Abstract

In 1988, I joined the vascular medicine department at Cleveland Clinic 2 years after working in private practice as a hematologist and internist at a large community hospital in Michigan. I became the fifth member of a group that was aligned closely with four vascular surgeons. I had no formal training in vascular medicine other than minimal exposure during medical school and residency and, entering the department much like a first-year fellow in vascular medicine, I had to learn on the job.
I had wonderful mentors including two future presidents of our Society for Vascular Medicine (Drs Jess R Young and Jeffrey W Olin) and two other clinicians (William F Ruschhaupt and Robert A Graor), all experts in vascular disease. I quickly learned what I did not know – that was, how to properly perform a vascular-related history and physical exam. I was also reminded of the importance of observation and to not be satisfied solely on what one expected to find based on the history, but to search for the unexpected while performing the physical examination.
That was over 32 years ago, and the importance of a thorough vascular-related history and physical examination has not changed since. Then as now, these skills are not always adequately taught in medical school or residency.
The physical examination
Vital signs are taken by the nurse in my clinic. Blood pressure must be taken in both arms and a difference of 15–20 mmHg or more between the arms is considered significant. 1 I also note patients’ temperature, pulse rate, and weight.
Patients are placed in an appropriate-sized gown in a warm room to minimize cutaneous vasoconstriction. If patients refuse (a common reason is that none of their other doctors requires them to disrobe), I ask for permission to examine them as I explain how important a properly performed physical examination is to their diagnosis and management.
Inspection
The physical exam should progress from inspection to palpation, percussion and auscultation, with few exceptions. Owing to time constraints, I often begin the physical exam while continuing to obtain additional history or the review of systems.
I assess the patient’s overall appearance, looking for signs of vascular disease. Are they apprehensive or breathless? Do they have a prominent temporal artery or a pulsatile mass in the neck that might suggest temporal arteritis, an aneurysm, or carotid body tumor? Is there xanthelasma (on the eyelids or periorbital skin), xanthomas of the plantar or palmar creases or arcus senilis (in a younger individual) to suggest premature atherosclerosis and/or hyperlipidemia? Do they have café au lait spots (neurofibromatosis) or yellowish orange papules over the neck or flexural areas (pseudoxanthoma elasticum)?
I look for a discrepancy in the size of the patient’s arms (unilateral swelling that might suggest an axillary-subclavian deep vein thrombosis (DVT) or bilateral swelling implying superior vena cava syndrome). Do they have telangiectasias on the hands or blue or pale fingers suggesting a connective tissue disorder? Do they have tobacco stains on the fingers that might suggest underlying arterial disease?
Is there edema or atrophy of the leg or changes in the color of the skin (pallor, rubor, cyanosis, or livedo reticularis)? Do they have squaring of the toes or a dorsal hump to suggest lymphedema or evidence of corona phlebectatica to suggest chronic venous insufficiency? Are both legs swollen – a result of heart failure, kidney, liver or thyroid disease, inferior vena cava thrombosis, lipedema or a side effect from a medication? Do they have a pale limb with or without ulceration that might suggest peripheral artery disease (PAD) or blue and painful toes implying atheroemboli or a vasospastic phenomenon.
Palpation, percussion, and auscultation
Examination of the pulses and listening for bruits and palpating for thrills is essential to the vascular examination. Palpation and auscultation should be practiced routinely over all large arteries including the carotid, subclavian, abdominal aorta, iliac, and femoral arteries. One should compare the strength of the pulse on one side of the body to the contralateral side. It is important to remember that the pulse may be decreased or not palpable in a stiff calcified vessel or in patients with cardiac failure, hypotension, or an edematous limb.
There are different grading scales for reporting pulses, including: 0 to 2 (absent, present, or normal); 0 to 4 (absent, markedly, moderate or mildly impaired, or normal). I prefer the AHA/ACC recommendations of 0 to 3 (0, absent; 1, diminished; 2, normal; or 3, bounding). 1 A bounding pulse may suggest aortic valve regurgitation, arteriovenous fistula, or may be encountered during emotional states, thyrotoxicosis, or hypertension, whereas an expansile pulse may infer an aneurysm.
Bruits should be rated for severity, location, and quality (mild, rumbling, blowing, harsh, or high pitched) and palpated to determine if there is an associated thrill. Both sides of the stethoscope should be used, although the diaphragm is best for high-pitch and the bell is best for low-pitch sounds. It should also be noted if a bruit is systolic or extends into diastole, as this may indicate severe arterial stenosis, arteriovenous malformation, or an arteriovenous fistula.
Head and neck
I generally start my exam with the temporal arteries, located anterior to the tragus of the ear. They should be palpated for symmetry, strength, tenderness, and induration, the latter suggesting temporal arteritis.
I proceed next to the fundoscopic examination. An abnormal exam may point towards underlying hypertension, diabetes, or atherosclerosis, while the presence of a Hollenhorst plaque suggests atheroembolism.
I have not routinely listened for a bruit over the eyeball, but, if heard, it may indicate internal carotid artery stenosis, a carotid-cavernous fistula, or arteriovenous malformation. 2 Auscultation is best performed by placing the bell of the stethoscope over the closed eye while asking the patient to stare at a fixed point with the opposite eye. This helps to minimize potential sound produced by eyelid tremor. If heard, the bruit is generally high-pitched. 3
The carotid arteries should be palpated lower in the neck, between the trachea and in front of the sternocleidomastoid muscles. Gentle pressure should be applied when palpating in the elderly because too much compression near the carotid sinus may result in bradycardia and hypotension. 4
A carotid artery bruit due to a nonobstructive lesion may produce a short bruit whereas one might hear a faint or high-pitched bruit of longer duration signifying a high-grade obstructive lesion. Total obstruction of the carotid artery may produce no bruit.5,6 It is also important for the clinician to recognize that a carotid artery bruit may result from too much external compression on the artery by the examiner. 6
A common mistake made when listening to a bruit over the carotid or subclavian arteries is to not check the aortic and pulmonic valves to determine if the sound is transmitted from the heart. A murmur from aortic stenosis will usually be transmitted upwards into all neck vessels (subclavian and common carotid arteries) but the intensity of the murmur diminishes as the stethoscope is moved up the neck. It is also important to remember that a loud murmur of aortic stenosis may mask a softer bruit of carotid artery disease. 6 To determine if a bruit over the carotid artery is from subclavian artery stenosis, compress the subclavian artery while listening over the carotid artery. If the stenosis is in the subclavian artery, the bruit disappears. 4
Vertebral arteries are not palpable, but bruits should be listened for with the bell posteriorly to the sternocleidomastoid muscles just above its clavicular origin with the head in the neutral position as well as flexed, extended, and rotated to each side. Arteriovenous malformations are sometimes heard more posteriorly in the neck and occipital areas as well. 4
A venous hum can be mistaken for a cardiac murmur and may be heard best in the supraclavicular fossa with the patient upright. It is a continuous soft or low-pitch sound caused by turbulence in the internal jugular veins and results from high velocity blood flow. It is generally loudest in diastole and can be differentiated from arterial bruits by lightly compressing the external jugular vein in the neck, stopping venous flow and eliminating the sound. It may also be eliminated by a Valsalva maneuver. In adults, the hum may result from anemia, thyrotoxicosis, pregnancy, cirrhosis, peripheral arteriovenous fistula, or an intracranial arteriovenous malformation.6,7 Other pertinent clinical findings include dilated jugular veins suggesting congestive heart failure or an arteriovenous fistula.
Upper extremities
I examine the chest wall and upper arms for dilated veins that may suggest superior vena cava syndrome or axillary or subclavian vein thrombosis. I observe for acrocyanosis of the hand or clubbing, ulceration, splinter hemorrhages, or pale white or blue fingers. A single blue and painful finger might suggest a spontaneous venous hemorrhage.
Subclavian, axillary, brachial, radial, and ulnar arteries should be palpated for strength and the presence of aneurysms while auscultated for an arteriovenous fistula or stenosis.
Atherosclerosis commonly involves the subclavian arteries. In younger individuals, this involvement may be a result of Takayasu’s arteritis. The origin of the left subclavian artery is the most common site of upper-extremity arterial stenosis. It is best felt using the thumb with the fingers placed behind the neck and heard in the supraclavicular fossa between the clavicle and pectoral muscles. 4 The presence of a bruit over the subclavian artery, a reduced blood pressure compared to the contralateral arm, and a weakened ulnar or radial pulse in the setting of arm claudication suggests subclavian artery stenosis. If the patient complains of dizziness, gait disturbance, dysphagia, or dysarthria, then vertebral-basilar occlusive disease must be considered. Subclavian steal syndrome results from the combination of retrograde vertebral flow and neurologic symptoms in response to upper-extremity exertion.
The axillary artery is generally felt deep in the axilla. It may be more easily palpated by externally rotating and elevating the arm. It can be felt along the middle of the clavicle below the insertion of the pectoralis major muscle. 8 The appearance of a pulsatile mass may suggest an aneurysm.
Comparing the timing of the pulsation of the radial arteries should be performed routinely. A delayed pulsation of one radial artery may suggest subclavian stenosis, whereas simultaneous palpation of the radial and femoral arteries with a delay in the latter may suggest coarctation of the aorta.
The Allen test is used to evaluate the vessels of the palmar arch and is helpful in assessing the patient for Raynaud’s phenomenon, hypothenar hammer syndrome, or thromboangiitis obliterans. It is also useful to perform prior to harvesting the radial artery for use as an autograft for coronary artery bypass. There are several variations to the procedure. I ask the patient to make a tight fist, emptying most of the blood from his or her hand, then swipe my thumbs down to drain blood from the thenar and hypothenar eminences. Next, I occlude the radial and ulnar arteries with my thumbs. A normal result occurs when pressure on the ulnar artery is released and pallor of the palm is replaced by a rapid return of color or a reactionary rubor. The Allen test is considered positive (abnormal) when no filling is seen upon release of the ulnar artery, and a positive reverse Allen test indicates occlusion of the radial artery. A false positive test may occur if the patient unclenches his or her fist and hyperextends the hand or widely separates the digits. 4
A digital Allen test has been described to determine if either or both digital arteries to a finger are occluded. 9 Ashbell and colleagues described the procedure where both digital arteries are compressed at the base of the fingers being examined. The patient flexes his or her fingers several times while the hand is elevated. Release of one side will result in continued pallor if arterial inflow is obstructed, and prompt return of color suggests the artery is patent (Figure 1). This procedure is repeated for the other digital artery.
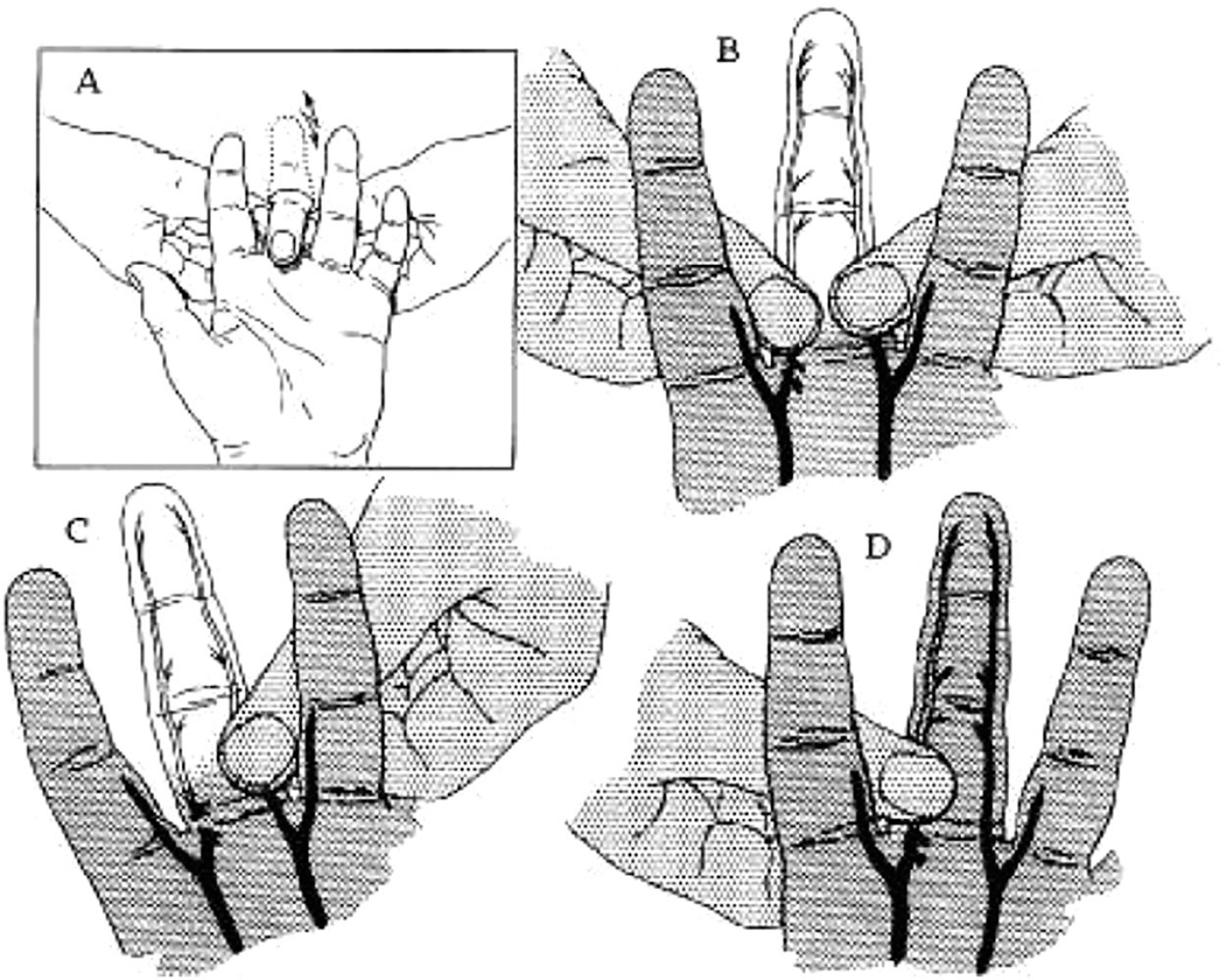
The digital Allen test. (
There are several maneuvers used to evaluate patients for thoracic outlet syndrome while recognizing any of the tests may be positive in asymptomatic individuals. I use the elevated arm stress test (EAST). It is performed by abducting and externally rotating the arms to 90 degrees as the patient repeatedly opens and closes his or her hands for 3 minutes. If the patient’s symptoms are reproduced, it is considered to be a positive test. Other commonly used exams include the costoclavicular maneuver (exaggerated military position), Adson’s maneuver (rotation and extension of the head with arms on the lap), or hyperabduction maneuver. These are considered positive with reduction or loss of the radial pulse and reflect compression of the axillary or subclavian artery in these extreme positions.
Abdomen
The abdomen should be inspected for surgical scars, engorged veins, striae or visible pulsating masses. Pulsation of the aorta may be a normal finding in very thin individuals. The abdomen should be auscultated before percussing and palpating using the bell to pick up vascular bruits over the aorta, mesenteric, renal, or iliac arteries. Abdominal bruits may be a common finding in older individuals but can result from stenosis of the aorta, renal, or mesenteric arteries. A systolic/diastolic bruit of the renal arteries may indicate severe stenosis, an arteriovenous fistula or aneurysm. These are best heard above and to the left of the umbilicus.
The abdominal aorta should be palpated (with warm hands) just to the left of the midline in the area between the umbilicus and costal margin. Asking the patient to raise his or her knees will help to relax the abdomen. I use both hands (palms down), placing an index finger on either side of the pulsating aorta to measure the aortic width. This may be difficult in obese patients or those with a low pain threshold, so it may be easier to probe one side of the aorta at a time.10,11 An aneurysm is considered to be present if the aorta is judged to be 3.0 cm or greater in diameter. There are no known risks associated with palpation of the abdominal aorta and no reports of aneurysm rupture attributed to physical examination. 11 The presence of tenderness over an abdominal aortic aneurysm is a warning sign that may indicate impending rupture or an inflammatory aneurysm. The right and left lower quadrants should also be evaluated for iliac artery bruits and aneurysms as well. The finding of large, tortuous, or dilated veins on the abdomen may indicate inferior vena cava thrombosis or occlusion.
Lower extremities
The legs should be examined with the patient supine, inspecting for discoloration (hemosiderin deposition, erythema, or pallor) while using the back of the hand to evaluate for temperature changes. A loss of the fat pad on the heel or calf muscle wasting may suggest chronic limb ischemia. The absence of hair on one limb only may suggest PAD if the other is normal. 8 In addition, I look for surgical scars from previous vascular procedures or amputations.
The location of an ulcer helps to determine if it is arterial, venous, or diabetic. Venous ulcers are generally located along the medial malleolus or distal medial calf. Arterial ulcers are commonly found on bony prominences such as the first and fifth metatarsal heads and heel. Neurotrophic ulcers are usually found on points of pressure, especially the plantar foot surfaces under the metatarsal heads or heel and may be surrounded by callus formation. It is also important to look for cracks or fissures in the skin and between the toes for kissing ulcers. 12 Motor function should be assessed by plantar and dorsiflexion of the foot as well as flexion and extension of the knee and hip. Evaluation for a neuropathy should also be part of the examination, and I commonly use both a tuning fork and a monofilament.
Varicose veins are best evaluated with the patient standing. 13 They are superficial veins that are dilated, tortuous, and generally greater than 3 mm in diameter. The presence of unilateral capillary malformations, limb hypertrophy, and venous malformations suggest Klippel–Trenaunay syndrome. A superficial thrombophlebitis will present as a linear, indurated, tender, and erythematous subcutaneous cord. Lower-extremity hemosiderin deposition, eczema, and a brawny appearance on the pretibial calf along with fibrosis is consistent with chronic venous insufficiency. Additional venous insufficiency clinical findings include corona phlebectatica (ankle flare sign) found along the medial malleolus or the circular and ivory-white atrophic skin surrounded by dilated red capillaries known as atrophie blanche. Advanced chronic venous insufficiency, referred to as lipodermatosclerosis, is recognized by its resemblance to an inverted bowling pin or champagne bottle. It results from subcutaneous atrophy lending to a concave appearance in the mid to distal calf. 13
Lymphedema will present with pitting edema (more likely in the early stages) and a Stemmer’s sign (inability to pinch the skin at the base of the second toe). Additional clinical findings of lymphedema include: squaring of the toes, toenails that are hypoplastic with up-turned concavity (referred to as ‘ski-jump’ toenails), exaggerated skin creases at the base of the toes, and the presence of a dorsal hump. 14 Peau d’orange, hyperkeratosis, papillomatosis, and cobblestoning are seen in more advanced stages of lymphedema.
The femoral, popliteal, dorsal pedis (DP), and posterior tibial (PT) pulses should be palpated. The femoral pulse is deep below the groin crease and may be difficult to feel in the obese individual. Rotating the leg laterally and pulling back the pannus may help. 15 Absent or diminished femoral pulses may suggest PAD including Leriche syndrome, coarctation of the aorta, or a dissecting aneurysm. If unable to feel a femoral pulse, an attempt should be made to palpate the external iliac artery that lies directly below the inguinal ligament.
The popliteal artery is generally considered the most difficult artery to palpate. I ask the patient to flex his or her knees slightly while using my index and middle fingers to palpate deep into the popliteal space as my thumbs apply pressure on top of the knee. A common mistake is to bend the knees too much or not palpate deep enough. If unable to palpate the artery, another approach I use is to have the patient lie face down with the knee flexed, holding the foot at the ankle. For the left leg, I hold the foot with my right hand and palpate with my left thumb. The opposite is done for the right leg. 17 If the pulse is widened with either procedure, it suggests an aneurysm.
The feet must be examined with socks off. The PT artery has a variable course posterior to the medial malleolus. The right PT pulse is best felt using the left hand and the left PT pulse is best felt using the right. Absence of the PT pulse suggests underlying PAD. 16 The DP arteries are usually found along the second metatarsal bone but may be congenitally absent in up to 12% of the population.15,16 It may be easier to feel if one lifts the great toe upward or slightly dorsiflexes the foot.18,19 If I cannot feel a DP pulse, I check for the anterior tibial pulse located more superficially at the lower third of the leg, just above the ankle.
McGee and Boyko described physical findings that may help clinicians consider the diagnosis of PAD beyond a lack of pedal pulses. They observed that a unilateral cool extremity, femoral bruit, or prolonged venous filling time were helpful. They also described several other physical signs helpful to determine the extent and distribution of vascular disease including an abnormal femoral pulse, lower-extremity bruits, warm knees, and a positive Buerger test (described in detail below). 20 Gaylis described the ‘warm knee sign’ in three patients with occlusion of their popliteal and/or their lower femoral arteries. He felt the increased warmth was a useful clinical sign for locating the obstruction, reasoning that this finding was due to increased blood flow through collateral vessels accompanied by an increase in local heat production. 21 He advised that the sign not be sought immediately after removing clothes or a blanket, but rather to first expose the limb at room temperature for about 10 minutes. O’Brien et al. also felt that finding the warm knee sign on a symptomatic patient with PAD was a valuable indicator of peripheral arterial occlusion or stenosis ending at the level of the adductor hiatus. 22
There are several exercises used to assess for severe limb ischemia in the patient with presumed severe PAD. A cold, pale, or mottled cyanotic foot noted while the patient is supine is indicative of critical PAD, but if the findings are less obvious, the Buerger test can be helpful. It is undertaken with the patient supine and the legs elevated approximately 60 degrees above the examining table for 2 minutes. The patient is then instructed to assume a sitting position and the legs dangled over the table for an additional 2 minutes. Patients with a positive Buerger test result have pallor with elevation, and the toes and foot will have a dusky red flush or rubor with dependency.20,23,24 There are modifications to this test including the angle of elevation or addition of exercise by asking the patient to flex and extend his or her ankles and toes while looking for pallor on the plantar surface, indicating severely impaired arterial flow.8,24 An approach I use is to elevate the legs above the patient’s heart and ‘milk’ the color from his or her feet by gentle pressure. Persistent pallor after 30 seconds suggests severe PAD. Following elevation, the patient assumes a sitting position with the extremities dependent and I observe for rubor. 4 For the frail patient, one may need to support the legs in this position.
A test that can be performed in conjunction with the Buerger test evaluates the veins on the dorsum of the foot. During leg elevation, the veins on the dorsum of the foot will collapse completely. When the patient is placed back in the sitting position, the time it takes for the veins on the dorsum of the foot to fill is recorded. This is referred to as the venous filling time, also known as Collens–Wilensky test. If there is normal arterial circulation, the veins will fill within 7–15 seconds; moderate obstructive arterial flow is likely if the veins fill slower (30–45 seconds), and severe obstruction if it takes 90 seconds.12,15
The capillary refill test was more commonly used in the past by physicians to assess circulation in the foot. It can be performed by firm digital pressure applied to the plantar skin of the great toe for 5 seconds. It is considered abnormal if it takes longer than 5 seconds for normal skin color to return. According to publications of McGee et al. and Pickering, the latter dating back to 1933, the capillary refill test is of questionable value.19,20,25
An ankle–brachial index (ABI) should be performed on all patients with suspected PAD. It is a simple, inexpensive, reproducible approach to diagnose PAD and easily done at the bedside or in the office. The only requirements are a sphygmomanometer, acoustic gel, and a hand-held continuous wave Doppler. The ratio of the systolic blood pressure at the ankle is compared to the brachial artery and is considered normal if the ABI is 1.0 to 1.40, borderline at 0.91 to 0.99, and abnormal if less than 0.91. 1 An ABI less than 0.40 suggests critical limb ischemia.
Conclusion
In today’s medical climate, there is often less time to perform a comprehensive history and physical exam, not only because it is time-consuming, but also because of administrative and financial requirements. This may be a result of more patients and the additional time spent with the electronic medical record (EMR).
Doctors may also shy away from making an educated guess on the basis of what they see and hear in part because of the fear of a lawsuit if their diagnosis is incorrect or peer pressure if they are wrong. 26 Furthermore, the advances and ready availability of ultrasound technology make it so much easier to order an imaging study to confirm their suspected diagnosis without relying on the physical examination.
Patients may also demand to be tested by newer imaging technologies because of information they have reviewed on the intranet or heard from other patients. 27
There may be other reasons for less emphasis placed on the history and physical exam. For instance, there may be fewer skillful clinicians as mentors or an impression that the physical exam will not change a patient’s management. Yet, there is so much information that can be derived from this exercise and we in vascular medicine know how important the history and physical is to make a correct vascular disease diagnosis. In addition, a properly performed history and physical exam may lead to increased patient trust, thus enhancing the doctor–patient relationship. 26 It is also cost-effective, can be performed anywhere, and thus is more easily used to make serial observations. It has also been shown to have a significant effect on the patient’s outcome. 28
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
