Abstract
Acute, painless, monocular vision loss (APMVL) usually has a vascular aetiology. We conducted a prospective observational study from 2011 to 2018 to analyse the added value of colour Doppler imaging to assess orbital vessel blood flow in the diagnosis of APMVL. The study included 67 patients (39 [58.2%] men; mean age, 65.9 years [SD 13.7]) with APMVL evaluated at the Neurosonology Laboratory within the first 5 days of symptom onset, who were classified as having either transient or persistent monocular blindness. The blood flow in the ophthalmic and central retinal arteries was assessed using colour Doppler ultrasound with a linear 7.5-MHz transducer. Thirty-three (49.3%) patients presented transient monocular blindness, with reduced blood flow in either the ophthalmic or central retinal artery. The group with persistent vision loss included 24 cases of central retinal artery occlusion (CRAO) and 10 cases of ischaemic optic neuropathy (35.8% and 14.9%, respectively, of the total sample). These patients were older and had a higher prevalence of hypertension and mild carotid atherosclerosis. Orbital colour Doppler ultrasound (OCDUS) clarified the mechanism/cause of the ischaemia in 11 (16.4%) patients and showed abnormal flow in 46 (68.7%) patients, confirming the vascular origin in 19 (57.6%) of the transient monocular blindness cases. Lower peak systolic velocity was observed in patients with CRAO (p < 0.001), and a velocity < 10 cm/s in the central retinal artery was independently associated with the diagnosis of CRAO. OCDUS can be helpful in confirming the vascular cause and identifying the aetiology of APMVL.
Introduction
Monocular visual field defects can be due to several conditions, although acute and painless episodes are usually related to vascular causes. Vision loss of vascular origin has been classified by a number of authors as transient or persistent, 1 others have distinguished between ocular ischaemia and ischaemic optic neuropathy (ION), 2 while another author described both embolic and nonembolic causes. 3 Between 35% and 98% of cases involving embolic events are associated with some degree of carotid artery atherosclerosis, which can result in transient or irreversible vision loss. Ocular ischaemic syndrome produced by reduced flow in orbital vessels has been linked to carotid artery stenosis greater than 70%. 4 However, a more recent study showed that monocular vision loss was related to significant carotid stenosis in only 18.9% of cases. 5
Transient or permanent occlusion of the ocular vessels is the most common mechanism underlying all ocular ischaemic events. However, vascular eye disorders include a wide range of clinical entities that vary in their pathophysiology, clinical features, diagnosis, and management 6 and can be due to haemodynamic phenomena, embolic events (produced by fibrin, cholesterol or calcium), focal thrombosis or hypercoagulability. The most frequent entities associated with these mechanisms are atherosclerosis, inflammatory diseases (giant cell arteritis), arterial dissections, atrial fibrillation, heart valve calcification, antiphospholipid syndrome, thrombocytosis, and vasospasm.1,6,7 Establishing the correct diagnosis of monocular vision loss is therefore a challenge for clinicians.
Vascular eye disease is diagnosed through a careful ophthalmological examination; for example, in 90% of central retinal artery occlusions (CRAOs), a cherry-red spot can easily be observed by indirect ophthalmoscopy in the acute phase. 3 However, fundoscopic examinations do not differentiate the cause of the lesion. Orbital colour Doppler ultrasound (OCDUS) is a useful cross-sectional imaging tool for assessing several pathological conditions of the orbit. Grayscale ultrasound imaging enables the study of the orbit anatomy, whereas colour Doppler imaging provides additional information on its vasculature. 8 This method can provide quantitative measurements of vascular flow and opens up a new range of diagnostic possibilities. OCDUS is a noninvasive, inexpensive, and well-tolerated tool widely used by radiologists and ophthalmologists, 9 although it is not commonly used in neurological practice.
Recently proposed protocols for acute vision loss management recommend routine blood testing, erythrocyte sedimentation rate measurements, electrocardiography, echocardiography, noninvasive imaging of the cervicocephalic vessels, brain magnetic resonance imaging, and admission to transient ischaemic attack (TIA) clinics or stroke units. 7 Although the most promising feature of OCDUS is the evaluation of orbital vessels, which might play a role in the diagnosis of ocular ischaemia, 8 this technique is not considered part of any protocol for evaluating vision loss. Retinal ischaemia is related to other recurrent vascular events, such as stroke, myocardial infarction, and vascular death within 1 year of onset. 10 Despite the fact that ultrasound is widely used among stroke neurologists, only a few of them have collaborated on publications related to ocular ischaemia.1,6,10 Given that acute painless monocular vision loss (APMVL) describes a group of syndromes or entities that have a vascular aetiology, the assessment of orbital vessels could help in the diagnosis. Based on our recent experience, the aim of this study was to analyse the results of vascular colour Doppler imaging of the orbital vessels and evaluate their added value in the diagnosis (cause/mechanism) of APMVL at our neurosonology laboratory.
Materials and methods
Study design
We conducted a prospective, observational study from 2011 to 2018 that included patients with APMVL evaluated at the neurosonology laboratory (part of the TIA clinic) of the neurology department of a stroke centre who were referred for ultrasound vascular evaluation within the first 5 days of symptom onset (to avoid cases in which the embolus had broken or disintegrated, or the arteries had already been recanalised). Patients who came to the emergency department were evaluated by an ophthalmologist using a Snellen chart (for visual acuity testing), a colour vision test, fundoscopy, visual field testing, and intraocular pressure measurement. We excluded patients with vision loss not attributable to arterial vascular causes based on the symptom profile, clinical characteristics, and physical examination (e.g., retinal detachment or haemorrhage, vitreous haemorrhage, optic or retrobulbar neuritis, and glaucoma). A neurologist then conducted a complete neurological examination of the patient and produced a differential diagnosis that considered the ophthalmological findings. Monocular vision loss was classified as either transient or persistent, with transient monocular blindness (TMB) considered synonymous with amaurosis fugax. We further divided persistent vision loss into CRAO or branch occlusion and ION. The diagnostic protocol included the erythrocyte sedimentation rate, electrocardiography, and neuroimaging (computer tomography scan/magnetic resonance imaging) for all patients. Extracranial and intracranial arteries were evaluated by duplex ultrasound, which included a carotid siphon assessment. In certain cases, we performed echocardiography, computed tomography-angiography, and colour duplex ultrasonography of the temporal artery to clarify the diagnosis. OCDUS examinations were performed on all patients as part of the routine diagnostic work-up, and no specific informed consent was obtained for this procedure. This study was approved by the Clinical Research Ethics Committee.
Orbital duplex ultrasound examination
The same observer (GRA) assessed the results of the OCDUS examinations, in accordance with previously published documents,9,11 which were performed using a Xario Diagnostic Ultrasound System (SSA-660A, software version 4.0; Toshiba Medical Systems, Tochigi, Japan) with a linear 7.5-MHz transducer, with patients lying in the supine position with their head at a 30-degree angle. A layer of acoustic gel was applied to the closed eyelids, after which the transducer was placed on the upper eyelid, with the examiner’s hand resting on the orbital margin to minimise pressure on the globe. Colour Doppler sonography was performed with the gain adjusted to avoid artefactual colour noise, thereby allowing the detection of low velocities. The Doppler sample gate (< 2 mm) was then placed on the detected vessel to record the blood flow signal. 12 The central retinal artery (CRA) was evaluated in horizontal sections, which were taken from the optic nerve head. The ophthalmic artery (OA) was identified immediately downstream from its crossing with the optic nerve. 9 The acoustic output of the ultrasound systems was adjusted to the requirements of the orbit ultrasonography according to the ALARA (‘as low as reasonably achievable’) principle to prevent harming the lens and retina. 13
The observer evaluated the ophthalmic and central retinal arteries and measured the peak systolic velocity (PSV), end diastolic velocity (EDV), and resistance index (RI). The following flow velocities (cm/s) were considered normal – OA: PSV 45.3 ± 10.5, EDV 11.8 ± 4.3, RI 0.74 ± 0.07; CRA: PSV 17.3 ± 2.6, EDV 6.2 ± 2.7, RI 0.63 ± 0.09. 12 Velocities under or over these values were considered abnormal (reduced or increased flow, respectively). We defined arterial stenosis as focal acceleration of the flow. The observer examined the flow direction and pattern in the OA.
Variables
The following parameters were recorded in a specific database: demographic characteristics (age, sex); vascular risk factors, such as arterial hypertension (defined as a previous clinical diagnosis of the condition, regular therapy with antihypertensive drugs, or two or more pre-APMVL blood pressure readings > 140/90 mmHg); diabetes mellitus (previously diagnosed and/or current therapy with insulin or oral hypoglycaemic drugs); hypercholesterolaemia (previously diagnosed and/or current therapy with lipid-lowering drugs); elevated low-density lipoprotein cholesterol levels (defined as levels > 130 mg/dL); cigarette smoking (current or within the last year); atheromatous complications such as coronary artery disease (angina, myocardial infarction, previous angioplasty or coronary artery bypass grafting); symptomatic peripheral arterial disease (intermittent claudication, critical leg ischaemia, previous surgery through amputation, reconstructive vascular surgery or angioplasty in one or both legs due to atherosclerotic disease); and previous stroke (TIAs or ischaemic lesions visible on brain computed tomography or magnetic resonance imaging). We assessed carotid disease by ultrasound (considering extracranial and intracranial circulation) and measured ipsilateral internal carotid atheroma following the Mannheim consensus. We defined the degree of atherosclerotic internal carotid artery stenosis as normal (no stenosis), < 50% stenosis, 50–69% stenosis, > 70% stenosis but less than near occlusion, near occlusion, and total occlusion. 14
Statistical analysis
We performed the statistical analysis using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA). Continuous data are presented as mean and SD or median and IQR (IQR 25th to 75th percentiles) if not normally distributed. We employed nonparametric tests to analyse the data and presented the discrete data as counts and percentages. We compared the proportions between the groups using the chi-squared and Fisher’s exact tests.
We performed a receiver operating characteristic curve analysis to determine the predictive value of the area under the curve (AUC) and a PSV cut-off that better distinguished a CRAO final diagnosis, according to the highest sensitivity and specificity combination. We constructed an adjusted logistic regression to adjust the PSV cut-off values for CRAO based on the factors that had a p-value less than 0.20 in the univariate testing.
Results
The study included 67 patients, 39 (58.2%) of whom were men. All participants were white, and their mean age was 65.9 years (SD 13.7). Table 1 summarises their baseline characteristics. Thirty-three (49.3%) patients were classified as having TMB or amaurosis fugax. The persistent vision loss group consisted of 24 cases of CRAO and 10 cases of ION (35.8% and 14.9% of the total sample, respectively). The patients with persistent vision loss were older and had a higher prevalence of arterial hypertension and ipsilateral internal carotid atherosclerosis. However, the frequency of carotid artery stenosis was 11.9% for > 50% stenosis and 6.0% for > 70% stenosis, with no differences between the groups. There were no differences in the other risk factors, such as atrial fibrillation, which was present in 10.4% of all patients (Table 1).
Baseline characteristics of patients.
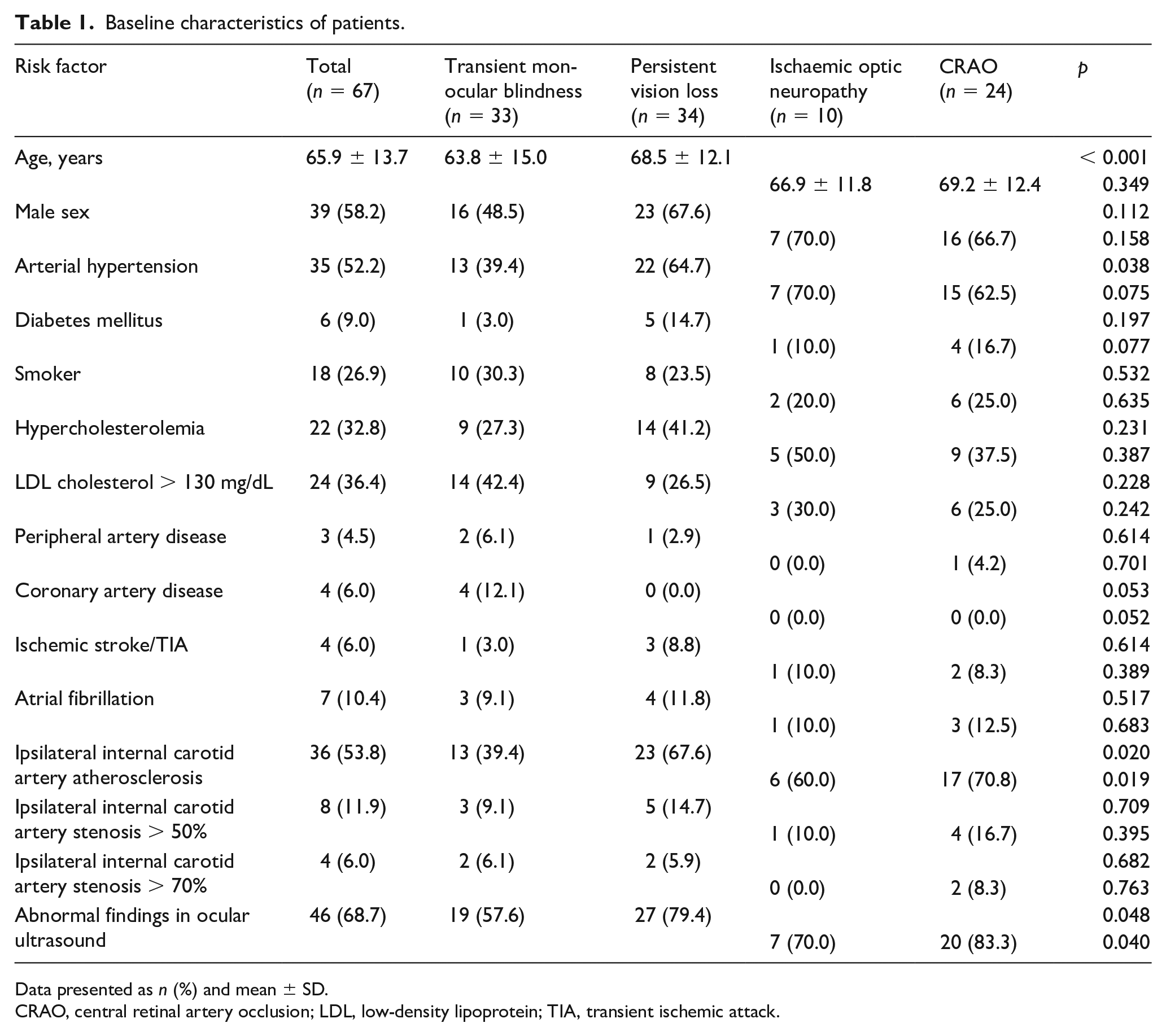
Data presented as n (%) and mean ± SD.
CRAO, central retinal artery occlusion; LDL, low-density lipoprotein; TIA, transient ischemic attack.
OCDUS showed abnormal flow (predominantly due to decreased flow) in 46 cases (68.7%) and was more frequently abnormal in the persistent vision loss group (79.4% vs 57.6% in the TMB group; p = 0.048). The comparison of blood flow measurements showed that CRA PSV was slower in the persistent vision loss group (8.7 [SD 5.5] cm/s) than in the transient group (12.5 [SD 3.4] cm/s; p = 0.001). The comparison between the diagnosis within the persistent vision loss group showed similar results, with slower velocities in CRAO (p < 0.001 ) (Figure 1A). Table 2 summarises the distribution of the diagnoses and the OCDUS findings. The receiver operating characteristic analysis identified a PSV < 10.0 cm/s as the best cut-off point, with a sensitivity of 80% and a specificity of 73% (AUC: 0.836; 95% CI 0.729–0.942; p < 0.001) for the final CRAO diagnosis (Figure 1B). The logistic regression model indicated that a CRA PSV < 10.0 was independently associated with the diagnosis of CRAO (odds ratio: 9.928; 95% CI 2.72–31.69; p < 0.001), after adjusting for age, high blood pressure, and internal carotid artery atherosclerosis. We observed atherosclerotic narrowing at the origin of the internal carotid artery in 36 (53.7%) patients, which was > 50% in eight (11.9%) of these patients. However, when comparing the carotid stenosis measured by ultrasound with the presence of abnormalities in the OCDUS, there were no significant differences among any of the patients with a diagnosis of vision loss.
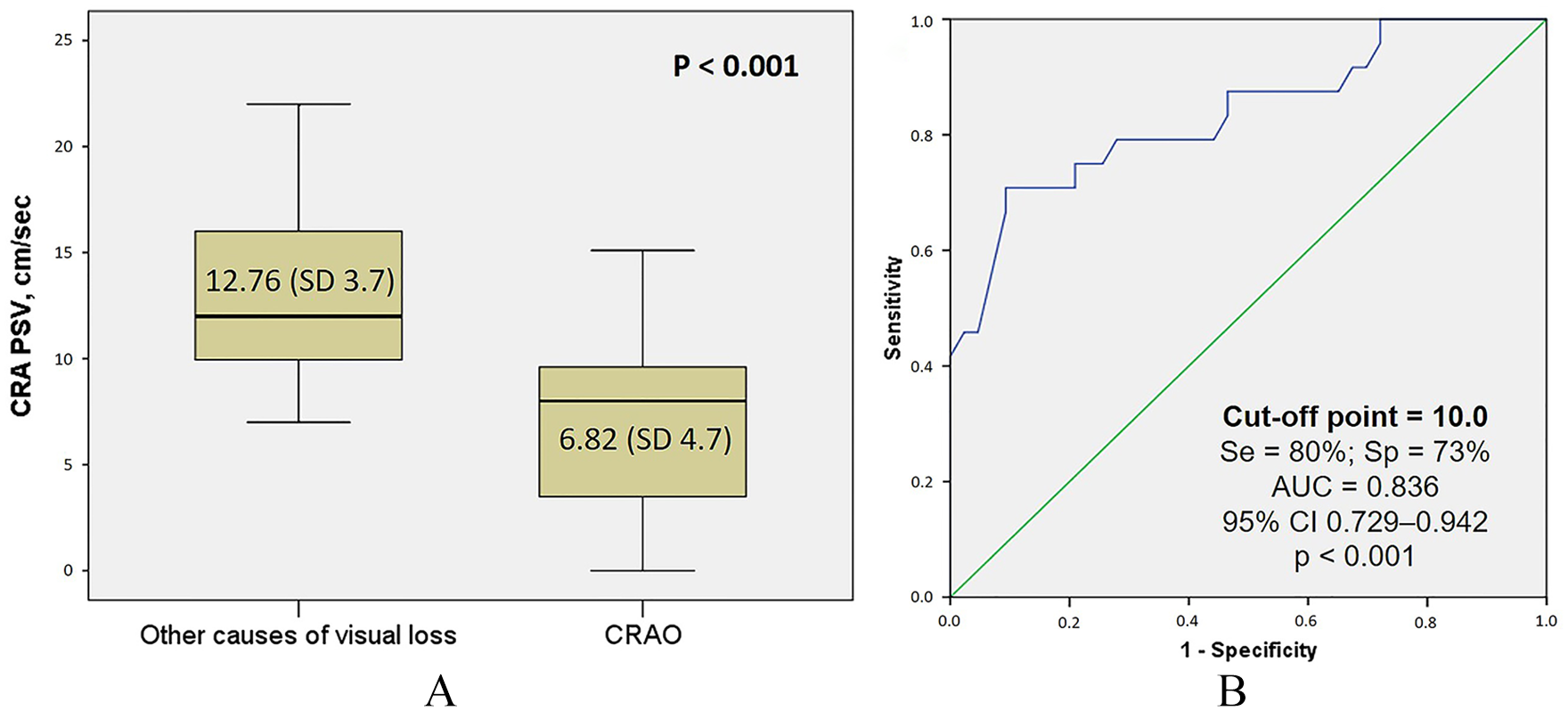
PSV analysis of central retinal artery occlusion (CRAO). (A) Box plot of central retinal artery (CRA) PSV (cm/s) distribution among CRAO and other causes of vision loss. (B) Receiver operating characteristic curve for CRAO diagnosis. PSV cut-off point is 10.0 cm2.
OCDUS findings and their distribution by diagnostic categories (n = 67).
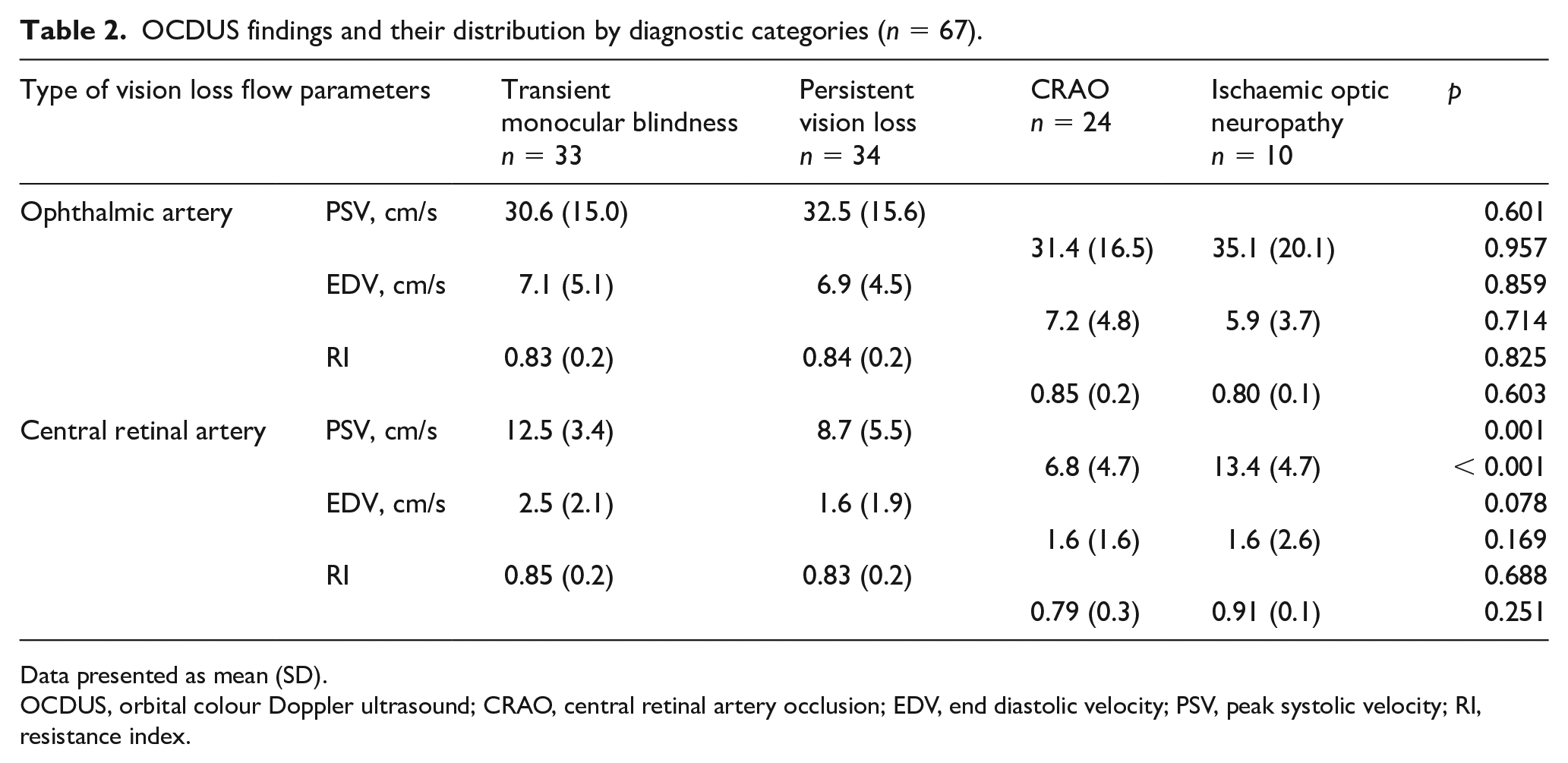
Data presented as mean (SD).
OCDUS, orbital colour Doppler ultrasound; CRAO, central retinal artery occlusion; EDV, end diastolic velocity; PSV, peak systolic velocity; RI, resistance index.
Transient monocular blindness
The TMB group was clearly associated with reduced blood flow in both the OA and CRA in approximately one-third of the patients. Two cases had accelerated flow in the OA and were diagnosed with focal stenosis (one of which was associated with internal carotid artery occlusion) (Figure 2). The other two cases showed a poststenotic waveform pattern associated with atherosclerosis at the origin of the OA at the carotid syphon. OCDUS showed pathological findings in 19 (57.6%) cases of TMB, confirming that the cause of the vision loss was vascular. Ten patients presented an increased RI, and one patient presented drusen. Other causes for the TMB were internal carotid artery stenosis with > 50% occlusion (three cases), atrial fibrillation (one case), valvular disease (one case), and aura without migraine (one case).
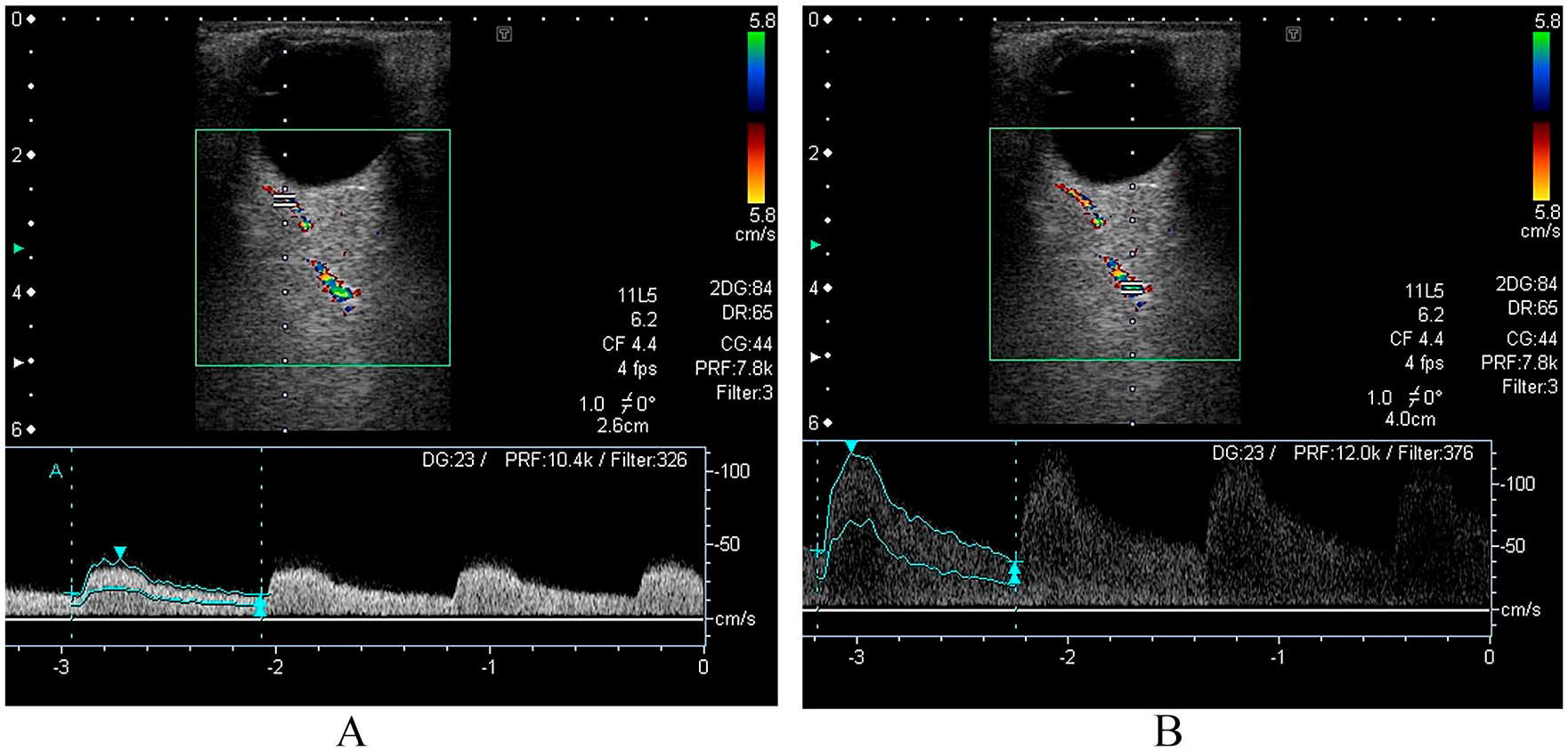
OCDUS images of a 71-year-old man with ipsilateral internal carotid artery occlusion and focal stenosis of the OA who presented TMB. (A) OA reversed flow with a PSV of 41 cm/s at 2.6 cm of depth as a prestenotic flow. The waveform is positive because the colour scale was inverted. (B) OA with PSV of 125 cm/s at 4.0 cm of depth that represented a focal stenosis.
Persistent vision loss
In the persistent vision loss group (n = 34), the OCDUS found abnormalities in 27 (79.4%) of the cases: 20/24 (83.3%) in the CRAO group and 7/10 (70.0%) in the ION group. Seventy-five per cent of the CRAO group presented with reduced or absent flow in the CRA, including four of the five patients with branch CRAO. The cause of the vision loss was related to OA stenosis in two cases (Figure 3), and in another case, a calcium embolus was observed within the shadow of the optic nerve, 4 mm behind the optic disc, suggesting an embolism from a calcified mitral valve, which was demonstrated by an echocardiogram. We observed internal carotid artery stenosis with > 50% occlusion in four patients (16.7%). Other causes for the vision loss included atrial fibrillation (one case), aortic valve calcification (one case), and antiphospholipid syndrome (one case). The ION group more frequently showed reduced flow in the OA and CRA. Two patients showed diffuse stenosis with a higher RI of the CRA; one of these patients also had OA stenosis and was ultimately diagnosed with giant cell arteritis (Figure 4). One patient presented a poststenotic waveform pattern with a reduced RI associated with atherosclerosis at the origin of the OA at the right carotid syphon. In one patient with posterior ION, we observed an arrhythmic and irregular wave with the disappearance of diastolic flow, suggesting a paroxysmal atrial fibrillation, which was immediately confirmed by an electrocardiogram. Twenty-five patients with persistent vision loss (73.5%) presented a pathological CRA flow, which represented the most frequent diagnostic sign (p = 0.001).
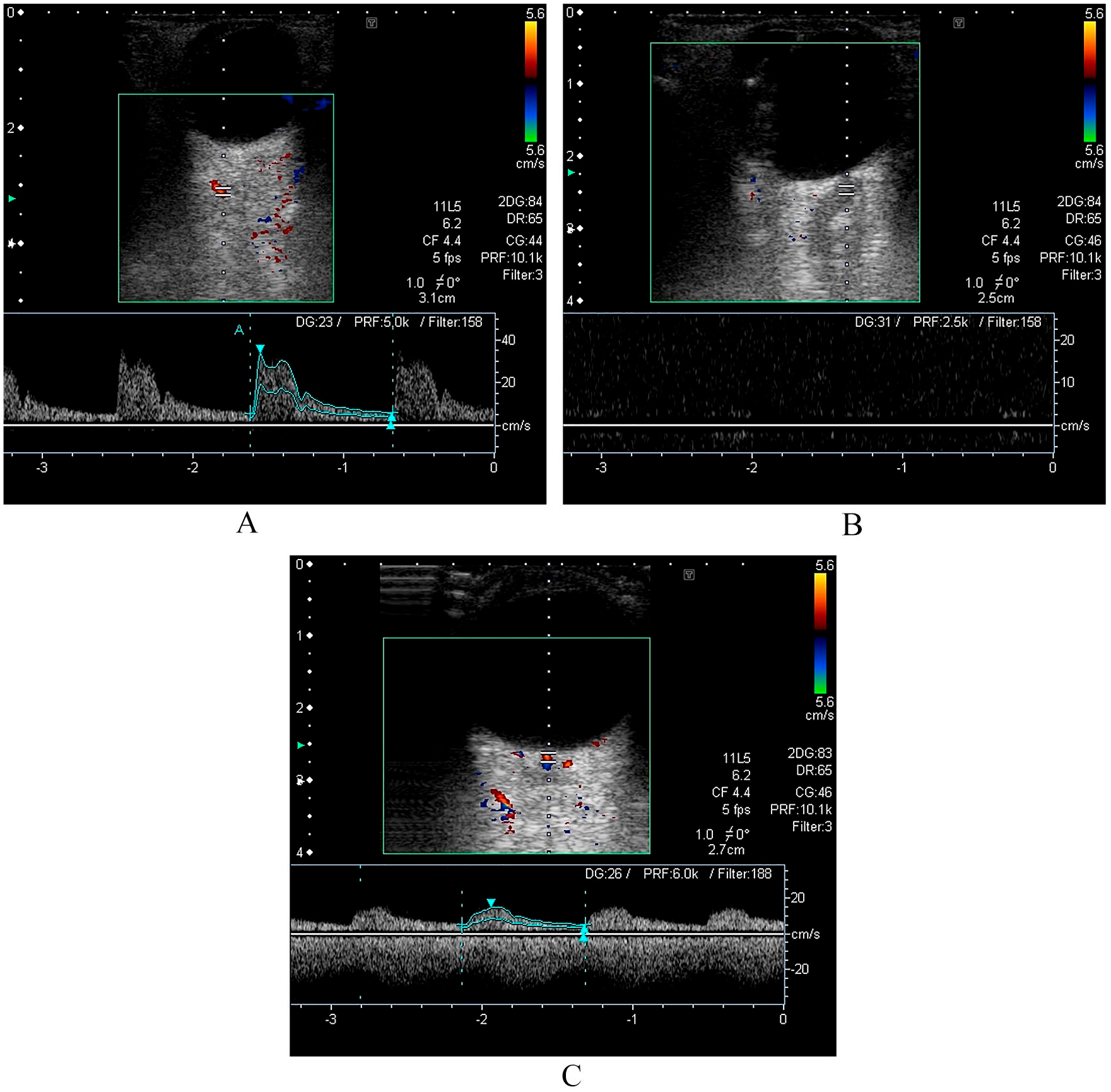
OCDUS images of an 85-year-old man with CRAO. (A) Normal blood flow in the right OA (PSV 37 cm/s); (B) almost absent flow in the right CRA; (C) normal blood flow in the left CRA (PSV 16 cm/s).
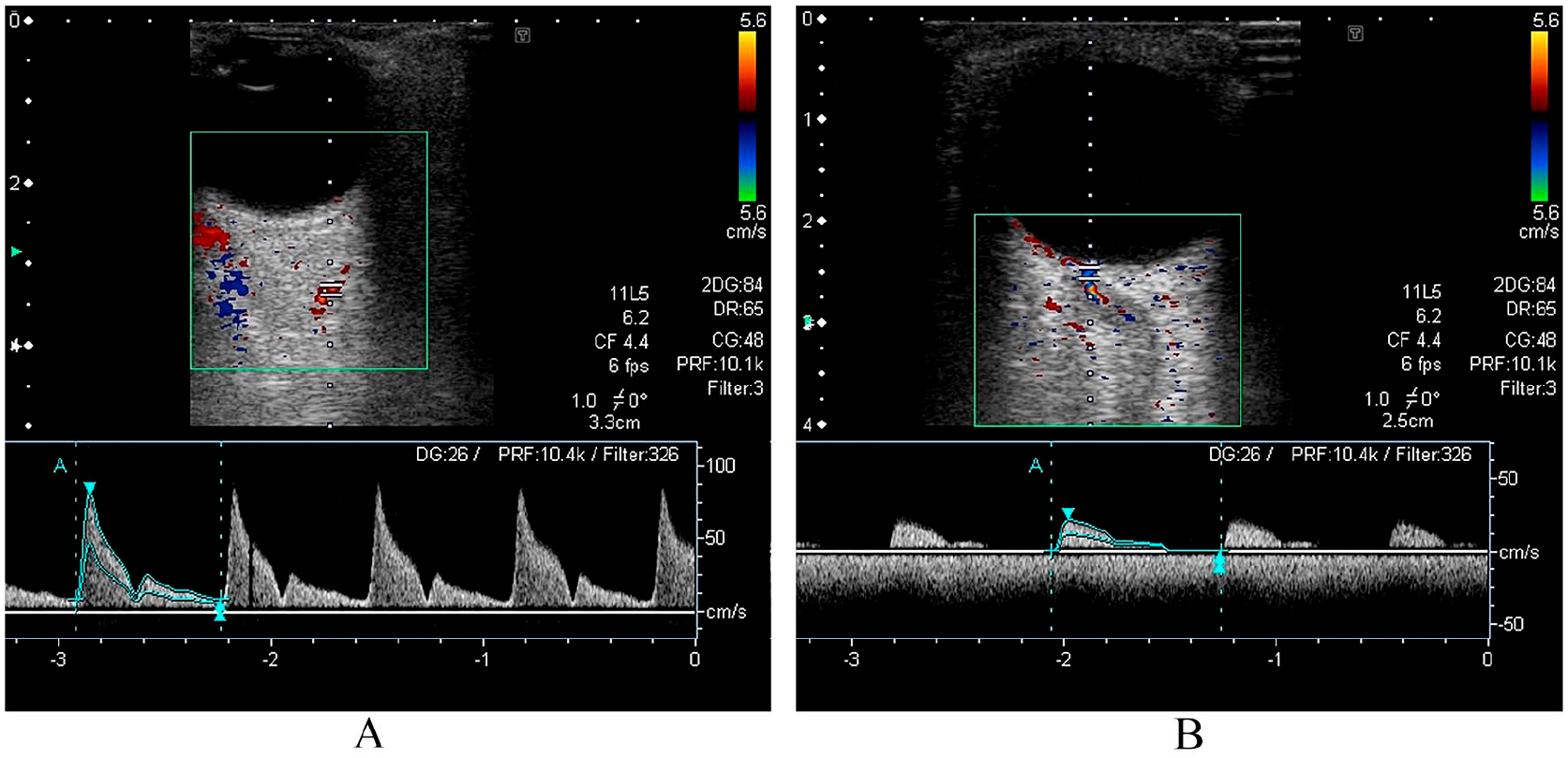
OCDUS images of a 67-year-old man with ION due to giant cell arteritis. (A) Accelerated flow (PSV 81 cm/s) with high RI (0.89) related to stenosis of the OA; (B) high PSV in the CRA with high RI (1.0) and without diastolic flow.
In summary, the mechanism/cause of the ischaemia was clarified in 11 (16.4%) patients after completing the OCDUS. The vascular origin of the vision loss in the TMB group (who underwent a normal but rigorous clinical examination) was confirmed in 19 (57.6%) of the cases.
Discussion
This prospective study showed the experience of a neurosonology laboratory in using OCDUS on patients with APMVL of suspected vascular cause, showing the importance of assessing the orbital arteries (especially the CRA) to clarify or confirm the diagnosis. Although relatively small, this study was one of the largest series conducted by neurologists, despite the high volume of patients with these diseases who are evaluated by stroke experts. The only other study on this topic was one published 30 years ago that included 105 patients. 15 Considering our results, OCDUS should be included in the diagnostic algorithm for APMVL. Acute retinal arterial ischaemia is a stroke equivalent and represents an ophthalmologic and neurologic emergency. Rapid evaluation in a stroke centre or a rapid-access TIA clinic is therefore recommended to perform the basic complementary examinations that include routine blood tests (including erythrocyte sedimentation rate and C-reactive protein levels), electrocardiogram, neuroimaging evaluation, carotid/transcranial ultrasound, and echocardiogram. 7 OCDUS should be included during the carotid and transcranial assessment.
In 1952, Fisher 16 indicated that TMB was associated with hemiplegia, with later publications confirming the relationship between TMB and atheromatous carotid disease. Several noninvasive studies were proposed to estimate carotid stenosis by combining duplex carotid ultrasound, ocular plethysmography, carotid phonoangiography, and supraorbital artery Doppler; however, these approaches did not replace arteriography. Transcranial Doppler subsequently enabled the evaluation of the OA. Advances in the equipment have enabled orbital evaluations with a 7.5-MHz transducer (the same employed for carotid arteries), achieving far better image resolution. Up-to-date ultrasound equipment provides high-quality images. Given that colour Doppler imaging of the eye and orbit has been shown to be useful for measuring blood flow, several publications have considered the application of colour Doppler in small series or isolated cases of diabetic retinopathy, psoriasis, 9 thyroid eye disease, and severe obstructive sleep apnoea syndrome. Ocular ischaemia is a potentially blinding disorder classically related to significant (> 50%) internal carotid artery stenosis. A number of series have reported abnormal blood flow in the orbital vessels as measured by ultrasound, 17 which improved following carotid endarterectomy. 18 An analysis of symptoms related to ocular ischaemic syndrome showed that photophobia, floaters, metamorphopsia, phosphenes, and diplopia might also be present. 19 More recent reviews have not only considered carotid atherosclerotic occlusive disease as the cause of ocular ischaemia but also embolus, arteritis, vasospasm, glaucoma, and antiphospholipid syndrome, emphasising the need to include these entities in the differential diagnosis.1,6,7
Giant cell arteritis deserves special consideration, given that several publications have considered ultrasound a useful tool for diagnosing this condition. 20 The most recent review and meta-analysis provides valuable positive and negative likelihood ratios for abnormal temporal artery ultrasound results compared with temporal artery biopsy. 21 We diagnosed two of our patients with giant cell arteritis, which fulfilled the classic criteria and ‘halo sign’ in the temporal artery ultrasound. These patients presented with stenosis in the CRA as measured by OCDUS, whereas we observed this finding in only one case of CRAO. Assessment of CRA flow changes associated with intimal hyperplasia could reveal a different mechanism for ocular ischaemia than that typically attributed to the posterior ciliary arteries.22,23
Several conditions can produce visual symptoms, thereby complicating the clinical diagnosis. We have shown our experience of how OCDUS can confirm the clinical diagnosis or, in some cases, help clarify the aetiology. Although accelerated flow in both the CRA and ophthalmic arteries throughout their entire route is characteristic of giant cell arteritis, only two patients in our sample were diagnosed with this condition, and more cases are needed to confirm this relationship. However, in the absence of an embolus in the CRA or in any of its branches, we showed that the absence of OCDUS signs was perhaps due to the suspected embolus having broken off or having been carried by the bloodstream, and therefore no visible signs were present when the patient arrived at the hospital.
Medical history and clinical examination help in diagnosing CRA occlusion in the vast majority of cases. However, colour Doppler imaging could be helpful when the clinical examination is difficult (e.g. due to media opacity) or when the results of the clinical examination are ambiguous. The hallmark of the colour Doppler imaging diagnosis of CRA occlusion is the lack of flow in the CRA, although there might be demonstrable flow in the CRA. In reported cases, CRA reperfusion occurred within 1 week of occlusion, 8 a situation that emphasises the importance of having a flow velocity cut-off to determine the diagnosis of CRAO (even when the artery is in the recanalisation phase), using a noninvasive diagnostic tool that provides complementary information to the intravenous fluorescein evaluation.
Study limitations
Given that the study objective was to show the applicability of OCDUS in evaluating monocular blindness, we assessed only those patients who were referred to the ultrasound laboratory or TIA clinic, and therefore the selection bias might explain some of the epidemiological disagreement with other studies. Nevertheless, putative mechanisms for ocular ischaemia appear to go beyond carotid artery atherosclerosis in our study. Another limitation of our study was the lack of a validation method for the OCDUS interpretations, which were made based on the experience of only one of the authors. An important detail shown in our results is that waveforms might differ between inflammatory stenosis and atherosclerotic OA stenosis and in ipsilateral carotid disease. We did not address whether the results of this diagnostic technique might have been affected by a learning curve, given that the same examiner (who has expertise in performing OCDUS) performed all of the evaluations during the study. However, all tests were performed by the same trained operator to eliminate interobserver variability.
Conclusion
In conclusion, this study suggests that the evaluation of orbital arteries using OCDUS might be helpful in determining the vascular cause and identifying the aetiology of ischaemic monocular vision loss. Considering that patients with persistent vision loss are more likely to present ocular vessel disorders, these results emphasise the usefulness of OCDUS in diagnosing ocular ischaemia, especially in CRAO, for which we found a PSV cut-off point showing high sensitivity and specificity. These results should, however, be validated with larger samples.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
