Abstract

Keywords
What are the different options for the COVID-19 vaccine?
SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) is the virus that causes coronavirus disease 2019 (COVID-19). Globally, numerous vaccines have been developed against COVID-19. In the United States, three vaccines for COVID-19 have been authorized or approved by the US Food and Drug Administration (FDA): Pfizer-BioNTech, Moderna, and Johnson & Johnson. The majority of patients in the US have received either the Pfizer-BioNTech or Moderna vaccines, but all three are highly effective in preventing serious disease, hospitalization, and death from COVID-19. Although certain side effects may occur, the benefits greatly outweigh the risks.
In the United Kingdom, Australia, and other countries (but not in the US), a vaccine produced by AstraZeneca has been widely administered. Though it is also effective, it has certain rare but unique side effects as discussed below. Nevertheless, its benefits still greatly outweigh its risks.
Important differences in the manufacturing process distinguish these vaccines from one another. The Pfizer-BioNTech and Moderna vaccines are both nucleoside-modified mRNA, encoding the viral spike (S) glycoprotein of SARS-CoV-2. The Johnson & Johnson vaccine comprises a recombinant, replication-incompetent Ad26 vector, encoding a stabilized variant of the S protein. Similarly, the AstraZeneca vaccine is a replication-deficient modified adenovirus ChAdOx1, containing the coding sequence of the S protein.
Put simply, the Pfizer-BioNTech and Moderna vaccines are mRNA vaccines, whereas the Johnson & Johnson and AstraZeneca vaccines are adenovirus-vector vaccines.
Are the COVID-19 vaccines safe?
Over 400 million doses of COVID-19 vaccine have been given in the US, with almost 7 billion doses administered worldwide. For the vast majority of people, including those with vascular disease, the benefits of vaccination against COVID-19 overwhelmingly outweigh any risks. These vaccines are undergoing continuous and intense safety monitoring. Across numerous clinical trials, the vaccines have been shown to be safe and effective, and they clearly reduce the risk of serious infection, hospitalization, and death related to COVID-19.
Although rare side effects have been reported with these vaccines, these events occur much less frequently than the often severe and life-threatening complications that occur with COVID-19. In summary, these vaccines are safe and effective.
What are the common side effects of COVID-19 vaccines?
The COVID-19 vaccines have been subjected to some of the most intense monitoring in medical history. In the US, this includes monitoring through the Vaccine Adverse Event Reporting System (VAERS), which is a national early warning system that the US FDA uses to monitor the safety of vaccines after they are authorized or licensed for use. VAERS helps to detect unusual or unexpected reporting patterns of adverse events for vaccines. Results from VAERS and other vaccine safety monitoring efforts are very reassuring.
Some people have no side effects at all, especially with the first dose. Others have common but minor side effects after COVID-19 vaccination. Symptoms may include swelling, redness, and pain at the injection site; fever; headache; tiredness; muscle pain; chills; or nausea. These symptoms usually go away on their own over a few days. They typically also respond well to simple analgesics such as acetaminophen (Tylenol; paracetamol).
What rare cardiovascular side effects have been reported with the COVID-19 vaccines?
Rare cardiovascular side effects have been reported with both the mRNA and the adenovirus-vector COVID-19 vaccines. However, most of these adverse effects are actually seen more frequently due to COVID-19, rather than after the vaccine (Figure 1).1,2
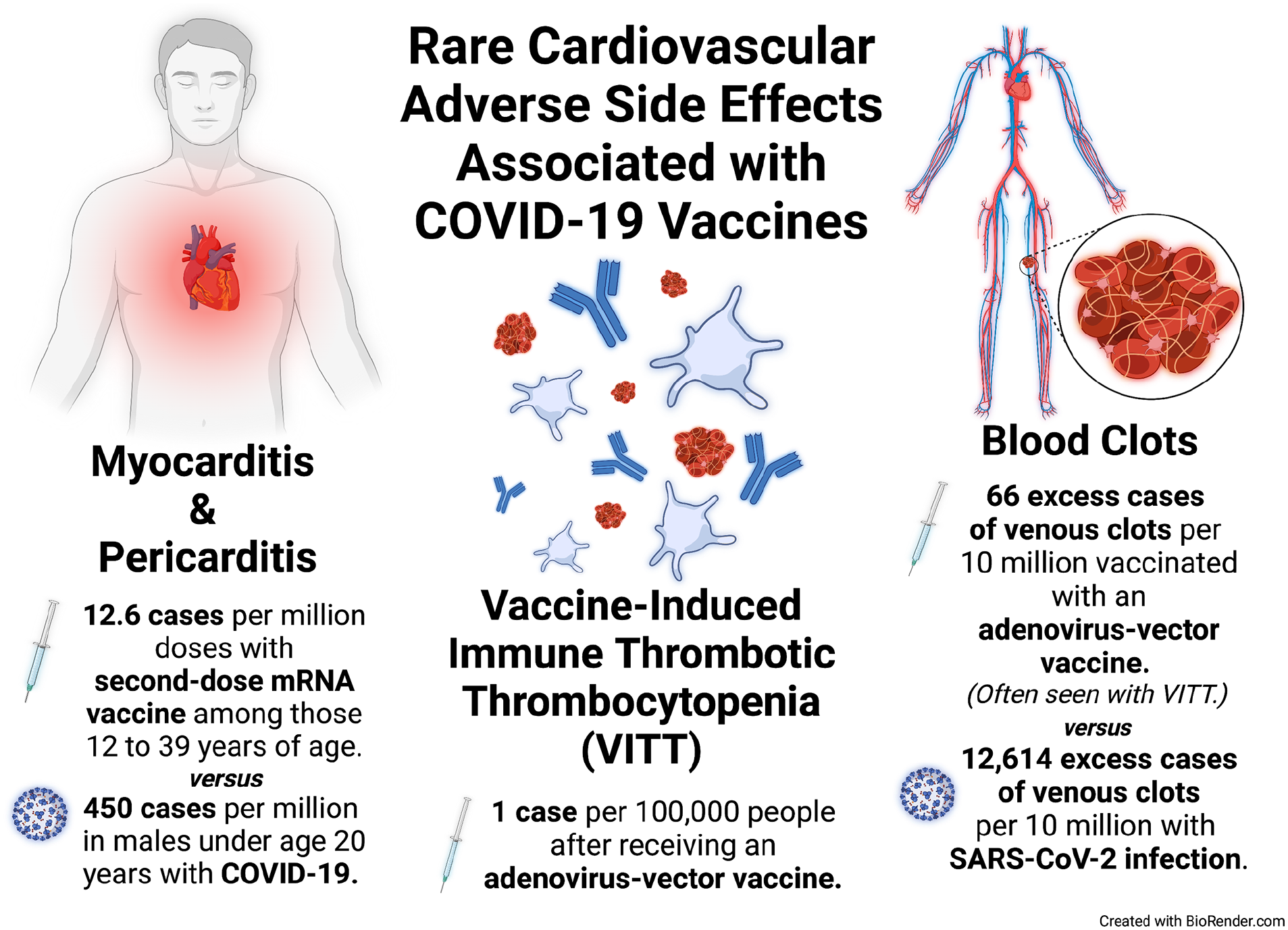
Occurrence of cardiovascular side effects associated with coronavirus disease 2019 (COVID-19) vaccines versus SARS-CoV-2 infection and COVID-19.
Adverse cardiovascular side effects of the COVID-19 vaccines include myocarditis/pericarditis, vaccine-induced thrombotic thrombocytopenia (VITT), and thrombosis (the formation of blood clots), which are often related to low platelet levels and VITT in this setting. 3 The exact causes of these conditions are not completely understood, but they are likely due to inflammatory immune system responses to components of the vaccines that attack cells and tissues in the body.4,5 These triggered immune system responses can make the blood cells clot more easily than usual and lead to low platelet levels or even inflammation of the heart (myocarditis/pericarditis). Blood clots (venous thromboses) can form in veins or arteries, in typical locations such as the blood vessels of the arms or legs, or even in less common locations like the blood vessels of the abdominal organs or brain.1,3
What is myocarditis/pericarditis and who is at risk?
The heart pumps blood to the rest of the body and is made up of a special type of muscle tissue called ‘myocardium’. The heart is covered by a protective layer of membrane called ‘pericardium’. The suffix ‘-itis’ refers to inflammation. Therefore, the terms ‘myocarditis’ and ‘pericarditis’ refer to inflammation of the heart muscle and its protective covering, respectively (Figure 2). Recent data have shown that both COVID-19 and the mRNA vaccines (Pfizer-BioNTech and Moderna) can lead to myocarditis/pericarditis due to a suspected inflammatory immune response, as described above. 4
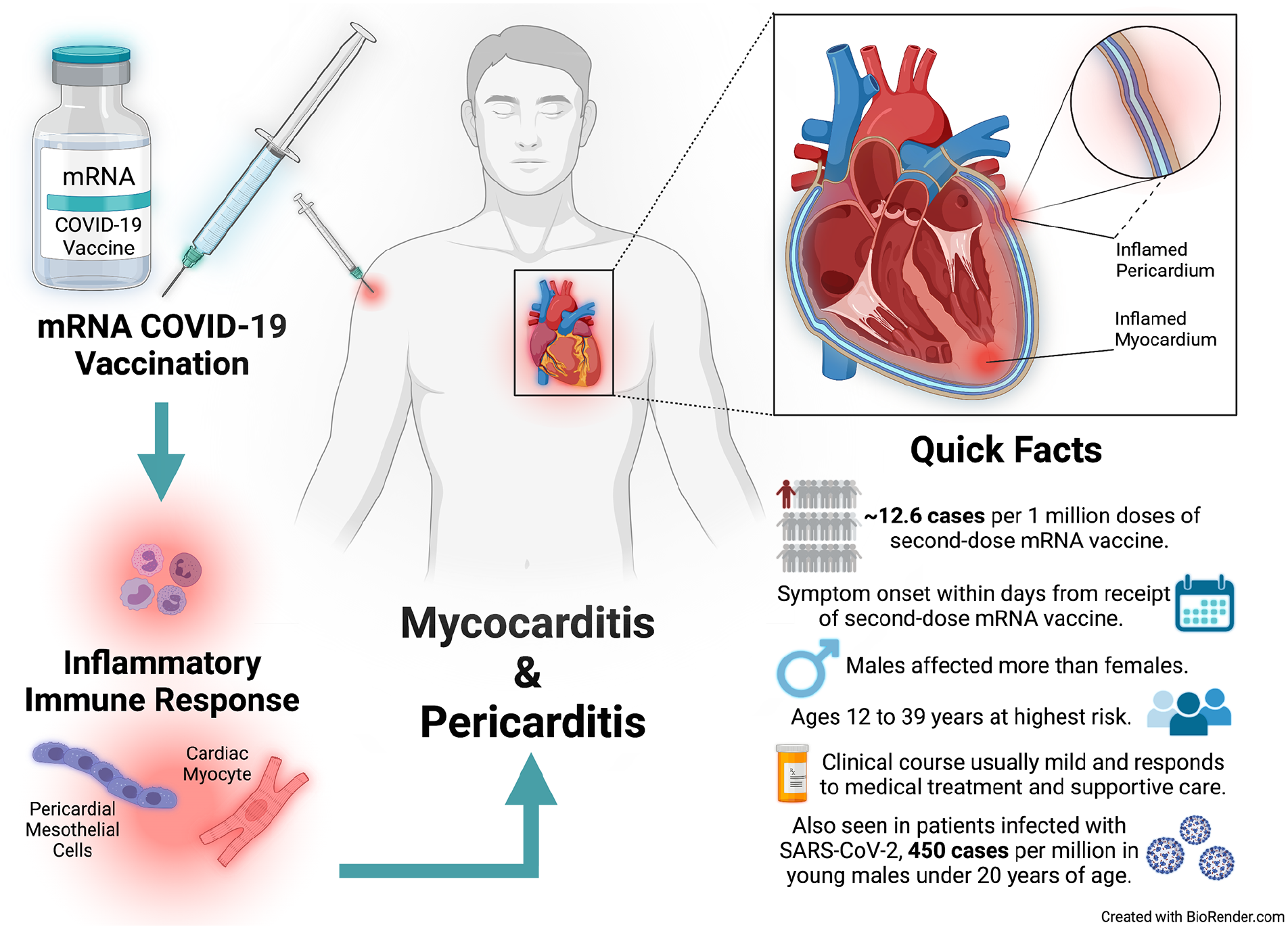
Summary of mRNA coronavirus disease 2019 (COVID-19) vaccine-associated myocarditis and pericarditis.
According to recent data, myocarditis/pericarditis were mostly seen after the second-dose mRNA vaccine but could be seen after the first dose as well. 4 Symptom onset was usually 2–3 days after the vaccine. 4 Chest pain was the most common symptom reported by most patients in the studies; fever, muscle aches, and fatigue were also reported. 4
A higher risk of myocarditis/pericarditis was seen among those 12–39 years of age, with males more likely to be affected than females. 4 Of note, myocarditis/pericarditis are rare adverse effects of the mRNA vaccines, with 12.6 cases per million doses of the second-dose mRNA vaccine among those 12–39 years of age. 4 Furthermore, myocarditis/pericarditis is more likely to be seen following COVID-19, with one study showing rates as high as 450 cases per million in males under 20 years of age. 2 Overall, vaccine-associated myocarditis/pericarditis is usually mild and responds well to medical treatment. 4 Those who are concerned that they may be experiencing myocarditis/pericarditis after a vaccine should seek prompt medical evaluation.
What is vaccine-induced immune thrombotic thrombocytopenia (VITT)?
Vaccine-induced immune thrombotic thrombocytopenia (VITT) is another rare, likely immune-mediated condition that generally occurs between 4 and 28 days after COVID-19 vaccination with an adenovirus-vector vaccine (the AstraZeneca and Johnson & Johnson vaccines).3,5 VITT is diagnosed in patients who have blood clots, low levels of platelets, an elevated blood marker of inflammation, and a positive blood test for an antibody called ‘antiplatelet factor 4 (PF4)’ in the weeks following an adenovirus-vector vaccine (Figure 3).3,5 Of note, patients with VITT can have blood clots in common locations such as the arms or legs (known as deep vein thrombosis or DVT), and they may also have blood clots in other locations, including the liver, gut, and brain (see below). 5 Recent data suggest that VITT occurs in about 1 per 100,000 people, and the majority of cases have been women under 50 years of age.3,5 VITT does not seem to be an issue among patients receiving the mRNA vaccines (Pfizer-BioNTech and Moderna).
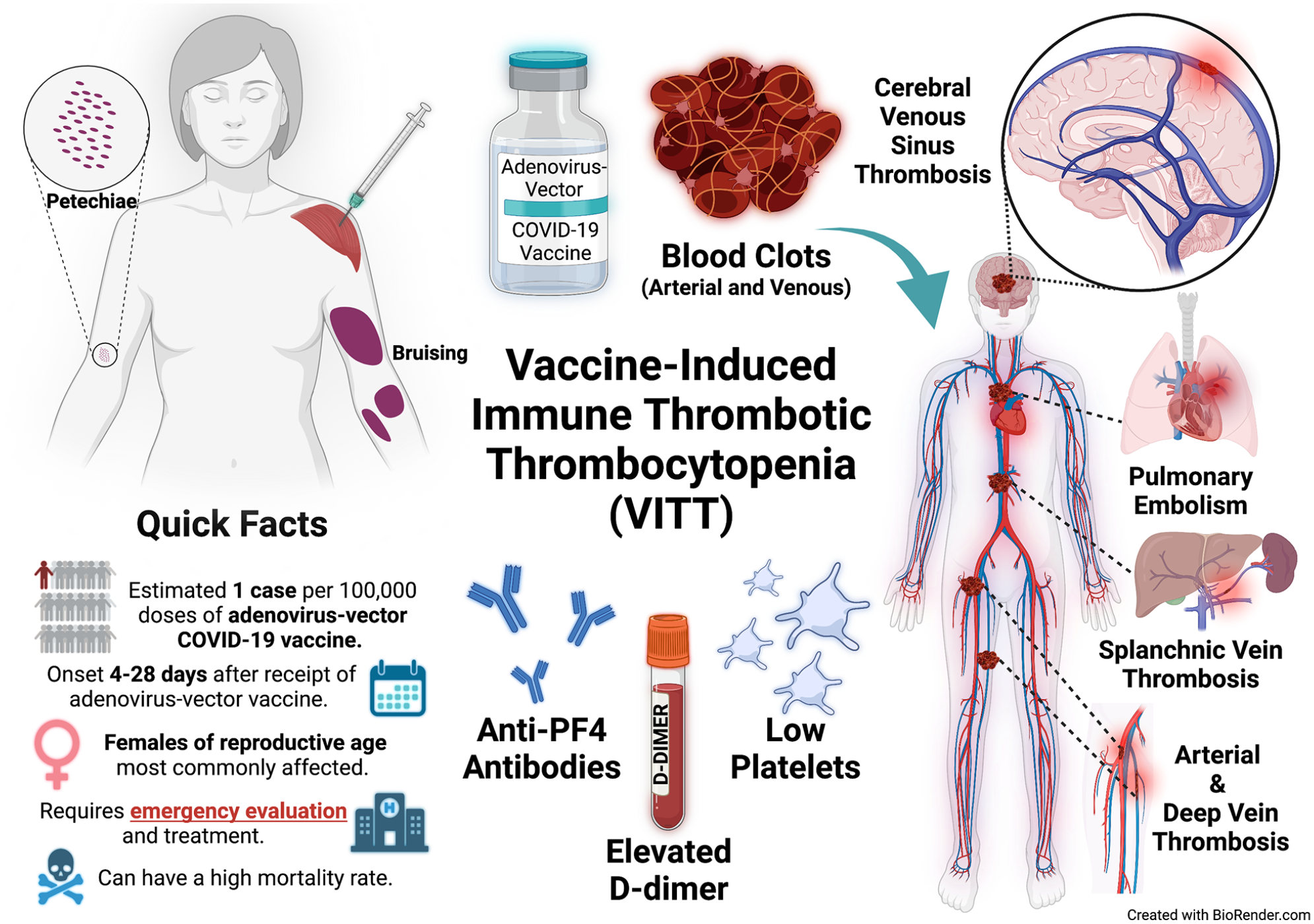
Summary of adenovirus-vector coronavirus disease 2019 (COVID-19) VITT, including cerebral venous sinus thrombosis.
Of note, symptoms that should raise concern for VITT in the weeks following an adenovirus-vector vaccine (AstraZeneca and Johnson & Johnson) include: ‘severe and persistent headache, abdominal pain, back pain, leg swelling or pain, and chest pain with or without shortness of breath’. 5 Owing to low platelet levels, some patients may also notice bruising or bleeding. 6 If there is suspicion for VITT, then in-hospital evaluation and treatment should not be delayed, as this condition can carry a high mortality rate even with treatment.3,6 Treatment generally consists of a nonheparin blood-thinning medication (for patients without bleeding and a sufficient number of platelets), intravenous immunoglobulins, and sometimes steroids. 5
What is cerebral venous sinus thrombosis (CVST)?
Cerebral venous sinus thrombosis (CVST) is a potential complication of VITT. 3 The cerebral venous sinuses are a network of blood vessels that drain blood and cerebrospinal fluid from the brain. 7 The term ‘thrombosis’ refers to the formation of a blood clot, or ‘thrombus’. If a blood clot forms in the cerebral venous sinuses and stops the flow of blood, then CVST occurs (Figure 3). Outside the context of COVID-19 and COVID-19 vaccination, CVST is a rare neurologic condition that is considered a type of stroke, though it only makes up about 1% of all strokes. 7 CVST has been seen in children, young adults, and females of reproductive age, with women three times more likely than men to develop the condition. 7 Overall, many factors may increase the risk of CVST, including infection, inflammation, trauma, and medications or conditions that make blood clot more easily. 7
More recently, CVST has also been seen in some patients with COVID-19 (207 cases per million), as well as in a smaller number of patients (0.9–3.6 cases per million, mostly women of reproductive age) who received an adenovirus-vector vaccine (the AstraZeneca and Johnson & Johnson vaccines) and who developed VITT. 8 CVST does not seem to be an issue among patients receiving the mRNA vaccines (Pfizer-BioNTech and Moderna).
Symptoms of CVST can vary depending upon the individual and the location of the blood clot. 7 The symptoms result from impaired drainage of blood and cerebrospinal fluid due to the clot, which can lead to brain injury and possible bleeding into the brain. Headache is the symptom most commonly reported and can be severe and occur suddenly. 7 Other symptoms of CVST can include seizures, vomiting, weakness, changes in mental status, eye pain, vision changes, and ringing in the ears. Overall, CVST, though rare, can be severe and can result in long-term neurologic symptoms or even death.
CVST of any cause is a life-threatening emergency that requires prompt medical evaluation. Once suspected, CVST can be diagnosed with imaging studies of the brain, generally by a specialized computed tomography (CT) scan and a magnetic resonance imaging (MRI) scan focused on imaging the veins of the brain. Typically, CVST requires close monitoring in a hospital setting and treatment with a blood-thinning medication.
What symptoms should prompt medical attention after the COVID-19 vaccine?
Mild symptoms can be treated at home with rest, hydration, and acetaminophen (Tylenol; paracetamol). These minor side effects should resolve within a day or two. More serious symptoms should prompt medical attention, including chest pain, shortness of breath, palpitations, leg swelling, persistent abdominal pain, easy bruising, stroke-like symptoms, blurry vision, or severe headaches.
Is there anyone who should not receive the COVID-19 vaccine?
There are exceptionally few patients who are not able to receive the vaccine. The only major reason not to be vaccinated is if a patient has a history of a severe allergic reaction (anaphylaxis) to one of the specific vaccine ingredients or if there was a prior immediate allergic reaction after a previous dose of a COVID-19 vaccine. In some countries, a history of prior CVST or other rare prior blood clotting events, heparin-induced thrombocytopenia, or antiphospholipid antibody syndrome with prior blood clots have been proposed as a reason to recommend preferentially receiving an mRNA vaccine (Pfizer-BioNTech or Moderna) rather than an adenovirus-vector vaccine (AstraZeneca and Johnson & Johnson).
Should patients with cardiovascular disease be vaccinated against COVID-19?
Patients with cardiovascular disease are at particularly high risk of complications from COVID-19, and vaccination will significantly reduce this risk. COVID-19 can lead to many different problems, including cardiovascular complications. The infection can lead to multiple blood clots, including DVT in the leg veins, or pulmonary embolism (PE) in which the DVT travels to the lungs. Blood clots can also occur in the arm or leg arteries, in the arteries of the brain leading to stroke, or in the arteries of the heart leading to heart attack.
Clotting is common with COVID-19 for multiple reasons. The virus may damage the blood vessels or the blood may clot more easily due to the infection. The clots may also be a result of the intense immune response to the infection. For more information, please see the Patient Information Page ‘COVID-19-related thrombosis’. 9 Vaccination can help to reduce the risk of these serious cardiovascular complications.
Summary
Rare cardiovascular complications have been reported with the COVID-19 vaccine. However, the frequency of these adverse events is significantly lower with vaccination than with COVID-19. For the large majority of people, including those with vascular disease, the benefits of the COVID-19 vaccine greatly outweigh any risks. Vaccination is safe and remains the best way to limit the spread of COVID-19 and to prevent severe infection.
The ‘Vascular Disease Patient Information Page’ is a regular feature of Vascular Medicine. All articles in the collection are available for free online at http://journals.sagepub.com/vmjpatientpage The Vascular Disease Patient Information Page is provided for educational purposes only and is not a substitute for medical advice
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Dr Alexandra Solomon currently holds the position of Anticoagulation Forum Ansell Fellow at Boston Medical Center. Boston Medical Center received an Anticoagulation Forum Ansell Fellowship Award, which was supported by an unrestricted educational grant from Janssen Biotech, Inc., administered by Janssen Scientific Affairs, LLC. Dr Elizabeth Ratchford’s work was supported in part by the generosity of David Kotick (1926–2021). Dr Jason Kovacic acknowledges support from NIH (R01HL130423, R01HL135093, R01HL148167), New South Wales health grant RG194194, and the Bourne Foundation.
