Abstract
Preeclampsia is a life-threatening multiorgan systemic disease with manifestations including gestational hypertension, oxidative stress, and vascular dysfunction. We aimed to evaluate the therapeutic effects of melatonin on an L-NAME (NLG-nitro-
Introduction
Preeclampsia is a high-risk disease in pregnant women and the clinical management of the disease is of paramount importance. 1 Hypertension, elevated oxidative stress, and cardiovascular system dysfunctions are manifestations of preeclampsia. 2 The cause of preeclampsia is multifactorial, such as an abnormal circadian blood pressure rhythm with elevated blood pressure during sleep. 3 There is a continuous search for therapeutics for preeclampsia. For example, aspirin has been used to reduce blood pressure and alleviate oxidative stress with remarkable improvement in gestational outcome. However, aspirin can cause gastric hemorrhage among other side effects, and a safe alternative is needed. 4
Melatonin (N-acetyl-5-methoxytryptamine) is a hormone secreted by the pineal gland during the night, which plays a critical role in a number of physiological functions of the body, including regulation of energy balance, circadian rhythm, epigenetic regulation, and control of reproduction. 5 However, the pineal gland is not the exclusive source of melatonin during pregnancy, since the placenta also secretes melatonin. 6 Preeclampsia results in suppressed melatonin secretion by the placenta, along with altered melatonin receptor expression, therefore leading to melatonin deficiency. 7 Numerous studies have focused on the correlation between serum melatonin levels and dysregulations during pregnancy, and the underlying mechanisms associated with the important role of melatonin.3,8–11
The clinical utility of melatonin therapy, which administers exogenous melatonin that compensates for a deficiency of endogenous melatonin, has been demonstrated.12,13 For example, exogenous melatonin was found to decrease blood pressure in normotensive patients, adolescents with diabetes mellitus type 1, and in those with essential hypertension. 14 It has been suggested that melatonin is also an indispensable regulator of immune responses. 15 In addition, melatonin was shown as a physiologically important antioxidant. 16 Consequently, melatonin may participate in the pathophysiology of a variety of diseases and its concentrations are being studied in various clinical settings. To further the application of melatonin in the clinical management of preeclampsia, the efficacies of melatonin in reducing blood pressure and improving the physiological parameters of pregnant women and gestational outcomes remain to be fully clarified.
The aim of our study was to evaluate the effects of melatonin treatment in attenuating blood pressure and oxidative stress in an L-NAME (NLG-nitro-
Materials and methods
Rat preeclampsia model
All animal experiments in our study were approved by Tianjin Central Hospital of Obstetrics and Gynecology. Sprague-Dawley rats (female, 5–7 weeks old, body weight 210–250 g) were acquired from Slaccas Inc. (Shanghai, China). The rats were housed in a temperature-controlled room (22–24°C) with a 12/12 light/dark cycle and fed with standard rat chow (LabDiet 5001) and tap water. Gestation was initiated by placing every two female rats with one male rat that was weight matched. Sperm and estrum were used for confirming gestational day (GD) 0. From GD8, L-NAME (Sigma, St Louis, MO, USA) was added to drinking water at the dose of 50 mg/kg/day until GD19. At GD19, the rats were euthanized using isoflurane overdose and blood was collected before sacrifice from the vena cava. Blood samples were centrifuged for 10 min at 3000 g to extract plasma. The placenta was excised after euthanization.
Melatonin and aspirin treatment
From GD8 to GD19, rats were either intraperitoneally injected with melatonin at the dose of 10 mg/kg/day or aspirin at the dose of 1.5 mg/kg/day mixed with rodent chow (Transgenic Dough Diet, Bio-Serv, Flemington, NJ, USA). Rats were divided into the following five treatment groups (n = 9 for each group): control group, control+melatonin group, L-NAME group, L-NAME+melatonin group, and L-NAME+aspirin group. In the control group and L-NAME group, rats were intraperitoneally injected with vehicle only.
Blood pressure and urine protein content analyses
The BP-2000 Analysis System (Visitech, Inc., Apex, NC, USA) was used to measure the systolic pressure of rats based on the noninvasive tail-cuff method according to manufacturer’s recommendations. The urine protein content was determined by applying Coomassie blue stain (Jiancheng Institute of Biotechnology, Nanjing, China) to urine samples collected from individually metabolic caged rats (Tecniplast, Buguggiate, Italy).
Plasma analysis
The malondialdehyde (MDA) levels in plasma were measured based on the thiobarbituric acid reaction according to a previous method. 19 A total antioxidant assay (Cayman Chemical, Ann Arbor, MI, USA) was used to determine the Trolox-equivalent antioxidant capacity of the plasma. The levels of the soluble fms-like tyrosine kinase (sFlt-1) and nuclear factor erythroid 2-related factor 2 (Nrf2) were determined using enzyme-linked immunosorbent assays (Quantikine; R&D Systems, Minneapolis, MN, USA) according to manufacturer’s recommendations.
Western blot
Placenta tissues were homogenized and measurement of protein concentration was performed using the bicinchoninic acid assay (Abcam, Cambridge, MA, USA). Next, 50 μg of protein loaded onto 4–20% sodium dodecyl sulfate polyacrylamide gels was separated by electrophoresis, and then transferred to nitrocellulose membranes. After blocking for 1 h using 1% bovine serum albumin, antibodies for sFlt-1, placental growth factor (PlGF), Nrf2, heme oxygenase-1 (HO-1) or β-actin (Abcam, Cambridge, MA, USA) were added to the membrane and incubated overnight at 4°C. PBS-T (phosphate-buffered saline, Tween-20, 0.1%) was used to wash the membranes, followed by incubation with horseradish peroxidase conjugated secondary antibodies for 1 h (Cell Signaling, Danvers, MA, USA). The primary antibodies were diluted as 1:500 and secondary antibodies were diluted as 1:2000. Chemiluminescent substrate (West-Femto, Pierce Biotechnology Inc., Rockford, IL, USA) was then added. The protein bands were digitized using an Alpha-Innotech digital imaging system and quantified using ImageJ (NIH, Bethesda, MD, USA). Beta-actin was used to normalize protein expression.
Statistical analysis
Data were presented as mean ± SD. One- or two-way analysis of variance (ANOVA) with a Bonferroni post hoc test was used for comparing the differences, and p < 0.05 was considered statistically significant. GraphPad Prism 7.01 (GraphPad Software, Inc., La Jolla, CA, USA) was used for data analysis.
Results
Melatonin lowers systolic blood pressure and urine protein content
We constructed a preeclampsia rat model by raising the blood pressure using L-NAME, which induces hypertension via inhibition of NO synthase activity 17 in rats under gestation. Blood pressure and urine protein content were monitored every 3 days. On GD19, blood samples were collected and mice were sacrificed. As shown in Figure 1A, L-NAME significantly elevated the blood pressure with increasing gestational days compared to the control group (p < 0.001, n = 9). Meanwhile, urine protein content was also elevated by L-NAME (p < 0.001 vs control group), and gradually increased over 12 days after L-NAME treatment (Figure 1B). Notably, rats treated with L-NAME+melatonin demonstrated significantly lowered systolic pressure (p < 0.001) and urine protein content (p < 0.001) compared with the untreated L-NAME group, similar to what was observed for L-NAME rats treated with aspirin (p < 0.001).
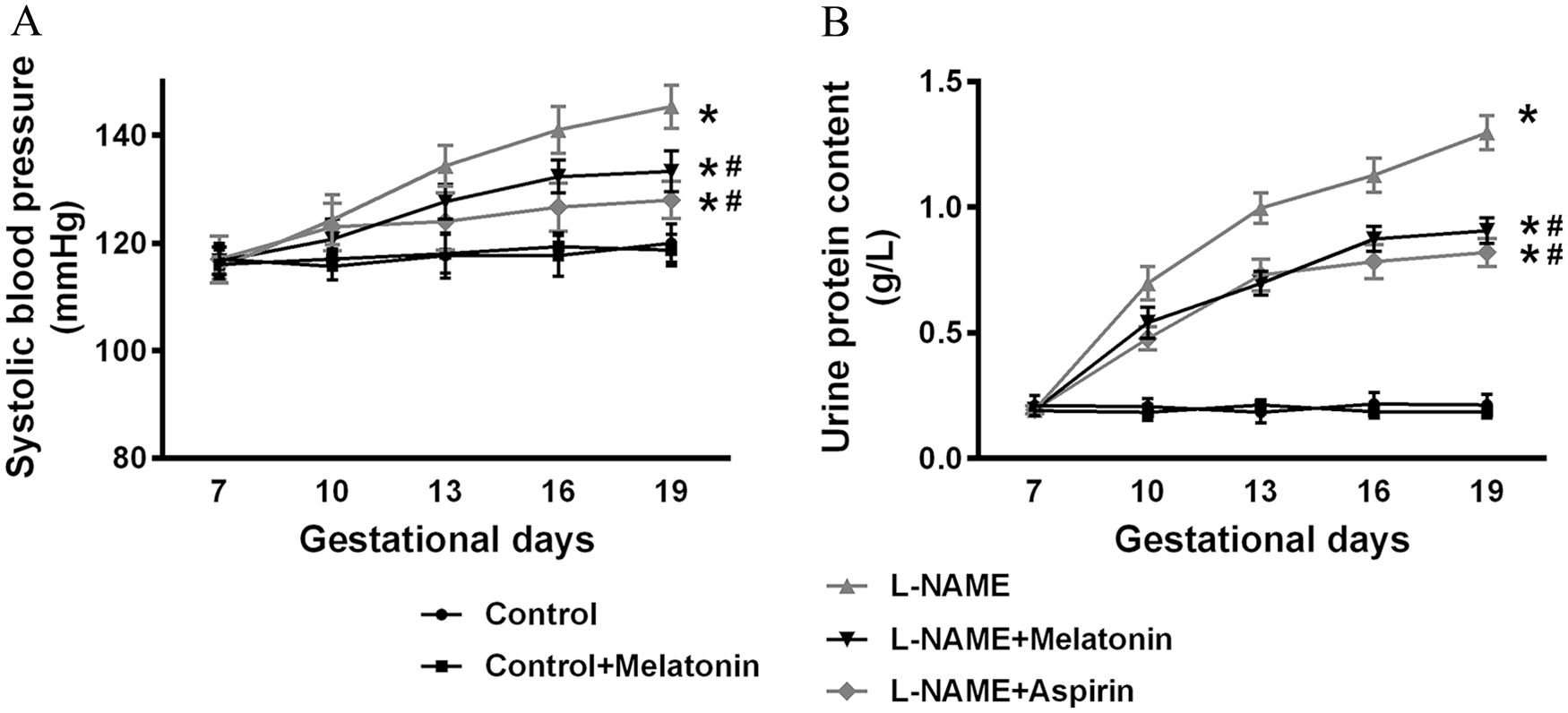
Melatonin attenuates systolic blood pressures (A) and urine protein content (B) in L-NAME-induced preeclampsia rats (n = 9 for each group). Data presented as mean ± SD.
Effects of melatonin on pregnancy outcomes in pregnant rats exposed to L-NAME
Next, we evaluated the effects of melatonin on pregnancy outcomes using the fetus alive ratio, fetal weight, placental weight, and the fetal weight/placenta weight ratio as the parameters. As shown in Figure 2, while L-NAME treatment exerted detrimental effects in pregnancy outcomes (i.e. decrease in the fetus live ratio (p < 0.001), fetal weight (p < 0.001), placental weight (p = 0.017), and the fetal weight/placental weight ratio (p < 0.001) compared to control and control+melatonin groups), treatment with melatonin resulted in ameliorating effects by improving these parameters. The effects were similar to those of aspirin. It should be noted that, while no significant improvement was seen in placental weight after melatonin or aspirin treatment, the improvements were significant in terms of the fetal weight/placental ratio (p = 0.029 in L-NAME+melatonin group and p = 0.030 in L-NAME+aspirin group) compared with the L-NAME group.
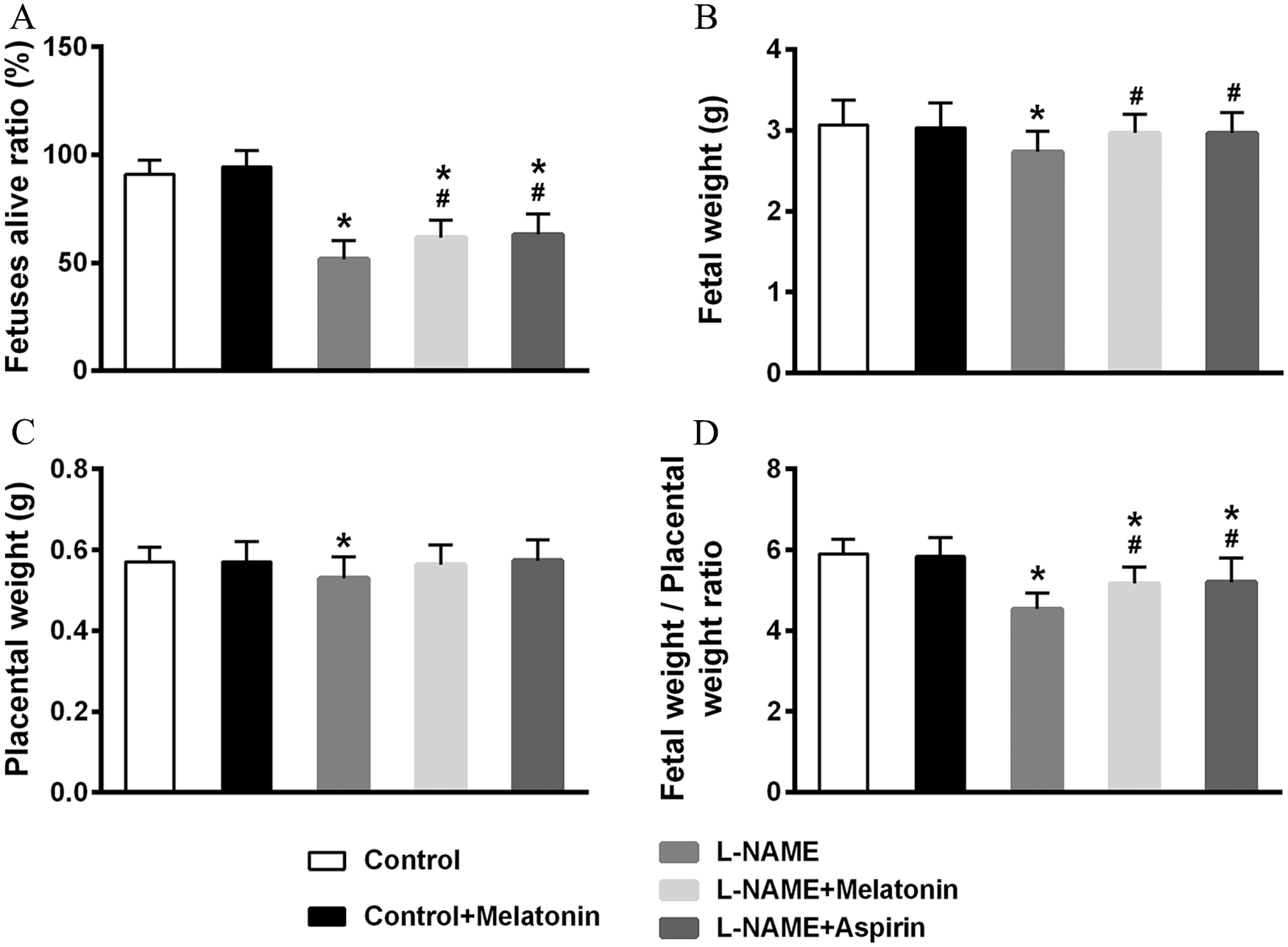
Effects of melatonin on pregnancy outcomes in pregnant rats exposed to L-NAME. The fetuses alive ratio (A), fetal weight (B), placental weight (C), and fetal weight/placental weight ratio (D) were evaluated on gestational day 19 (n = 9 for each group). Data presented as mean ± SD.
Melatonin increases antioxidant capacity and decreases oxidative stress
Further, we sought to evaluate how melatonin affected the oxidative stress of rats. The Trolox-equivalent antioxidant capacity of plasma was decreased significantly by L-NAME (p < 0.001), which was partially recovered by melatonin and aspirin treatment (p = 0.049 and p = 0.017, respectively) (Figure 3A). Plasma MDA was upregulated by L-NAME, suggesting elevated oxidative stress (p < 0.001) (Figure 3B), while treatment with melatonin and aspirin reversed this upregulation (p = 0.045 and p = 0.016, respectively). These data indicated the effects of melatonin in increasing antioxidant capacity and decreasing oxidative stress.
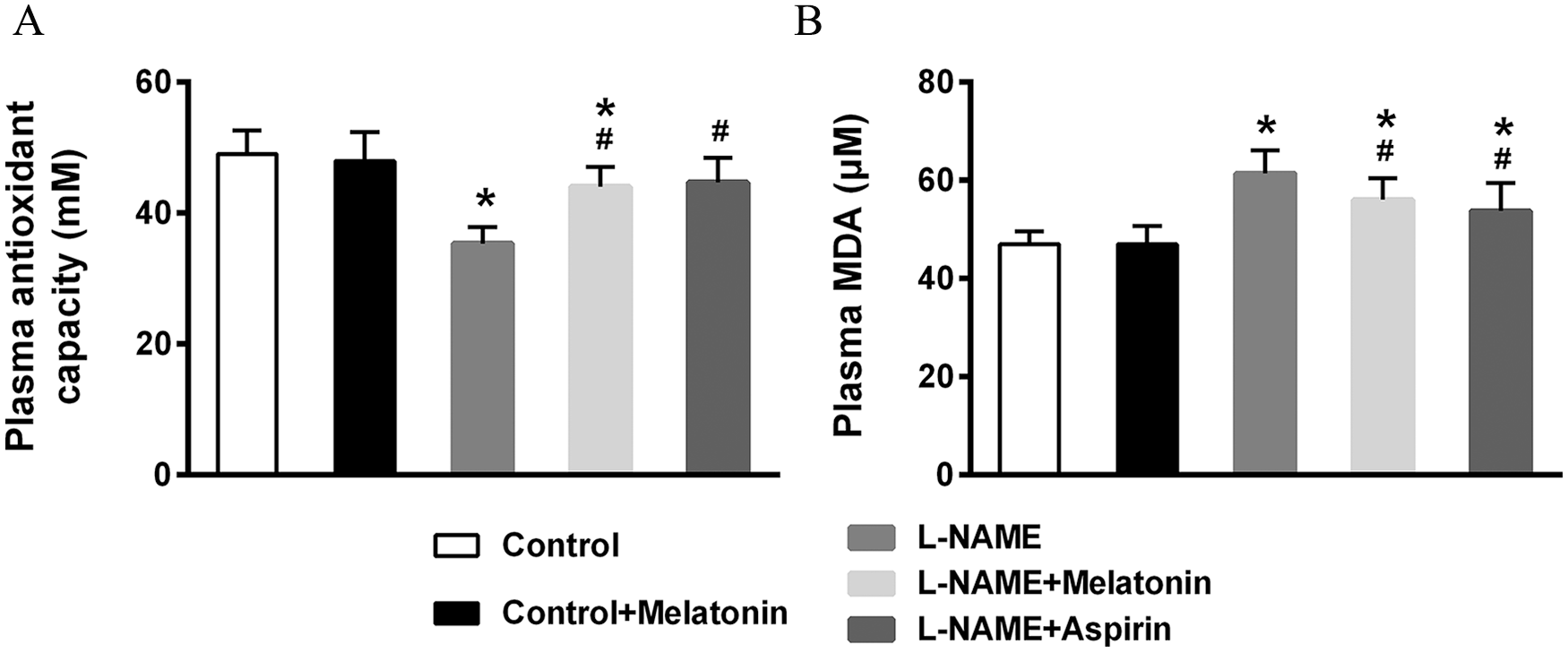
Effects of melatonin on plasma Trolox antioxidant capacity (A) and MDA (B) in L-NAME-induced preeclampsia rats (n = 9 for each group). Data presented as mean ± SD.
Melatonin exerts protective effects on cardiovascular parameters
sFlt-1 and PlGF are essential factors for the onset of preeclampsia. 20 To further elucidate the benefits of melatonin, the levels of sFlt-1 and PlGF in the serum were evaluated using biochemical analyses, which suggested that while L-NAME increased sFlt-1 levels (Figure 4A), decreased PlGF levels (Figure 4B), and increased the ratio of sFt-1/PlGF (Figure 4C), treatment with melatonin and aspirin reversed these effects.
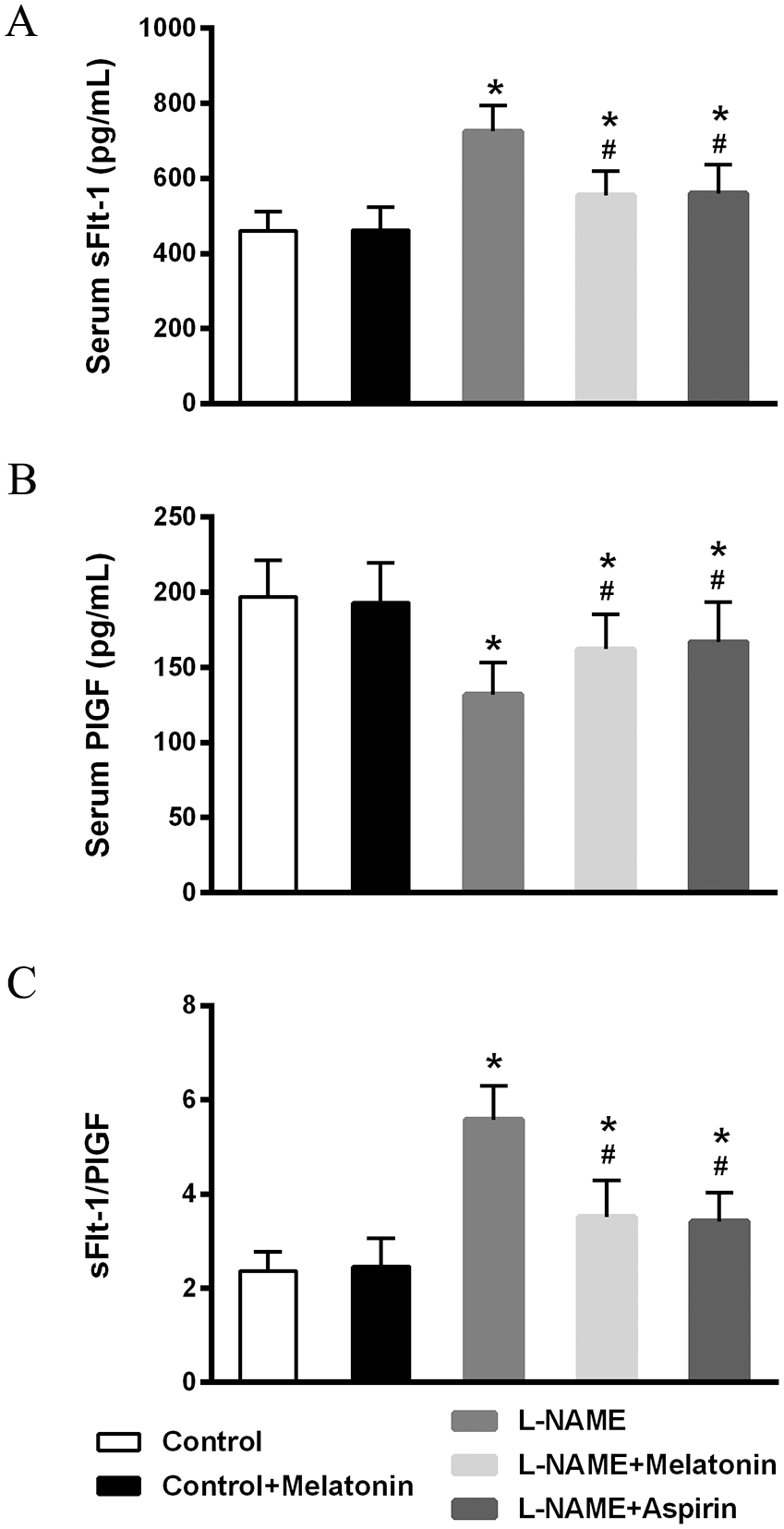
Effects of melatonin on serum levels of sFlt-1 (A), PlGF (B), and the sFlt-1/PlGF ratio (C) in L-NAME-induced preeclampsia rats (n = 9 for each group). Data presented as mean ± SD.
As Nrf2 and HO-1 are beneficial factors against oxidative stress, their protein levels, as well as those of sFlt-1 and PlGF, were evaluated using Western blot in placental tissues. Similar to the observations in the serum, melatonin and aspirin treatment reversed the effects of L-NAME in upregulating sFlt-1 (p < 0.001 for L-NAME group compared to control and control+melatonin groups; p = 0.042 for L-NAME+melatonin group compared to L-NAME group; and p = 0.049 for L-NAME+aspirin group compared to L-NAME group) (Figure 5A and B) and downregulating PlGF (p < 0.001 for L-NAME group compared to control and control+melatonin groups; p = 0.045 for L-NAME+melatonin group compared to L-NAME group; and p = 0.037 for L-NAME+aspirin group compared to L-NAME group) (Figure 5A and C). Nrf2 and HO-1 were suppressed by L-NAME (p < 0.001 compared to the control group) (Figures D–F), but melatonin (p = 0.040 for Nrf2 and p = 0.046 for HO-1) and aspirin (p = 0.027 for Nrf2 and p = 0.037 for HO-1) treatment increased the expression of these proteins.
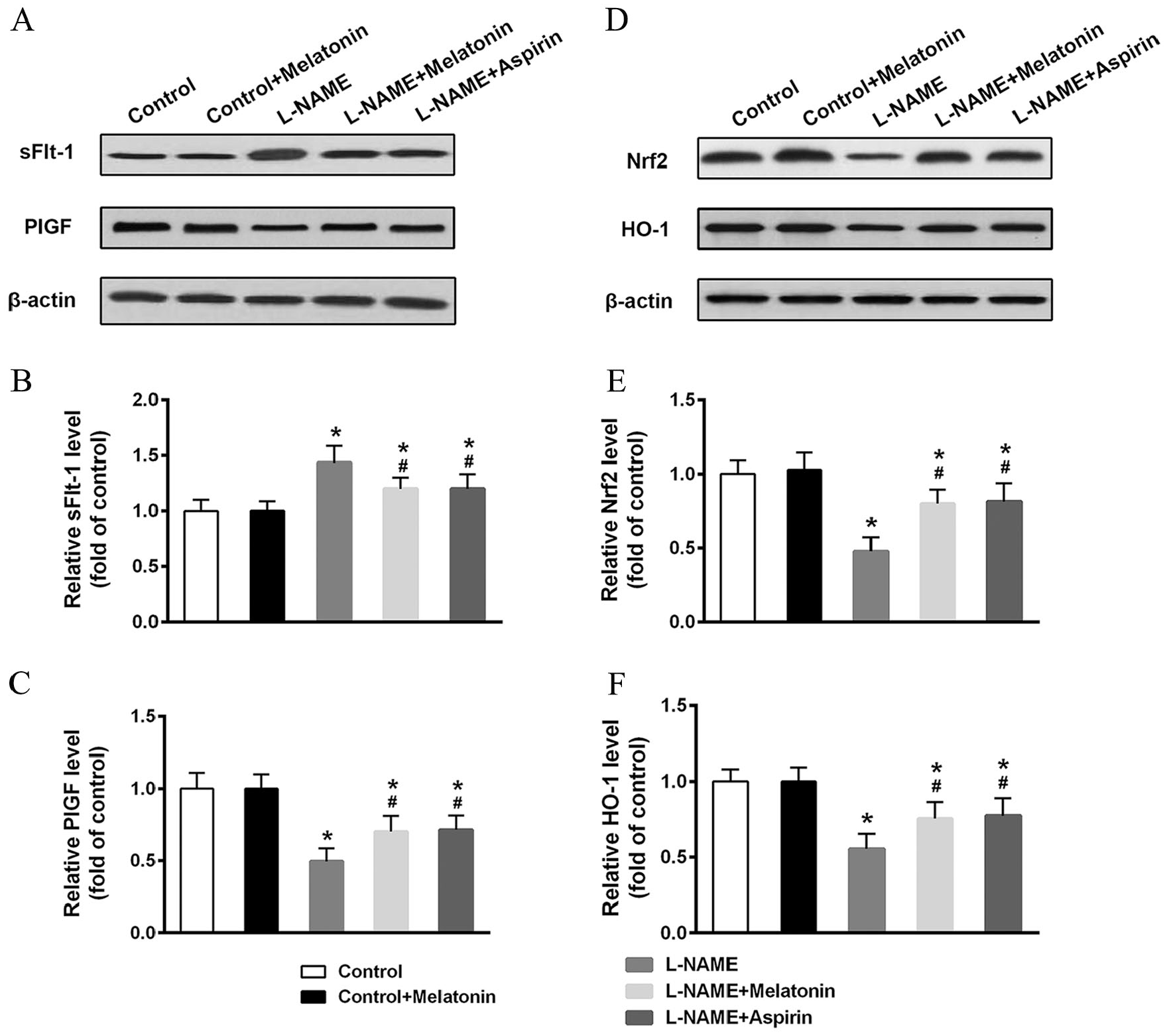
Effects of melatonin on placental protein levels of sFlt-1, PlGF, Nrf2, and HO-1. Placental protein levels of sFlt-1and PlGF (A) and Nrf2 and HO-1 (D) were determined by Western blot and relative levels (B, C, E, F) were analyzed (n = 9 for each group). Data presented as mean ± SD.
Discussion
In the present study, we investigated the efficacy of melatonin therapy on an L-NAME-induced rat preeclampsia model. Previously, melatonin has been verified to attenuate hypertension in a number of experimental animal models. 21 The L-NAME-induced preeclampsia model is a good representation of the NO-related models of preeclampsia, 22 characterized by hypertension, proteinuria, glomerular sclerotic injury, reduced glomerular filtration rate, intrauterine growth restriction, and thrombocytopenia. This model has been adopted by many recent studies to assess the effects of anti-preeclampsia drugs23–25 and placenta physiology. 26 Besides, melatonin has been reported as a versatile therapeutic drug with a key role in, for example, cancer,27,28 kidney diseases, 29 cardiovascular diseases, 30 and obesity. 31 Melatonin also is used for inducing sleep (e.g. to counter jet lag and for restoring the inherent sleep rhythm).
We hereby demonstrated that melatonin treatment led to prominently lowered blood pressure and proteinuria in L-NAME rats compared to untreated L-NAME rats. These blood pressure lowering effects are consistent with findings in the study of melatonin in kidney models.32,33 However, it is worth noting that we did not observe the pressure lowering effects of melatonin in healthy control rats, which is different from previous reports that melatonin also lowered blood pressure in normotensive subjects. 34 This is presumably due to differences in the melatonin’s effects on humans and rats, as well as blood pressure measurement approaches. Indeed, in animal experiments, the effects of melatonin are focused on ocular blood pressure rather than systolic blood pressure.35,36 Further, the antioxidative role of melatonin was confirmed by measuring the Trolox antioxidant capacity and MDA levels in the serum. Indeed, melatonin plays an important role in reducing oxidative stress by both serving as an antioxidant itself and stimulating antioxidant enzymes and suppressing pro-oxidative enzymes. 37 Therefore, these beneficial properties of melatonin support the use of this drug in attenuating symptoms of preeclampsia in pregnant women.
Moreover, melatonin has been found to be rapidly transferred from maternal to fetal circulation. 38 This property has been exploited to prevent oxidative stress damage and fructose intake-induced programmed hypertension in the offspring.39,40 Consistently, we found that melatonin therapy in the rat preeclampsia model could improve gestational outcomes, manifested as increased fetus alive ratio, fetal weight, placental weight, and fetal weight/placental weight ratio. The improved gestational outcomes were also supported by the reduced sFlt-1 and increased PlGF, suggesting the attenuation of preeclampsia.
We have also demonstrated that melatonin exerts significant effects in restoring Nrf2 and HO-1 levels. Here we attributed the effects to the antioxidative role of melatonin. It is also likely that these effects stemmed from the improved circadian rhythms induced by melatonin treatment, as suggested by previous studies.3,10 Further studies are needed to verify this hypothesis.
Aspirin is the most common drug for cardioprotection. 41 For instance, a low dose of aspirin is able to prevent one-fifth of atherothrombotic vascular complications. 42 Aspirin is also a commonly used drug with excellent efficacy in reducing mortality and morbidity in women with preeclampsia. 18 However, aspirin is known to cause gastric hemorrhage, prompting the search for a safer alternative cardioprotective drug. In this context, we also demonstrated a comparable efficacy of melatonin in lowering blood pressure and proteinuria, as well as reducing oxidative stress, using aspirin as a control. Our data have solidified that melatonin is a potential alternative to aspirin in protecting against preeclampsia. Although not tested in our study, melatonin is recently thought of as a new partner to aspirin,4,43 whereby melatonin could confer protection against gastric injury, and the ameliorating effects of both drugs could greatly benefit patients with preeclampsia. Currently, the use of melatonin therapy in preeclampsia women is being investigated by several clinical trials.44,45 Our results could provide further evidence on the benefits of melatonin on these women.
In conclusion, our study demonstrated that melatonin is a potent drug in lowering blood pressure and proteinuria, reducing oxidative stress, while improving gestational outcomes in a rat model of preeclampsia.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
