Abstract
Recent studies suggest the use of topical nitroglycerin (NTG) application in systemic sclerosis (SSc)-associated Raynaud phenomenon (RP). With the current study, we aimed to characterize for the first time the microvascular response to a NTG patch (Trinipatch® 5 mg/24 h) applied to the hand dorsum in patients with SSc using Laser Doppler imaging (LDI) at baseline and following a cold challenge. The study included 21 patients with SSc and 13 controls. Blood flow was evaluated by LDI at the level of the fingertips and metacarpus. Microvascular morphology was evaluated by nailfold capillaroscopy (NC). LDI revealed decreased fingertip baseline perfusion and a stronger vasoconstrictor response to a cold challenge in patients with SSc versus control. Metacarpal application of a NTG patch led to an increase in blood flow and hand temperature in patients with SSc. Furthermore, NTG administration led to a faster reperfusion after cold challenge. Correlation analyses revealed that the magnitude of the vasodilatory response was inversely related to baseline fingertip perfusion and hand temperature, but unrelated to the number of capillaries/mm assessed using NC. In conclusion, we provide evidence of a vasodilatory reaction following NTG patch application in patients with SSc using LDI and a protective effect against cold challenge. The magnitude of the response to NTG was related to functional, but not structural features. Our results support a further evaluation of the NTG patch as a possible therapeutic agent in SSc-associated RP.
Keywords
Introduction
Systemic sclerosis (SSc) is a multisystem autoimmune disease, characterized by vascular abnormalities, chronic inflammation, and skin and internal organs fibrosis. 1 It is currently hypothesized that SSc is primarily a vascular disease. 2 Fibrosis, the clinical hallmark of SSc, appears as a consequence of disrupted tissue repair in response to vascular injury. 3 Raynaud phenomenon (RP) is the main clinical manifestation of impaired functioning of cutaneous vessels involved in thermoregulation. It occurs in more than 95% of patients with SSc. 4 Furthermore, RP is associated with progressive structural capillary damage, which can lead to development of digital ulcers and critical digital ischemia, causing important functional impairment. 5
Structural and functional evaluations of cutaneous microcirculation have important implications in the understanding, diagnosis, and management of microvascular pathology associated with SSc. 6 Various noninvasive techniques were developed to this end. Nailfold capillaroscopy (NC) allows the investigation of morphological microvascular changes in the nailfold bed, and can be used to identify capillaroscopic abnormal features associated with SSc (giant capillaries, microhemorrhages, avascular areas, and loss of capillaries).6,7 NC patterns have also been more recently used to stratify the microvascular alterations in SSc. 8 Laser Doppler imaging (LDI) is a technique able to provide objective measurements of superficial cutaneous blood flow.7,9 Recent reports indicate that LDI can be used to detect with good sensitivity changes in blood flow at baseline, or after a cold challenge in patients with SSc compared to healthy volunteers.9,10
A previous study indicated that topical nitroglycerin (NTG) application in primary and secondary RP leads to vasodilatory effects, and might be of therapeutic interest in the local management of RP. 11 Using LDI, Anderson et al. found that topical application of NTG ointment on the dorsum of the finger led to significant vasodilatory effects in primary RP and SSc-associated RP. 12 Similarly, topical application of NTG gel on the fingers of patients with RP was found to lead to a faster blood flow recovery after a cold challenge as assessed by LDI, 13 and to a significant improvement in the Raynaud’s Condition Score. 14 Transdermal administration of NTG patches was otherwise investigated due to ease of administration and interruption in case of any limiting side effect, as well as commercial availability. An earlier report indicated that NTG patch application to the chest wall for 7 days led to less frequent and less severe Raynaud’s attacks in patients with primary and secondary RP. 15 Using thermography, Kan et al. further demonstrated that application of a NTG patch to the wrist of patients with SSc led to a significant increase in finger temperature. 16 Despite these promising findings, and the use of NTG patches in clinical practice, surprisingly little is known regarding the vasodilatory effects of NTG patch application in patients with SSc. In particular, it is not well understood whether the vasodilatory reaction to NTG is homogeneous, and whether it can be predicted by functional and/or structural microvascular parameters.
In this study, we aimed at investigating the effects of NTG patch application on baseline local skin blood flow and the response to cold challenge in patients with SSc-associated RP and healthy volunteers, using LDI. Furthermore, we investigated predictive markers of the response to NTG, by correlating the magnitude of the vasodilatory effect with functional (LDI) and structural (NC) microvascular parameters.
Methods
Study participants and design
Following assessment for eligibility (Figure 1), 21 patients with SSc and 13 healthy volunteers were included in this study. All patients recruited from the Belgian Cohort of Systemic Sclerosis of Erasme Hospital met the American College of Rheumatology criteria for SSc, 17 and presented with secondary RP. Healthy volunteers were recruited among employees of the same hospital and were included in order to better define the characteristics of the microcirculation following microvascular challenges in patients with SSc compared to control conditions. Informed consent was obtained from all patients and healthy volunteers, and the study was approved by the Ethical Committee of Erasme Hospital (reference number P2014/040). Patients were asked to interrupt all vasoactive treatment 1 day prior to the measurements.
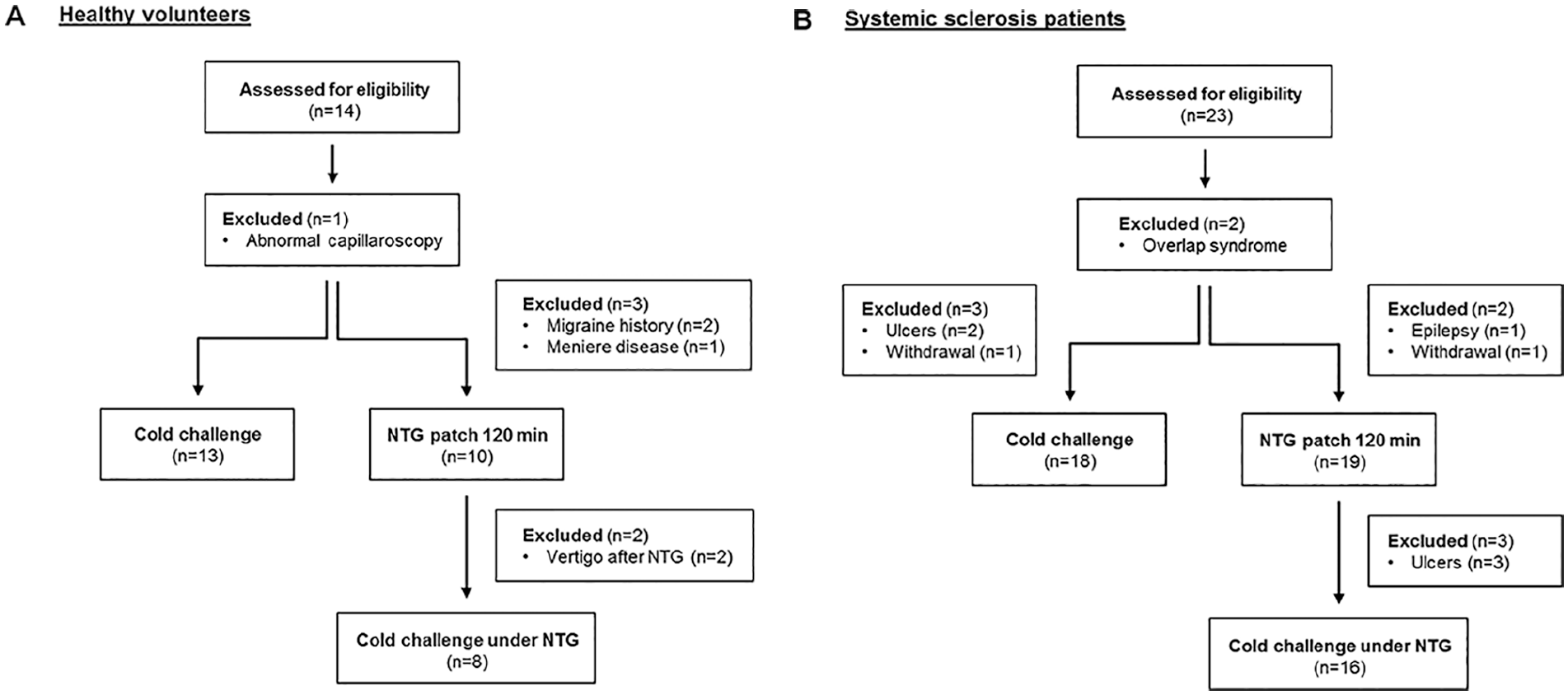
Flow chart showing selection and exclusion criteria for healthy volunteers (A) and patients with SSc (B).
The study was realized over 2 days. On both days, subjects were asked to refrain from caffeine and nicotine 4 hours prior to the study, and were acclimatized for 20 minutes at a constant temperature of 23.5 ± 0.5°C. On the first day, a morphological assessment of the microvasculature was performed by NC. Thereafter, the subjects underwent three repeated baseline LDI perfusion measures (averaged to obtain a single baseline measure), followed by a cold challenge, which consists in a complete immersion of both hands in 15°C cold water for 1 minute. Subsequently, LDI measures were performed at 1, 5, 10, 15, 20, 25, and 30 minutes post cold challenge. On the second visit, three repeated baseline LDI perfusion measures were performed (averaged to obtain a single baseline measure), followed by application of a NTG patch (Trinipatch® 5 mg/24 h; Abbott, Wavre, Belgium). The patch was applied on the dorsum of the left hand for a total of 120 minutes, as NTG from the patch is believed to reach steady state plasma levels after 2 hours of application. 18 Blood flow was measured by LDI every 10 minutes immediately after application of the patch, with heart rate continuously monitored. Afterwards, a cold challenge was performed (as described above), with the NTG patch still applied on the dorsum of the hand. Blood pressure was measured on the left arm 1 hour before and at the end of NTG application, but not during the measurements to avoid interfering with blood flow assessment.
Nailfold capillaroscopy
NC was performed on all fingers of both hands, except the thumbs. The following parameters were investigated: number of capillaries/mm, presence of enlarged and giant capillaries, hemorrhages, loss of capillaries, and signs of neoangiogenesis. Based on these observations, patients were categorized as having an early, an active, or a late NC pattern, as reported previously. 8
Laser Doppler imaging
Blood flow was measured at the fingertip and metacarpal level of both hands using LDI (moorLDI2; Moor Instruments, Axminster, UK), as described previously 9 with minor modifications. All images were obtained at a scan speed of 4 ms/pixel and at maximum scan resolution, leading to a time of image acquirement of 3 minutes and 19 seconds. The distance between the examined surface and the photodetector was 70 cm. Fingertip blood flow was measured between the distal interphalangeal joint up to and including the nailbed. Metacarpal blood flow was measured on both hands in an area of 3.2 cm × 2.3 cm overlapping on one side the area of NTG application. Each finger blood flow except the thumb was integrated (Moor LDI software, V5.3D), and averaged to obtain a mean blood flow score, in perfusion units (PU), for the entire hand. Hand temperature was monitored using a VHP3 probe connected to the LDI, positioned at the carpal level of both hands. An overview of temperature changes following the different experimental protocols is given in online Supplementary Figure 1.
Statistical analysis
For all data analysis, nonparametric tests were chosen, as normality cannot be assumed due to the small sample size. Unless stated otherwise, data are presented as median, interquartile range. When analyzing measures obtained at various time points after an intervention, we employed the Friedman test, followed by Dunn’s post-hoc test, comparing each time point to the corresponding group baseline. When analyzing two independent groups we employed the Mann–Whitney U-test, while for paired observations we used the Wilcoxon signed-rank test. When analyzing multiple groups, we employed the Kruskal–Wallis test. Categorical variables were compared using the chi-squared test. Correlation analyses were performed using Spearman’s correlation test. The significance level was set as 0.05. Statistical analysis was performed using GraphPad Prism 4.03 (GraphPad Software, San Diego, CA, USA).
Results
Cohort characteristics
Baseline clinical characteristics of the participants in the study are indicated in Table 1. There was no statistical difference in age or sex between the two groups. The two groups were also matched for dominant hand, history of smoking, and diabetes.
Clinical and epidemiological characteristics of the subjects in the SSc and control groups.
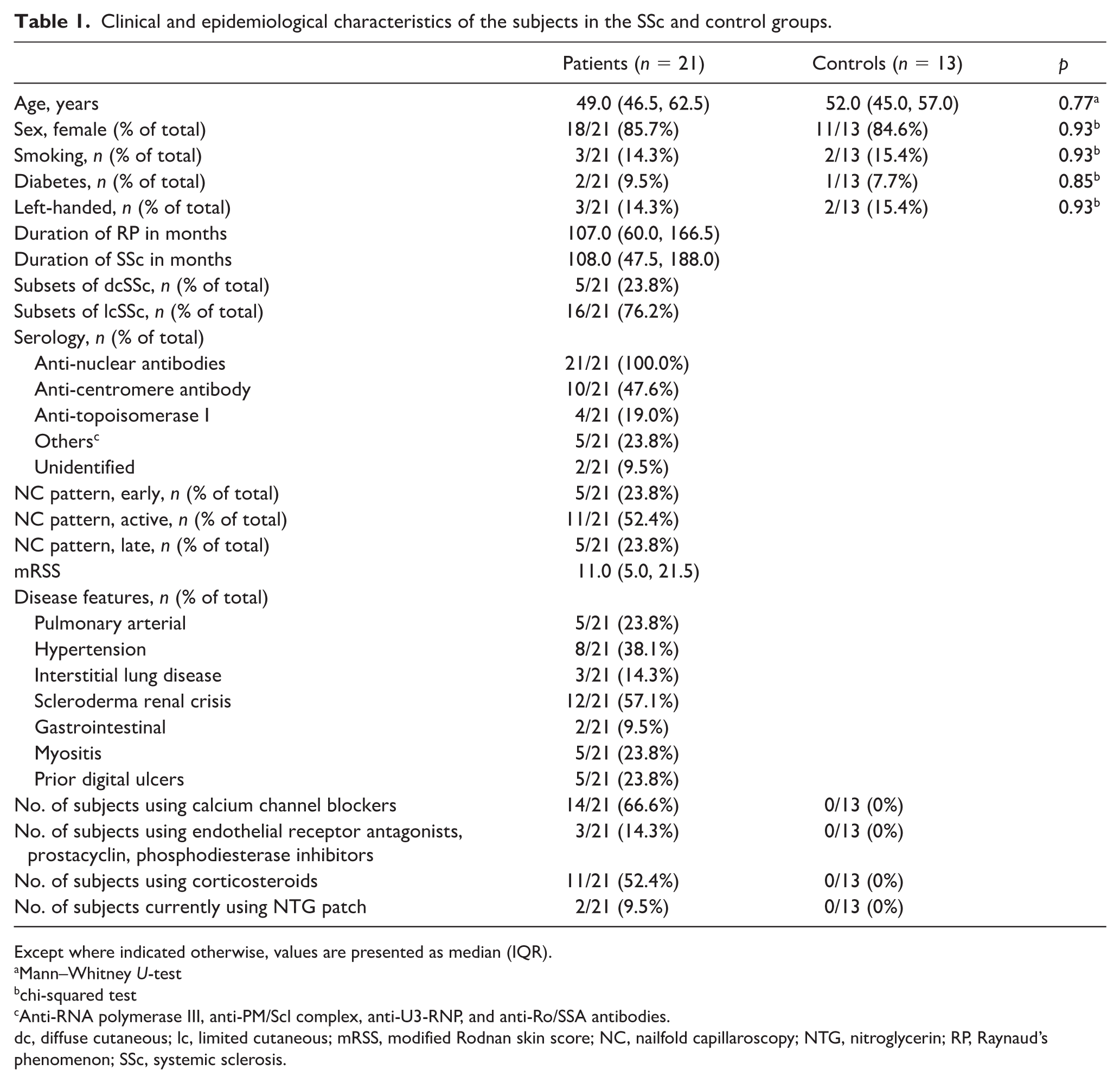
Except where indicated otherwise, values are presented as median (IQR).
Mann�Whitney U-test
chi-squared test
Anti-RNA polymerase III, anti-PM/Scl complex, anti-U3-RNP, and anti-Ro/SSA antibodies.
dc, diffuse cutaneous; lc, limited cutaneous; mRSS, modified Rodnan skin score; NC, nailfold capillaroscopy; NTG, nitroglycerin; RP, Raynaud’s phenomenon; SSc, systemic sclerosis.
Baseline measurements
Baseline LDI measurements indicate that patients with SSc have lower baseline perfusion compared to healthy volunteers at the fingertip level but not at the metacarpus level (Table 2). Furthermore, patients with SSc have lower baseline hand temperatures, as measured at the carpal region, compared to healthy volunteers (Table 2). Based on NC findings, 23.8% of patients with SSc had an early SSc pattern, 52.4% an active pattern, and 23.8% a late pattern. When averaged over the two hands, fingertip perfusion was found to be higher in patients with an early NC pattern, compared to patients with a late pattern (611.0 (505.6, 663.1) PU early pattern vs 404.1 (315.5, 563.3) PU active pattern vs 256.8 (122.9, 490.7) PU late pattern; p = 0.024 Kruskal–Wallis test; p = 0.028 Dunn’s post-hoc comparison early vs late pattern).
Baseline characteristics of the subjects in the SSc and control groups.
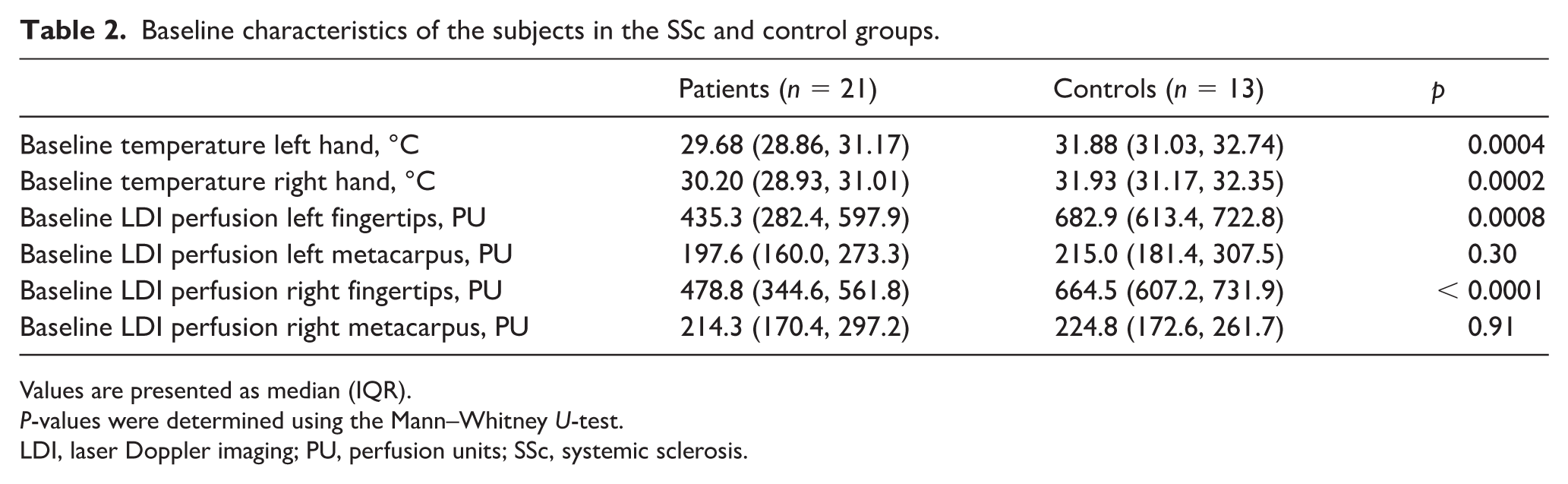
Values are presented as median (IQR).
P-values were determined using the Mann–Whitney U-test.
LDI, laser Doppler imaging; PU, perfusion units; SSc, systemic sclerosis.
Cold challenge
After immersion of both hands in cold (15°C) water for 1 minute, the blood flow of healthy volunteers decreased, but perfusion recovered to baseline within 5 minutes at the fingertips and right metacarpus and within 10 minutes at the left metacarpus (Figure 2A). In contrast, hand perfusion of patients with SSc decreased profoundly after cold challenge, and did not recover within the 30-minute observation period at all regions analyzed (Figure 2A). By integrating the area under the perfusion-time curve normalized for baseline flow, we could observe that patients with SSc showed a stronger vasoconstrictive response following cold challenge, compared to healthy volunteers, of about 33% at the level of the left fingertips (p < 0.01), 22% at the right fingertips (p < 0.001), 15% at the left metacarpus (p < 0.01), and 23% at the right metacarpus (p < 0.001) (online Supplementary Table 1).
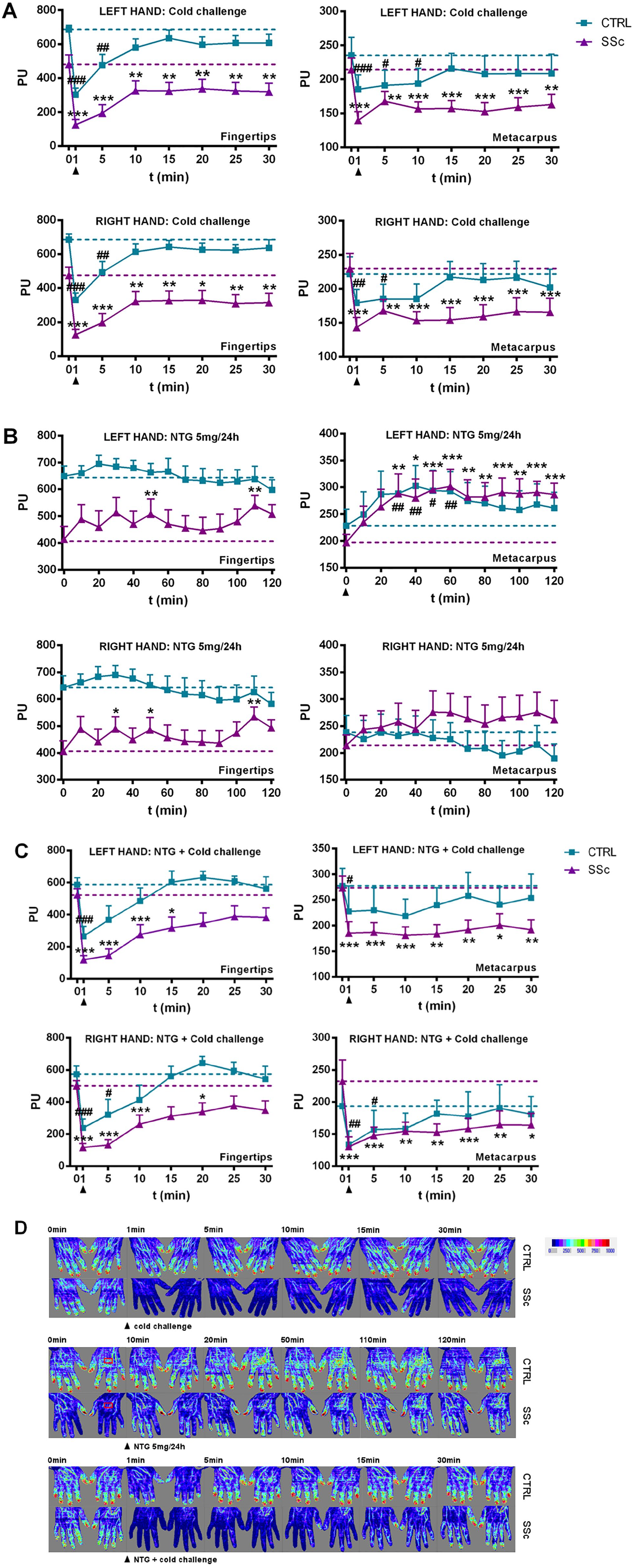
Fingertip and metacarpal blood flow evaluated using LDI in SSc and CTRL groups. Data are presented as mean PU + SE of the mean, following: (A) a cold challenge, (B) application of a NTG 5 mg/24 h patch on the left metacarpus, and (C) a cold challenge performed at the end of NTG application. (D) Representative laser Doppler images.
Nitroglycerin (NTG) patch
Following application of a NTG 5 mg/24 h patch for 120 minutes in healthy volunteers, local skin blood flow transiently increased in the region of NTG application on the left metacarpus (T30–60; p < 0.05), while no significant blood flow changes were observed at the right metacarpus, or at the level of the fingertips (Figure 2B). Application of a NTG patch to patients with SSc led to a sustained increase in local skin blood flow at the site of application on the left metacarpus, starting from 30 minutes, while no significant changes in blood flow were observed at the right metacarpus (Figure 2B). In addition, the fingertips also demonstrated increased perfusion at selected time points during NTG in both the left (T50 and T110; p < 0.05) and right (T30, T50, and T110; p < 0.05) hands. Application of the NTG patch led to an increased vasodilatory effect in patients with SSc compared to healthy volunteers of about 18% at the right metacarpus (p < 0.05) and 21% at the left fingertips (p < 0.05), but not at the right fingertips (p > 0.05) or the site of NTG application on the left metacarpus (p > 0.05) (online Supplementary Table 1). Application of the NTG patch increased the temperature of the left hand in patients with SSc (p < 0.001) (Figure 3B), but not in healthy volunteers (p > 0.05) (Figure 3A).
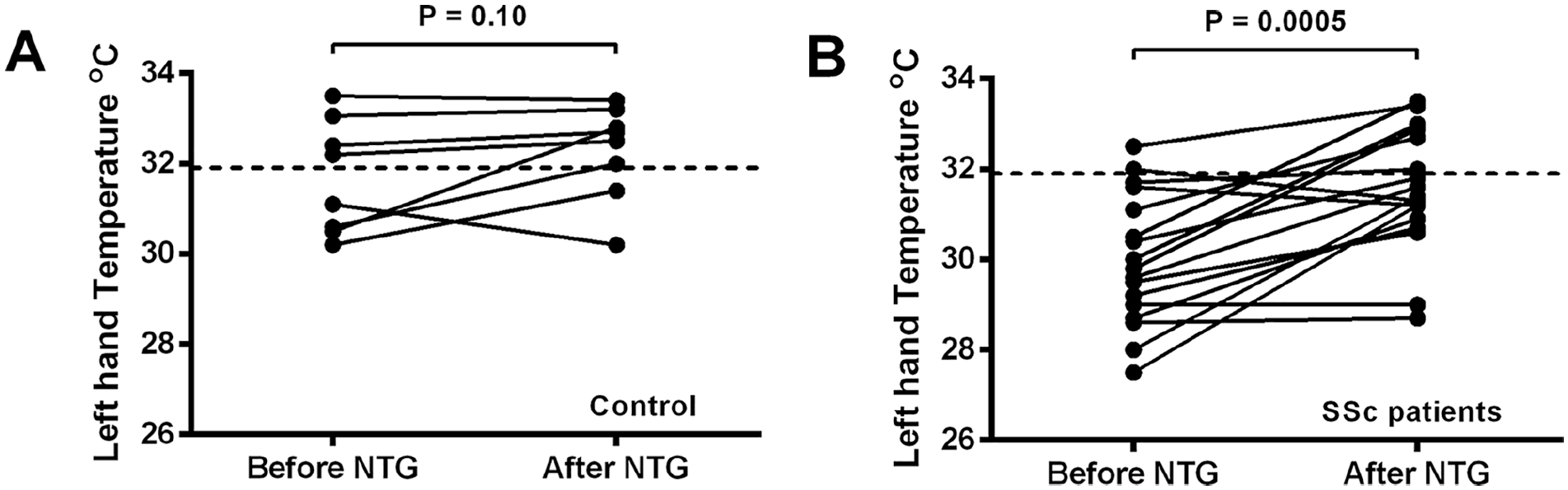
Change in hand temperature in healthy controls (A) and patients with SSc (B) following application of a NTG 5 mg/24 h patch on the left metacarpus for 120 minutes.
NTG administration decreased the mean arterial pressure, of approximately 5%, both in patients with SSc (86.7 (73.3, 93.3) mmHg vs 80.0 (70.0, 86.7) mmHg after NTG, p < 0.05), as well as in healthy controls (85.8 (75.8, 98.3) mmHg vs 86.7 (70.0, 95.42) mmHg after NTG, p < 0.05). Furthermore, application of NTG induced headaches and vertigo, with a significantly higher frequency in healthy volunteers (frequency of headaches: 36.8% SSc vs 80.0% control, p < 0.05; frequency of vertigo: 0.0% SSc vs 20.0% control, p < 0.05).
Cold challenge with NTG patch
A cold challenge at the end of NTG application in healthy volunteers revealed a faster recovery of blood flow, as baseline perfusion was reached within a maximum of 10 minutes of cold stimulus at all regions analyzed (Figure 2C). NTG application in patients with SSc led to recovery of perfusion at the level of the fingertips within a maximum of 20 minutes after cold stimulus (Figure 2C), compared to no recovery within 30 minutes of cold stimulus without the patch (Figure 2A). On the other hand, no effect from the NTG patch could be observed at the level of the metacarpus in patients with SSc, where the cold challenge induced a profound decrease in perfusion without recovery in the 30-minute period (Figure 2C). When performing a direct comparison between the groups, patients with SSc demonstrated a stronger decrease in blood flow after cold stimulus, compared to healthy volunteers, of about 34% at the left fingertips (p < 0.05), 35% at the right fingertips (p < 0.01), and 19% at the right metacarpus (p < 0.05), but not at the site of NTG application on the left metacarpus (p > 0.05) (online Supplementary Table 1).
Predictive markers for NTG response in patients with SSc
At the level of the fingertips, the area under the normalized perfusion-time curve after NTG was inversely correlated with baseline fingertip perfusion (r = −0.752, p < 0.001) (Figure 4A). Moreover, the percentage temperature increase after 120 minutes of NTG application was inversely correlated with baseline hand temperature (r = −0.489, p < 0.05) (Figure 4B). However, no relation was found between the total vascular response to NTG and baseline perfusion, when both parameters were evaluated at the level of the metacarpus (r = −0.235, p > 0.05). Regarding structural parameters, the number of capillaries per millimeter, as evaluated by NC, was directly correlated with the fingertip baseline blood flow (r = 0.473, p < 0.05) (Figure 4C). Similarly, the number of capillaries per millimeter was inversely correlated with disease activity (r = −0.590, p < 0.01), disease severity (r = −0.558, p < 0.05), and the modified Rodnan skin score (mRSS) (r = −0.501, p < 0.05), while it was not correlated with the total vascular response to NTG (r = −0.222, p > 0.05) (Figure 4D). Furthermore, no difference in NTG response was observed when patients with SSc were stratified based on their NC pattern (early vs active vs late pattern; data not shown).
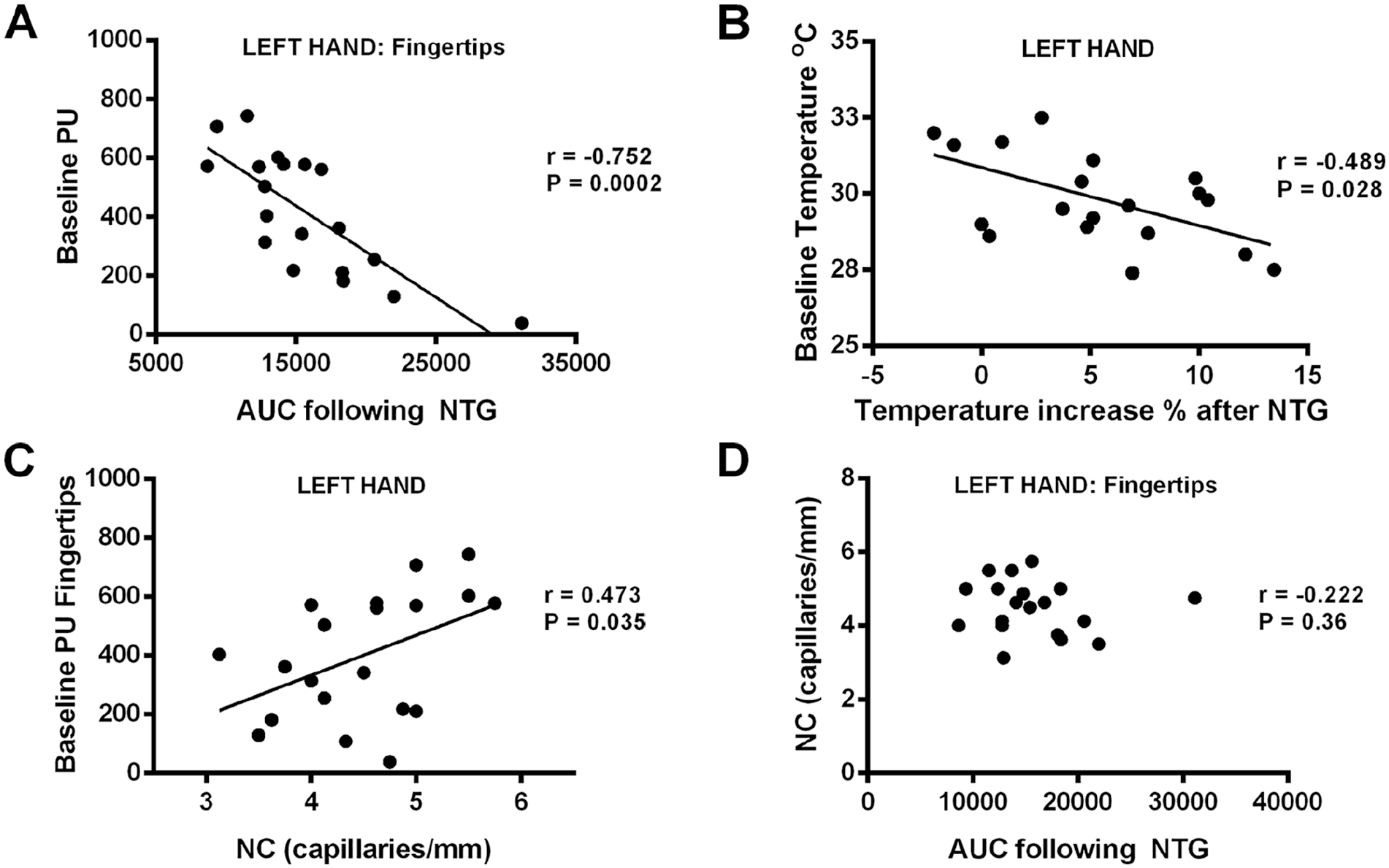
Predictive markers of the NTG response in patients with SSc. The AUC following NTG was found to correlate inversely with the baseline fingertip perfusion evaluated before NTG application (A). Similarly, the % increase in hand temperature after NTG administration correlated inversely with the baseline hand temperature before NTG (B). On the other hand, while the number of capillaries/mm, as evaluated using NC, correlated with the baseline fingertip perfusion (C), no significant correlation could be found between the number of capillaries/mm and the subsequent response to NTG (D).
Discussion
In order to characterize the microvascular features of patients with SSc, we employed two complementary noninvasive techniques that can give information regarding microvascular function (LDI) and structure (NC), 7 and compared their response with similar evaluations performed in age-matched healthy volunteers. LDI measurements at baseline confirmed a decreased baseline fingertip blood flow in patients with SSc compared to controls, in line with findings of an altered vasomodulatory balance in patients with SSc in favor of vasoconstriction. 4 This decrease was specifically observed at the level of the fingertips, but not at metacarpal level, and indicates altered baseline fingertip perfusion in patients with SSc. Our results are in line with previous observations supporting specific microvascular damage at the level of the fingertips, but not the hand dorsum in SSc.19–21
In this study, we applied dynamic tests inducing vasoconstriction (cold challenge) or vasodilation (left metacarpal application of a NTG patch), and evaluated the response of the blood vessels in patients with SSc compared to age-matched healthy volunteers. In line with previous reports,9,19,22 our results confirmed a stronger vasoconstriction response at the level of the fingertips in patients with SSc. Interestingly, we could also observe stronger vasoconstriction of the hand dorsum in response to cold challenge. This finding extends recent observations of the impaired microvascular reaction of the hand dorsum after cold challenge in patients with SSc with a late nailfold video capillaroscopy pattern. 19
In order to evaluate vasodilatory microvascular function, we applied a NTG patch at the left metacarpal level for 2 hours. NTG is a nitric oxide (NO) donor and has an endothelial-independent vasodilatory effect that can also occur in patients whose endothelial function is compromised, such as patients with SSc. Our results indicate that application of a NTG patch in healthy volunteers induces a local vasodilatory response restricted to the site of application. A similar local vasodilatory reaction was observed in patients with SSc; however, this also seemed to extend to the fingertips toward the end of application. These findings support a systemic vasodilatory effect of the NTG patch that was hypothesized in previous studies,15,16 but not yet demonstrated using LDI. Moreover, our study indicates that there is an added local vasodilatory effect of the NTG patch at the site of application. Intriguingly, in some areas, the normalized vasodilatory effect observed in patients with SSc was higher than that observed in healthy controls, hinting at a potentially stronger vascular response to NTG in patients with SSc. Furthermore, the hand temperature increased significantly after application of the NTG patch in patients with SSc, but not in healthy volunteers. These findings are in contrast with Anderson et al., who observed comparable increases in digital blood flow by LDI after topical application of NTG in patients with SSc compared to healthy volunteers. 12 On the other hand, using a similar set-up to us, Kan et al. demonstrated that application of a NTG patch leads to a significant increase in finger temperature in patients with SSc, but not healthy volunteers. 16 Furthermore, a recent study observed that sublingual NTG increased the rate of rewarming and skin temperature of the coldest toe after a cold challenge in cold-sensitive healthy individuals compared to naive participants. 23 The authors hypothesized that NTG might have replenished NO levels in cold-sensitive individuals, but not in healthy volunteers that do not have NO deficiency. 23 Consistent with this hypothesis, the magnitude of the response to NTG in our study was correlated with baseline perfusion and hand temperature. In particular, we found that patients with a lower baseline fingertip perfusion and temperature reacted more strongly to the vasodilatory effects of NTG. Although it is currently unclear whether NO levels are decreased 24 or increased25–27 in patients with SSc, NO supplementation (using a NO-generating gel) induces vasodilation when applied on the skin of patients with RP. 28 Alternatively, NTG could have reversed the vasoconstrictor tone present in patients with SSc via NO-independent vasodilatory effects. 29 We hypothesize that the particularly strong response to NTG patch application in patients with SSc with decreased baseline function might be due to a ‘rescue’ effect from a highly constrictive vascular state, resulting from either impaired vasodilation or increased vasoconstriction. Further studies are needed to evaluate the vasodilatory response to NTG in relation to NO bioavailability, 30 as serum NO levels might not be homogenously distributed in patients with SSc. 26
Although we observed that the number of capillaries/mm correlated significantly with baseline fingertip perfusion, as well as with disease activity, disease severity, and mRSS, the response to NTG did not correlate either with NC patterns or with the number of capillaries/mm. This finding indicates that functional, but not structural parameters predict the subsequent vasodilatory effect of NTG administration. Our findings are in line with recent nailfold video capillaroscopy studies indicating an inverse relation between nailfold microangiopathy severity and fingertip blood perfusion,8,20,21,31 and a direct relation between nailfold microangiopathy severity and mRSS.20,31
Two hours after NTG application, we investigated the functional response of the microvasculature to a cold challenge under NTG. Interestingly, in this condition, we observed improved perfusion after cold challenge in healthy volunteers, as well as in patients with SSc at the level of the fingertips, indicating a potential protective effect of NTG on microvascular changes following cold challenge. Our findings are consistent with a recent study indicating that application of MQX-503, a topical gel formulation of NTG, leads to a faster recovery of blood flow after cold challenge in patients with RP. 13
Our observations that NTG application in patients with SSc led to a vasodilatory effect, which seemed to recover microvascular function following a cold challenge, supports the hypothesis that topical application of NTG may represent an interesting pharmacological approach for RP secondary to SSc.4,13,15,16,32–34 The preferential local effect on the microvasculature indicates a potential benefit in placing the NTG patch in the proximity of strongly affected areas. Future placebo-controlled cross-over studies are warranted re-visiting the therapeutic utility of a NTG patch, 15 particularly in patients with decreased baseline function, in line with our current observations. The potential therapeutic efficacy of NTG should also be balanced with its pro-oxidant nature, as the released NO can form damaging peroxynitrite radicals in the presence of an oxidative environment, 35 such as it occurs in SSc. 36 Concomitant antioxidant treatment37,38 could be an interesting approach in this direction in order to potentiate the effect of NTG and to protect against the damaging effect of excess NO.
Study limitations
Our study has a series of limitations. We included a small number of participants and did not take into account the possible placebo effects of patch application. Taking these limitations into account, we provide novel insight into microvascular changes occurring after NTG patch application in SSc.
Conclusion
In conclusion, this is the first study demonstrating the vasodilatory effects of NTG patch application in patients with SSc using LDI and a potential protective effect against cold challenge. The magnitude of this response was related to functional (baseline perfusion), but not structural, parameters. Our results support NTG patch application as a potential therapeutic strategy in SSc-associated RP.
Supplemental Material
Supplementary_Material – Supplemental material for Laser Doppler imaging evaluation of nitroglycerin patch application in systemic sclerosis patients
Supplemental material, Supplementary_Material for Laser Doppler imaging evaluation of nitroglycerin patch application in systemic sclerosis patients by Georgiana Pintea Bentea, Aurélien Wauters, Jean-Claude Wautrecht and Elie Cogan in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
