Abstract
Kawasaki disease (KD) is a systemic inflammatory process that affects the medium-sized arteries, causing various cardiovascular complications. However, it is not clear if the vascular sequelae following KD can predispose to the development of atherosclerosis later in life. Our aim was to examine the macrophage phenotypes in the coronary arteries forming giant aneurysms after KD to gain insight into the pathogenesis of vascular lesions in KD. We examined histological sections of the coronary arteries from five patients with KD who underwent coronary bypass grafting procedure as treatment for giant aneurysms and subsequent stenosis. Immunohistochemical expression of M1- and M2-macrophage markers was assessed to determine the macrophage phenotype of KD to compare with that of atherosclerosis in eight adult patients. All the KD specimens showed a mild to moderate degree of intimal thickening consisting of mature fibrous tissue and distortion of elastic fibers, mimicking the histological features of atherosclerosis. The total number of CD68 positive macrophages was higher in atherosclerosis than in KD specimens. Among the CD68 positive macrophages, the proportion of M1 phenotype, detected by CD86 or SOCS3, was higher in KD than in atherosclerosis. In contrast, the proportion of M2 phenotype, detected by CD163 or MRC1, was higher in patients with atherosclerosis. Despite similar histological features, KD and atherosclerosis appear to have a different immunological etiology for progression of the chronic vascular lesions. A further study enrolling a larger number of cases is required to delineate underlying mechanisms of vascular complications in KD.
Introduction
Kawasaki disease (KD) is an acute inflammation of the systemic medium-sized arteries that commonly affects children under the age of 5 years in developed countries.1,2 In the acute phase of KD, the coronary artery is infiltrated by numerous active inflammatory cells, which results in the destruction of elastic tissue of the vascular media with subsequent formation of artery aneurysms.3–5 Although the administration of intravenous γ-globulin has been shown to diminish the vascular inflammation, the coronary aneurysms still develop in about 10% of patients with KD, which can trigger myocardial ischemia or sudden death.6,7 In addition, the resulting coronary arteritis can increase the risk of long-term cardiovascular complications, such as high blood pressure and greater arterial stiffness.8–10 Yet, whether these KD-related changes in the arterial properties predispose to the development or acceleration of atherosclerosis remains an issue of debate.11–14
Macrophage heterogeneity is characterized by two different phenotypes, namely a classically activated macrophage (M1) and an alternately activated macrophage (M2).15,16 The M1 macrophages exert antimicrobial and tissue-destructive properties, via secretion of pro-inflammatory cytokines, whereas the M2 macrophages reduce the inflammatory reaction to promote angiogenesis and tissue repair. Atherosclerosis is a chronic inflammatory disorder of the arterial wall, where the phenotype and functions of macrophages are dictated by microenvironmental stimuli.17,18 Despite controversies regarding their role, both M1 and M2 macrophages can be found in the arterial walls of patients with atherosclerosis. Some studies have reported an increase in the number of M2 macrophages in vulnerable plaques of human arteries whereas others found a predominance of M1 macrophages in atherosclerotic plaques.19–23 To the best of our knowledge, however, no previous studies have examined the macrophage phenotype in arterial lesions of patients with KD. Therefore, our aim was to examine the macrophage phenotype in the wall of coronary arteries, in which giant aneurysms have formed after KD, and compare it to the phenotype in adult patients with atherosclerosis to gain insight into the pathogenesis of vascular lesions associated with KD.
Materials and methods
Case selection
Five patients with KD underwent coronary artery bypass grafting (CABG) for treatment of giant coronary aneurysms and subsequent artery stenosis at Nippon Medical School Hospital between January 1998 and August 2006 (Table 1). These patients also had aneurysm size-reduction procedures (plication) as an adjunct operation. The patients’ ages ranged from 1 year 1 month to 8 years 7 months (mean 3.7 ± 3 years), and the mean time from KD onset to CABG ranged from 12.2 to 122.7 months (mean 44.8 ± 44 months). The excised coronary artery specimens were subjected to histological analysis. For comparison, eight patients with a diagnosis of atherosclerosis were randomly selected from the archives of pathology at our hospital, and their carotid artery (n = 5) and aortic (n = 3) sections were investigated using the same technique (Table 2). Coronary arteries of three autopsied patients with no significant histological findings of atherosclerosis were used as negative controls.
Profiles of patients with Kawasaki disease.
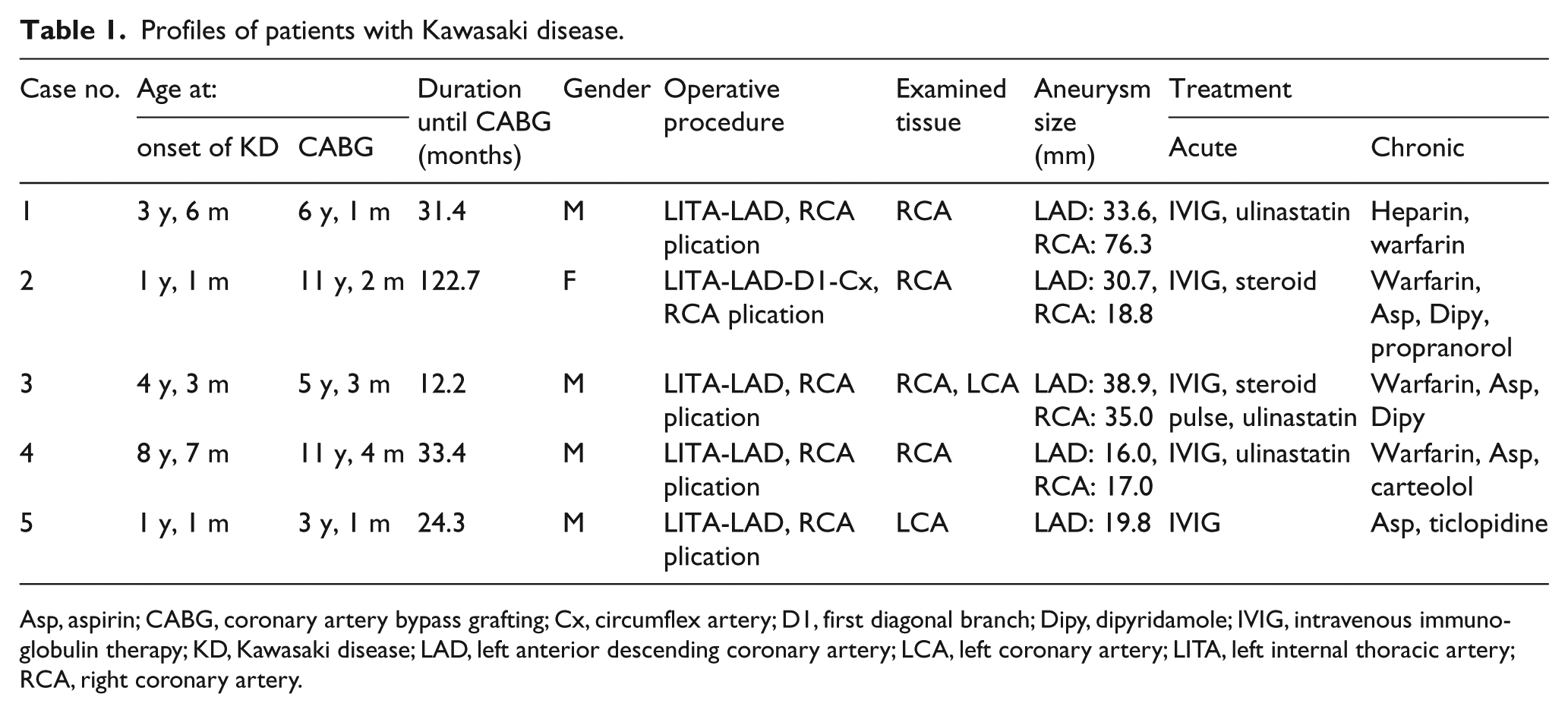
Asp, aspirin; CABG, coronary artery bypass grafting; Cx, circumflex artery; D1, first diagonal branch; Dipy, dipyridamole; IVIG, intravenous immunoglobulin therapy; KD, Kawasaki disease; LAD, left anterior descending coronary artery; LCA, left coronary artery; LITA, left internal thoracic artery; RCA, right coronary artery.
Profiles of atherosclerosis (cases 1–8) and control (cases 9–11) patients.
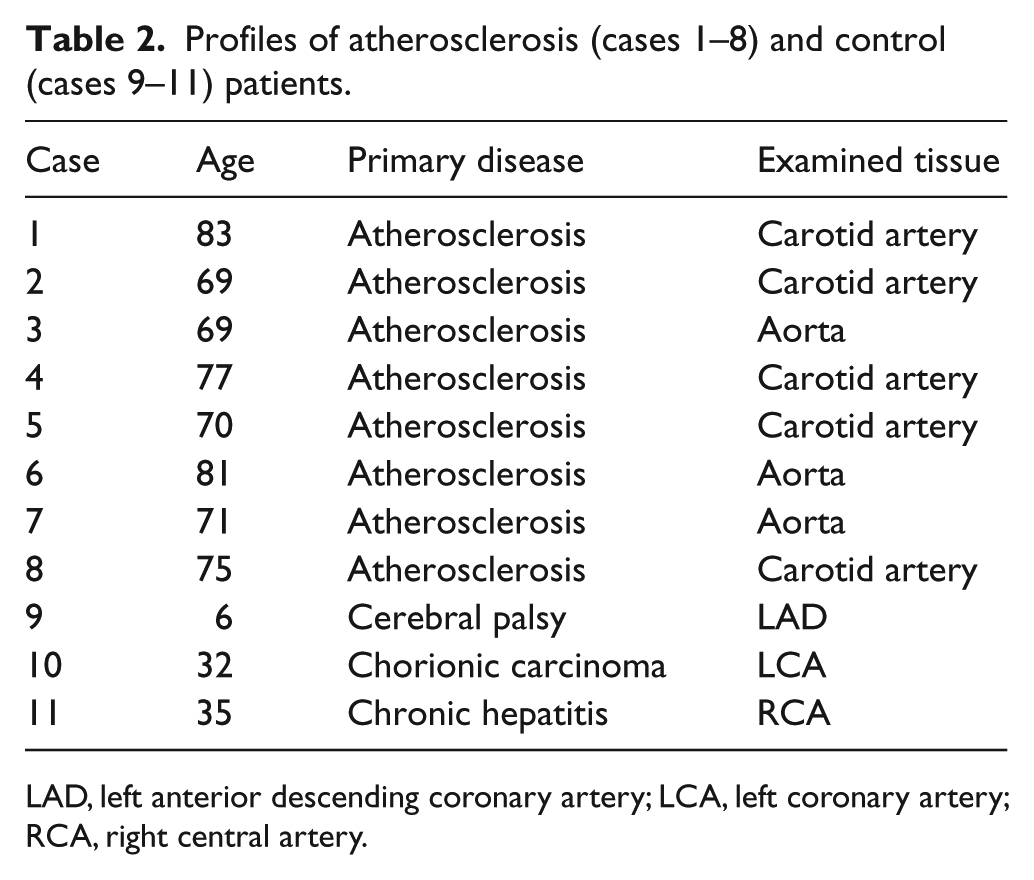
LAD, left anterior descending coronary artery; LCA, left coronary artery; RCA, right central artery.
The study was performed in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Nippon Medical School Hospital (#28-06-593). Written consent for using the sample for research purposes was obtained from all the patients.
Histological and immunohistochemical assessment
Resected tissues were fixed in formalin and subjected to standard processing and paraffin embedding. The tissues were sliced into 3-μm thick sections for hematoxylin and eosin (HE) staining. Elastica-Masson Goldner (EMG) staining was also performed to further investigate the vascular architecture. We reviewed all the HE and EMG sections to assess morphological characteristics of the vasculature. Immunohistochemical staining was performed on formalin-fixed histological sections with primary antibodies using the standard avidin-biotin-peroxidase complex technique (Table 3). For the identification of macrophages, we used: monoclonal mouse anti-human CD68 (PG-M1, dilution 1:100; Agilent, Santa Clara, CA, USA) as a pan-macrophage marker; polyclonal rabbit anti-human SOCS3 (dilution 1:100; Abcam, Tokyo, Japan), and monoclonal rabbit anti-human CD86 (EP1158Y, dilution 1:100; Abcam) as M1 macrophage markers; polyclonal rabbit anti-human MRC1 (dilution 1:200; Sigma-Aldrich, St Louis, MO, USA) and monoclonal mouse anti-human CD163 (10D6, dilution 1:100; Abcam) as M2 macrophage markers.18,22,24–26 The specificity of each antibody as M1 and M2 markers was assessed in serial sections of the atherosclerosis cases (online supplementary figure).
Primary antibodies used for immunohistochemical analysis.
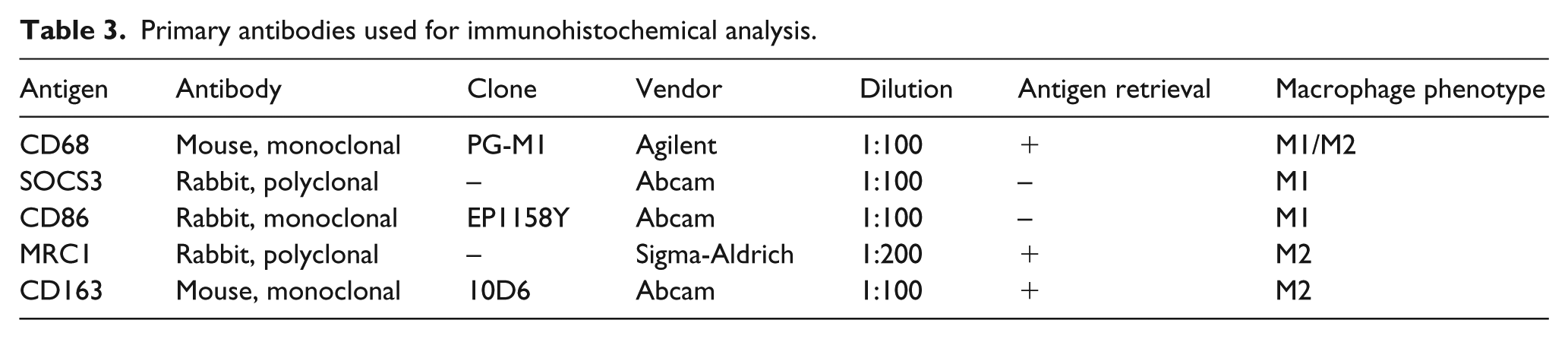
After deparaffinization, endogenous peroxidase activity was blocked by incubating sections in 0.3% hydrogen peroxide in methanol for 30 min. Antigen retrieval treatment was performed for some antibodies by using Immunosaver (Nisshin EM, Co., Ltd, Tokyo, Japan) for 45 minutes at 95°C. The tissue sections were then incubated with the primary antibodies diluted in phosphate-buffered saline containing 1% bovine serum albumin for 16 h at 4°C. Bound antibodies were detected with the Histofine Simple Stain MAX PO reagents (Nichirei Biosciences INC, Tokyo, Japan). Positive and negative controls for each antibody were used.
The number of CD68 positive cells was assessed in at least 10 high-power fields (HPF) of each layer of the vascular wall and expressed as mean per HPF. In assessment of other macrophage-associated markers, the number of positive cells was expressed as per total number of CD68 positive cells (%).
All immunohistochemically stained specimens were blindly and independently assessed by two investigators (RO and AS).
Statistical analysis
All data are shown as mean values ± SD of the mean. The Mann–Whitney U-test was used to compare variables between two groups. Statistical analyses were performed using JMP statistical software, version 11 (SAS Institute Inc., Cary, NC, USA). A p-value of < 0.05 was considered significant.
Results
Comparison of histological features of coronary arteries between Kawasaki disease and atherosclerosis cases
All the KD cases showed significant stenosis of the arterial lumen, associated with aneurysmal change of the vascular wall, in which no thrombus formation was identified. A mild to moderate degree of intimal thickening consisting of mature fibrous tissue was observed in KD cases 1, 2, and 4, while this finding was mild in KD cases 3 and 5 (Figure 1A). Only KD case 2 showed deposition of calcified material in the fibrous intima. In the arteries of KD cases 2 and 4, the tunica media was significantly thinner with distortion of elastic fibers, revealed by EMG staining (Figure 1B). The artery wall of all the KD specimens showed a mild to moderate degree of mononuclear cell infiltration on HE staining (Figure 1C, D). In all the atherosclerosis specimens, there was a moderate to marked intimal thickening of arterial walls associated with atheroma formation and deposition of cholesterol crystals with disruption of elastic fibers (Figure 1E, F). In at least six of the atherosclerosis cases, there was a variable degree of foamy macrophages in the intima, focally extending into the media. None of the KD cases exhibited significant foamy macrophage infiltration and deposition of cholesterol crystals in the arterial wall.
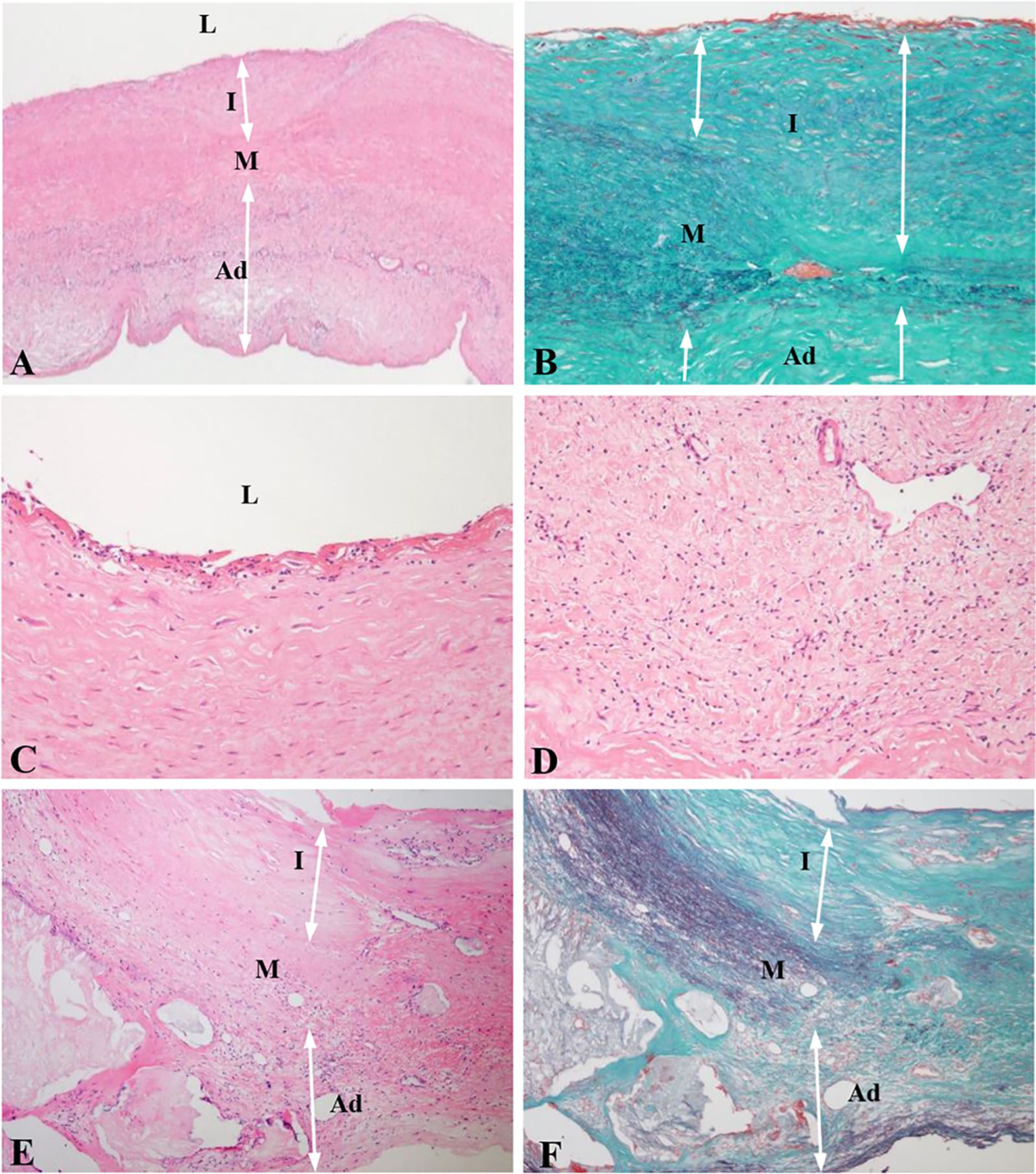
Histological findings of arteries from patients with KD (A–D) and atherosclerosis (E, F). In specimens in the KD group, there was a moderate degree of intimal thickening consisting of mature fibrous tissue (A) with distortion of elastic fibers (B). A varying degree of mononuclear cell infiltration in the subendothelial area was identified in all KD specimens (C) with a widened adventitial region (D). In the atherosclerosis group, there was moderate to marked intimal thickening of arterial walls with an atheromatous change characterized by deposition of cholesterol crystals, mononuclear cell infiltration, and accumulation of foamy macrophages, causing disruption of elastic fibers (E, F).
Comparison of M1 and M2 macrophage infiltration in Kawasaki disease and atherosclerosis cases
Representative histological findings of each macrophage marker are presented in Figure 2. The number of pan-macrophages, detected by CD68 staining, and the ratio of M1 and M2 macrophages out of CD68 positive macrophages are shown in Table 4, and in Figures 3 and 4. Although there was a varying degree of CD68 positive macrophages in each section of arteries from both patients with atherosclerosis and patients with KD, the macrophage infiltration was more pronounced in atherosclerosis than KD specimens in each layer of the arterial wall (p < 0.001) (Figure 2A, B, Table 4). The infiltration of CD68 positive macrophages was minimal or absent in all the layers of the arterial walls in control specimens.
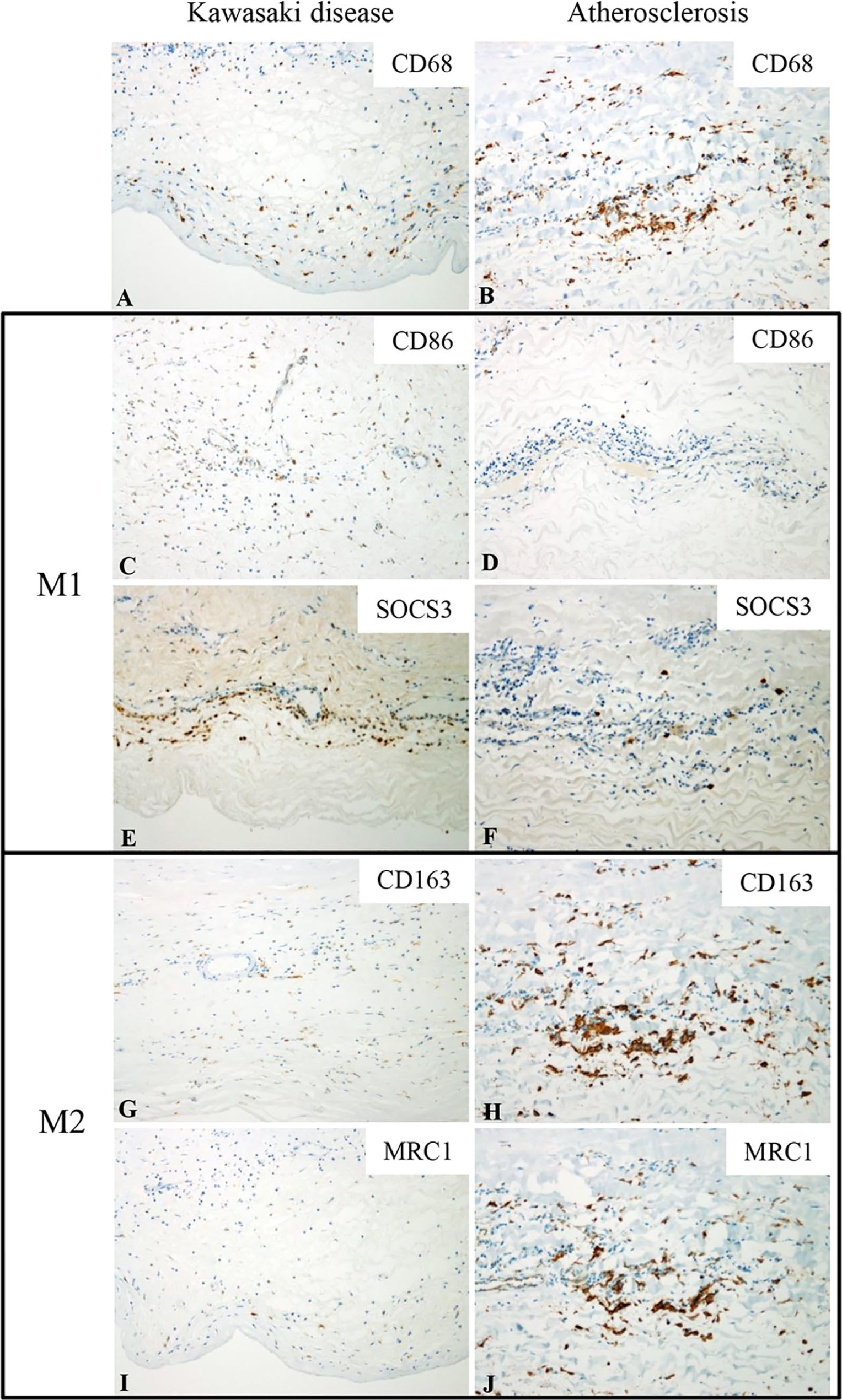
Immunohistochemical expression of CD68 (A, B), CD86 (C, D), SOCS3 (E, F), CD163 (G, H), and MRC1 (I, J) in the adventitia of arterial specimens of the KD (A, C, E, G, and I) and atherosclerosis (B, D, F, H, and J) groups. Although CD68 positive macrophage infiltration can be identified in KD (A), it is more pronounced in atherosclerosis (B). M1 markers CD86 and SOCS3 are positive in many of the macrophages in KD (C, E, respectively), but their expression is scarce in atherosclerosis (D, F). By contrast, M2 markers CD163 and MRC1 are highly expressed in the macrophages of atherosclerosis (H, J, respectively), while their expressions were limited to a few scattered cells in KD (G, I). Original magnification: ×100 (A–J).
Expression of macrophage markers in patients with KD and atherosclerosis.
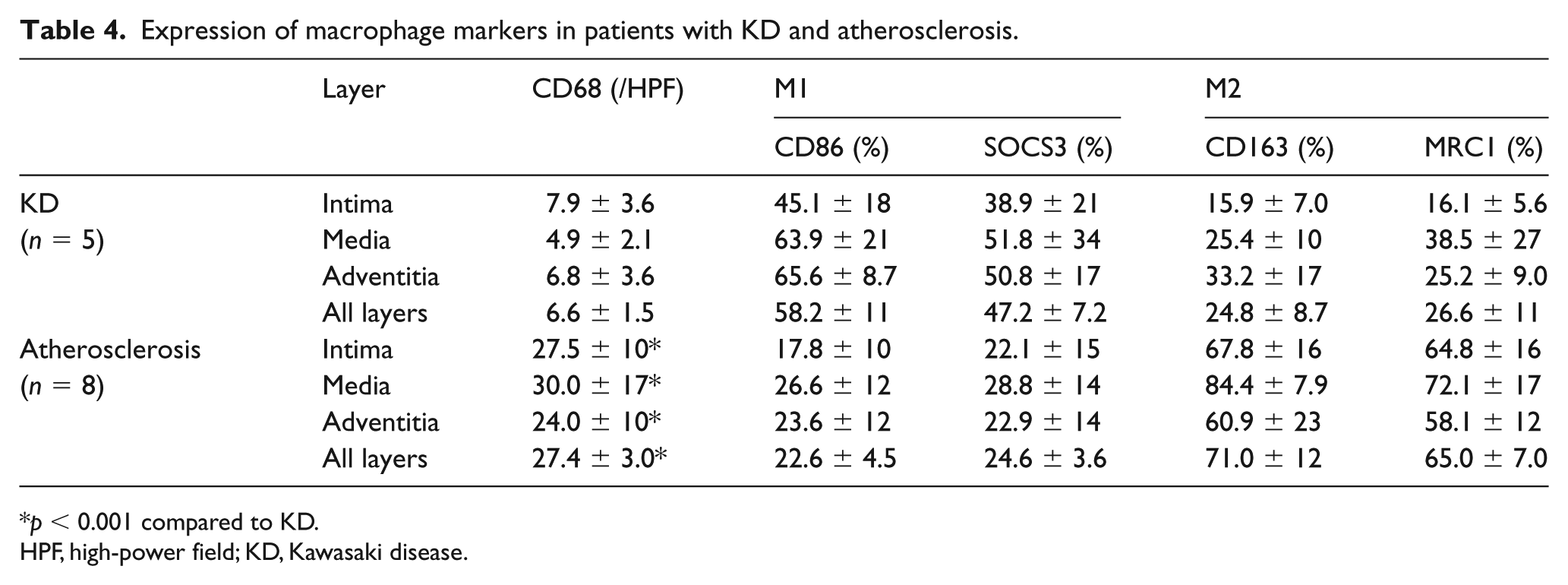
p < 0.001 compared to KD.
HPF, high-power field; KD, Kawasaki disease.
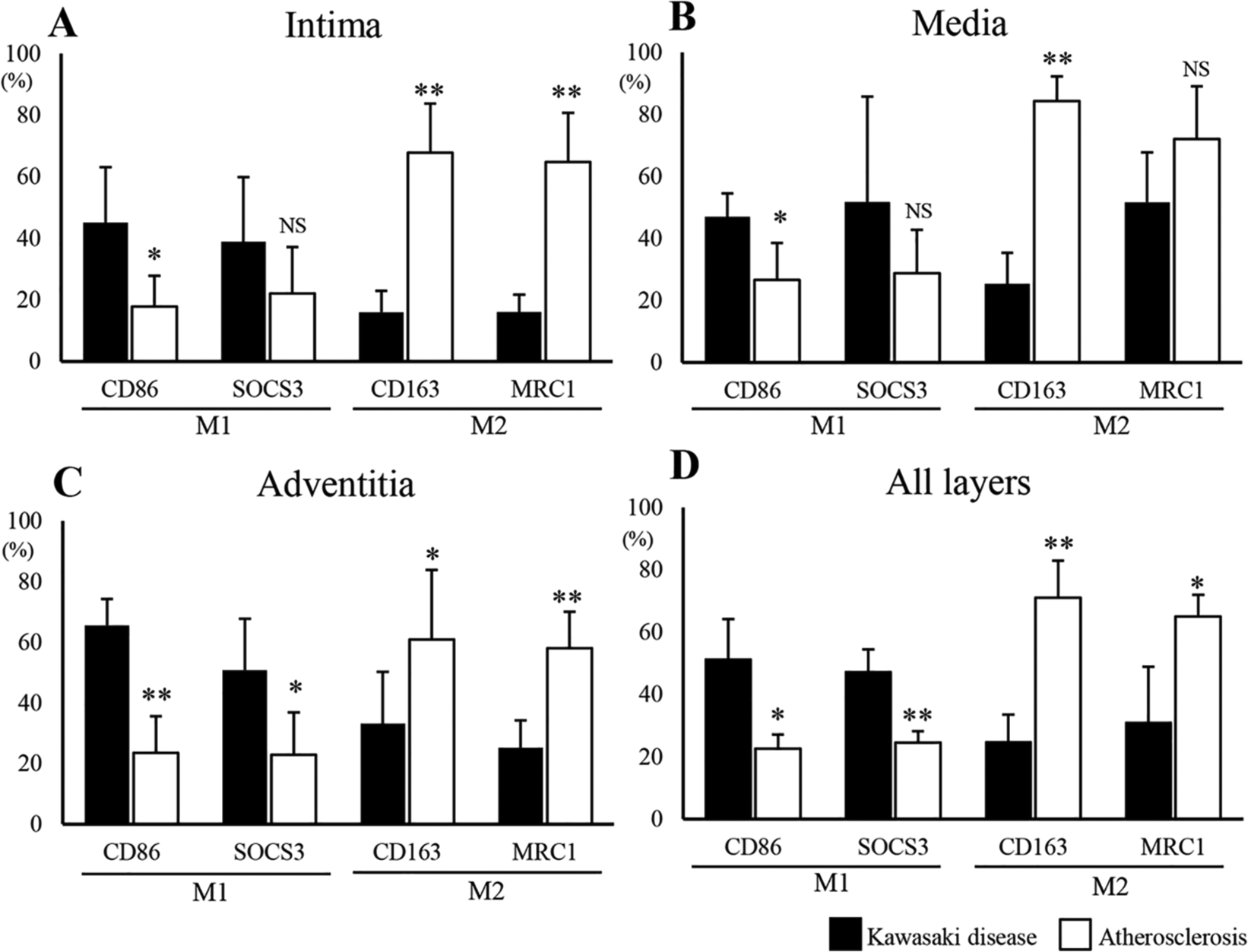
Comparison of each macrophage marker expression between KD and atherosclerosis in the three layers of the arterial wall (A–C) and all the layers (D). The degree of positivity for each marker is expressed per total number of CD68 positive cells (%).
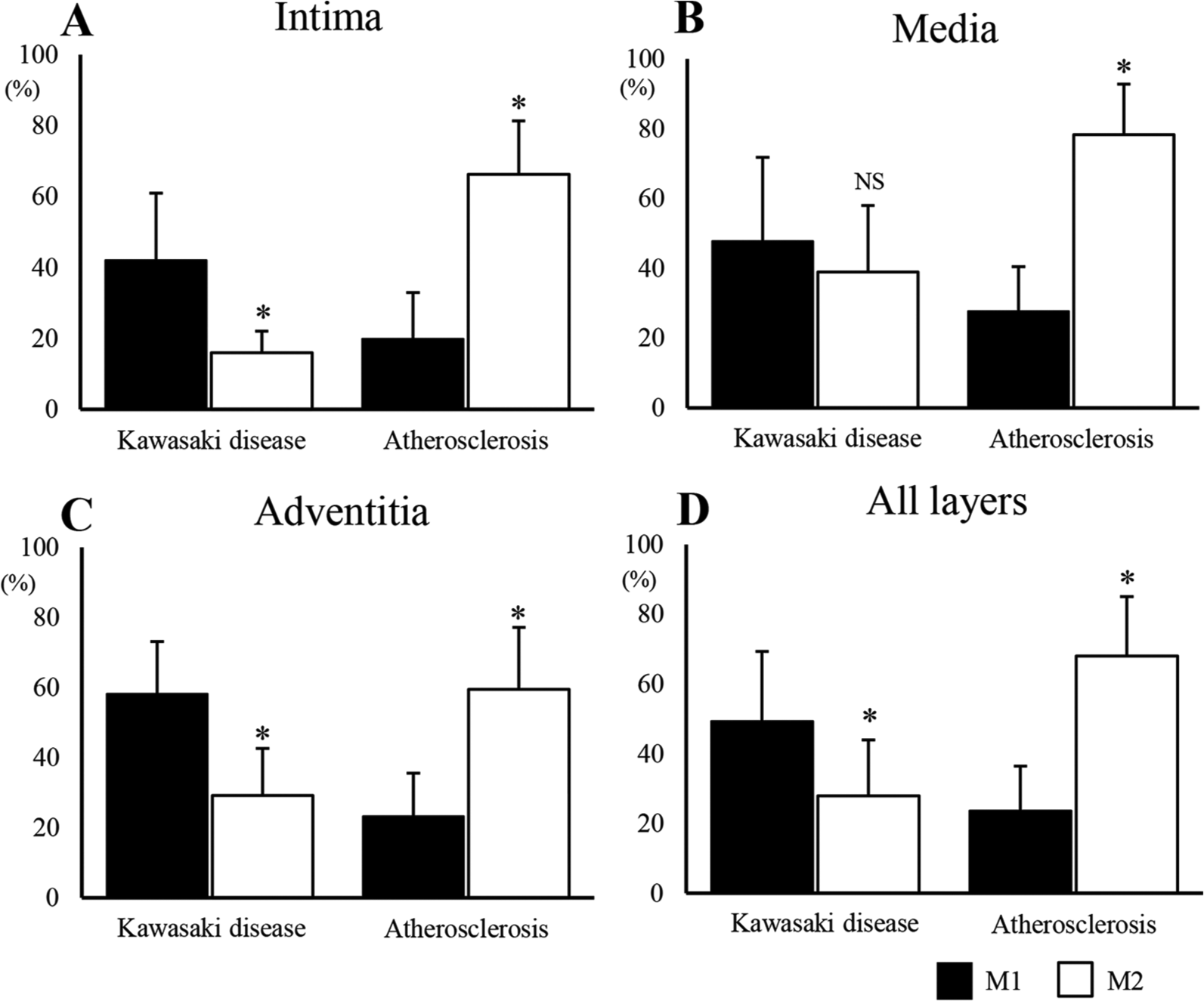
Comparison between M1 and M2 macrophage marker expression for KD and atherosclerosis in the three layers of the arterial wall. The degree of positivity for each marker is expressed per total number of CD68 positive cells (%).
The CD86 positive M1 macrophage ratio was predominantly higher in the intima of KD than atherosclerosis specimens (Figure 3A, p = 0.005), although there was no significant difference in the SOCS3 positive M1 macrophage ratio between the KD and atherosclerosis groups (Figure 3A, p = 0.12). In CD163 and MRC1 staining, the ratio of M2 macrophage in the intima was significantly higher in the atherosclerosis group than in the KD group (Figure 3A, p < 0.001 for both analyses).
In the media, the CD86 positive M1 macrophage ratio was higher in the KD than atherosclerosis group (Figure 3B, p = 0.03), with no significant difference between the two groups using SOCS3 staining (Figure 3B, p = 0.1). Although the CD163 positive M2 macrophage ratio was higher in the atherosclerosis than the KD group (Figure 3B, p < 0.001), the difference in the MRC1 positive M2 macrophage ratio was marginal between the two groups (Figure 3B, p = 0.057).
In the adventitia, the CD86 and SOCS3 positive M1 macrophage ratios were higher in the KD than the atherosclerosis group (Figure 2 C–F) (Figure 3C, p < 0.001 and p = 0.009, respectively). In contrast, the CD163 and MRC1 positive M2 macrophage ratios were higher in the atherosclerosis than the KD group (Figure 2 G–J) (Figure 3C, p = 0.004 and p < 0.001, respectively).
On a comparative analysis of all the layers of the vascular wall, the CD86 and SOCS3 positive M1 macrophage ratios were higher in the KD than the atherosclerosis group (Figure 3D, p = 0.02 and p < 0.001, respectively). Conversely, the CD163 and MRC1 positive M2 macrophage ratios were higher in the atherosclerosis than the KD group (Figure 3D, p < 0.001 and p = 0.04, respectively).
In comparison, between the KD and atherosclerosis groups for macrophage phenotypes in each vascular layer, the M1 macrophage ratio was higher in the intima and adventitia in the KD group (Figure 4A, C, p < 0.001), but not in the media (Figure 4B, p = 0.3). On the other hand, the M2 macrophage ratio was higher in the atherosclerosis group in each layer of the arterial wall (Figure 4 A–C, p < 0.001). In the analysis of all three arterial wall layers, the M1 macrophage ratio was higher in the KD than the atherosclerosis group, whereas the M2 macrophage ratio was higher in the atherosclerosis group (Figure 4D, p < 0.001 for both analyses).
Discussion
It has been under debate whether KD is associated with acceleration or development of atherosclerosis. Several studies using ultrasonography have shown an increase in the thickness and stiffness of the intima-media of the aortic and carotid arteries in patients with a history of KD compared to that in patients without a history of KD, suggesting that coronary arteritis in KD may predispose to an acceleration of atherosclerosis development.8–10,13,27 Histological assessment of autopsied specimens from patients with KD revealed a variety of alterations in coronary arteries, such as new intimal thickening, calcification, and thrombotic occlusion, most of which mimic histological features common in adult atherosclerosis. 12 On the other hand, a comparative study using B-mode ultrasound reported different textural patterns of the coronary arterial wall between KD and atherosclerosis of familial hypercholesterolemia, suggesting that the remodeling process in KD may differ from that of atherosclerosis. 28 Orenstein et al. performed a histological analysis of coronary arteries obtained from 41 patients with KD, and found no histological features of atherosclerosis even in patients who died later in life. 29 However, none of the above studies included an extensive delineation of constituents of inflammatory cells infiltrating in arteries with an emphasis on the macrophage phenotype. Herein, for the first time, we explored the inflammatory cellular components in coronary arteries of patients with KD, describing a heterogenous prevalence of M1 and M2 macrophages in each vascular layer.
M1, which are classical macrophages, possess a pro-inflammatory function in killing microbes and triggering inflammation. M1 macrophages are activated by microbial products, including lipopolysaccharide or pro-inflammatory cytokines such as interferon-γ.15,16 On the other hand, M2, which are alternately activated macrophages, have a critical role in the reduction of inflammation and promotion of angiogenesis and tissue repair. M2 macrophages are stimulated by cytokines, such as interleukin (IL)-4, IL-10, or IL-13, and produce IL-10 and the macrophage colony-stimulating factor. In atherosclerosis, previous studies reported an association between a higher expression of M2 macrophage markers and worse cardiovascular outcome.21,23,30 For instance, Jager et al. observed a higher accumulation of M2 macrophages in a vulnerable area of atherosclerotic plaques compared to other regions of arterial walls. 19 In agreement with these reports, we identified a significant number of pan-macrophages possessing a higher proportion of M2 macrophage in each layer of the vascular wall in atherosclerosis specimens. Conversely, KD specimens showed a modest degree of macrophage infiltration, in which the M1 phenotype was more pronounced than M2. Our findings collectively indicate a different immunological etiology between atherosclerosis and arterial lesions in KD.
Previous studies have investigated macrophage phenotypes in a variety of systemic inflammatory diseases, with variable results depending on the disease. Macrophages in synovial fluid collected from patients with rheumatoid arthritis (RA) have a higher M1/M2 ratio, suggestive of a predominant role of M1 macrophages in RA. 31 Peripheral blood analyses of patients with RA identified a positive correlation between a M1/M2 ratio and the promotion of osteoclastogenesis. 32 In contrast, M2 macrophages were major constituents of granulomatous inflammation in the antineutrophil cytoplasmic antibody (ANCA)-associated crescentic glomerulonephritis, a form of systemic vasculitis. 33 In the respiratory tract of patients with granulomatosis with polyangiitis affecting small to medium-sized arteries, M2 was also identified as the predominant macrophage phenotype. 26 Taking these findings and our results together, we can postulate that the immunological process of KD may be similar to that of RA. This hypothesis should be tested in a future study enrolling a larger number of patients with RA and KD, as our study included only five patients with KD.
In KD, the vascular lesions develop through three stages: (1) necrotizing arteritis, (2) subacute/chronic vasculitis, and (3) luminal fibroblastic proliferation. 29 As the current study employed tissue samples obtained over 1 year after disease onset, we cannot determine if the macrophages observed are residues of early inflammatory reaction or a new phenomenon appearing from a later stage. This is a limitation of our study, and thus it will be necessary to assess patients with KD during the acute phase of the disease to determine the macrophage phenotype significantly contributing to the progression of arteritis in KD in the future.
The large number of pro-inflammatory macrophages can be distributed into some regions of the artery that are more susceptible to rupture (e.g. coronary and carotid arteries), and thus some may postulate the inflammatory response may differ among different regions of the artery. In the present study, atherosclerosis samples were obtained from two regions, such as aorta (n = 3) and carotid artery (n = 5). We compared these two groups but found no difference in macrophage phenotypes (data now shown). However, there is still a possibility that inflammatory reaction may differ depending on the regions of arteries. This hypothesis should be assessed in the future using a number of cases with atherosclerosis. Patients suffering from atherosclerosis are usually under treatment with various kinds of drugs including statins, aspirin, calcium-blockers, and β-blockers, raising a concern that the inflammatory response in the vasculature may be modified by the drug. In fact, five out of eight patients with atherosclerosis in our study had been treated with statins at least for over 3 years, which could have exerted an anti-inflammatory effect. 34 Therefore, it should be noted that the results of atherosclerosis lesions should be interpreted with caution, taking into account the effect of drugs that have been taken.
Conclusion
To conclude, although a higher proportion of M2 macrophages was noted in atherosclerosis, the M1 phenotype was predominant in KD. These results suggest a different immunological etiology underlying the formation of vascular lesions in KD and atherosclerosis. As the current study included a limited number of cases, a further study enrolling a larger number of patients with KD is warranted to reveal underlying mechanisms of vascular complication in KD.
Supplemental Material
10.1177_1358863X19878495_Supplementary_figure – Supplemental material for M1 macrophage is the predominant phenotype in coronary artery lesions following Kawasaki disease
Supplemental material, 10.1177_1358863X19878495_Supplementary_figure for M1 macrophage is the predominant phenotype in coronary artery lesions following Kawasaki disease by Ryuji Ohashi, Ryuji Fukazawa, Akira Shimizu, Shunichi Ogawa, Masami Ochi, Takashi Nitta and Yasuhiko Itoh in Vascular Medicine
Footnotes
Acknowledgements
The authors thank Mr Shigeki Namimatsu for excellent technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by a grant from a Grant-in-aid for Scientific Research in Japan (16K10079) from the Ministry of Education, Culture, Sports, Science and Technology.
Supplemental material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
