Abstract
The objective of this study was to investigate the correlation between the amount of blood flow in the area of neovascularization within a carotid atherosclerotic plaque by superb microvascular imaging (SMI) and the microvessel density (MVD) determined by histopathological staining. Twenty-eight carotid atherosclerotic plaques were detected by SMI in 28 patients who underwent carotid endarterectomy. SMI was graded according to the visual methods as follows: grade I: no appearance of neovascularization within the plaque; grade II: punctate neovascularization; grade III: one or two linear neovascularizations within the plaque; and grade IV: multiple (> 2) linear neovascularizations throughout the plaque. The neovascularization density was determined by the CD31 complex staining method. There was a significant correlation between the density of neovascularization in histopathologic plaques and the blood flow grade found by SMI (r = 0.788, p < 0.001). A significant difference was observed in SMI blood flow grade between the symptomatic and asymptomatic groups (χ2 = 2.634, p = 0.036). The MVD of plaques in the symptomatic group was significantly higher than that in the asymptomatic group (t = 2.530, p = 0.018). The SMI-based classification was positively correlated with plaque thickness. SMI, which is a new nonultrasound contrast-enhanced imaging method, can effectively evaluate neovascularization in carotid atherosclerotic plaques and can be used as a novel method for the clinical prediction of stroke risk.
Keywords
Introduction
Approximately 30% of cerebrovascular events are caused by atherosclerotic plaques. 1 Unstable plaques are one of the important causes of cerebrovascular events. An unstable plaque is an atherosclerotic plaque that is prone to rupture, thus activating platelets and leading to thrombosis and cerebrovascular events. An unstable plaque is characterized by an inflammatory reaction, a thin fibrous cap, a large lipid core, and neovascularization. Unstable plaques are closely associated with the occurrence of neovascularization in plaques;2,3 therefore, the stability of a plaque can be better evaluated by accurately observing neovascularization using imaging technology.
Contrast-enhanced ultrasound (CEUS), dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI), optical coherence tomography (OCT), computed tomography angiography (CTA), and other techniques can be used to detect carotid plaques and intraplaque neovascularization. CEUS is the main method used to evaluate intraplaque neovascularization, but this technique has clinical limitations. For example, this technique is not convenient and is costly. In recent years, Toshiba Medical Systems has developed a new Doppler technique called superb microvascular imaging (SMI).4,5 SMI is a microvascular flow imaging mode that is designed to improve blood flow visualization, especially of slow flow signals from microscopic vessels, using a new adaptive algorithm that dramatically removes clutter while maintaining very high frame rates.
The aim of our study was to investigate whether SMI could be used to effectively evaluate neovascularization in carotid atherosclerotic plaques and whether it is a potential new method for the clinical prediction of stroke risk.
Materials and methods
Subjects
The study subjects were 62 vascular surgery inpatients (65 carotid atherosclerotic plaques) who were admitted to our hospital with carotid artery stenosis detected by ultrasound (US) and were scheduled to undergo carotid endarterectomy (CEA) from January 2015 to April 2016. Our study was reviewed by the institutional review board (IRB), and we received informed consent from each participant in our study. The flow chart of the patient selection process is shown in Figure 1.
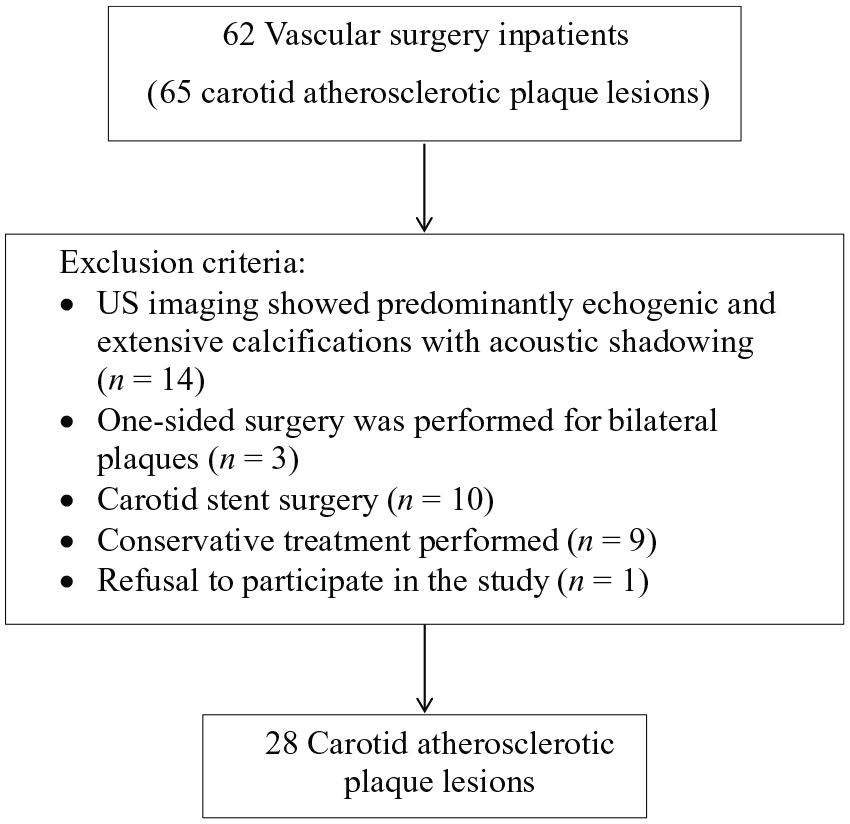
Flow chart of the patient selection process.
Enrollment criteria
The diagnosis of stenosis was classified as follows: (i) normal: when the internal carotid artery (ICA) peak systolic velocity (PSV) was < 125 cm/sec and no plaque or intimal thickening was visible, or ICA end-diastolic velocity (EDV) was < 40 cm/sec; (ii) 50% stenosis: when the ICA PSV was < 125 cm/sec and plaques or intimal thickening was visible or ICA EDV was < 40 cm/sec; (iii) 50–69% stenosis: when the ICA PSV was 125–230 cm/sec and plaques were visible or ICA EDV was 40–100 cm/sec; (iv) 70% stenosis to near occlusion: when the ICA PSV was > 230 cm/sec and visible plaques and lumen narrowing were seen or ICA EDV was > 100 cm/sec; (v) near occlusion: when there was a markedly narrowed lumen on color Doppler US; and (vi) total occlusion: when there was no detectable lumen on grayscale US and no flow on spectral, power, and color Doppler US. 6
For asymptomatic patients, at least one side exhibited carotid artery stenosis with more than 70% stenosis; for symptomatic patients, at least one side exhibited more than 50% carotid artery stenosis. 6
Exclusion criteria
The exclusion criteria were as follows: (i) US imaging showed predominantly echogenic and extensive calcifications with acoustic shadowing (carotid plaque surface calcifications, posterior echo attenuation, or poor imaging effects) (n = 14); (ii) one-sided surgery was performed for bilateral plaques (n = 3); (iii) carotid stent surgery (n = 10) and conservative treatment (n = 9) were performed; and (iv) refusal to participate in the study (n = 1).
Finally, 28 carotid atherosclerotic plaque lesions were selected as the objects of study; 22 of the patients were male, and six were female. The patients were 50–84 years of age, with an average age of 63.4 ± 6.3 years. The clinical characteristics of the subjects are shown in Table 1.
Clinical profile of the entire study population and the surgical subgroup.
Ultrasound examination
All US examinations were performed with a high-frequency transducer (9–4 MHz Aplio 500; Toshiba Medical Systems Corp., Tochigi, Japan). The study used SMI software, and the subjects were in a supine position with their heads tilted slightly backward, fully exposing the neck.
Standard US imaging: continuous scanning was conducted to examine the bilateral common carotid artery, common carotid artery bifurcation, and internal and external carotid arteries. We also searched for atherosclerotic plaques in the arteries (intima–media thickness (IMT) > 1.5 mm). If a plaque was present, continuous scanning was performed on the short axis of the carotid artery, and the maximum thickness of the plaque was measured. If there were multiple plaques in the carotid artery, the largest plaque at the site of carotid artery stenosis was selected for the study. The location, size, and echo of the plaque were recorded, and the rate of lumen stenosis and peak velocity of the blood flow were recorded by standard US.
SMI examination: An Aplio 500 ultrasonic diagnostic system that can perform a new flow imaging technique, SMI, was used. The region of interest for SMI was examined using a double display showing both grayscale US and SMI findings. The target area was placed in the center of the screen, the sampling range was adjusted, the SMI color velocity scale was adjusted to 1.5–3.0 cm/s, and the vascular information was enhanced by adjusting the time smoothing. The new blood vessels in the plaque were observed in real time in the multisection plane. A section with a large amount of blood flow was selected. The frequency spectrum of neovascularization was measured using spectral Doppler imaging. Dynamic and static images were stored for post-imaging analysis.
Image analysis
Both standard US and SMI data were analyzed offline by two investigators (with approximately 15 and 2 years of experience in carotid US) who were blinded to the pathological findings. The echogenicity of each lesion was objectively classified according to the Gray–Weale (GW) visual classification, as follows 7 : uniformly echolucent (class I); predominantly echolucent (class II); predominantly echogenic (class III); or uniformly echogenic or extensively calcified (class IV). SMI was used to visually grade the microvessel density (MVD), as follows 8 : grade I: no appearance of neovascularization within the plaque; grade II: punctate neovascularization; grade III: one or two sites of linear neovascularization within the plaque (Figure 2); and grade IV: multiple (> 2) sites of linear neovascularization throughout the plaque (Figure 3) (see the supplementary online video).
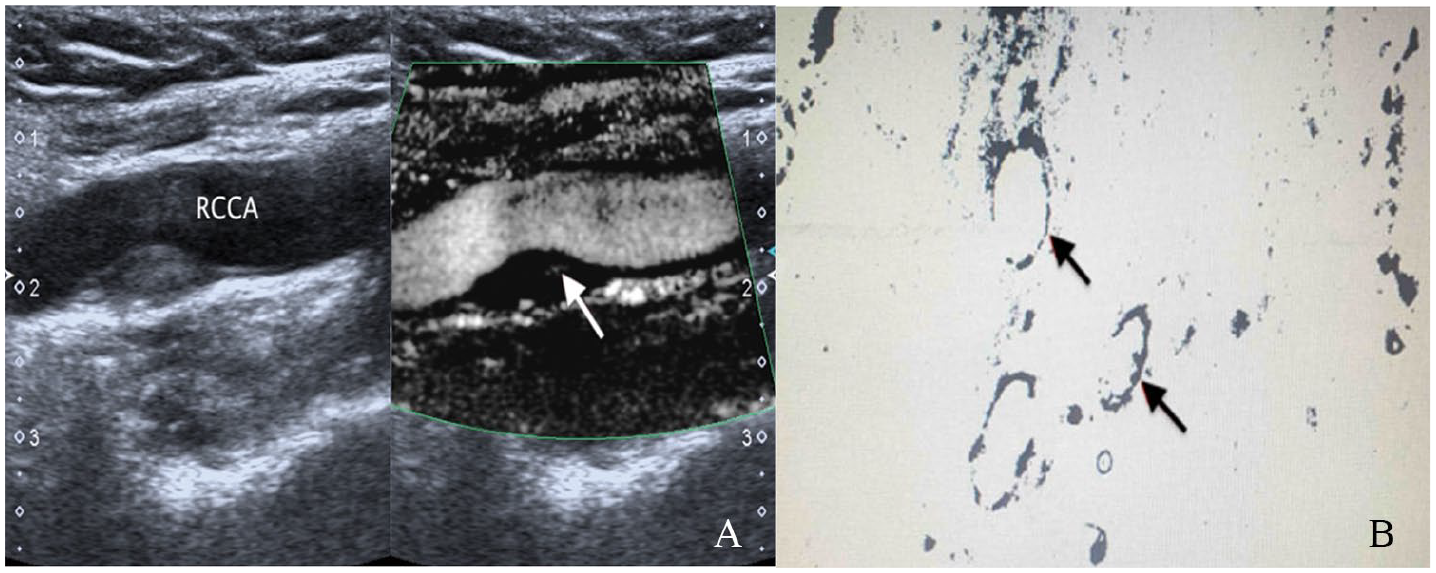
(A) Hypoechoic plaque at the posterior wall of the bifurcation of the right common carotid artery (RCCA). Several areas of neovascularization in the plaque are shown by SMI (arrow). (B) Quantitative analysis of a small amount of neovascularization indicated by histology (arrows).
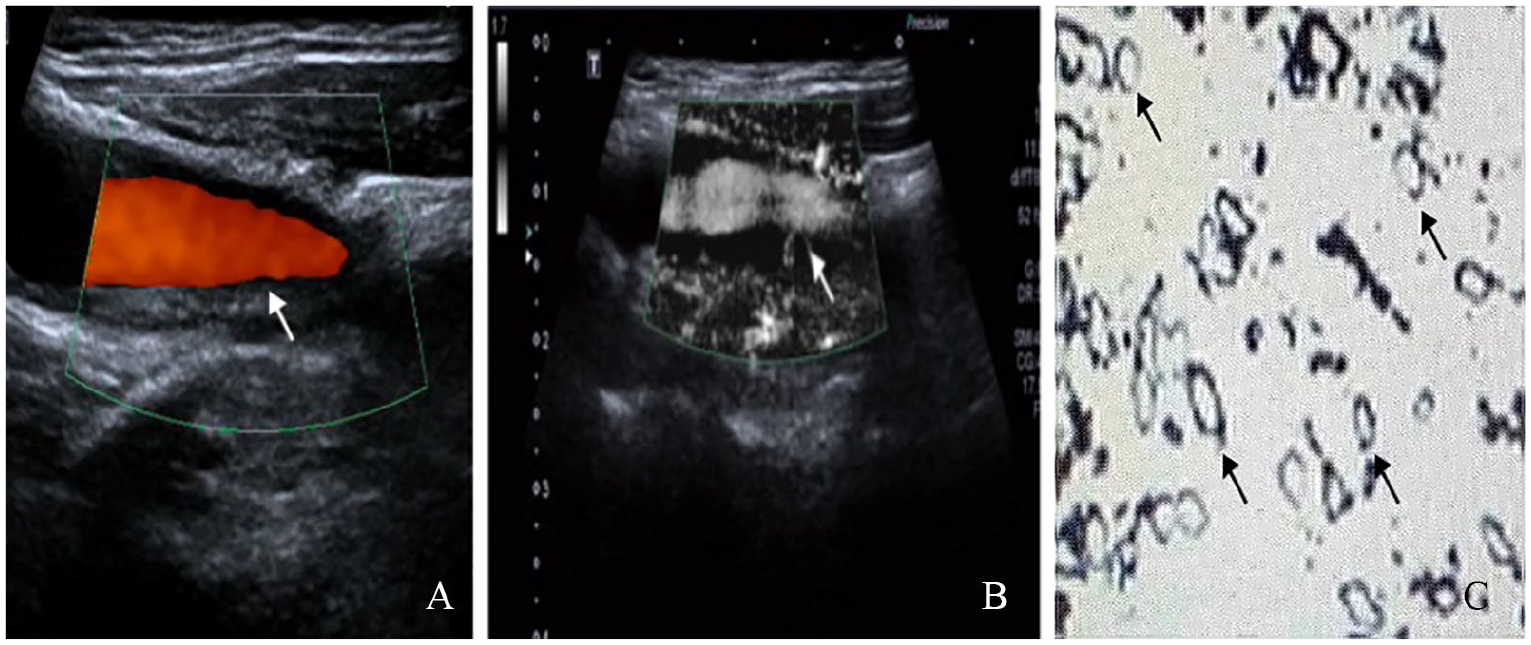
Hypoechoic plaque at the left carotid bifurcation in a 64-year-old man. (A) Hypoechoic plaque at the posterior wall of the bifurcation of the left common carotid artery by color Doppler ultrasound (arrow). (B) Vertical path of neovascularization in the plaque shoulder as shown by SMI (arrow). (C) Quantitative histopathological analysis of many sites of neovascularization (arrows) (see the supplementary online video).
Surgical procedures
CEA was performed using standard techniques. The intimal hyperplasia and plaques of the carotid artery were visualized. The largest patch section was taken to ensure the integrity of the plaque, and the sample was sent to pathology. If there were more than two plaques, the larger plaque was taken. The ICA and the common carotid artery were then anastomosed. Alternatively, an artificial blood vessel patch was used after hemostasis was achieved. A drainage tube was placed in the wound, the incision was sutured layer by layer, and the operation was completed.
Pathological staining and analysis
After CEA, the plaque was removed by flushing the area with saline. The samples were embedded in tissue-freezing medium and cut into cryosections 3–5 μm thick. Hematoxylin and eosin (H&E) staining was performed, and the microvessels were stained with CD31 monoclonal antibody (1:4000 dilution) for immunohistochemistry. Positive staining was considered neovascularization. The entire section was first observed at low magnification (100×), and three regions with the highest vascular densities were selected (i.e. the hot spots). Then, the vessels stained brown with CD31 antibody were counted at high magnification (200×). Quantitative analysis of MVD was performed by Image-Pro Plus (IPP) image analysis software (Media Cybernetics, Inc., Rockville, MD, USA). The mean MVD of neovascularization per square millimeter was used for correlation with the imaging data.
Statistical methods
The MVD data were not normally distributed. A logarithmic transformation was carried out to conform the data to a normal distribution. Spearman’s rank coefficient was used to test the correlations among study variables. The relationship between the SMI grade and clinical symptoms was evaluated by chi-squared analysis. The relationships between plaque MVD and clinical symptoms were evaluated using t-tests. The consistency of the intraplaque neovascularization grades determined by two analysts was tested using the kappa coefficient (good consistency, kappa ⩾ 0.7; general consistency, 0.4 ⩽ kappa < 0.7; poor consistency, kappa < 0.4). Statistical analyses were conducted using SPSS software version 19.0 (IBM Corp., Armonk, NY, USA). A p ⩽ 0.05 corresponded to a statistically significant difference. Quantitative analysis of MVD was performed by IPP image analysis software (Media Cybernetics) and by SMI software (Aplio 500; Toshiba Medical Systems Corp.).
Results
Correlation between histopathological MVD and plaque neovascularization as detected by SMI
Neovascularization was detected in 28 plaques by SMI, as follows: grade I: 5 (18%); II: 5 (18%); III: 8 (28%); and IV: 10 (36%). The SMI blood flow grade was positively correlated with the histopathological MVD (r = 0.788, p < 0.001) (Figure 4).
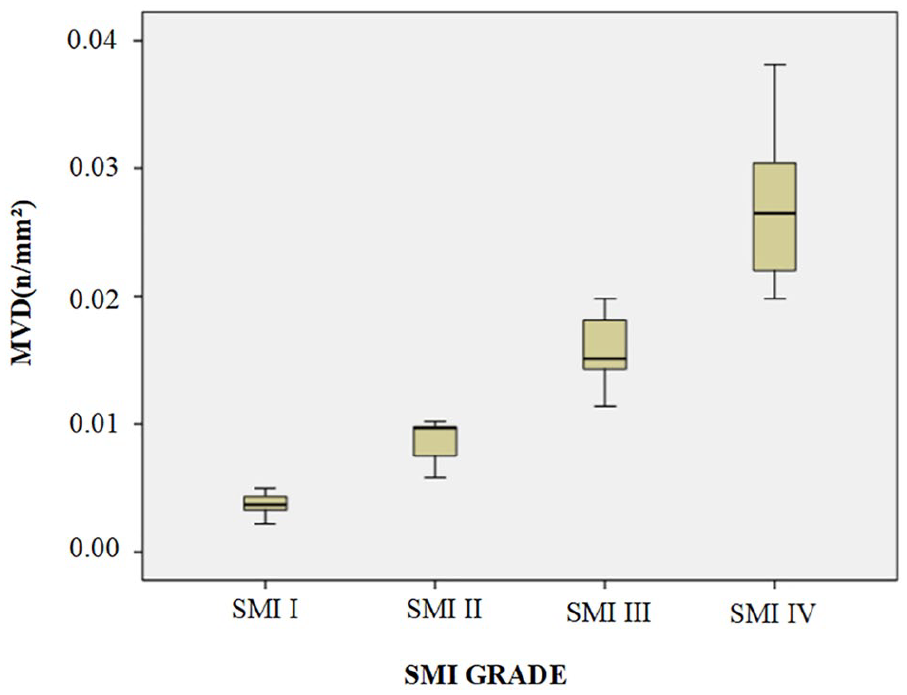
Relationship between the SMI grade of neovascularization in carotid atherosclerotic plaques and histopathological MVD. The SMI grade was positively correlated with the histopathological MVD.
Relationship between SMI grade and clinical symptoms
The patients were grouped according to their clinical symptoms. Twenty-three patients had a history of a clinically diagnosed transient ischemic attack or stroke. Five patients had no history of treatment for a cerebrovascular event and had no clinical symptoms. No significant difference in sex or age was observed between the two groups (p > 0.05). No significant difference was observed in the degree of stenosis between the symptomatic and asymptomatic groups (p > 0.05).
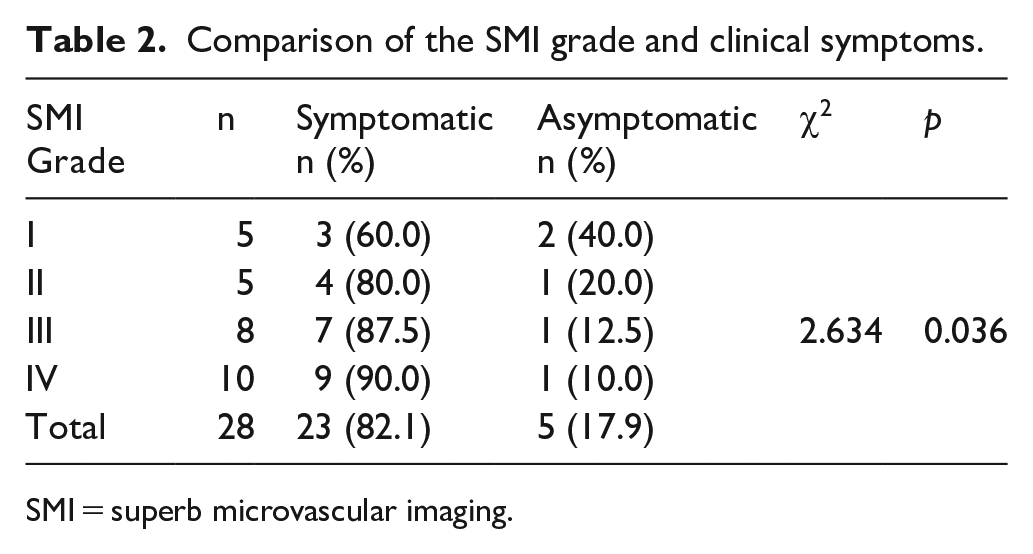
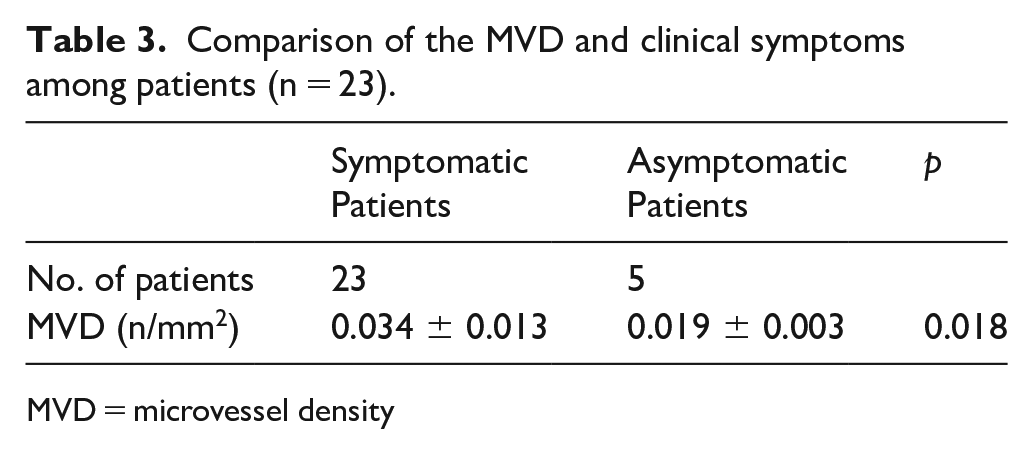
A significant difference in the SMI blood flow grade was observed between the symptomatic and asymptomatic groups (χ2 = 2.634, p = 0.036) (Table 2). The plaque MVD in the symptomatic group was significantly higher than that in the asymptomatic group (t = 2.530, p = 0.018) (Table 3).
Comparison of the SMI grade and clinical symptoms.
SMI = superb microvascular imaging.
Comparison of the MVD and clinical symptoms among patients (n = 23).
MVD = microvessel density
Relationship between SMI grade and plaque thickness
The plaque volume range was 0.086–1.776 cm3. The mean plaque thickness in the 28 patients was 0.415 ± 0.160 cm (min: 0.190 cm, max: 0.800 cm). The SMI-based grade was positively correlated with plaque thickness (r = 0.686, p = 0.002).
Reproducibility of SMI findings
To establish the reproducibility of our qualitative assessment, the interobserver agreement was determined by applying Cohen’s kappa statistic to the intraplaque neovascularization grade determined by two different readers. Substantial interobserver agreement (kappa coefficient: 0.72) in the grade of intraplaque neovascularization was found.
Discussion
To date, the most widely accepted predictor of stroke risk is the degree of stenosis in carotid atherosclerosis. However, in asymptomatic patients, the degree of stenosis cannot provide a sufficient basis for predicting the risk of stroke, so other additional imaging features for assessing atherosclerotic plaque morphology and composition are becoming recognized as significant factors.9–11 Therefore, only a comprehensive objective assessment of the arterial lumen, wall structure, and plaque characteristics can more accurately stratify patients by stroke risk and guide individualized treatment. Neovascularization plays an important role in the development of atherosclerotic plaques and in the process of plaque rupture and hemorrhage. 12 Several pathology studies have shown that more extensive plaque neovascularization is associated with the features of plaque vulnerability and with clinically symptomatic disease.13–15
Most previous studies16–19 have used CEUS to observe intraplaque neovascularization. CEUS increases the detectability of blood flow and is thus able to overcome some limitations; however, it does have some drawbacks. CEUS is not readily available everywhere, and it is subject to certain restrictions regarding contrast agent use. CEUS cannot measure the direction or velocity of blood flow. DCE-MRI represents an indirect measure of angiogenesis since it mainly reflects leakage of the vascular bed by measuring the transfer of contrast agent into the interstitial space. In terms of drawbacks, DCE-MRI is not only invasive but also places an additional cost burden on the patient. 20 In this study, the new technology of SMI was used to detect neovascularization in carotid plaques. By analyzing clutter motion and using a new adaptive algorithm to identify and remove tissue motion, SMI is designed to improve the visualization of microvascular blood flow. SMI depicts this information as a color overlay or as a monochrome map of flow, 21 and SMI findings were compared with pathological MVD findings for the first time in this study.
The purpose of this study was to evaluate the availability and accuracy of the neovascularization information obtained using the SMI technique in carotid atherosclerotic plaques. The results show that the SMI classification was positively correlated with histopathological MVD. Therefore, the SMI technique is a nontraumatic imaging method that can be used to safely and accurately identify neovascularization in vulnerable plaques and facilitate timely prevention and treatment.
In our study, the SMI technique and the neovascularization density were used to analyze plaques in symptomatic and asymptomatic patients. The results show that the neovascularization density in the symptomatic group was significantly different from that in the asymptomatic group. This result occurred because neovascularization can easily lead to plaque blood leakage and inflammatory cell aggregation, as well as plaque hemorrhage and fibrous cap thinning, eventually leading to plaque rupture and causing them to slough off. Mofidi et al. 22 conducted a similar study; in 73 patients with clinically symptomatic carotid dissection, the plaque MVD was significantly elevated, and these plaques sloughed off to form emboli, resulting in ischemic stroke due to blood vessel blockage. The abundance of blood flow signals in the carotid plaques on SMI was significantly higher in patients with symptoms than in those without symptoms. The higher resolution and lower overflow of SMI contributed to its superior detection of flow details. The capability and accuracy of SMI for detecting carotid plaque neovascularization were demonstrated.
In this study, the plaque neovascularization classification and the neovascularization density were positively correlated with plaque thickness. In addition to the hypoxia and nutrient depletion caused by plaque growth, plaque neovascularization can further facilitate plaque growth.23,24 Intraplaque neovascularization occurs prior to carotid plaque formation (i.e. during fatty streak formation (intima–media thickening)), which plays a key role in plaque formation, development, and rupture.25–27 These features eventually promote plaque growth and instability. Plaque enlargement can lead to luminal stenosis, and an unstable plaque combined with luminal stenosis is more likely to cause cerebrovascular events. Therefore, the detection of unstable plaques may provide the basis for clinical intervention.
Study limitations
The limitations of this study are as follows: (1) the number of samples in the groups is relatively small, so it is necessary to expand the sample size in future studies; and (2) SMI technology uses continuous scanning to find the sections with the most neovascularization. The pathological sections with the highest MVD were also identified. The biases of the statistical results need to be minimized but cannot be completely avoided.
Conclusion
In conclusion, this initial experience indicates that the SMI technique can be used to evaluate intracarotid atherosclerotic plaque neovascularization.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: we confirm that there are no relationships with Toshiba Medical Systems.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by The National Natural Science Foundation of China (81601517) and The Beijing Natural Science Foundation (7202156).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
