Abstract
Heparin-induced thrombocytopenia (HIT) is a life and limb-threatening complication of heparin exposure. Here, we review the pathogenesis, incidence, diagnosis, and management of HIT. The first step in thwarting devastating complications from this entity is to maintain a high index of clinical suspicion, followed by an accurate clinical scoring assessment using the 4Ts. Next, appropriate stepwise laboratory testing must be undertaken in order to rule out HIT or establish the diagnosis. In the interim, all heparin must be stopped immediately, and the patient administered alternative anticoagulation. Here we review alternative anticoagulation choice, therapy alternatives in the difficult-to-manage patient with HIT, and the problem of overdiagnosis.
The University of Virginia School of Medicine (UVASOM) is accredited by the ACCME to provide continuing medical education for physicians. UVASOM designates this journal-based CME activity for a maximum of one AMA PRA Category 1 Credit™. Participants should claim only the credit commensurate with the extent of their participation in the activity. UVASOM, as an accredited provider, awards 1 hour of participation (consistent with the one AMA PRA Category 1 Credit™) to participants who successfully complete this educational activity. Successful completion of this CME activity, which includes participation in the evaluation component, enables the participant to earn up to one MOC II point in the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) program. It is the CME provider’s responsibility to submit participant completion information to ACCME for the purpose of granting ABIM MOC II credit. UVASOM maintains a record of participation for 6 years.
The faculty, staff, and planning committee of the University of Virginia Office of Continuing Medical Education have no financial affiliations to disclose. The CME planning committee disclosed the following: Heather Gornik receives intellectual property rights and equity from FlexLife Health; Aditya Sharma is supported by National Institutes of Health, AstraZeneca, and Biomet Biologics; and Valerie Clark has nothing to disclose. The authors disclosed no financial relationship or interest with any proprietary entity producing healthcare goods or services.
Introduction
Heparin is a century-old, ubiquitous medication that remains exceptionally relevant to modern medicine. Unfractionated heparin (UFH) and low molecular weight heparin (LMWH) continue to be widely used in a variety of inpatient and outpatient settings. Heparin-induced thrombocytopenia (HIT) remains one of the most important complications, and clinicians must remain vigilant to the development of this adverse event. HIT is caused by antibodies directed toward heparin–platelet protein platelet factor 4 (PF4) complexes. While these antibodies occur with some frequency in many patients, a minority of patients develop clinically relevant complications, including HIT with thrombosis, which is referred to as HITT. Regardless of whether a patient has an identified thrombus in the setting of HIT, HIT alone is a hypercoagulable state and alternative anticoagulation must be started promptly.
Incidence of heparin-induced thrombocytopenia (HIT) in general and special populations
HIT has been estimated to occur in 0.1–5% of patients receiving therapeutic doses of heparin.1–3 Its incidence has been based on the results of modest sample sizes until recently when Dhakal et al. reported the results of a large study through the Nationwide Inpatient Sample (NIS), with the caveat that this population-based study relied on coded cases of HIT in an all-payer inpatient United States healthcare database, and therefore may not fully represent the true incidence of HIT. 4 HIT was diagnosed in 0.065% of inpatients, or 1/1500 hospital admissions. Patients over age 50 were more likely to be diagnosed with HIT, with each advancing year of age conferring significantly higher adjusted odds of developing the disease. In the NIS study, women were less likely to develop HIT compared to men overall; however, this is in contrast to previous studies demonstrating a higher incidence of HIT in females. 5 The NIS study did report that females had significantly higher odds in subsets of cardiopulmonary bypass (CBP) and hemodialysis subgroups 4 as well as in the vascular surgery setting. 1
It is well known that patients undergoing cardiac surgery have high rates of HIT compared to the general medical hospital population. In the NIS study, 0.63% of CPB patients developed HIT and represented the highest risk group; however, previous studies have shown higher rates with estimates ranging from 1% to 3%.6–8 Given that the development of HIT is associated with a 50% increase in early mortality, 2 this finding is of particular importance; however, prevention of HIT proves difficult.
In delineating the risk of HIT in patients undergoing vascular surgeries, the NIS data demonstrated that 0.31% of patients undergoing vascular procedures developed HIT. The highest incidence of HIT in the vascular surgery population occurred in thoracic subclavian procedures (0.74%) followed by vein reconstructions (0.67%). The lowest incidence of HIT in this population was seen in patients undergoing carotid endarterectomy. 1
Heparin formulation also affects rates of HIT and it is well known that HIT is less likely to occur in the setting of LMWH compared to UFH when utilized for thromboprophylaxis. 9 The ultralarge complexes of PF4 and heparin required for HIT form much less efficiently with the smaller LMWH species.10,11 Heparin-induced antibodies are more frequent among patients exposed to UFH compared to LMWH, as measured by both immunoassays 12 and functional assays. 13 A Cochrane review demonstrated a lower prevalence of HIT when LMWH was used over UFH for thromboprophylaxis. The risk with UFH was found to be 22 per 1000, whereas it was only 5 per 1000 in patients receiving LMWH. Additionally, HIT complicated by venous thromboembolism (VTE) occurred significantly less (4 per 1000 vs 17 per 1000) in patients receiving LMWH compared to UFH. 14 Some institutions have made quality efforts to reduce the burden of HIT through such schemes as the ‘Avoid-Heparin Initiative’, where UFH was replaced with LMWH and resulted in a 79% reduction in HIT. 3
Pathogenesis of HIT and the HIT antibody
The pathophysiology of HIT is complex. It is initiated by immunoglobulin G (IgG) against the PF4–heparin complex, which in turn activates platelets via FcγRIIa; it is propagated by activated platelets, endothelial cells, monocytes, and coagulation proteins, as described by McKenzie and Sachais in their recent comprehensive review of HIT pathogenesis. 15
PF4 is a cationic protein stored within platelet alpha granules that is released upon platelet activation. PF4 interacts with negatively charged glycosaminoglycans (GAGs), including heparin, leading to the formation of an immunogenic complex that is the target of HIT antibodies.16,17 These pathogenic PF4-heparin ‘ultralarge complexes’ form both in the circulation and on the platelet surface. When HIT antibodies, now attached to the PF4–heparin complex, bind to platelet FcγRIIa, platelet activation occurs via intracellular signaling involving spleen tyrosine kinase and the release of procoagulant microparticles (Figure 1).16,18–20 Thrombin is generated and platelet consumption occurs likely both through clearance and platelet-fibrin thrombi. Although thrombocytopenia is the hallmark of HIT, its underlying mechanisms are not completely understood.21,22 A positive feedback loop propagates the prothrombotic loop. The roles of additional cell types in the pathogenesis of HIT are currently being investigated, including B and T-cells. 23 Recent studies have suggested that neutrophil extracellular traps (NETs) also contribute to thrombosis in patients with HIT.24,25
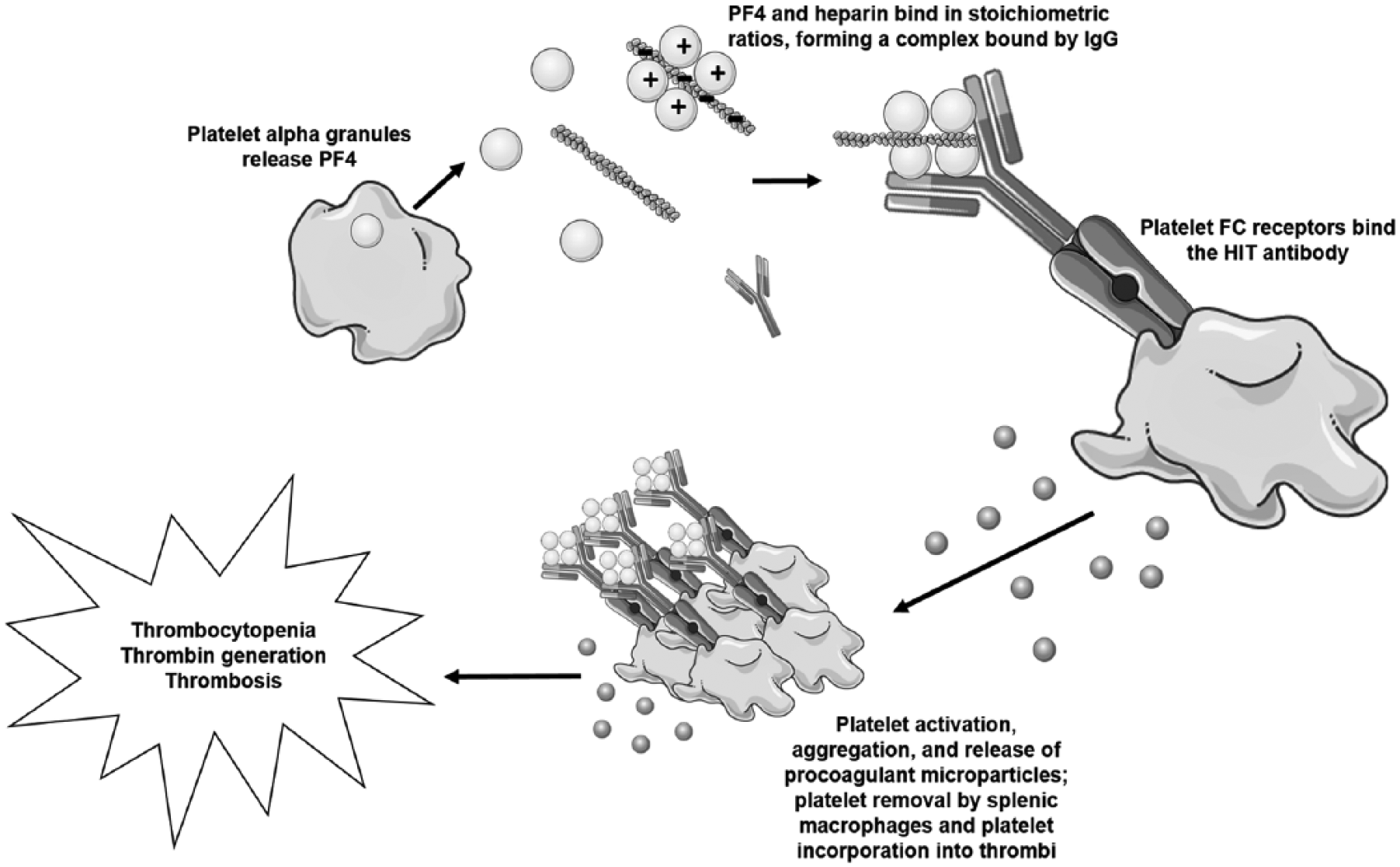
Pathogenesis of HIT.
It is worth noting that while antibodies to the PF4–heparin complex are rare in healthy populations, occurring in the range of 0.3–0.5%,26,27 antibody formation occurs in medical/surgical patient populations with some frequency. One study noted an incidence of antibody formation of 17% in patients receiving UFH and 8% in patients receiving LMWH. 28 This may be due to immune system priming: antibodies are developed when PF4 binds to bacteria, consistent with the known association between gingival disease and the presence of anti-PF4/heparin antibodies.27,29 Additional investigations have suggested a role for the postoperative inflammatory milieu and mechanical thromboprophylaxis in the development of anti-PF4/heparin antibody. 30 The incidence of anti-PF4/heparin antibodies is even higher in postoperative CPB patients, occurring in approximately 22–50% of patients.31–37 The reason for this is not fully understood; one study evaluated the role of atherosclerosis in this population given that PF4 is deposited into plaques; however, atherosclerosis was not found to be a major risk factor for anti-PF4/heparin seroconversion. 36 As Rauova et al. note in their recent review of HIT pathogenesis, ‘pathologic’ molar ratios of PF4 and UFH are required for the development of the HIT antigen and formation of ultralarge complexes, and may explain why HIT occurs more frequently in post-CPB patients, and why not all heparin exposures lead to HIT. 11
Diagnosis of HIT
The diagnosis of HIT may be challenging and requires the combination of both clinical features and laboratory testing.
Clinical presentation and complications of HIT
The hallmark of HIT is thrombocytopenia, which is the most common manifestation of HIT and is temporally related to heparin exposure. Exposure may be as extensive as anticoagulation during CBP or as trivial as a heparin flush. The most common presentation of thrombocytopenia in HIT is a drop in platelet count at least 4 days following heparin exposure and usually occurs 5–14 days after exposure.38,39 A minority of patients have rapid onset thrombocytopenia within hours of receiving heparin; however, these patients are thought to have pre-existing circulating HIT antibodies due to previous heparin exposure within the previous 100 days.38–40 Postsurgical patients have been noted to sometimes demonstrate a bimodal presentation of thrombocytopenia in which there is an immediate platelet drop following surgery that rebounds, but then drops again.13,38,41,42 Of note, the majority of patients undergoing CPB will demonstrate a postoperative reduction in platelet count secondary to hemodilution and platelet consumption, requiring astute clinical and diagnostic acumen to distinguish true HIT in this population.7,43 This is discussed further in the pre-test probability: the 4Ts and other clinical scoring systems.
While reduction in platelet count may occur as ‘true thrombocytopenia’ (i.e. < 150 × 109/L), the decrease may also be relative and manifest as a 30–50% reduction in baseline platelet count. 39 Thus, a baseline platelet count followed by careful notation of a patient’s platelet count trend is paramount in early recognition of HIT rather than relying on an ‘abnormal’ lab value.
Paradoxically, HIT is a prothrombotic state, with thromboembolic complications occurring in approximately one-third to a half of all patients with HIT.4,13,44,45 Risk of thromboembolic complications are related to the magnitude of platelet decrease, although some complications may precede the drop in platelet count, 44 and thrombosis may be the presenting symptom. VTE is the most common thrombotic complication in patients with HIT, but arterial thrombosis and thrombotic stroke also occur. Of note, it has been demonstrated that arterial thromboses occur with equal frequency as VTE in the cardiac and vascular surgery populations. 44 Patients with HITT are more likely to have serious complications, including additional thromboses, major bleeding, limb amputation/gangrene, and death compared to patients without HIT. 46 HIT-associated disseminated intravascular coagulation (DIC) may occur, manifesting as microthrombosis, decreased fibrinogen, and increased fibrin degradation products.47–49 Case reports of HIT-related DIC have been described as being treated with both typical agents like argatroban, 47 as well as hirudin and intravenous immunoglobulin (IVIG). 50
Despite thrombocytopenia, bleeding is not classically associated with HIT. However, a recent study of 310 patients with suspected HIT found that major bleeding occurred in 40.6% of patients and was no less common in HIT+patients (40.9%). 51 This surprising result highlights the need for further investigation. The most common type of bleeding in HIT is gastrointestinal, although intracranial hemorrhage, retroperitoneal bleeding, and other types of bleeding may occur. 4 Less frequently, patients with HIT may develop heparin-induced skin necrosis, limb gangrene, or an anaphylaxis-type reaction following a bolus of heparin. Rarer and perhaps under-recognized manifestations of HIT include adrenal failure/adrenal hemorrhage secondary to adrenal thrombosis52–55 and flap failure.56–61 A summary of HIT manifestations is listed in Table 1.
Clinical manifestations of HIT.
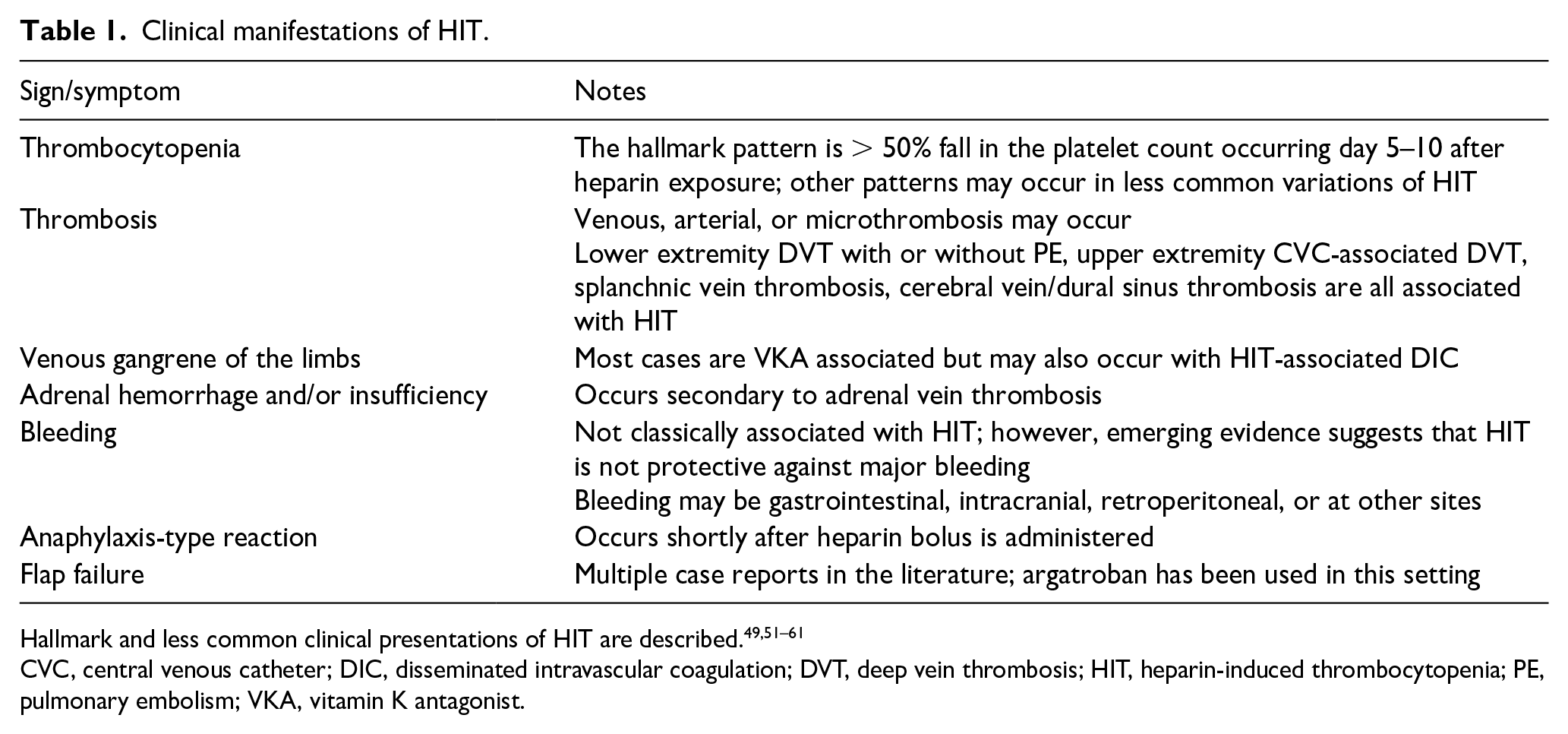
CVC, central venous catheter; DIC, disseminated intravascular coagulation; DVT, deep vein thrombosis; HIT, heparin-induced thrombocytopenia; PE, pulmonary embolism; VKA, vitamin K antagonist.
Variants of HIT
Two described variants of HIT are delayed onset HIT and spontaneous HIT; these are sometimes referred to as autoimmune heparin-induced thrombocytopenia syndromes. The true incidence of these entities is not clearly established and they have mainly been described in case reports. 39 Delayed onset HIT may present days to weeks after cessation of heparin, and is driven by antibodies that cross-react with PF4–GAG complexes.16,62 While delayed onset HIT accounts for less than 5% of HIT cases, 62 it is an important clinical entity and should be considered in any patient presenting with thrombocytopenia or thrombosis who recently received heparin. Spontaneous HIT is an incompletely understood condition characterized by a HIT-like picture in the absence of heparin exposure, likely due to anti-P4–heparin antibodies triggered by GAGs or other polyanions. 63 In the case of spontaneous or delayed HIT, the patient will have a positive functional assay, even in the absence of current heparin exposure.
Pretest probability: The 4Ts and other clinical scoring systems
The 4Ts
First introduced in 2006, the ‘4Ts’ has become the most widely used and extensively studied pretest clinical scoring system to identify HIT (Table 2).
64
The 4Ts assessment accounts and gives points for
The 4Ts scoring system.
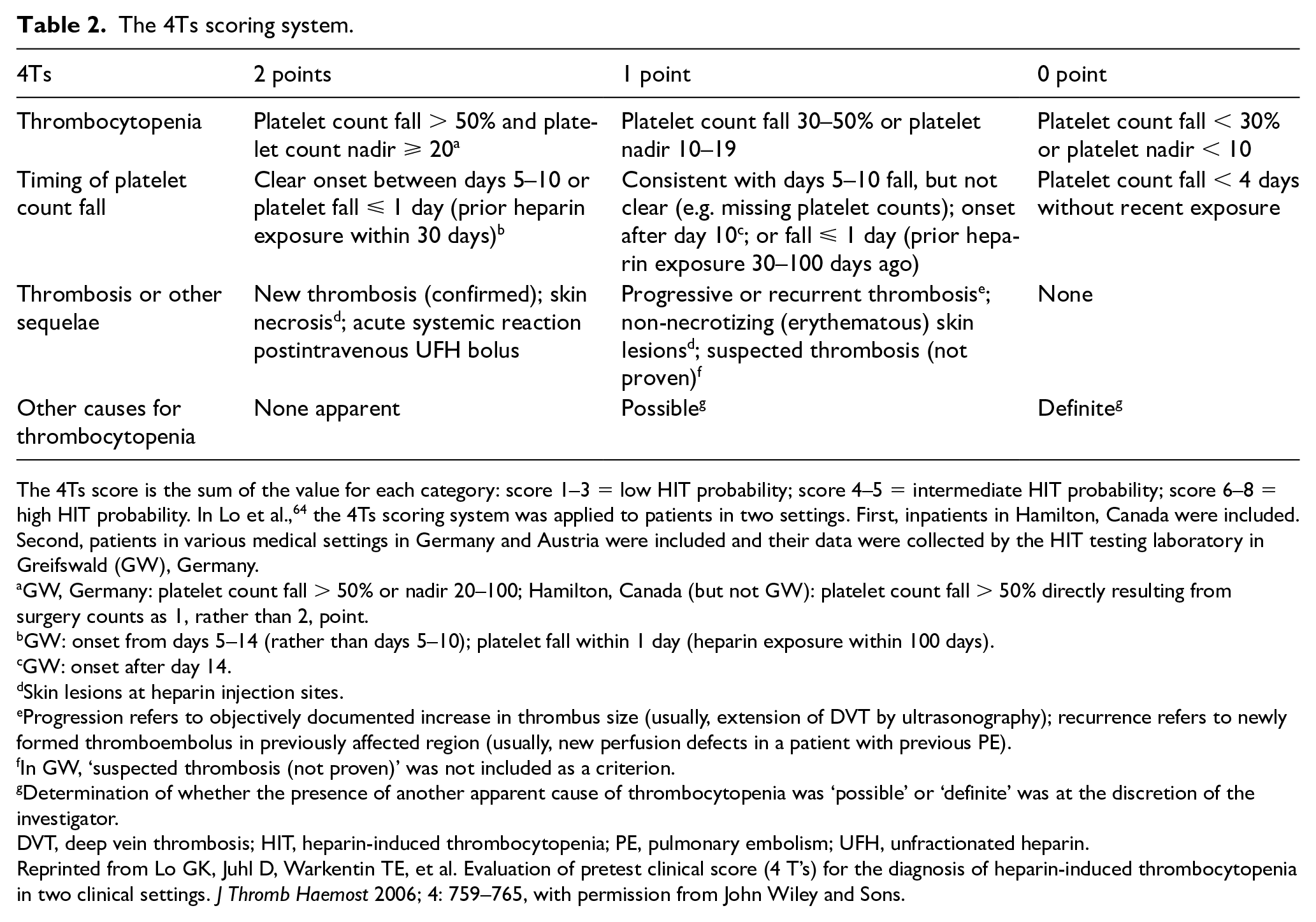
The 4Ts score is the sum of the value for each category: score 1–3 = low HIT probability; score 4–5 = intermediate HIT probability; score 6–8 = high HIT probability. In Lo et al., 64 the 4Ts scoring system was applied to patients in two settings. First, inpatients in Hamilton, Canada were included. Second, patients in various medical settings in Germany and Austria were included and their data were collected by the HIT testing laboratory in Greifswald (GW), Germany.
GW, Germany: platelet count fall > 50% or nadir 20–100; Hamilton, Canada (but not GW): platelet count fall > 50% directly resulting from surgery counts as 1, rather than 2, point.
GW: onset from days 5–14 (rather than days 5–10); platelet fall within 1 day (heparin exposure within 100 days).
GW: onset after day 14.
Skin lesions at heparin injection sites.
Progression refers to objectively documented increase in thrombus size (usually, extension of DVT by ultrasonography); recurrence refers to newly formed thromboembolus in previously affected region (usually, new perfusion defects in a patient with previous PE).
In GW, ‘suspected thrombosis (not proven)’ was not included as a criterion.
Determination of whether the presence of another apparent cause of thrombocytopenia was ‘possible’ or ‘definite’ was at the discretion of the investigator.
DVT, deep vein thrombosis; HIT, heparin-induced thrombocytopenia; PE, pulmonary embolism; UFH, unfractionated heparin.
Reprinted from Lo GK, Juhl D, Warkentin TE, et al. Evaluation of pretest clinical score (4 T’s) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost 2006; 4: 759–765, with permission from John Wiley and Sons.
The HEP score
More recently, the HIT expert probability (HEP) score (Table 3) was developed. Created by 26 HIT experts, the HEP score uses detailed language within the assessment to elicit certain clinical features. 66 The HEP score showed similar diagnostic accuracy compared to the 4Ts in both retrospective and prospective studies.66,67 Further prospective evaluations of the HEP score are needed.
The HIT expert probability (HEP) probability score.

ATE, arterial thromboembolism; DIC, disseminated intravascular coagulation; ECMO, extracorporeal membrane oxygenation; HIT, heparin-induced thrombocytopenia; IABP, intra-aortic balloon pump; VAD, ventricular assist device; VTE, venous thromboembolism.
Reprinted from Cuker A, Arepally G, Crowther MA, et al. The HIT Expert Probability (HEP) Score: a novel pre-test probability model for heparin-induced thrombocytopenia based on broad expert opinion. J Thromb Haemost 2010; 8: 2642–2650, with permission from John Wiley and Sons.
Alternative scoring and representative patterns in postcardiac surgery patients
Given that post-CPB patients commonly have thrombocytopenia in the setting of heparin for many reasons, diagnosing true HIT may be challenging. 7 In 2004, Lillo-Le Louët et al. described a model for estimating the probability of HIT specifically in post-CPB patients. 8 Based on retrospective analysis of 84 patients, the authors found that a biphasic platelet count (described as an initial fall in platelet count immediately after CPB, then a rise of at least 30% within 5 days followed by a second subsequent fall; Figure 2) from CPB to the first day of suspected HIT, an interval of ⩾ 5 days from CPB to the first day of suspected HIT, and a CPB duration of ⩽ 118 minutes were independent risk factors for HIT. Each of these variables is assigned a score of 0, 1, or 2, with a score of ⩾ 2 assigned a high probability of HIT (Table 4). Using this scoring system, the authors were able to determine HIT with a PPV of 62% and a NPV of 97%. A score of 5 had a diagnostic specificity of 95%. This scoring model has not been independently validated. Somewhat similarly, Selleng et al. performed a prospective study of 581 patients undergoing cardiac surgery and found that early onset, persistent thrombocytopenia following CPB was rarely due to HIT, in contrast to a thrombocytopenia pattern marked by a platelet count fall by > 50% between days 5 and 10 following surgery. 68
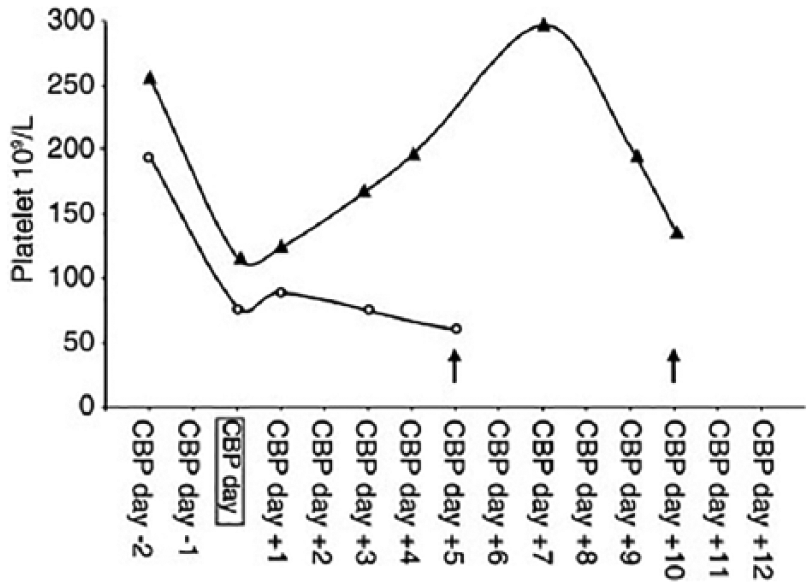
Patterns of thrombocytopenia following cardiac bypass surgery.
Probability score for post-CPB HIT.
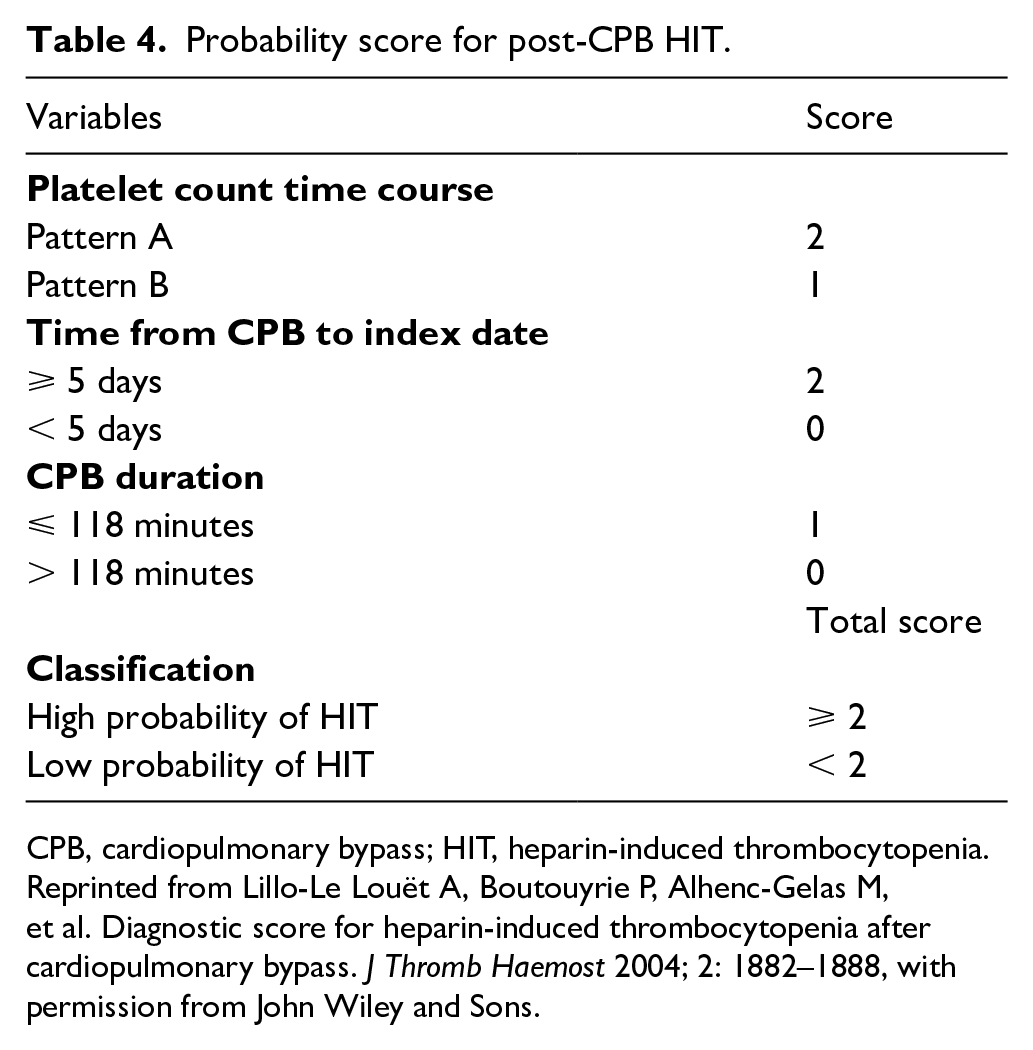
CPB, cardiopulmonary bypass; HIT, heparin-induced thrombocytopenia.
Reprinted from Lillo-Le Louët A, Boutouyrie P, Alhenc-Gelas M, et al. Diagnostic score for heparin-induced thrombocytopenia after cardiopulmonary bypass. J Thromb Haemost 2004; 2: 1882–1888, with permission from John Wiley and Sons.
Laboratory tests for HIT
The pretest clinical assessment score by the 4Ts or HEP should guide the clinician in pursuing the next step: laboratory testing. There are two general categories of laboratory assays to diagnose HIT: immunoassays and functional assays. The immunoassay is the first step and acts as a screening test to rule HIT out. Next, a functional assay establishes the diagnosis.
Immunoassays measure antibodies against PF4–heparin complexes and are the first line laboratory test for HIT. The PF4/heparin enzyme-linked immunosorbent assay (ELISA) assay is a commonly used immunoassay; others include particle gel immunoassay, lateral flow assays, automated latex-based assays, and automated chemiluminescence-based assays. Immunoassays are accessible, highly sensitive (97%), 69 and offer a rapid result, making them a reasonable laboratory screening tool for patients in whom HIT is suspected. Unfortunately, immunoassays are plagued by variably poor specificity in the range of 30–80% depending on the assay.69,70 Because of its high NPV, a negative immunoassay rules out HIT. A positive result, on the other hand, does not confirm the diagnosis, given the low specificity. If the immunoassay is positive, further testing to confirm the diagnosis, usually with a functional assay, is warranted.
Functional assays measure platelet activation due to PF4/heparin antibody binding and crosslinking platelet FcγRIIa receptors. The serotonin release assay (SRA) is the most common functional assay in the diagnosis of HIT, acts as a confirmatory test, and is considered the gold standard. However, the SRA is technically challenging and time-consuming to perform 71 and is a ‘send out’ test for most institutions. Depending on the laboratory, it is resulted as ‘positive’ or ‘negative’, or expressed as percent release with correlating interpretation guidance. It has a sensitivity of > 95% and superior specificity for HIT compared to immunoassays of over 95% in most cases. 71 A positive SRA confirms the diagnosis of HIT.
While the SRA is widely used, some institutions employ the heparin-induced platelet-activation (HIPA) as the functional assay of choice. The HIPA measures platelet aggregation of washed platelets from healthy donors combined with serum from patients suspected to have HIT. These samples are then exposed to buffer or varying concentrations of heparin followed by measurement of platelet aggregation. It is time-consuming, given its requirement of specialized equipment and fresh platelets from at least four donors. 69 Other functional assays include light transmission aggregometry,69,72 the platelet viability assay, 73 the PF4-dependent P-selectin expression assay (PEA), 74 and flow cytometry. 69 Currently, none of these is as widely employed as the SRA.
Putting it together: clinical suspicion in concert with laboratory tests
The diagnosis of HIT requires clinical suspicion with accurate risk assessment via the 4Ts or HEP tool and a positive laboratory test. Figure 3 summarizes the diagnostic and interventional steps for the patient with suspected HIT.
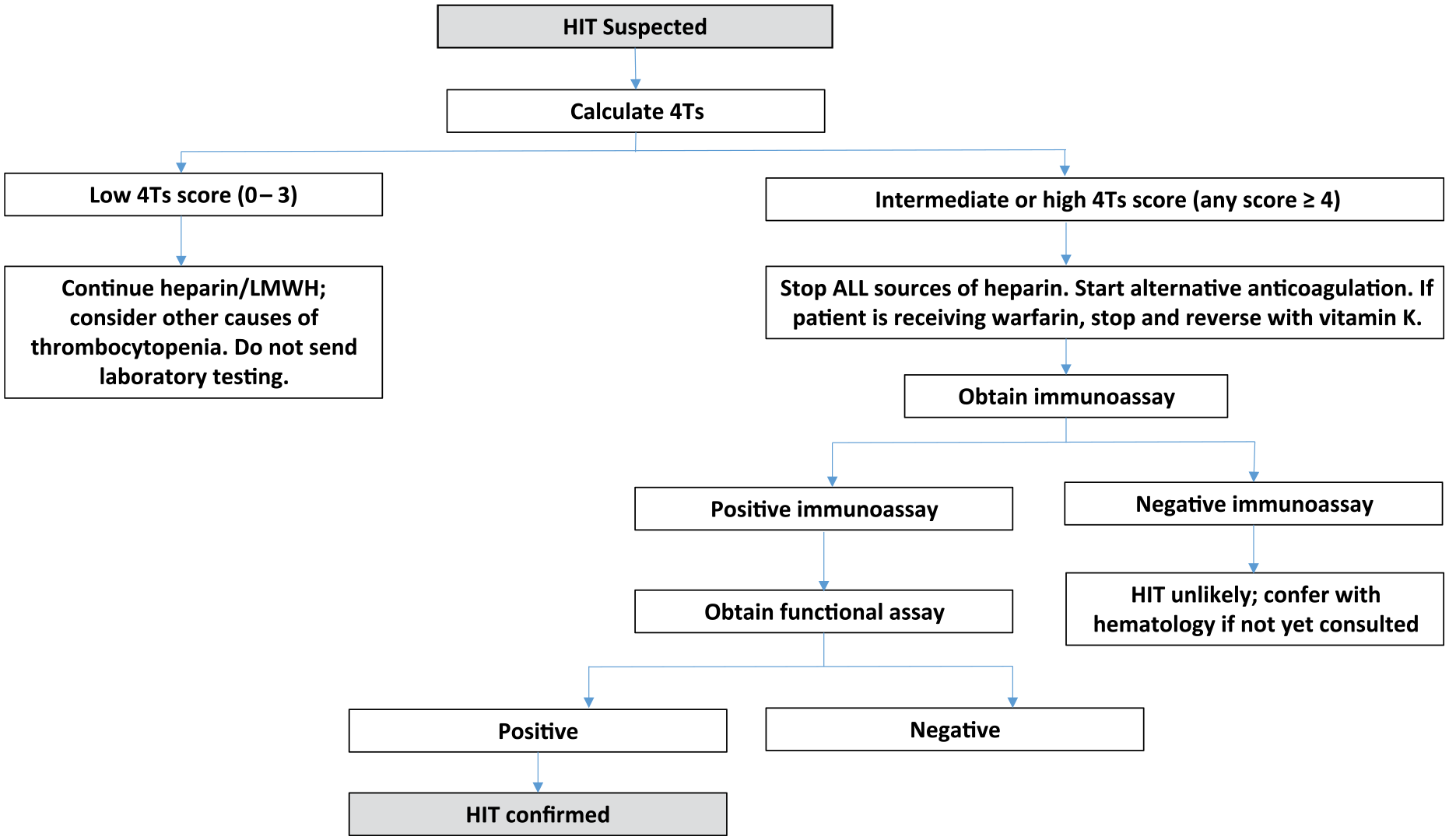
An algorithm for diagnostic and clinical management when HIT is suspected.
When HIT is suspected, the first step is to calculate the 4Ts score; most algorithms are based on this assessment. If the 4Ts score is low probability for HIT, further testing is not recommended. If the 4Ts assessment results in an intermediate or high clinical probability score (⩾ 4), all heparin should be stopped immediately, and a nonheparin anticoagulant should be initiated.
The next step is to send an immunoassay for HIT, such as the PF4/heparin ELISA assay, which is sometimes simply referred to as the PF4. If that is positive, continue cessation of heparin and continue alternative anticoagulation. It has been suggested that for patients with a high 4Ts score and a clear positive PF4 result, additional testing may not be needed;75,76 however, approaches vary. Per clinician discretion, and certainly if the PF4 result is intermediate (optical density: 0.4–2.0), the next step is to obtain a functional assay for HIT, such as the SRA. If the SRA is positive, HIT is confirmed. In the case of a negative SRA, HIT is unlikely; however, false negatives are possible, and the complete clinical and laboratory picture must be considered in the decision to resume heparin.
General principles and acute management of HIT
It is critical to stop all heparin exposure at the first suspicion of HIT rather than waiting for laboratory testing. This includes both UFH and LMWH, heparin flushes, and any heparin-coated lines or infusions. Given the highly prothrombotic state of HIT, it is paramount to immediately initiate alternative anticoagulation at specific treatment dosing for HIT, regardless of the presence of thrombosis. While all HIT guidelines recommend heparin cessation and initiating alternative anticoagulation, the choice of an alternative anticoagulant varies. Typically, heparin is stopped, alternative parenteral anticoagulation is initiated, and a transition to a vitamin K antagonist (VKA) is made later if anticoagulation needs to be continued, such as in the case of thrombosis.
Alternative anticoagulation for the patient with HIT
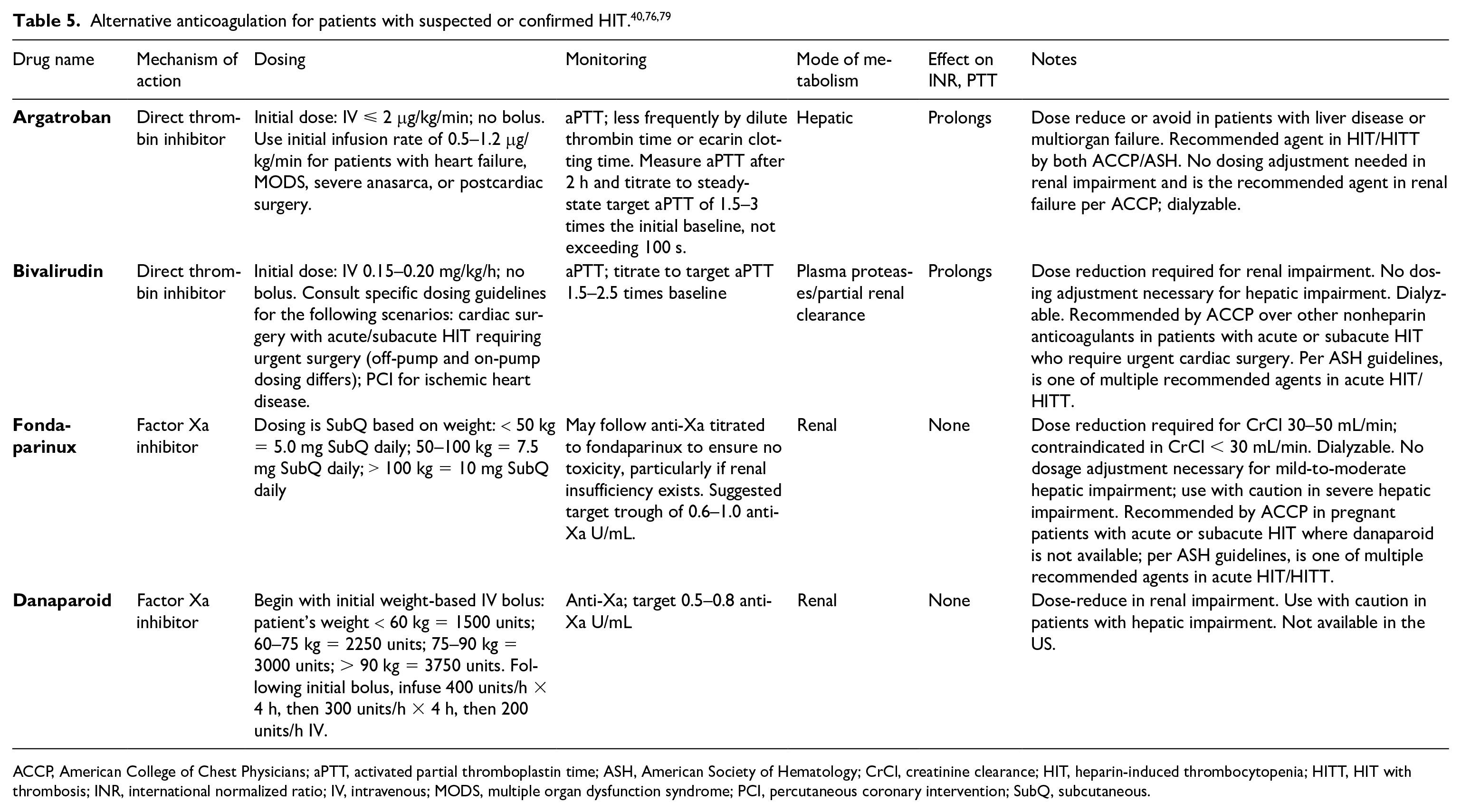
Alternative anticoagulation includes direct thrombin inhibitors (DTIs), direct Factor Xa inhibitors, and fondaparinux. More recently, direct oral anticoagulants have gained consideration for the treatment of HIT. Selection is multifactorial and must consider additional planned procedures, the mode of metabolism for patients with compromised hepatic or renal function, and drug cost and availability, as well as clinician experience. Nonheparin anticoagulation is dosed at therapeutic, not prophylactic, intensity, and there are dosing guidelines specific for HIT. Table 5 summarizes approaches to alternative anticoagulation.
ACCP, American College of Chest Physicians; aPTT, activated partial thromboplastin time; ASH, American Society of Hematology; CrCl, creatinine clearance; HIT, heparin-induced thrombocytopenia; HITT, HIT with thrombosis; INR, international normalized ratio; IV, intravenous; MODS, multiple organ dysfunction syndrome; PCI, percutaneous coronary intervention; SubQ, subcutaneous.
Argatroban
Argatroban is a DTI administered as a continuous intravenous infusion with a short duration of effect, making it appealing for those patients who may be undergoing additional surgical procedures or who are at high hemorrhagic risk.77,78 It is US Food and Durg Administration (FDA) approved for use in HIT, and is recommended by the American College of Chest Physicians (ACCP) and American Society of Hematology (ASH) for use in HIT with and without thrombosis.76,79 Of note, argatroban use in HIT has been criticized as it requires PTT monitoring that is often in a critical care setting, where an underlying coagulopathy may lead to PTT confounding and hence underdosing;80,81 additionally, because argatroban affects the INR, care must be taken when transitioning the patient to warfarin for longer-term anticoagulation. 79 Argatroban is metabolized in the liver, making it an appealing choice for those with renal compromise. Argatroban should be avoided in patients with hepatic impairment.
Bivalirudin
Bivalirudin is another DTI that is administered as a continuous IV infusion, and although it is not FDA approved for the medical treatment of HIT, it is commonly used.76,82,83 Like argatroban, its short duration of effect makes it appealing for those with bleeding risks or those who are undergoing additional procedural interventions and need rapid cessation of drug effect. It is FDA approved for patients with current or previous HIT/HITT during percutaneous coronary intervention (PCI). ACCP guidelines recommend the use of bivalirudin for patients with acute or subacute HIT who require urgent cardiac surgery. 79 Of note, there are specific dosing parameters for off-pump and on-pump time, as well as for PCI. Bivalirudin is cleared by serum proteases as well as renal excretion, and requires dose adjustment based on creatinine clearance.
Fondaparinux
Fondaparinux is a synthetic pentasaccharide and Factor Xa inhibitor that is administered subcutaneously. It has been frequently used off-label for treatment of HIT. However, it has a history of controversy as it has rarely been noted to cause or worsen HIT, and it has been suggested that cross-reactivity with HIT antibodies may rarely occur when treating HIT with fondaparinux. 84 It has been speculated that although fondaparinux is capable of inducing HIT antibodies, clinical manifestations are rare because antibodies typically do not recognize PF4 in the presence of fondaparinux, therefore preventing the required platelet activation.85–88 The HIT-potential of fondaparinux has been disputed, 79 and it is considered a reasonable choice for alternative anticoagulation with growing popularity. 89 It is recommended as a safe, effective choice for a nonheparin anticoagulant by many groups, as well as in a recent systematic review.84,90 However, it is not favored in the most recent ACCP guidelines except in pregnant patients when danaparoid is unavailable, such as in the United States. 79 Fondaparinux is renally cleared and kidney function must be considered.
Danaparoid
Danaparoid is a naturally occurring low molecular weight heparinoid that is a mixture of heparin, dermatan, and chondroitin sulfates. 91 Administration is intravenous and monitoring is via an anti-Xa assay. 81 Danaparoid is no longer available in the United States, but is approved for HIT therapy in other countries, where it is frequently incorporated into national guidelines.92–94
Vitamin K antagonists
For patients receiving a VKA antagonist at the time of diagnosis, reversal with vitamin K should be administered given the risk of warfarin-induced skin necrosis and gangrene in the setting of HIT.79,95,96 When warfarin is the long-term anticoagulation of choice following resolution of HIT, it is important to bear in mind that initiation of VKAs should not be undertaken prior to the platelet count recovering to ⩾ 150 × 109/L. Transition to warfarin requires a period of overlap with a nonheparin anticoagulant. 79 Care should be taken not to overshoot the INR, as this has been associated with warfarin-related complications. 96
Direct oral anticoagulants
Direct oral anticoagulants (DOACs) include the direct Factor Xa inhibitors rivaroxaban and apixaban, as well as the DTI dabigatran. DOACs, particularly rivaroxaban, have gained traction as off-label therapy for HIT. DOACs encompass several desirable qualities including short onset of action, no required monitoring, and ease of administration. These qualities, as well as their lack of any potential interaction between HIT antibodies, make them an attractive option both as a primary therapy and as an alternative to warfarin for long-term anticoagulation. However, there is yet to be robust data describing DOAC use in HIT. A recent systematic review of the literature, which included 54 total patients, suggested that DOACs appear to be a viable, safe option to treat HIT following an initial short course of parental therapy with a nonheparin anticoagulant. 80 The optimal length of parental nonheparin therapy prior to DOAC initiation is unclear, and it is possible that DOACs may be used as first line alternative anticoagulation after stopping heparin. The Hamilton study, a small prospective observational study, included 12 patients with HIT, five of whom received rivaroxaban as primary therapy (i.e. without a period of alternative nonheparin anticoagulation). 97 Only one of these patients went on to have an additional thrombotic event, a possible extension of an upper extremity deep vein thrombosis (DVT) associated with a central venous catheter. The same group published follow-up data including additional cases and a literature review demonstrating an overall new, progressive, or recurrent thrombosis rate of 2.2% in patients treated with rivaroxaban during acute HIT (either as primary therapy or following a variable course of nonheparin/non-DOAC anticoagulation), with a bleeding rate of 0%. 98 An additional small retrospective study of 12 patients treated for acute HIT with apixaban or rivaroxaban demonstrated no major bleeding or thrombotic events; five of these patients received a DOAC as primary therapy without first receiving nonheparin parenteral therapy. 99 The 2018 ASH guidelines conditionally support the use of DOACs in isolated HIT and acute HITT in stable patients at average risk of bleeding. 76 However, DOAC use in HIT has not yet been widely adopted as there remains limited published data thus far.
Alternatives and adjuvants to alternative coagulation
There are two main alternatives to consider for treatment of the patient with refractory HIT: plasmapheresis and IVIG. Both approaches lack systematic evidence but may be of utility in challenging cases.
Plasmapheresis in the setting of HIT has been described in a number of reports, both in difficult to treat cases and as a neoadjuvant approach prior to CPB in patients with HIT antibodies.100–105 The ASH guidelines suggest the possibility of using plasma exchange in cardiovascular patients in which surgery cannot be delayed, noting that availability, cost, and clinician experience may affect this choice as opposed to other strategies. 76
There are nearly 30 reported cases of IVIG used to treat HIT.106–108 IVIG is hypothesized to work by saturating platelet FcγRIIa receptors, thereby inhibiting platelet activation via antibodies. 106 IVIG has been described as an effective therapy for cases of severe and refractory HIT. It has also been described as a novel, preventative approach in patients formerly diagnosed with HIT with persistent antibodies who need urgent surgical intervention requiring intraoperative heparin such as in cardiac transplantation and vascular surgery.109,110
Of note, IVIG has a potential risk of thrombosis and carries an FDA black box warning for such. There are no cases of apparent thrombus extension or new thrombus in any of the reported cases of HIT treated with IVIG,106–108 and although the true incidence is poorly understood, this risk must be considered.
Indications for platelet transfusion
In patients with HIT or strongly suspected HIT, prophylactic platelet transfusion for thrombocytopenia in the absence of bleeding is not recommended. 111 Platelet transfusion may be warranted in the case of bleeding or very high bleeding risk.76,111
Re-exposure to heparin
In general, if it is possible to avoid heparin-containing products in patients with a history of HIT, one should do so. However, for those patients in which heparin is needed, such as CPB, vascular surgeries, as well as hemodialysis, it is possible to re-introduce heparin without inducing a second episode of HIT under certain conditions. Clinical trials are lacking in this domain, however, and guidance on this topic is based on mainly retrospective data and expert opinion.112–115
There are multiple factors that make re-exposure to heparin possible in the patient with a history of HIT: anti-PF4/heparin antibodies are transient; patients with a remote history of HIT (weeks to months) who are currently antibody-negative should not retain immune memory to heparin; and, finally, it takes 5 days of heparin exposure to develop antibodies, leaving it possible to re-introduce heparin for < 4 days without eliciting a second episode of HIT.79,114 Detailed recommendations and guidance related to this matter should be sought for guidance prior to re-initiating heparin in the patient with HIT.79,114,116 Consideration of alternative approaches to anticoagulation in the patient with a history of HIT are also available.76,92,116,117
A note about HIT guidelines
A number of groups have published guidelines for the diagnosis and management of HIT, including ACCP, 79 ASH, 76 British Society for Hematology (BSH), 92 University of Wisconsin Health (UW Health), 118 and others.93,94,119 A recent systematic review evaluated 11 different HIT clinical practice guidelines using the Appraisal of Guidelines for Research and Evaluation (AGREE) II instrument, which assesses the rigor of clinical practice guideline development (CPG), 90 outlining discrepancies among the various CPGs for HIT. While all HIT guidelines recommend heparin cessation and starting alternative anticoagulation, the details of timing, choice of nonheparin anticoagulant, duration of therapy, and other practices vary. Elements that make the development of universal HIT guidelines difficult include local variability in availability of both laboratory assays and nonheparin anticoagulant drugs.
The problem of HIT overdiagnosis
Given the potential catastrophic complications associated with HIT as well as increased education surrounding the matter, clinicians are increasingly vigilant for this entity. However, it has been suggested that there is an ‘epidemic of overdiagnosis of HIT’, 120 with repercussions including increased cost, risk of bleeding, and unnecessarily delayed medical interventions. Furthermore, the misdiagnosed patient carries this forward as heparin is documented as an allergy in their medical chart. In one study, 42% of patients with a documented heparin allergy were actually HIT-negative, and, of those, 68% received alternative anticoagulation. Of those patients who received alternative anticoagulation with argatroban and/or lepirudin, 66% went on to meet International Society on Thrombosis and Haemostasis (ISTH) criteria for major bleeding. 121 Suggestions for avoiding overdiagnosis of HIT include evaluation of institutional HIT diagnosis and management, development of a HIT management protocol, and multidisciplinary collaboration. 122
Summary
HIT is a potentially catastrophic, life-threatening complication of one of the ubiquitous medications used in hospitals today, heparin. Diagnosis may be challenging and relies on clinical suspicion followed by stepwise testing. It is paramount to stop all forms of heparin exposure if HIT is strongly suspected, and to start alternative anticoagulation.
Key points to remember
HIT is manifested by a ⩾ 30% drop in platelet count from baseline ⩾ 5 days after heparin exposure
One-third to a half of patients develop HIT with thrombosis (HITT), which may be the presenting symptom
The ‘4Ts’ is the most common clinical assessment tool to assess the probability of HIT
For intermediate or high clinical probability for HIT, all heparin should be stopped immediately and nonheparin anticoagulation should be initiated while laboratory testing is underway
The most common diagnostic assays for HIT are the PF4/heparin ELISA and the SRA, which should be performed sequentially
Choice of alternative anticoagulation is multifactorial and must be tailored to the patient.
Conclusions
In summary, HIT is a rare but important cause of morbidity and mortality in patients exposed to heparin-containing products. A high index of suspicion is paramount to early recognition and abatement of complications. Understanding of appropriate testing is key to identifying true HIT. In those patients with HIT, decision making about alternative anticoagulation must be based on unique patient characteristics. As new nonheparin anticoagulants are developed, as well as novel assays with improved sensitivity, specificity, and availability, the future burden of HIT is likely to be reduced.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.