromboprophylaxis, whereas it remained satisfactory (AUC = 0.88) for inpatients without.
In 2015, Silveira et al. 14 found a very low discriminatory rate of Wells score for all inpatients, regardless of their thromboprophylaxis status. The authors reported an AUC = 0.56, almost equal to the threshold of 0.5, corresponding to a model with no class separation capacity. For inpatients, D-dimer tests have a very low level of efficiency to exclude thromboembolism,24,25 with a cut-off of 500 µg/L. Hence, calculating the failure rate and efficiency of the low probability score to rule out DVT is of little relevance.
Published data support the efficacy and accuracy of the Wells score to predict DVT among inpatients.15,26 However, these studies have modest sample sizes (273 and 150 inpatients). A meta-analysis by Geersing et al. 7 studied the robustness of the Wells score in different subgroups, including the care setting (primary or hospital), presence of active cancer, sex, and history of DVT, and concluded that the model based on the Wells prediction score is effective in all subgroups except for patients with active malignancy. Despite this conclusion, data showed higher proximal DVT rates for secondary care or hospital settings compared with outpatients in the low pre-test probability group (4.6–6.4% vs 2.2–4.0%, respectively) and lower proximal DVT rates in the high pre-test probability group (22.9–64.9% vs 34.4–92.6%, respectively). In our study, the actual observed proximal DVT rate in the high pre-test probability group (16.4%) was much lower than in the Geersing et al. study (43.2%). 7 Finally, when the Wells score was accessed by the ordering physician, the efficiency rate (proportion of patients in the low probability group) was low in agreement with Silveira et al. (0.5% and 11.9%, respectively) and disagreeing with Geersing et al. (23.1%). On the other hand, the efficiency rate of the Wells score was maintained (23.4%) when assessed by thromboembolism specialist physicians.
The AUCs between inpatients with and without pharmacological thromboembolism prophylaxis were 0.72 and 0.88, respectively. On the one hand, this result suggests that the Wells score as a standalone tool, assessed by a thromboembolism specialist, remains useful to exclude DVT in inpatients with no thromboprophylaxis or long-term anticoagulation. This is consistent with recent data. With an 8% proximal DVT prevalence, Sartori et al. 36 emphasized that the Wells score remains reliable when assessed by a physician experienced in venous thromboembolism management (AUC = 0.75).
But on the other hand, our results suggest that the Wells score risk stratification is insufficient to rule out DVT in inpatients with thromboprophylaxis. For inpatients with thromboprophylaxis, prevalence of proximal DVT showed no significant difference (p = 0.22) among the three pre-test probability groups, whereas it showed great difference (p < 0.001) for inpatients with no thromboprophylaxis or for less than 72 hours. In their prospective study, Silveira et al. 14 highlighted that patients hospitalized in medicine and/or hematology/oncology services had a prevalence of proximal DVT two times higher than surgical patients. Whilst the cause is likely multifactorial, use of pharmacological thromboprophylaxis was lower in medicine/hematology/oncology than in surgery (40–57% vs 67%). In our study, surprisingly, thromboprophylaxis prescription did not significantly differ between inpatient subgroups, such as presence of an active cancer (p = 1) and presence or not of history of documented DVT (p = 0.36), although there was a trend for recently bedridden patients and/or major surgical procedures (p = 0.19). Hence, the presence of cancer seems to have no influence on the attending physician’s decision process regarding thromboembolism pharmacological prevention, even if cancer is well recognized as a major risk factor for DVT and pulmonary embolism prevalence in the medical20,27,28 and surgical29,30 setting.
Our study underscores the limitation of utilization of the Wells score by a nonthrombosis specialist, as concordance rates were modest for items concerning patient clinical settings and unacceptable for items regarding patient clinical signs of DVT. This indicates misknowledge or misunderstanding of measurement techniques – or indeed lack of measurement at all – and highlights the importance of the vascular specialist’s expertise as well as education for nonvascular specialists in assessing the Wells score.
In the original study by Wells et al., 6 all included patients were assessed for pre-test probability scoring by one of the study physicians experienced in thromboembolic disorders. Our study suggests that the accuracy and efficiency of the Wells score may depend on a clinician’s knowledge and clinical experience of DVT. It should be noted that, in French hospitals, specialized exams are mostly requested by residents who may tend to present their patient’s case in an unfavorable light (higher Wells score) in order to obtain the exam more quickly. 31 In 2005, Oudega et al. conducted a prospective study to validate the accuracy of the Wells score when performed by general practitioners in a primary care setting. 32 With over 1200 included patients, the prevalence of DVT among low, medium, and high pre-test probability groups was 12.0%, 16.5%, and 37.5%, respectively; the negative likelihood ratio was 0.48 versus 0.16 in the original study by Wells et al. 5 In contrast to the study by Oudega et al., 32 based on the calculation of the original Wells rule, our study is based on the modified Wells score.
Study limitations
Finally, this study presents some limitations. It was designed as a cross-sectional study, so that no causality links can be concluded. In a hospital setting with easy access to LEUS, we hypothesized that all patients with clinical suspicion of DVT underwent LEUS for definite diagnosis, and it would be unlikely in our practice that patients could be empirically treated for DVT without confirmation. Second, in both recruiting centers, D-dimer testing is restricted to outpatients and emergency departments where its efficacy has been validated.8,33 Hence, we could not analyze the accuracy and efficacy of the modified Wells prediction rule with D-dimer testing for inpatients in the low or medium probability group, nor the failure rate of the Wells score. 7 Third, recruitment from oncology/hematology departments was not strictly consecutive because part of the activity of Doppler ultrasounds was delegated to private practitioners that were not part of the study. Fourth, the Wells score was assessed within 24 hours following DVT suspicion by a study physician (blind to the Wells score by the ordering physician) who then performed LEUS. Using the same physician to perform the LEUS can introduce a bias but this study was designed as real-life management of VTE in the hospital setting. Every LEUS was performed as recommended by the French Vascular Society with a systematic approach which significantly reduces any bias of misdiagnosis. Fifth, only a minority of patients had a DVT risk stratification (by Padua or Caprini scores), owing to insufficient clinical description in the medical record. Last, prevalence of proximal DVT was lower than expected, although sufficient to reach statistical significance for our primary outcome.
Conclusion
In our inpatients group, prevalence of DVT across the low, medium, and high probability groups was much lower than reported for outpatients in the literature. Wells score accuracy and efficiency are greatly dependent on the assessing physician’s experience in thromboembolic disease. As currently framed, the Wells score assessed by a nonthrombosis specialist physician is barely better than chance in discrimination of risk for proximal DVT in inpatients. In addition, anticoagulation clearly affects Wells score performance. Indeed, Wells score risk stratification for detection of DVT (assessed by a VTE specialist) remains efficient for inpatients with no thromboprophylaxis but is not sufficient to rule out DVT in hospitalized patients on anticoagulation treatment. A candidate item ‘presence of thromboprophylaxis for at least 72 hours or long-term anticoagulation therapy’ could be added to the Wells score (possibly to replace the item ‘alternative diagnosis at least as likely as DVT’). Even if it could represent a step forward, further validation is required.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X21994672 – Supplemental material for Performance of the Wells score in predicting deep vein thrombosis in medical and surgical hospitalized patients with or without thromboprophylaxis: The R-WITT study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X21994672 for Performance of the Wells score in predicting deep vein thrombosis in medical and surgical hospitalized patients with or without thromboprophylaxis: The R-WITT study by Jean-Eudes Trihan, Michael Adam, Sara Jidal, Isabelle Aichoun, Sarah Coudray, Jeremy Laurent, Laurent Chaussavoine, Sebastien Chausserie, Jerome Guillaumat, Damien Lanéelle and Antonia Perez-Martin in Vascular Medicine
Footnotes
Acknowledgements
We would like to thank Sarah Kabani (University Hospital of Nîmes, France) for valuable editing assistance and Hugo Potier (University Hospital of Nîmes, France) for his help in data management.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
