Abstract
Trends in prescription for venous thromboembolism (VTE) prophylaxis following total hip (THR) and knee replacement (TKR) since the approval of direct oral anticoagulants (DOACs) and the 2012 guideline endorsement of aspirin are unknown, as are the risks of adverse events. We examined practice patterns in the prescription of prophylaxis agents and the risk of adverse events during the in-hospital period (the ‘in-hospital sample’) and 90 days following discharge (the ‘discharge sample’) among adults aged ⩾ 65 undergoing THR and TKR in community hospitals in the Institute for Health Metrics database over a 30-month period during 2011 to 2013. Eligible medications included fondaparinux, DOACs, low molecular weight heparin (LMWH), other heparin products, warfarin, and aspirin. Outcomes were validated by physician review of source documents: VTE, major hemorrhage, cardiovascular events, and death. The in-hospital and the discharge samples included 10,503 and 5722 adults from 65 hospitals nationwide, respectively (mean age 73, 74 years; 61%, 63% women). Pharmacologic prophylaxis was near universal during the in-hospital period (93%) and at discharge (99%). DOAC use increased substantially and was the prophylaxis of choice for nearly a quarter (in-hospital) and a third (discharge) of the patients. Aspirin was the sole discharge prophylactic agent for 17% and 19% of patients undergoing THR and TKR, respectively. Warfarin remained the prophylaxis agent of choice for patients aged 80 years and older. The overall risk of adverse events was low, at less than 1% for both the in-hospital and discharge outcomes. The low number of adverse events precluded statistical comparison of prophylaxis regimens.
Introduction
Venous thromboembolism (VTE) is the third most common cause of cardiovascular death in the US after myocardial infarction and stroke. It occurs in approximately 350,000–900,000 persons each year and accounts for at least 100,000 deaths annually in the US.1,2 Total hip and knee replacement (THR and TKR) pose a particularly high risk for VTE. Without prophylaxis the prevalence of VTE is between 40% and 60% following hip or knee arthroplasty. 3 Given the acute and long-term morbidity and mortality associated with VTE, routine prophylaxis following THR and TKR has been the standard of care for more than 30 years.3–6 However, optimal prophylaxis strategy has been uncertain, and little is known about adverse outcomes following prophylaxis.
Until recently, the American College of Chest Physicians (ACCP) has recommended higher potency pharmacologic options. In 2008, the recommended options included fondaparinux, low molecular weight heparin (LMWH), or warfarin with the international normalized ratio (INR) target of 2.0–3.0, while the ACCP recommended against use of aspirin. 3 Many orthopedic surgeons disagreed with the ACCP guidelines, believing that the recommendations for prophylaxis should be based on prevention of pulmonary embolus (PE) and not deep vein thrombosis (DVT). In 2008, the American Academy of Orthopedic Surgeons (AAOS) developed its own guidelines. Importantly, the AAOS recommended aspirin 325 mg twice daily combined with mechanical devices or warfarin with the target INR < 2.0 on equal footing as fondaparinux and LMWH. 7 In 2012, the ACCP updated its guidelines to include DOACs – rivaroxaban, apixaban, and dabigatran – in addition to the previously recommended regimens, and aspirin was added without specification of the dose and frequency. 8 Adding to the confusion, the new AAOS guidelines published in 2011 stated that there is inconclusive evidence to be able to recommend for or against any specific pharmacologic agent. 9 Although ACCP singled out LMWH as perhaps the best option in most cases because it has the most extensive safety information available, these most recent guidelines opened the number of acceptable regimens so broadly that they left the average front line clinician virtually unguided in making a prophylaxis selection.
The uncertainty surrounding the optimal prophylaxis regimen has led to wide variation in clinical practice. Registry data from 3292 patients who underwent THR and/or TKR from 2001 to 2004 demonstrated that at least 39% of patients received a VTE prophylaxis regimen that was not in accordance with the ACCP guideline. 10 Given that elective joint replacements are projected to increase substantially over the next several decades, and that participants in randomized trials of VTE prophylaxis were younger than most joint replacement patients (i.e. average age of trial participants was 61–68 years),11–21 there is now a pressing need to investigate the safety and efficacy of VTE prophylaxis, particularly in older, medically complex patients. We therefore sought to report the practice patterns of in-hospital and discharge prophylaxis and their associated in-hospital and 90-day adverse outcomes across community hospitals in the US during the years 2011 to 2013.
Methods
Patient selection
We assembled a cohort of patients 65 years of age or older who underwent elective THR or TKR at a hospital in the Institute for Health Metrics (IHM) database over a 30-month period (January 1, 2011 – September 30, 2013). The IHM provides information technology support to community hospitals to facilitate reporting of federally mandated performance and quality measures. The IHM extracts clinical data directly from hospitals and delivers formatted data back to those hospitals in an effort to support quality and process improvement at community hospitals across the US. The data collected include demographics, medical diagnoses, operative and radiology reports, clinical electronic notes from physicians, nurses, and physical therapists, laboratory results, and drugs administered.
Patients were eligible if they underwent elective THR or TKR identified by primary International Classification of Diseases (ICD)-9 procedure codes (81.51 for THR and 81.54 for TKR) and detailed chart review. We excluded patients with hip fracture, revision surgery, or multiple joint surgeries. For the in-hospital sample, we limited our study sample to those from hospitals with the bedside barcode medication validation system and to those with a hospital length of stay of 2 days or greater. For the discharge sample, we excluded patients who experienced VTE, major hemorrhage, cardiovascular events, and death prior to hospital discharge and limited our analysis to the patients with an electronic medical record (physician’s discharge text summary) of discharge prophylaxis medication. Figure 1 depicts the patient selection process for the in-hospital and discharge samples.
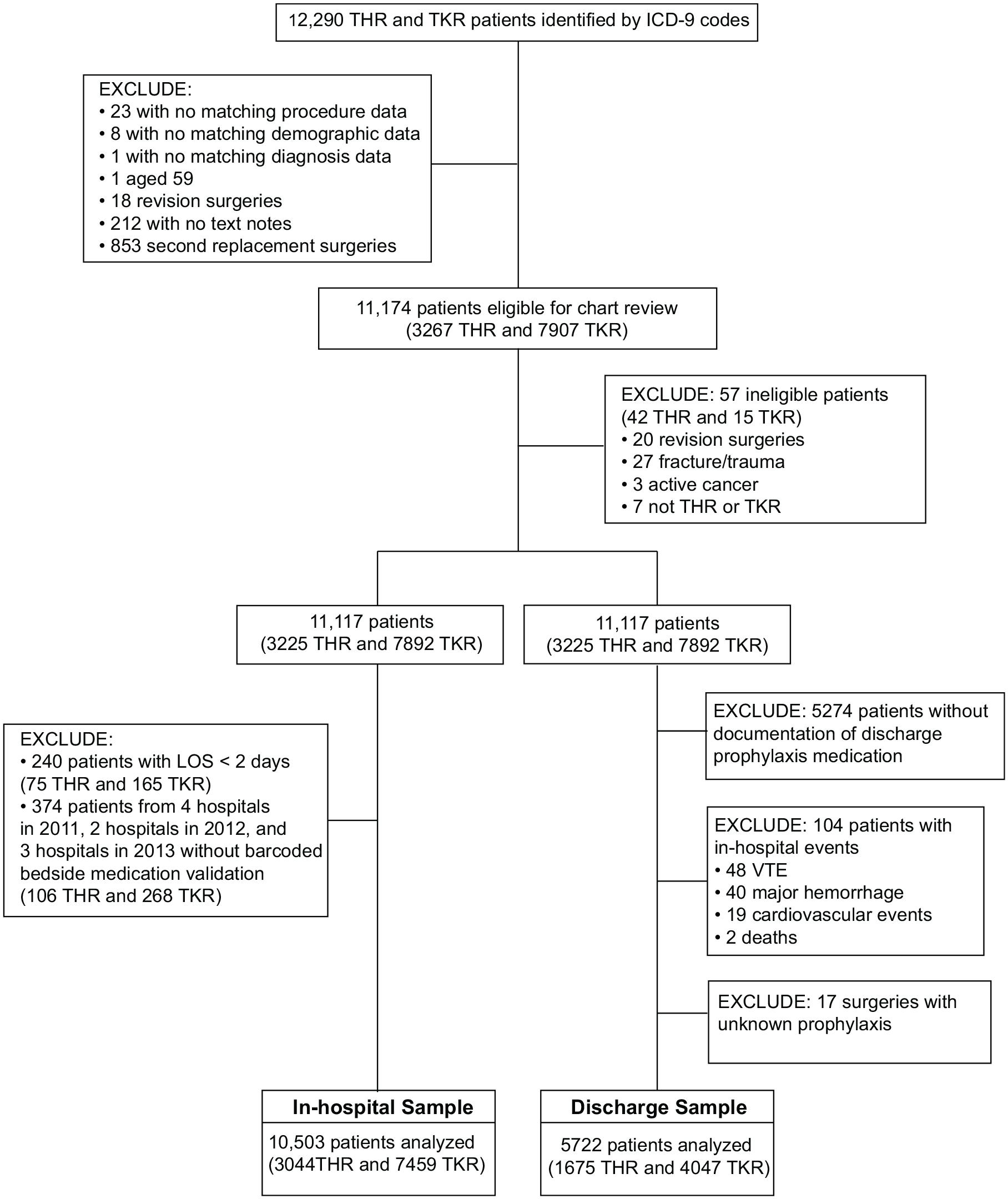
Flow chart of study sample selection for the in-hospital and outpatient samples.
Ascertainment of patient characteristics and prophylaxis medications
Patient demographics were extracted from the medical record and baseline medical comorbidities were obtained using ICD-9 codes (Table S1). For the in-hospital sample, VTE pharmacologic prophylaxis was determined from the bedside barcodes on the day of surgery or during the postoperative period prior to discharge. For the discharge sample, information on the pharmacologic agents was determined from the physician’s discharge text summary. Eligible medications included aspirin (including other antiplatelet agents), warfarin, dabigatran, rivaroxaban, apixaban, LMWH (including other heparin products), and fondaparinux. In cases where there were multiple eligible medications administered (e.g. aspirin plus an anticoagulant), we assigned a single prophylaxis regimen category based on the following rank order consistent with the literature: fondaparinux, DOACs, LMWH, warfarin, and aspirin.22,23
Outcomes
Outcome information was obtained through reviewing the ICD-9 codes and the clinical information available within the IHM data system, which includes information on laboratory results, drugs administered, radiology reporting, nursing, physical therapy, and physicians’ notes (e.g. admissions, discharge, and daily progress notes), as well as claims data. All outcome events (VTE, major hemorrhage, cardiovascular events, and death) were validated by independent physician reviewers. Disagreements were resolved by consensus or by a third investigator.
VTE – patients with imaging evidence of PE (ventilation and perfusion scan or computed tomography (CT) pulmonary angiogram) or DVT (ultrasound with Doppler) were classified as having experienced VTE.
Major hemorrhage – was defined using the International Society of Thrombosis and Hemostasis definition of major hemorrhage, which includes clinically overt hemorrhage (1) leading to death or (2) occurring in a critical anatomical site such as intracranial, intraspinal, intraocular, retroperitoneal, intra-articular, pericardial, or intramuscular with compartment syndrome, or (3) leading to a decline in hemoglobin level of ⩾ 2 g/dL or transfusion of two or more units of packed red blood cells. 24
Cardiovascular event – patients who experienced a stroke, myocardial infarction, or cardiac arrest were classified as having experienced a cardiovascular event. For stroke, we included all types and required imaging evidence with CT or magnetic resonance imaging. For myocardial infarction, we required documentation of troponin elevation, electrocardiogram changes, or interventions for myocardial infarction.
Death – defined as all-cause mortality prior to hospital discharge or during the 90-day period.
Statistical analysis
We compared the baseline characteristics between THR and TKR using the t-test for continuous variables and the chi-squared test for the categorical variables. We tested for a linear trend using the Mantel–Haenszel chi-squared test. For subgroup analyses, we stratified the prophylaxis medication use by surgery type, study year, and four different age groups (65–69, 70–74, 75–79, ⩾ 80 years). We then combined the prophylaxis medications into the following categories and compared their use by age group: anticoagulant only, aspirin only, aspirin plus anticoagulant, and none. All analyses were performed separately for the in-hospital and discharge samples. The low number of adverse events precluded multivariable analyses. All analyses were conducted using SAS software, Version 9.4 (SAS Institute Inc., Cary, NC, USA).
The Boston University Institutional Review Board approved the study. IHM operates under a HIPAA (Health Insurance Portability and Accountability Act) Business Associates Agreement and is permitted by contract to use aggregated de-identified data for clinical research.
Results
Patient demographics and baseline comorbidities
We initially identified 12,290 patients with primary ICD-9 codes for THR (81.51) and TKR (81.54) (Figure 1). Of these, 10,503 patients met the inclusion and exclusion criteria for the in-hospital sample, after excluding patients with a length of stay of less than 2 days (n = 240) and those from hospitals without barcoded bedside medication validation (n = 374). The discharge sample consisted of 5722 patients that had an electronic medical record of discharge prophylaxis medication and did not have any in-hospital events. Our study samples represented 65 community hospitals within the IHM network representing 31 states.
Table 1 provides the descriptive characteristics of the in-hospital and discharge samples. The mean ages of the samples were 73 and 74 years, respectively, and about 16% (TKR) and 24% (THR) were 80 years of age or older. Women comprised 61–63% of the samples and 95–96% were white.
Baseline demographics and comorbidities among 10,503 in-hospital and 5722 discharge sample patients receiving THR or TKR in the IHM network, 2011–2013.
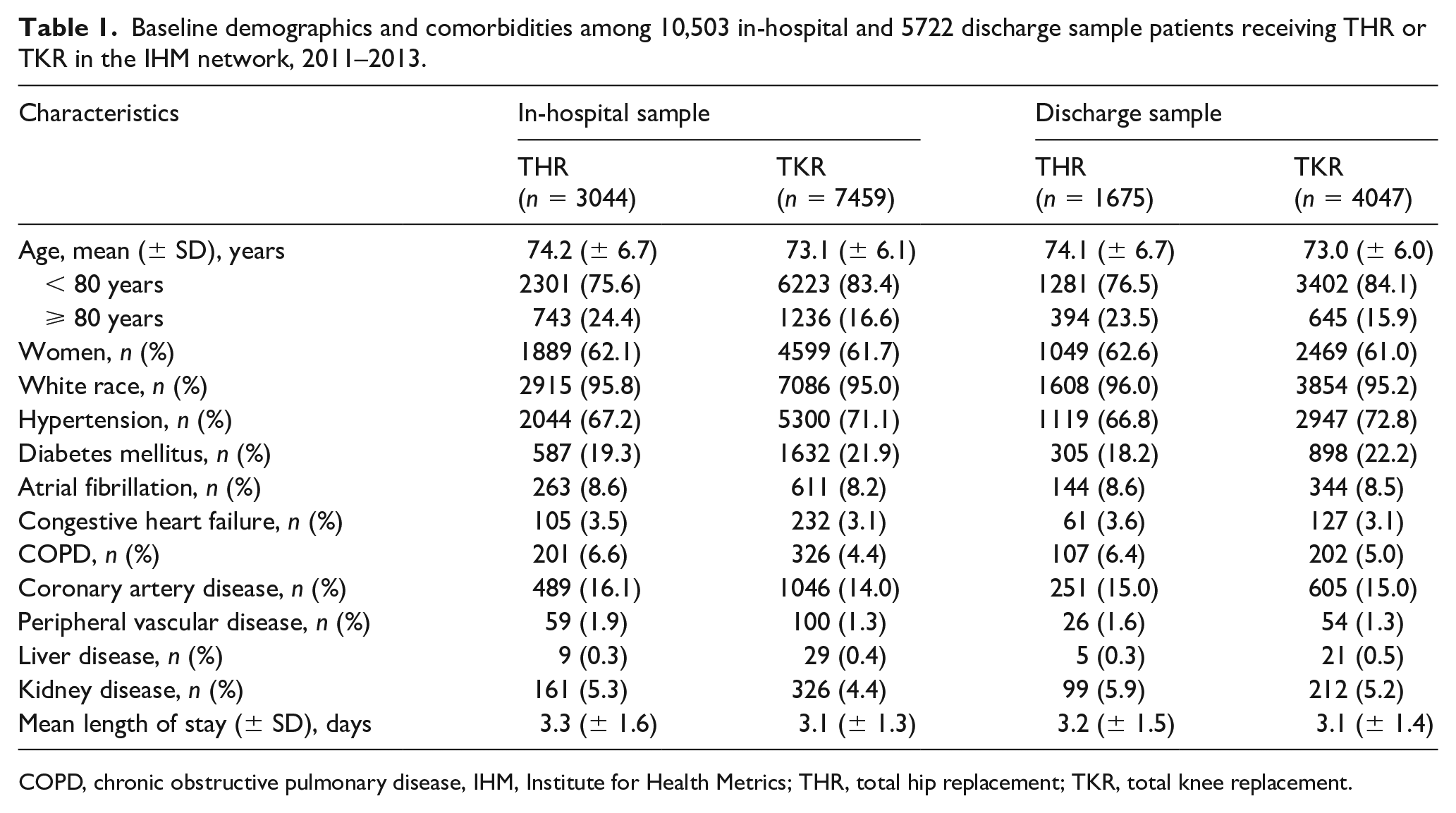
COPD, chronic obstructive pulmonary disease, IHM, Institute for Health Metrics; THR, total hip replacement; TKR, total knee replacement.
Trends in VTE prophylaxis during 2011 to 2013
During the study period, greater than 90% of patients received some type of pharmacologic VTE prophylaxis during the hospital stay and nearly all patients were prescribed pharmacologic VTE prophylaxis at the time of discharge (Tables 2A and 2B). In 2011, warfarin was the most often administered in-hospital and discharge prophylaxis for both hip and knee replacement. The use of a DOAC, almost exclusively rivaroxaban (in-hospital sample 98.3%, discharge sample 98.0%), changed dramatically during the study period for both THR and TKR, accounting for a quarter to a third of prophylaxis in 2013. The in-hospital use of LMWH remained stable during the study period whereas the prescription of LMWH at discharge dropped by 4–5% from 2012 to 2013. For the in-hospital sample, the use of aspirin as sole pharmacologic prophylaxis remained constant for TKR across the study period (13%), but declined significantly for THR from 12% in 2011 to 6% in 2013. At discharge, aspirin as the sole prophylaxis accounted for 17% of THR and 19% of TKR in 2013.
In-hospital prophylaxis by surgery type and year.
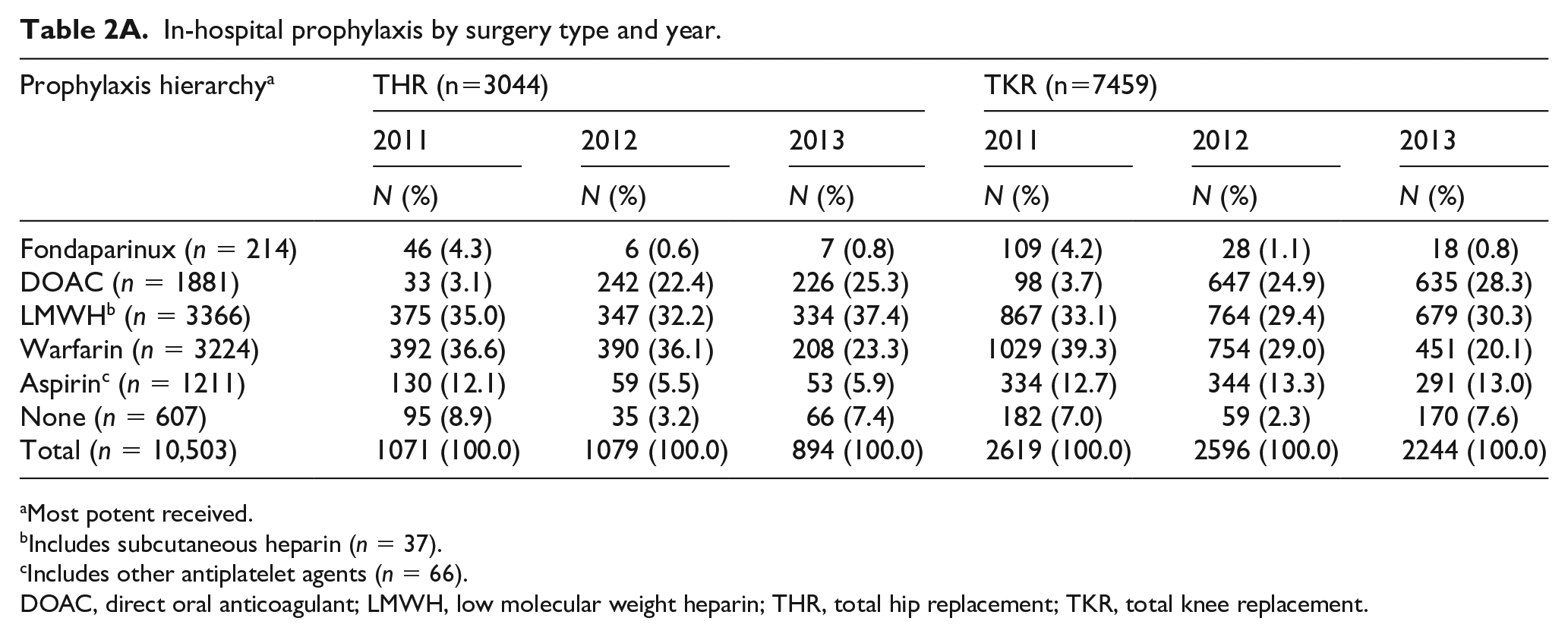
Most potent received.
Includes subcutaneous heparin (n = 37).
Includes other antiplatelet agents (n = 66).
DOAC, direct oral anticoagulant; LMWH, low molecular weight heparin; THR, total hip replacement; TKR, total knee replacement.
Discharge prophylaxis by surgery type and year.
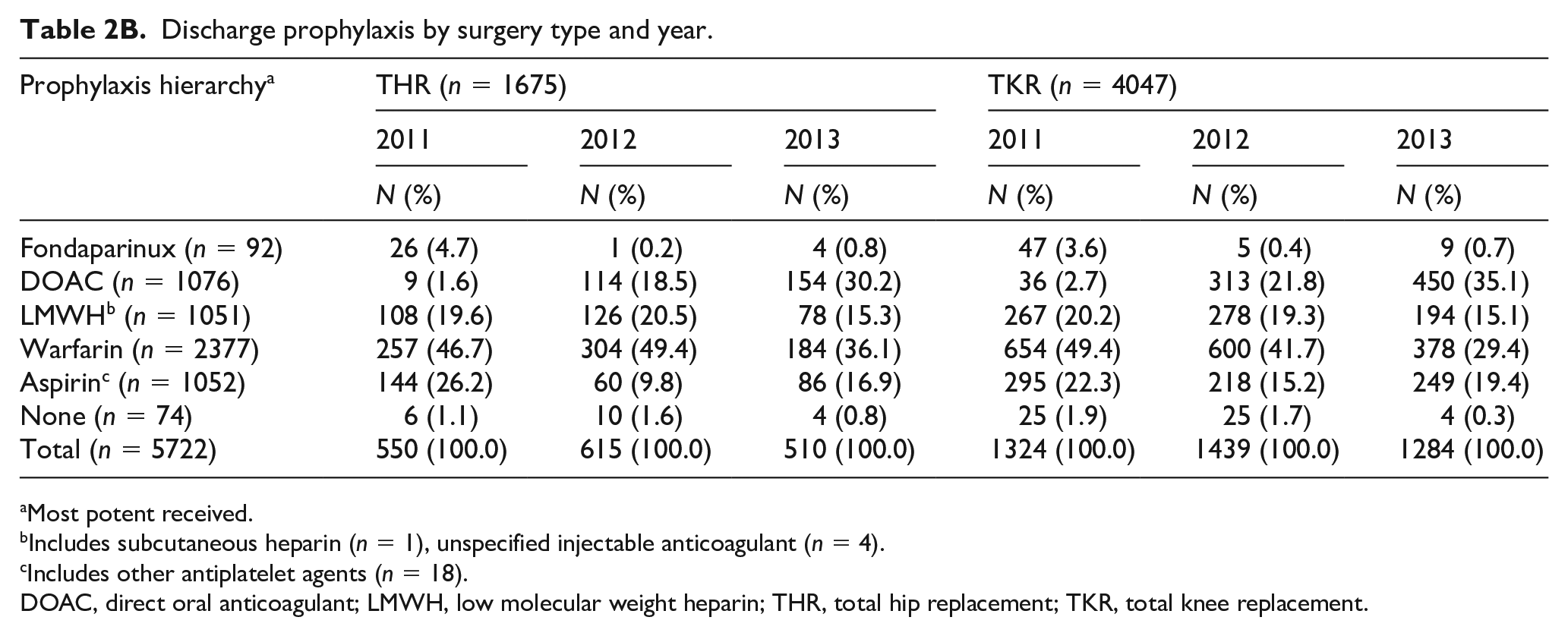
Most potent received.
Includes subcutaneous heparin (n = 1), unspecified injectable anticoagulant (n = 4).
Includes other antiplatelet agents (n = 18).
DOAC, direct oral anticoagulant; LMWH, low molecular weight heparin; THR, total hip replacement; TKR, total knee replacement.
VTE prophylaxis by age
The proportion of patients who received aspirin only or no prophylaxis was nearly identical across the four age groups for both in-hospital (83%) and the discharge (80%) samples (Tables 3A and 3B). Patients aged 80 years and older more often received warfarin as prophylaxis (p-values for trend < 0.0001 for both the in-hospital and discharge samples) and less often received a DOAC (p-values for trend < 0.001 for both the in-hospital and discharge samples) compared to the younger groups. Combination therapy with aspirin plus an anticoagulant was more frequent among patients aged 80 years and older compared to the younger age groups (Tables S2A and S2B).
In-hospital prophylaxis stratified by age group for all arthroplasty.
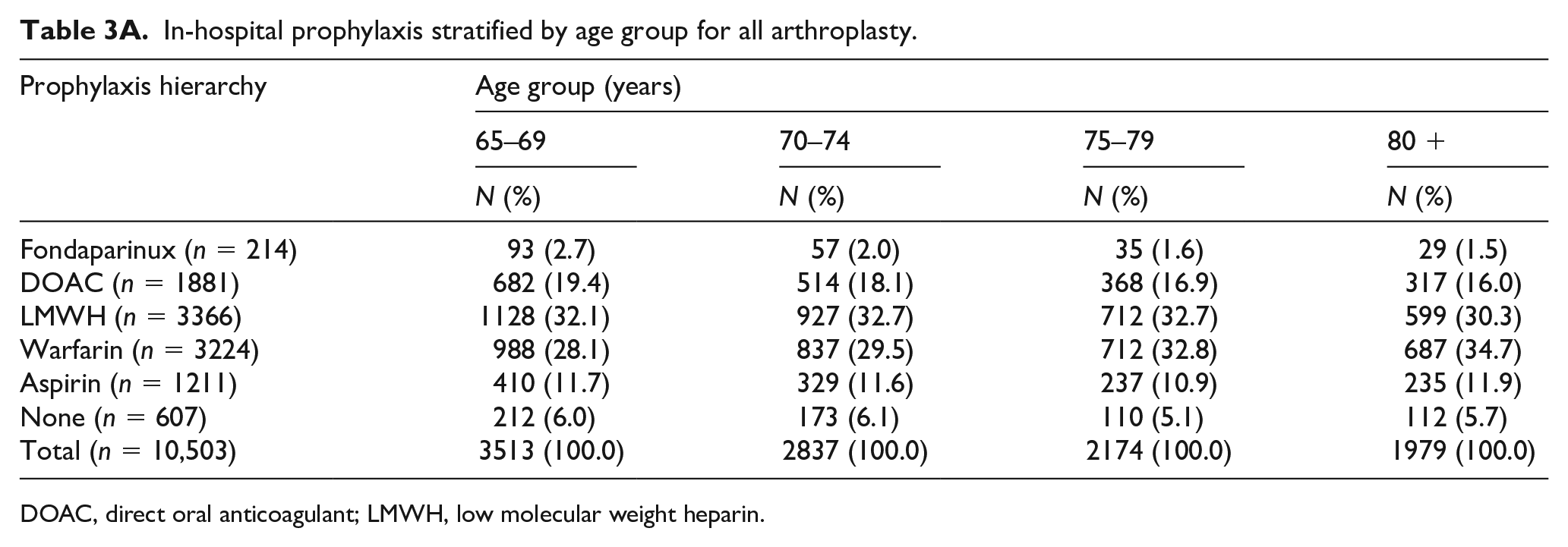
DOAC, direct oral anticoagulant; LMWH, low molecular weight heparin.
Discharge prophylaxis stratified by age group for all arthroplasty.
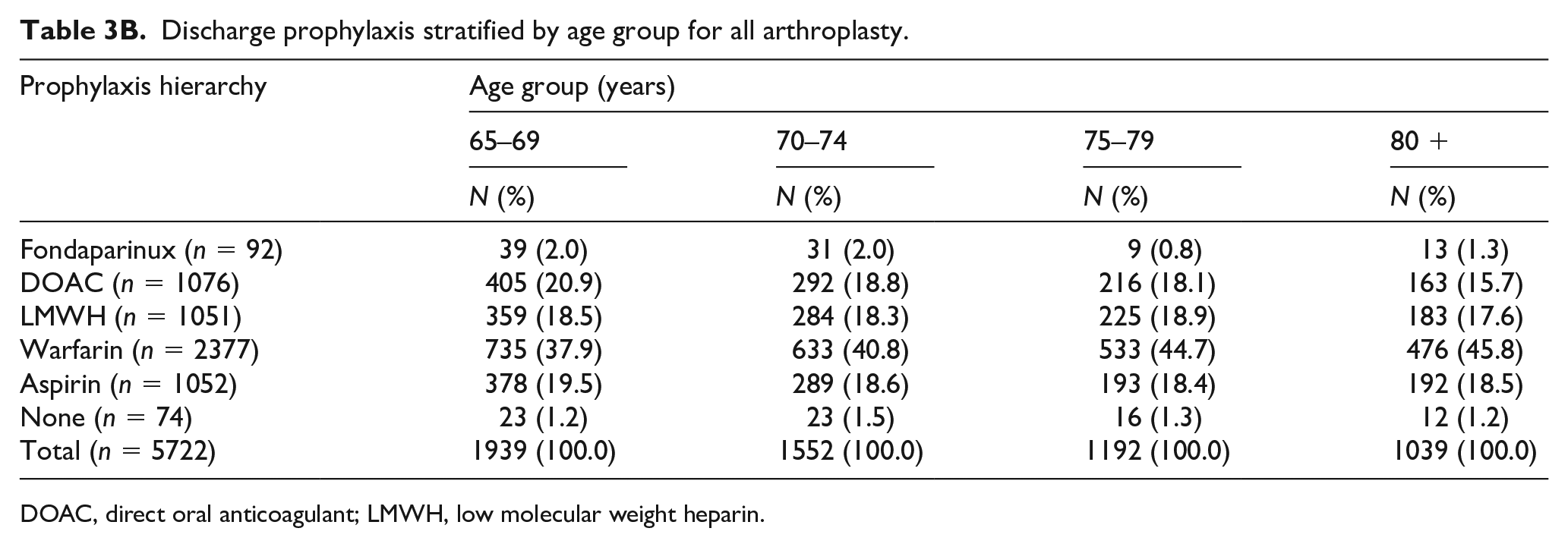
DOAC, direct oral anticoagulant; LMWH, low molecular weight heparin.
Variations in practice by hospital
Hospitals varied widely in terms of their prophylaxis strategies as shown for those hospitals with 50 or more THR or TKR surgeries during 2012 to 2013 (Figures 2A and 2B). The proportion of patients receiving a DOAC ranged from 0% to 90%. Aspirin use ranged from 0% to nearly 50%. There were also wide variations in the VTE prophylaxis strategies by hospital size and the overall volume of surgical operations (Tables S3A and S3B).
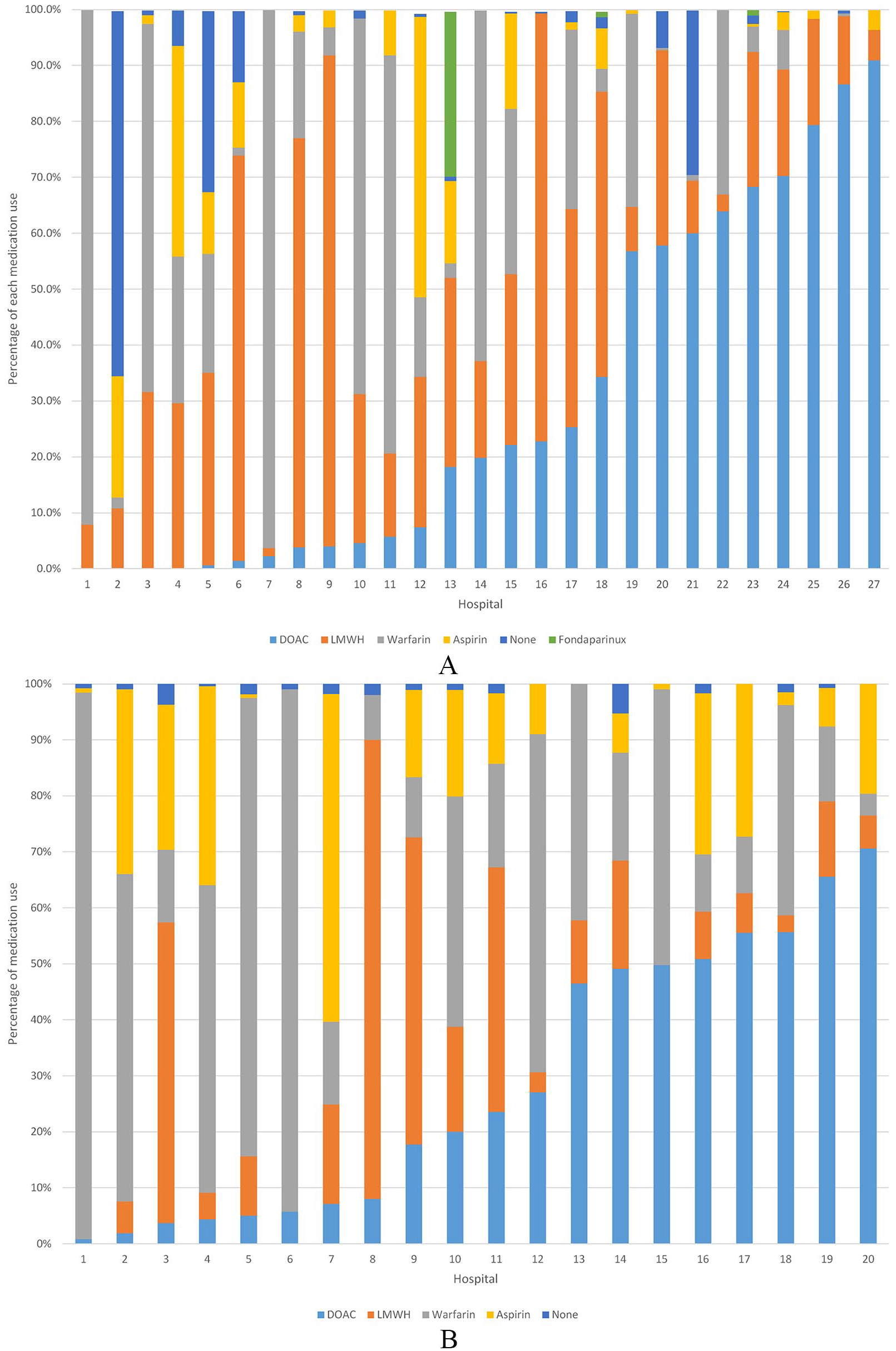
(A) In-hospital prophylaxis for VTE following THR and TKR by hospitala. (B) Discharge prophylaxis for VTE following THR and TKR by hospitala. Light blue = DOAC, orange = LMWH, gray = warfarin, yellow = aspirin, dark blue = none, green = fondaparinux.
In-hospital and 90-day discharge outcomes stratified by prophylaxis
The overall risk of both in-hospital and 90-day discharge adverse events was low (Tables 4A and 4B). Among the 10,503 surgeries in the in-hospital sample, there were 14 deaths (0.13%), 57 VTE events (0.54%), 45 cardiovascular events (0.43%), and 26 major hemorrhages (0.25%). Among the 5722 surgeries in the discharge sample, there were 18 deaths (0.31%), 35 VTE events (0.61%), 15 cardiovascular events (0.26%), and 17 major hemorrhages (0.30%). The low number of adverse events precluded statistical comparison of adverse events associated with prophylaxis regimens.
In-hospital outcomes stratified by prophylaxis for all arthroplasty a .
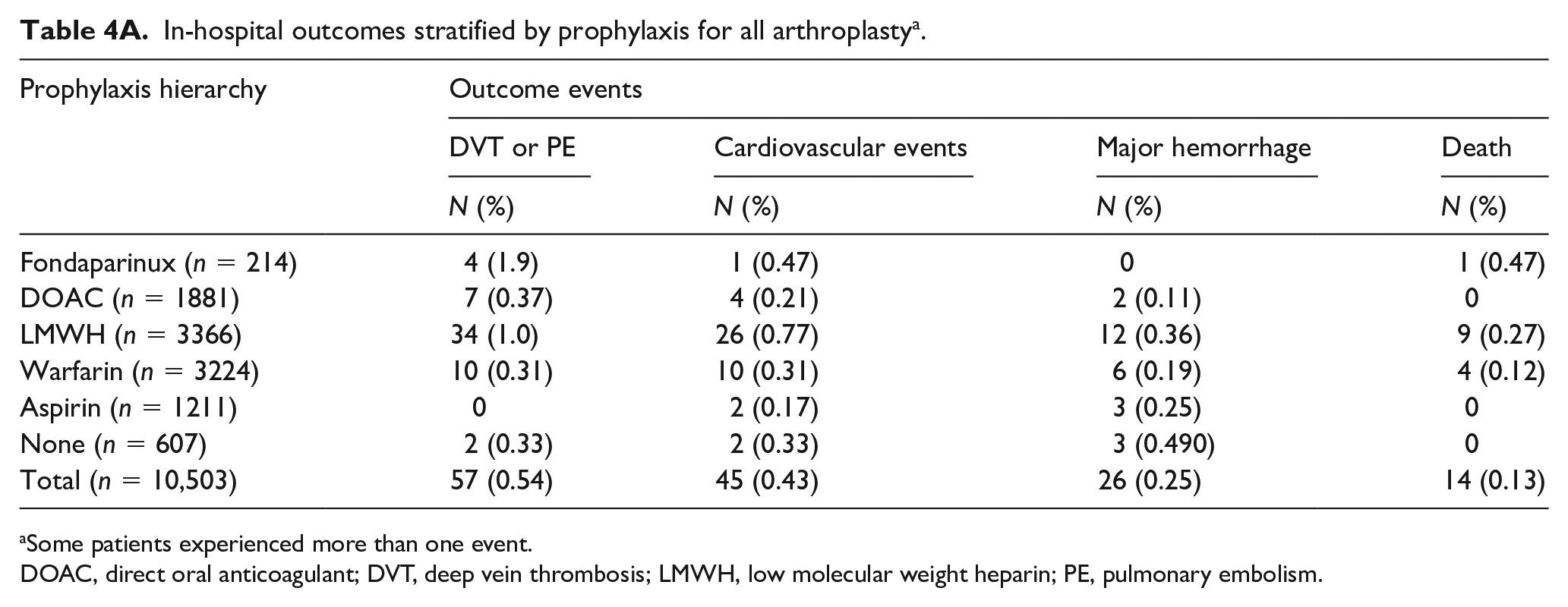
Some patients experienced more than one event.
DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; LMWH, low molecular weight heparin; PE, pulmonary embolism.
The 90-day discharge outcomes stratified by prophylaxis a .
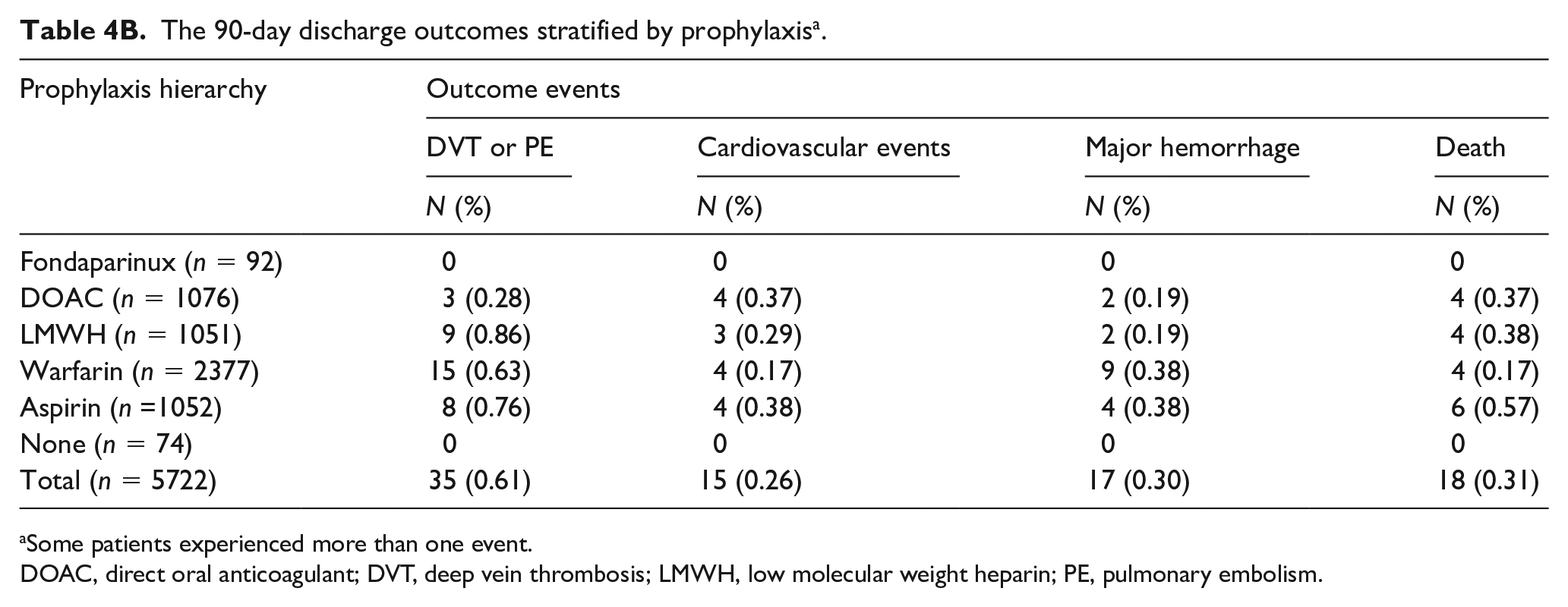
Some patients experienced more than one event.
DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; LMWH, low molecular weight heparin; PE, pulmonary embolism.
Discussion
In our analysis of older adults undergoing THR and TKR, we note several important trends in VTE prophylaxis. First, nearly 93% and 99% of patients received some form of pharmacologic prophylaxis in-hospital and at discharge, respectively, affirming the recognition of this high-risk surgery and the effectiveness of the long-running campaign for VTE prophylaxis in this setting. Second, during the study period 2011–2013, DOAC use increased substantially following the first approval for this indication in 2011. 25 By 2013, DOAC was the prophylaxis choice for 25% and 30% of patients with THR and 28% and 35% of patients with TKR during the in-hospital period and at discharge, respectively. Third, aspirin was the sole discharge prophylactic agent for 17% and 19% of patients undergoing THR and TKR, respectively. Fourth, warfarin remained the prophylaxis agent of choice for patients aged 80 years and older, and despite their higher bleeding risk, this age group was also more often prescribed aspirin plus an anticoagulant. Lastly, the overall rate of adverse outcomes was low in this contemporary population of older adults undergoing elective arthroplasty.
Although our study documents a significant shift in prophylactic choice, it reaffirms wide variation in clinical practice. In addition, our study demonstrates a substantial uptake of aspirin as a sole prophylaxis agent, which may reflect endorsement of aspirin by professional guidelines. The AAOS guideline incorporates data from observational studies due to concerns that the rates of adverse outcomes in randomized controlled trials of VTE prophylaxis11–21 do not accurately reflect the true bleeding risk in the sicker, frailer ‘real-world’ patient population.7,26 In an attempt to balance the efficacy and the bleeding risk of antithrombotic treatments, the first AAOS guideline published in 2008 recommended aspirin 325 mg twice daily in conjunction with mechanical prophylaxis as a relatively effective and safe prophylaxis strategy compared to anticoagulants for the prevention of PE. 7 However, the orthopedic guideline has been criticized for not recognizing the clear pathophysiological mechanism linking (symptomatic and asymptomatic) DVT and PE, and for lack of scientific evidence supporting the efficacy and safety of the high-dose aspirin and its twice daily dosing. 27 In a controversial decision, the ACCP guideline was revised in 2012 to recommend aspirin based on the results of the PEP (Pulmonary Embolism Prevention) trial. 28 Although the trial showed the benefit of aspirin (160 mg once daily) compared to placebo for prevention of symptomatic DVT in combined results (arthroplasty and hip fracture), it did not show benefit among the elective hip arthroplasty patients. 28 Subsequent trials reported the non-inferiority of aspirin compared to dalteparin 29 or rivaroxaban 10 mg once daily, 30 but the studies have major limitations including failure of randomization 29 and the trial events occurring prior to randomization and initiation of the trial medications. 30
The preference for warfarin among patients aged 80 years and older may reflect a hesitance to use DOACs due to reduced kidney function, increased risk of bleeding, and lack of data for this age group. Whether rivaroxaban increases the risk of bleeding compared to LMWH is inconclusive. A recent meta-analysis of randomized trials demonstrated that although rivaroxaban reduced the risk of VTE significantly compared to enoxaparin, it increased the risk of major/clinical relevant bleeding. 31 However, a network meta-analysis of randomized trials reported that the risk of major hemorrhage was not significantly different between once-daily LMWH and direct oral factor Xa inhibitors. 32 More data are needed on the comparative effectiveness of these agents, particularly in the oldest patient groups.
The overall number of adverse events during the hospital stay in our study was low and consistent with other recent estimates.30,33 A meta-analysis of studies published between 1996 and 2011 reported that among randomized clinical trials, the rates of in-hospital symptomatic VTE were 0.51% and 0.31% for LMWH and DOACs, respectively. Among observational studies of LMWH, the rate of VTE was similarly very low at 0.83%. 33 The low number of events in current studies may reflect early mobilization, improved surgical techniques, shorter hospital stay, and greater use of perioperative anticoagulation and mechanical prophylaxis. Our results confirm significant advances toward the Centers for Medicare & Medicaid Services’ goal of ‘VTE being a never event post-orthopedic surgery’. 34
Limitations and strengths
Our study has several limitations. We were unable to extend our study period beyond 2013, and there may have been further changes in the medication utilization for VTE prophylaxis after the joint replacements. A review of about 22,000 THR and TKR cases performed during 2014 to 2016 indicates that aspirin use in combination with sequential compression devices has increased substantially in the past few years. 35 Whether there have been further increases in the use of DOACs and decreases in the use of other anticoagulants after hospital discharge is unknown and should be the subject of future investigation. Owing to our use of hospital records only, we were unable to accurately ascertain some baseline characteristics such as prior history of VTE or major bleeding. We did not have accurate information on the use of mechanical prophylaxis, and we were unable to obtain information about the exact dosage of aspirin, which is most often purchased over the counter. We were unable to account for patients who may have been lost to follow-up because they did not receive follow-up care within the IHM network. Finally, the small number of adverse events in our study, similar to reported estimates in previous studies,30,33 precluded comparative analyses.
Nonetheless, our study has several important strengths. Inclusion of only the hospitals with the barcoded medication validation system for the in-hospital sample allowed the most accurate ascertainment of the prophylaxis medication as the codes are scanned at the patient bedside at the time of medication administration. In addition, we were able to accurately ascertain the discharge prophylaxis medication, particularly aspirin, by including only the surgeries with an electronic medical record (physician’s discharge text summary) of discharge prophylaxis medication. Our study sample presents a major opportunity to examine practice patterns of VTE prophylaxis after THR and TKR in an older, higher-risk patient population outside of the academic setting. Among the studies included in the systematic review of in-hospital VTE prophylaxis, 33 only four of 20 studies for TKR and one of 23 studies for THR had a sample whose mean age was over 70 years. In comparison, the mean age of our study sample was 73–74 years and 19% were 80 years of age or older. Physician validation of the outcome events using documentation of imaging evidence and other source documents is an additional strength.
Conclusion
In conclusion, pharmacologic prophylaxis following elective joint replacement is nearly universal in this contemporary cohort of older patients, with wide variation in surgeon choice of agent. Overall the rates of both in-hospital and 90-day adverse outcomes were low. Given the projected increase in THR and TKR among older adults due to the growth of the older population, more studies are needed on the comparative effectiveness of different prophylaxis strategies during hospitalization and following discharge for this age group.
Supplemental Material
VTE_AJMed_eSupplement_resubmission – Supplemental material for Temporal trends in pharmacologic prophylaxis for venous thromboembolism after hip and knee replacement in older adults
Supplemental material, VTE_AJMed_eSupplement_resubmission for Temporal trends in pharmacologic prophylaxis for venous thromboembolism after hip and knee replacement in older adults by Darae Ko, Alok Kapoor, Adam J Rose, Amresh D Hanchate, Donald Miller, Michael R Winter, Joseph N Palmisano, Lori E Henault, Lisa Fredman, Allan J Walkey, Yorghos Tripodis, Anita Karcz and Elaine M Hylek in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Karcz is the Chief Medical Officer for the Institute for Health Metrics. Dr Hylek has served on advisory boards for Armetheon, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Janssen, Medtronic, Pfizer, and Portola. The other authors had nothing to disclose.
Funding
This study was supported by the National Heart, Lung, and Blood Institute awards 5T32HL007224-41 and 5R01HL106029.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
