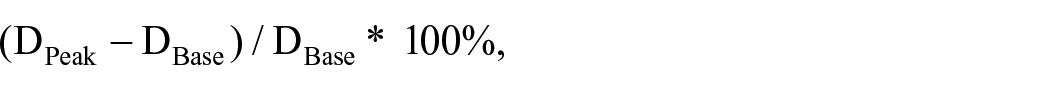Abstract
Exposure to prolonged sitting increases blood pooling and the risk of lower-extremity vascular arterial and venous complications, including deep vein thrombosis. Compression garment stockings (CGS) may prevent pooling, thereby mitigating the associated vascular complications. Three aims were addressed: (i) does use of CGS help to prevent blood pooling; (ii) does blood pooling correlate with decreased stroke volume; and (iii) does use of CGS preserve leg arterial flow-mediated dilation and reactive hyperemia response. Twenty inactive participants (22 ± 4 years, 30% female, 22.1 ± 2.0 kg/m2) were randomized to sit for 3 hours with (CGS) and without use of CGS. Blood pooling was determined using medial gastrocnemius total hemoglobin. Stroke volume was estimated using finger photoplethysmography, respectively. Macrovascular and microvascular function were measured using popliteal artery flow-mediated dilation and reactive hyperemia, respectively. In response to 3 hours of sitting: (i) there was an interaction effect for total hemoglobin (p < 0.001); the condition without use of CGS increased 10.5% (95% CI: 6.7 to 14.3) and CGS increased 4.3% (95% CI: 0.1 to 7.7); (ii) blood pooling was negatively associated with stroke volume (r = −0.58, 95% CI: −0.68 to −0.45); (iii) reactive hyperemia and flow-mediated arterial dilation was impaired after prolonged sitting, and use of CGS was not associated with attenuation of this impairment. Use of CGS decreases blood pooling but does not preserve arterial macrovascular and microvascular responses to prolonged sitting. Further study is warranted to determine whether CGS has additive benefits when combined with sitting interruption strategies.
Introduction
High levels of sedentary behavior, particularly regular prolonged exposure to sitting, may increase the risk of lower-extremity vascular arterial and venous complications, including deep vein thrombosis (DVT).1–3 For example, the risk of DVT is four times higher in older adults with a sedentary lifestyle, 1 and 2.8 times higher in younger and middle-aged adults who sit for prolonged periods at work. 2 Considering one in 12 middle-aged adults will develop DVT in their lifetime, 4 and that the prevalence of sedentary behavior is increasing, 5 identifying simple strategies to mitigate the risks associated with repeated exposure to prolonged sitting is important. One strategy is the use of simple physical activities such as walking breaks to interrupt prolonged sitting, which has been reported to decrease or prevent impaired leg arterial function. 6 However, such strategies may not be suitable for use in all contexts (e.g. some work environments), or for all populations (e.g. those confined to bedrest or wheelchairs). A strategy which may compliment or serve as an alternative to physical activities is the use of compression garment stockings (CGS).
For both venous and arterial complications, an important contributory factor may be blood pooling in the lower extremities. 7 Blood pools due to increased hydrostatic pressure, and is augmented when the muscle pump is not regularly activated to aid venous return. 8 On the venous side, the blood pooling results in venous stasis, which increases the risk of blood clotting and subsequent DVT. 9 On the arterial side, the decreased venous return compromises stroke volume 10 and leads to decreased leg blood flow-induced shear stress, the stimulus for maintaining endothelial function. 11 Indeed, a meta-analysis of 17 studies (n = 269) indicated that uninterrupted sitting decreased leg flow-mediated dilation (FMD), a measure of macrovascular endothelial function, by 2.4%. 12 Additionally, prolonged sitting reduced leg microvascular function, assessed using reactive hyperemia, by ~40–90%. 13 Minimizing blood pooling may reduce the negative effects of prolonged sitting on the lower-extremity venous and arterial systems.
Activating the muscle pump is likely the most effective strategy for preventing blood pooling. However, as discussed above, this strategy may not always be a viable option. Alternatively, the use of CGS attenuated blood pooling compared to a control condition (~165% vs ~71% increase, respectively) when young, health males were asked to stand. 14 Additionally, wearing CGS while standing mitigated the decrease in stroke volume compared to standing without CGS. 15 However, no studies have investigated whether wearing CGS mitigates lower-extremity venous and arterial complications when inactive adults sit for a prolonged period. Therefore, the aim of the present study was to investigate the effects of uninterrupted prolonged (3 hours) sitting, with CGS and without CGS (CON), on the lower-limb venous and arterial system. More specifically, three specific aims where addressed: (i) does the use of CGS help to prevent blood pooling (increased medial gastrocnemius hemoglobin); (ii) does blood pooling correlate decreased stroke volume; and (iii) does the use of CGS preserve lower-limb macrovascular (popliteal artery FMD) and microvascular (popliteal artery reactive hyperemia) function.
Methods
This study is reported in accordance with CONSORT (Consolidated Standards of Reporting Trials) guidelines. 16 All study procedures were approved by the ethical committee of the Mount Fuji Research Institute and were performed in accordance with the guidelines of the Declaration of Helsinki (ECMFRI-02-2017). All participants provided written informed consent prior to participating in the study.
Participants
We recruited healthy men and women between the ages of 18 and 30 years. Exclusion criteria included pregnancy, regular engagement in moderate-to-vigorous physical activity such as jogging, running, stationary bike use (120 ⩾ min/week), current smoker, any known cardio-metabolic disorders, or use of medications known to affect cardiovascular health.
Experimental design
As shown in Figure 1, this study was a randomized, controlled cross-over trial with two experimental conditions: prolonged (3 hours) sitting (i) without use of CGS (CON), and (ii) with use of CGS. Participants were stratified by sex and randomized to CON or CGS using a lottery. Participants reported to the laboratory on three occasions: one familiarization visit and two experimental visits. A minimum of 2 days separated visits to minimize potential carryover effects (e.g. blood pooling in the lower limbs), with a maximum of 4 days to minimize within-subject variation.
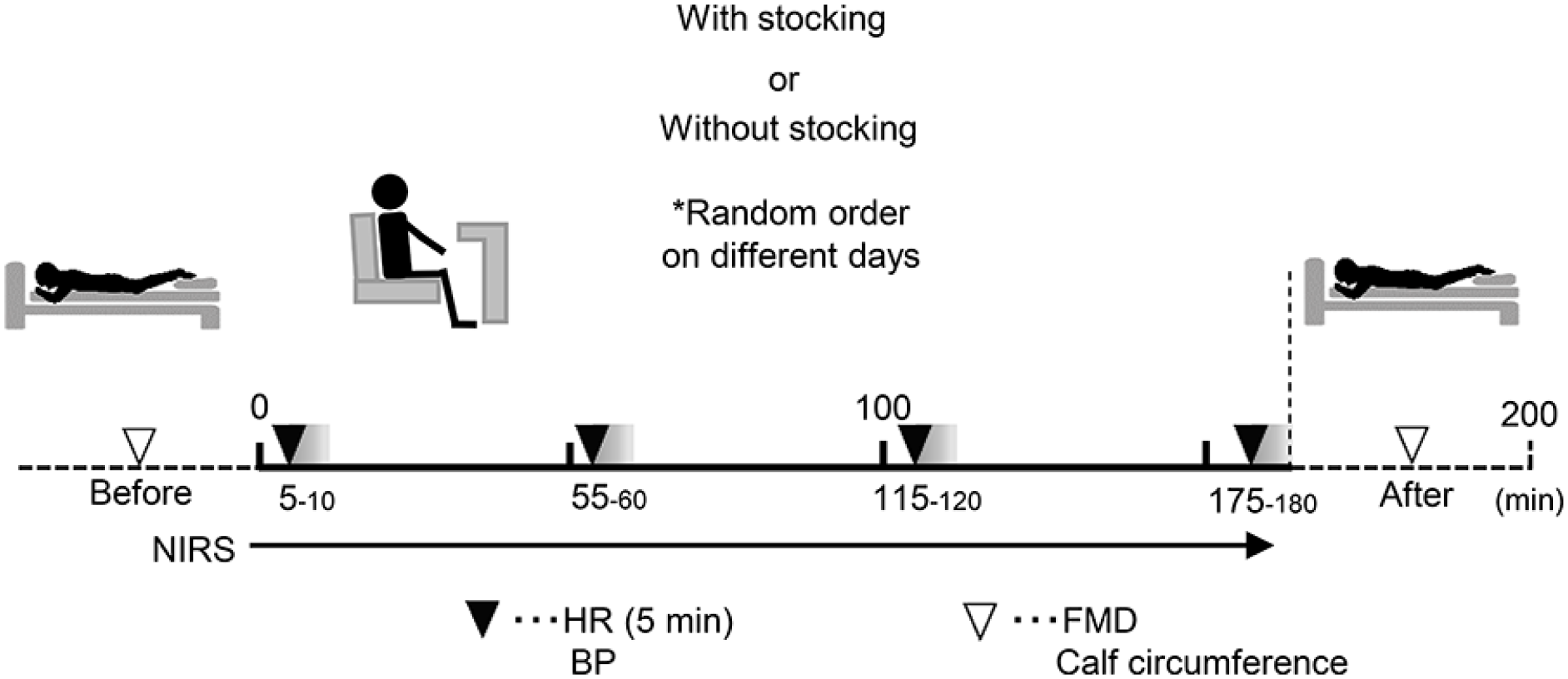
Experimental protocol.
For the experimental visits, participants arrived between 07:00 and 11:00, and at the same time for between-visits. Participants were asked to fast for 12 hours, consuming only water, and having refrained from supplement intake that morning. Additionally, participants were asked to avoid strenuous physical activity and alcohol for 24 hours prior to experimentation. Experimental procedures were conducted in a quiet, environmentally controlled room. After being asked to empty their bladder, participants were asked to rest supine for at least 20 minutes while being connected to measurement devices to assess heart rate and beat-to-beat mean arterial pressure (MAP). Following instrumentation, calf circumference, popliteal artery FMD, and reactive hyperemia were assessed in the prone position in accordance with a published report. 17 Subsequently, participants were positioned in a comfortable chair and near infrared spectroscopy (NIRS) (BOM-L1TRW; Omegawave, Tokyo, Japan) probes were attached to enable continuous assessment of blood pooling.18–20 Participants remained in the seated posture for 3 hours. They were allowed to use their arms to read or use a laptop, but their feet were placed on a non-slip mat to help keep feet in place and avoid muscle contraction. Study personnel monitored the participants to ensure they remained seated and did not fidget.
Following the sitting bout, calf circumference was re-assessed and then the participants were returned to the bed and placed in the prone position. The participants were carried by the study personnel to the bed in order to avoid leg muscle contraction and to maintain the sitting-induced effects on hemodynamics. Following 5 minutes of quiet rest, popliteal FMD was re-assessed.
Compression garment stocking
The length of the stocking was from the top of the instep to below the knee. The applied pressure of the graduated compression stockings was 29 hPa at the ankle and 20 hPa at the calf. Several sizes of stockings were prepared, and an appropriate garment was selected for each participant according to their ankle width.
Experimental measures
Aim 1: calf blood volume
Blood pooling was assessed using the total hemoglobin (tHb) and deoxygenated hemoglobin (HHb) signals from continuous-wave NIRS (BOM-L1TRW; Omegawave)7,21 and by measuring calf circumference. Bilateral calf circumference was measured at the widest point of the calf using a non-elastic tape, with the participant in the upright seated position, and marked for repeat measurements. The NIRS probe was placed unilaterally along the vertical plane of the central belly of the medial gastrocnemius at the position of maximum circumference,18–20 proximal to the CGS. The location was marked, and between-visits participants were asked to re-mark themselves each day to ensure identical site placement. Subcutaneous adiposity and muscle thickness at this site were assessed using B-mode ultrasound (Logic-E; GE Healthcare, Tokyo, Japan).
The continuous-wave NIRS instrument emits infra-red wavelengths of 780, 810, and 830 nm to detect relative tissue levels of oxygenated and deoxygenated hemoglobin (HHb), the total of which is tHb. Whereas tHb reflects blood pooled on both the arterial and venous, the predominant continuation to HHb is likely the venous compartment. 22 The three wavelengths were emitted from two transmitters at 2 cm (detector 1) and 4 cm (detector 2) from the photodiode detector, allowing for a theoretical penetration distance of up to 2 cm. 23 The hemoglobin concentrations received by detector 1 were subtracted from those received by detector 2 to minimize the influence of skin blood flow. 24 A path-length factor of 4.0 was used to correct for photon scattering within the tissue. 20 Considering the device uses continuous-wave NIRS, the scattering of light in tissue cannot be measured and therefore only relative changes in hemoglobin concentration can be detected. 25 NIRS values are presented as percentages from the resting baseline. 20 The use of continuous-wave NIRS for measuring blood pooling is reliable (intraclass correlation coefficients: 0.75). 7
Aim 2: central hemodynamics
Heart rate was measured continuously using a standard lead II electrocardiogram (Daily Care BioMedical, Chungli, Taiwan) and beat-by-beat MAP was determined using finger photoplethysmography (MUB-101; MediSens Inc., Tokyo, Japan) placed on the middle or index finger of the left hand 20 at a sampling frequency of 200 Hz using rate of using a commercially available analog-to-digital converter (es8; TEAC, Tokyo, Japan). Additionally, stroke volume was estimated using the Modelflow method 26 and cardiac output was calculated by multiplying stroke volume by heart rate.
Aim 3: leg arterial flow-mediated dilation and reactive hyperemia response
To evaluate FMD and reactive hyperemia responses, a custom-made tourniquet was placed approximately 5 cm distal to the popliteal fossa on the nondominant leg and inflated to 220 mmHg using a custom device. Popliteal artery diameter and shear rate was measured using brightness-mode imaging and pulsed Doppler waveforms, captured using a 10 mHz linear array probe (Logic-e; GE Healthcare). The probe was placed distal to the popliteal fossa while the participant rested in the prone position, with the leg being held in place using a vacuum pack to prevent movement (Vacform; Muranaka Medical Instruments Co. Ltd, Osaka, Japan). The probe placement position was marked within and between-visits and participants were asked to re-mark themselves each day to ensure identical probe placement. The insonation angle was kept constant between 45° and 60° and the sample volume included most of the vessel. Using a commercial video capture device (AMCap; Microsoft, WA, USA), recordings were made for 2 minutes at baseline and then from 30 seconds prior to tourniquet deflation and continued for 3 minutes post-tourniquet deflation. The videos were analyzed offline using custom-designed edge detection and wall-tracking software (version 2.0.1, S-13037, Takei Kiki Kogyo, Japan). 27 FMD (%) was calculated as:
where DBase is the mean diameter averaged over 2 minutes preceding the cuff inflation period, and DPeak is the peak diameter recorded in response to reactive hyperemia.
For microvascular reactivity, area under the curve (AUC) means hyperemic blood flow (blood flowAUC) and shear rateAUC were calculated from cuff release to the point of peak diameter to capture the entire hyperemic response above baseline. AUC measures were calculated using the sum of trapezoids method. Blood flow (mL/min) was calculated as (3.14 × mean velocity × [artery diameter/2] 2 × 60) and shear rate (s−1) as (4 × mean velocity/artery diameter). Resting blood flow is also presented.
Sample size
Previously, we reported that the typical error for change in the tHb signal is 4 μM. 7 To estimate the sample size required to detect the smallest detrimental (or beneficial) effect in a cross-over study 28 with a Type I error rate of 0.05 and 80% power, approximately 18 participants are required to detect a small (2.5 μM) change in tHb. Twenty participants completed the experimental protocol to account for potential missing data.
Statistics
Statistical analyses were performed using the R programming language (RKWard, version 0.7.1; KDE e.V., Berlin, Germeny). Only participants who had complete data for the primary outcomes were included in the analyses. Raw data are presented as mean (SD) and mixed model data are presented as mean (95% CI). Effect size (ES) was calculated as Cohen’s d where ⩽ 0.2, 0.2, 0.5, and 0.8 were defined as trivial, small, moderate, and large. 29 Correlation (r) estimates of 0.1, 0.3, and 0.5 were defined as small, medium, and large, respectively. 29
To test Aim 1, the effects of time (pre vs post) and condition (CON, CGS) on tHb, HHb, and calf circumference were analyzed using linear mixed models, with random effects of time (slope) and fixed effect of condition. To limit within-subject variance, the models were adjusted for baseline values as specified by Kenward and Roger. 30 To test Aim 2, the effects of time (10, 60, 120, 180 min) and condition (CON, CGS) on central hemodynamic variables were analyzed using linear mixed models, as described above. Intra-individual associations (i.e. change in one measures vs change in the other measure measure) between tHb and HHb with heart rate and estimated stroke volume were analyzed using rmcorr package for R. 31 To test Aim 3, the effects of time (pre vs post) and condition (CON, CGS) on FMD, diameter change (∆D), and blood flowAUC were analyzed using linear mixed models, as described above. Additionally, to control within-subject variation, ∆D was adjusted for DBase and shear rateAUC. The adjustment for DBase accounted for allometric scaling (i.e. the known dependence of FMD on baseline diameter)32,33 and shear rateAUC accounted for the reactive hyperemic shear rate response, which is the vasodilatory stimulus. 34
Results
We recruited 20 adults (30% female) aged 22 (SD: 4) years with a body mass index of 22.1 (SD: 2.0) kg/m2, MAP of 77 (SD: 9) mmHg, and heart rate of 70 (SD: 6) bpm. All participants successfully completed both experimental trials, and there were no missing data.
Aim 1: blood pooling
Compression garment stockings (CGS) decreased the blood pooling response to prolonged sitting (interaction effect p < 0.001). tHb increased 4.3% (ES = 0.20) for CGS versus 10.5% (ES = 0.44) for CON (Figure 2A), and HHb increased 6.8% (ES = 0.17) for CGS versus 14.9% (ES = 0.38) for CON (Figure 2B). We also measured calf circumference, which decreased by 0.6% for CGS (∆ = −0.24 cm, 95% CI: −0.40 to −0.07; ES = 0.28) and increased by 2.3% for CON (∆ = 0.84 cm, 95% CI: 0.68 to 1.01; ES = 1.01).
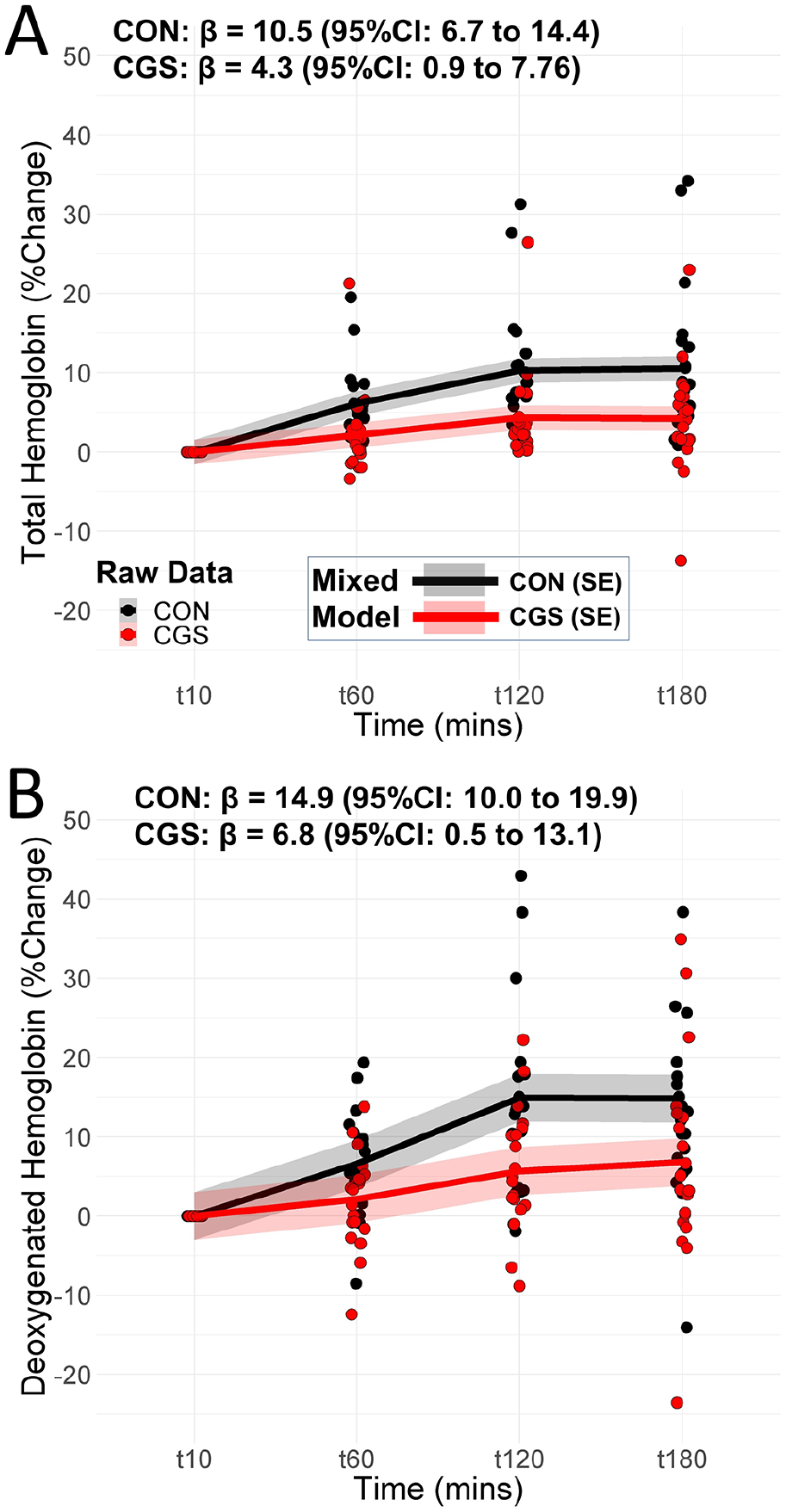
(A) The tHb and (B) HHb responses to prolonged sitting (3 hours) for CON and CGS groups.
Aim 2: relationship between hemoglobin concentrations and central hemodynamics
There were no statistical baseline differences for the central hemodynamic variables reported in Table 1. The MAP increased by 3.5% with time, but the difference was nonsignificant (p = 0.710) between conditions. However, CGS resulted in a 1.3% increase in estimated cardiac output (CO) compared to CON (0.08 L/min, 95% CI: 0.01 to 0.15; ES = 0.17). The higher estimated CO for CGS was driven by the greater preservation in estimated stroke volume, with estimated stroke volume decreasing 7.6% (−7.1 mL, 95% CI: −10.2 to −3.9; ES = −0.36) for CGS versus 14.8% (−13.7 mL, 95% CI: −16.8 to −10.5; ES = −0.61) for CON. There was a large association between tHb with SV (r = −0.58, Figure 3A), and a moderate association between tHb and HR (r = 0.47, Figure 3B). There were large associations between HHb with SV (r = −0.59, Figure 3C) and HR (r = 0.50, Figure 3D).
Central hemodynamics responses to prolonged sitting with (CGS) and without (CON) use of compression garments (n = 20).
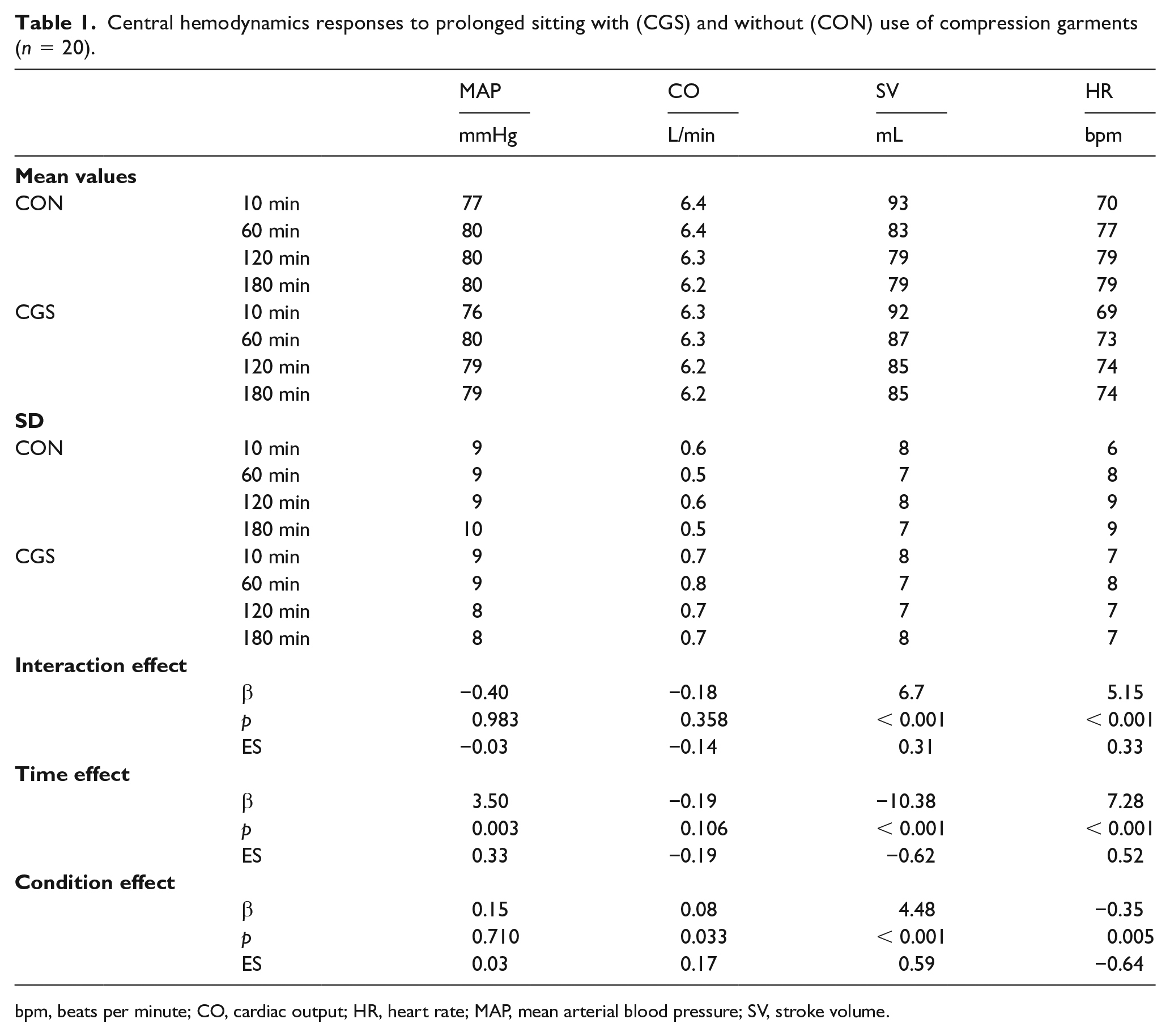
bpm, beats per minute; CO, cardiac output; HR, heart rate; MAP, mean arterial blood pressure; SV, stroke volume.
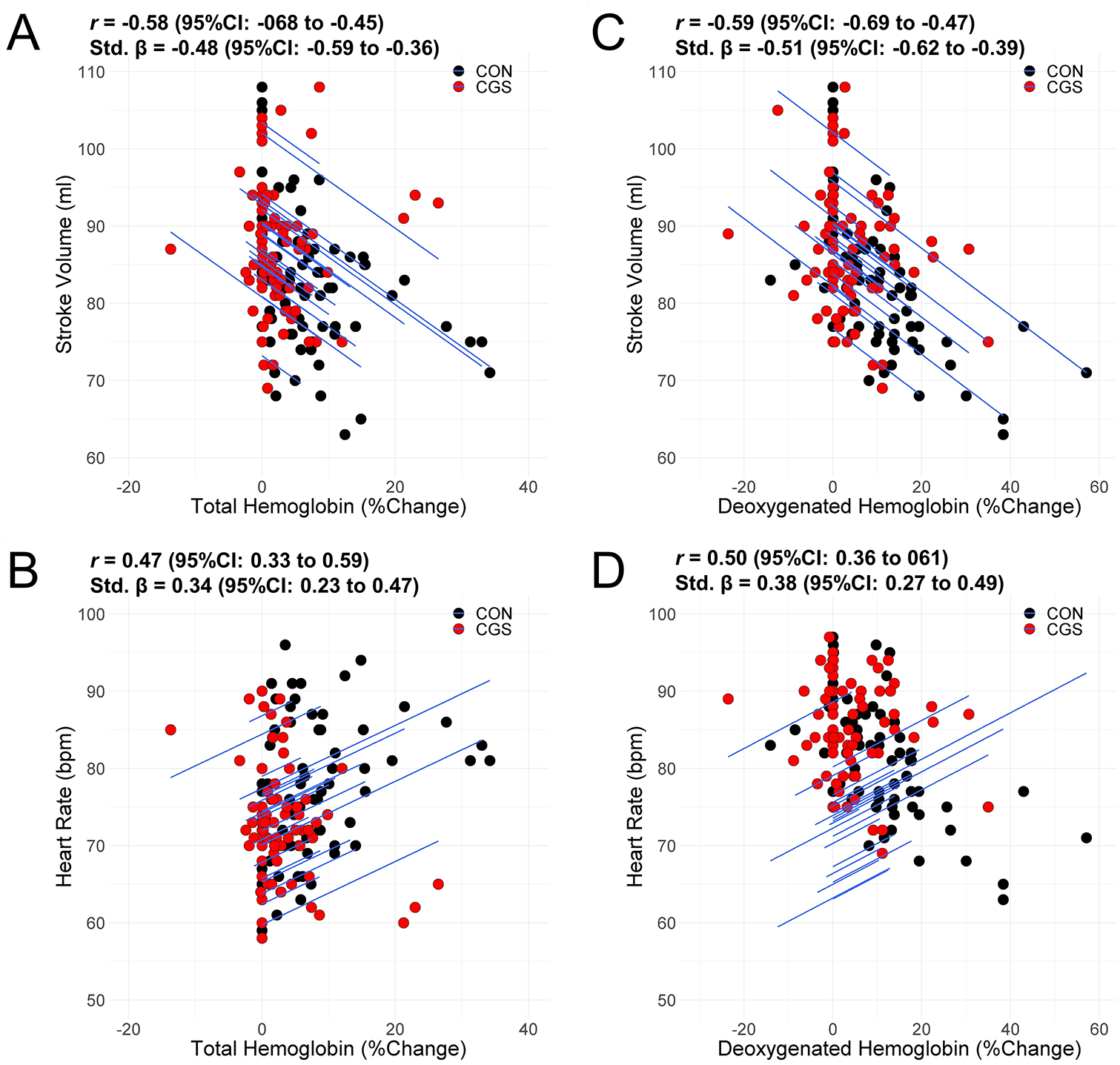
Intra-individual associations between total hemoglobin with (A) stroke volume and (B) heart rate, and between deoxygenated hemoglobin with (C) stroke volume and (D) heart rate.
Aim 3: leg arterial flow-mediated dilation and reactive hyperemia response
The use of CGS did not prevent impairments in leg macrovascular or microvascular function (Table 2). Across both conditions, BFAUC decreased by 25.9% (−8464 a.u., 95% CI: −13255 to −3672) and FMD by 39.5% (−2.40%, 95% CI: −2.99 to −1.81).
Popliteal artery flow-mediated microvascular and macrovascular responses to prolonged sitting with (CGS) and without (CON) use of compression garments (n = 20).
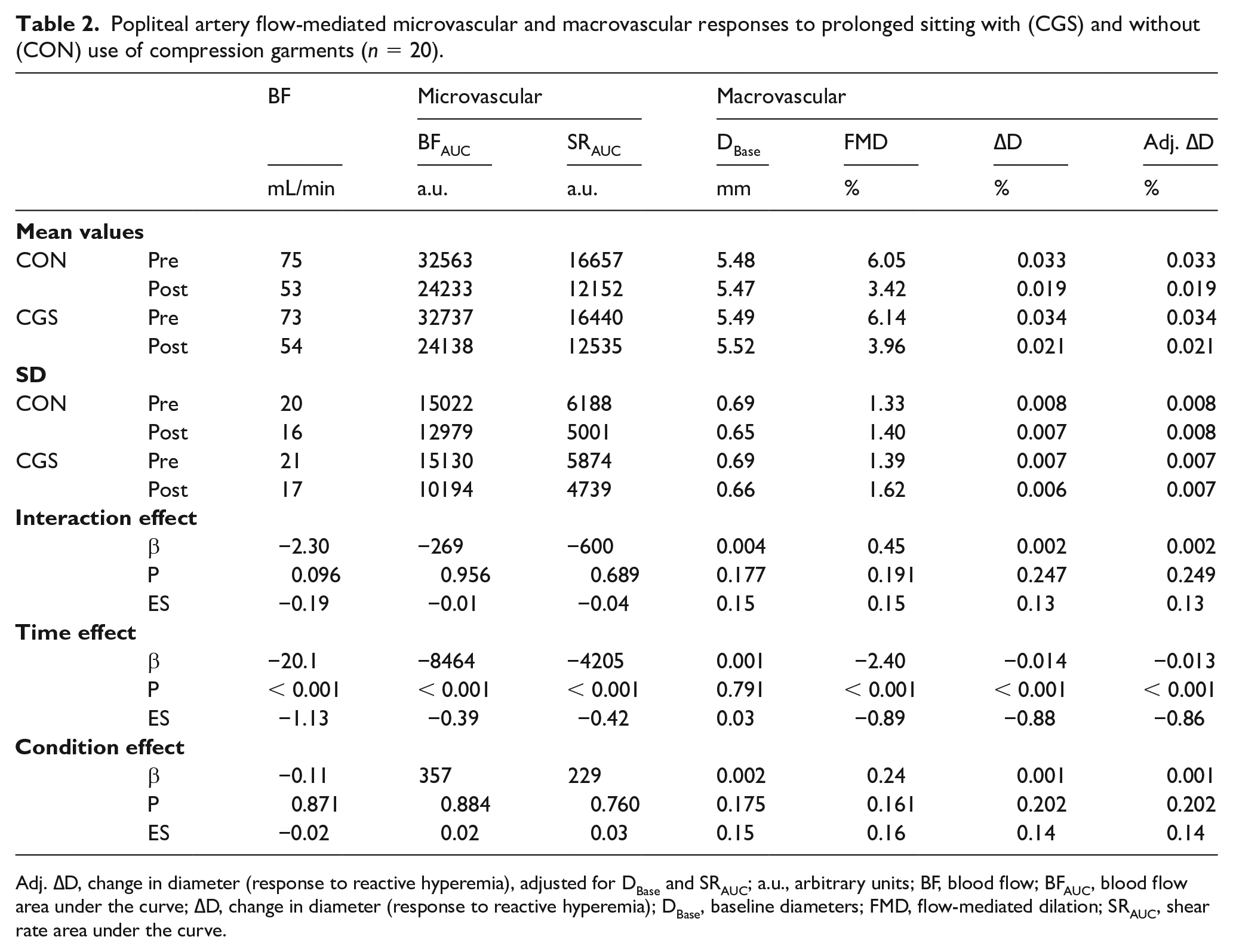
Adj. ∆D, change in diameter (response to reactive hyperemia), adjusted for DBase and SRAUC; a.u., arbitrary units; BF, blood flow; BFAUC, blood flow area under the curve; ∆D, change in diameter (response to reactive hyperemia); DBase, baseline diameters; FMD, flow-mediated dilation; SRAUC, shear rate area under the curve.
Discussion
The major findings of the present study are: (i) use of CGS mitigated the blood pooling response to prolonged sitting; (ii) blood pooling was associated with the increase in heart rate and the decrease in estimated stroke volume; and (iii) reactive hyperemia and flow-mediated arterial dilation was impaired after prolonged sitting, and use of CGS was not associated with attenuation of this impairment. Collectively, these preliminary findings suggest that use of CGS may decrease blood pooling, but not prevent the impairment in FMD/reactive hyperemia – at least in young, healthy participants.
Comparison to the literature
Venous system
We found that CGS helped mitigate the blood pooling response to prolonged sitting. Each of the tHb, HHb, and calf circumference measures indicated a reduced blood pooling response. These findings support previous studies reporting increased calf circumference following prolonged sitting compared to a supine control position.13,35 We extend these findings by reporting the efficacy of CGS in preventing this blood pooling response. While the precise mechanisms for reduced blood pooling are unclear, CGS may increase venous wall shear stress, thereby drawing blood from the lower limbs back towards the heart. 36 These findings indicate that, at least in young, healthy participants, CGS may help to prevent venous stasis, and thereby potentially decrease the risk of blood clotting and subsequent DVT. 9
Arterial system
Our second aim was to investigate whether blood pooling is inversely correlated with estimated stroke volume. If CGS preserves stroke volume, the previously reported impairments to macrovascular and microvascular function with prolonged sitting may be mitigated.6,13 There was a statistically significant association between blood pooling (tHb) and the increase in heart rate and decrease in estimated stroke volume. These findings support a previous study reporting that wearing CGS helps to prevent the decrease in stroke volume and increase in heart rate when standing. 15 Collectively, these previous findings and our findings suggest that CGS does help to maintain venous return, which ensures adequate ventricular filling and maintains the effect of preload on stroke volume for a given contractility. 10 The maintenance of stroke volume will negate the need to increase heart rate to maintain cardiac output.
There was no association between condition and leg macrovascular (popliteal artery FMD) or microvascular (popliteal artery reactive hyperemia) function. In response to the CON condition, FMD deceased by 2.7%, which is in accordance with a recent meta-analysis reporting that prolonged sitting reduces leg FMD by 2.4%. 12 We hypothesized that CGS would maintain venous return, which in turn would maintain stroke volume and leg blood flow. The decrease in leg blood flow-induced shear stress is thought to be the mechanism leading to leg endothelial dysfunction with prolonged sitting. 11 However, while CGS did mitigate the decrease in estimated stroke volume, the decrease was not completely eliminated, and leg blood flow did not significantly differ between conditions. It may be that, at least in young, healthy participants, the use of CGS does not sufficiently maintain stroke volume at a reasonable enough level to maintain leg blood flow, and/or additional hemodynamic or local factors play a role. For example, one study had young, healthy participants lie supine for 3 hours, with one leg straight and the contralateral leg bent. 37 Leg FMD did not decrease in the straight leg (5.6 % vs 7.1%, p = 0.24) but did in the bent leg (6.3% vs 2.8%, p < 0.01). The rationale provided was that the bent leg resulted in a turbulent shear stress profile. With prolonged sitting both the decrease in stroke volume and leg bending may play prominent roles, along with additional hemodynamic factors.
Study limitations and strengths
A major strength of this study is that it was internally robust and the first to comprehensively assess the effects of prolonged sitting with CGS on both the venous and arterial systems. However, there are several limitations that should be considered when interpreting the findings. First, the generalizability of the findings may be limited as we purposefully elected to recruit a small, homogeneous group of adults. The focus of this initial study was internal rather than external-validity, and thus we ensured our study was powered to detect a change in venous pooling under internally robust conditions. For this reason, we also purposefully elected to recruit healthy adults, to minimize the potential confounding of cardiometabolic diseases. Further studies are now warranted to generalize the findings to older populations and those with existing cardiometabolic disorders. Second, we were not adequately powered to detect sex interactions. While the findings are mixed, there is evidence that the prevalence of DVT is greater in women. 38 Additionally, prolonged sitting has been reported to similarly decrease microvascular function in both men and women, but men, and not women, were found to have reduced popliteal artery FMD following prolonged sitting. 13 Last, we must acknowledge that our study protocol lasted for 3 hours. It is possible that at-risk adults may remain seated without interruption for periods longer than 3 hours. Future study is warranted to determine the effects of longer periods of uninterrupted sitting.
Implications and conclusions
Evidence has demonstrated that prolonged, uninterrupted sitting leads to blood pooling13,35 and subsequent arterial macrovascular and microvascular dysfunction. 13 The blood pooling may increase the risk of DVT, 9 and the macrovascular and microvascular dysfunction may increase cardiovascular disease risk. 39 The current findings suggest that CGS, a simple and likely highly feasible strategy, decreases blood pooling but does not preserve leg arterial FMD and reactive hyperemia response. As such, CGS may help to protect against DVT risk, but not cardiovascular disease risk. Additional studies are also warranted to better generalize the current findings, including to older generations and the sex-specific effects.
Footnotes
Acknowledgements
The authors would like to thank the participants for their time. We also thank Yoko Kirihara and Misato Watanabe (Mount Fuji Research Institute, Fuji-yoshida City, Yamanashi, Japan) for their technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported in part by Japan Society for the Promotion of Science (JSPS; KAKENHI, Grant No. JP18K11012 to MH).