Abstract
Optimal management of patients with internal carotid artery (ICA) stenosis concurrent with severe cardiac disease remains undefined. The aim of this study is to evaluate the safety and feasibility of the one-day, sequential approach by carotid artery stenting (CAS) immediately followed by cardiac surgery. The study included 70 consecutive patients with symptomatic > 50% or ⩾ 80% asymptomatic ICA stenosis coexisting with severe coronary/valve disease, who underwent one-day, sequential CAS + cardiac surgery. The majority of patients (85.7%) had CSS class III or IV angina and 10% had non-ST elevation myocardial infarction. The EuroSCORE II risk was 2.4% (IQR 1.69–3.19%). All CAS procedures were performed according to the ‘tailored’ algorithm with a substantial use of proximal neuroprotection devices of 44.3%. Closed-cell (75.7%) and mesh-covered (18.6%) stents were implanted in most cases. The majority of patients underwent isolated coronary artery bypass grafting (88.6%) or isolated valve replacement (7.1%). No major adverse cardiac and cerebrovascular events (MACCE) occurred at the CAS stage. There were three (4.3%) perioperative MACCE: one myocardial infarction and two deaths. All MACCE were related to cardiac surgery and were due to the high surgical risk profile of the patients. Up to 30 days, no further MACCE were observed. No perioperative or 30-day neurological complications occurred. In this patient series, one-day, sequential CAS and cardiac surgery was relatively safe and did not result in neurological complications. Thus, a strategy of preoperative CAS could be considered for patients with severe or symptomatic ICA stenosis who require urgent cardiac surgery.
Keywords
Introduction
Optimal management of patients with severe carotid and cardiac disease remains to be established. Significant internal carotid artery (ICA) stenosis is a risk factor for stroke associated with cardiac surgery in the embolic (unstable carotid plaque) and cerebral hypoperfusion mechanisms.1–3 The risk of perioperative stroke ranges between 3.2% (unilateral, asymptomatic ICA stenosis) and 8.5% in patients with symptomatic ICA stenosis.2,3 Stroke is a serious and devastating complication of cardiac surgery with in-hospital mortality reaching 22%. 4
About 8–13% of patients accepted for coronary artery bypass grafting (CABG) and 5% of patients with severe aortic valve stenosis have significant (⩾ 70–80%) ICA stenosis.5–8 There are several possible treatment strategies for severe, concurrent carotid and cardiac disease. The surgical approach includes staged or simultaneous carotid endarterectomy (CEA) and cardiac surgery is associated with a high perioperative combined event rate: death, cerebrovascular accident, and myocardial infarction (MI) of 10.2–11.5%. 9
Another treatment option is carotid artery stenting (CAS), either staged or with simultaneous cardiac surgery. In the staged procedure, CAS is performed in the first stage with delayed cardiac surgery about 30 days later. This has been associated with a major adverse event (MAE) rate of 6.7–9.4% (death, stroke, MI) when ICA was neurologically asymptomatic and 12.3% in patients with a history of stroke or transient ischaemic attack (TIA).10–12
In one-day, sequential CAS + cardiac surgery, CAS performed in the first stage is immediately followed by cardiac surgery. This might be an option for patients with unstable cardiac disease and severe/symptomatic ICA stenosis. According to the previously published registries, this strategy has a periprocedural outcome of about 2.2–4.5% (death/any stroke, no MI),13,14 but it requires further evaluation.
The aim of this study is to evaluate the safety and outcome of the one-day, sequential strategy of CAS according to the ‘tailored’ algorithm followed by immediate cardiac surgery, performed in a group of patients with coexisting severe/symptomatic ICA stenosis and severe coronary/valve disease with unstable cardiac symptoms.
Materials and methods
Study population
In a tertiary, high-volume cardiovascular centre, performing over 2000 open heart surgeries and over 200 CAS procedures per year, between the years 2009 and 2017, 130 consecutive patients had severe/symptomatic ICA stenosis coexisting with multivessel coronary artery disease (MVD) and/or severe valvular heart disease (VHD). The primary strategy in this population was CAS combined with cardiac surgery. CEA was performed if ICA anatomy was not suitable for an endovascular procedure. Patients with CCS (Canadian Cardiovascular Society) class III/IV or NYHA (New York Heart Association) class III symptoms and non-ST elevation MI (NSTEMI), requiring urgent treatment, who underwent one-day, sequential CAS + cardiac surgery (70, 54%), were included in the prospective, non-randomized registry. All patients were individually assessed by a multidisciplinary, joint neurovascular and heart team, including a cardiac surgeon, invasive and non-invasive cardiologist, neurologist, vascular surgeon, angiologist, and anaesthetist, to determine the correct treatment modality, as shown in Figure 1. All patients were treated by the ‘hybrid team’, which consisted of all the above-mentioned specialists.
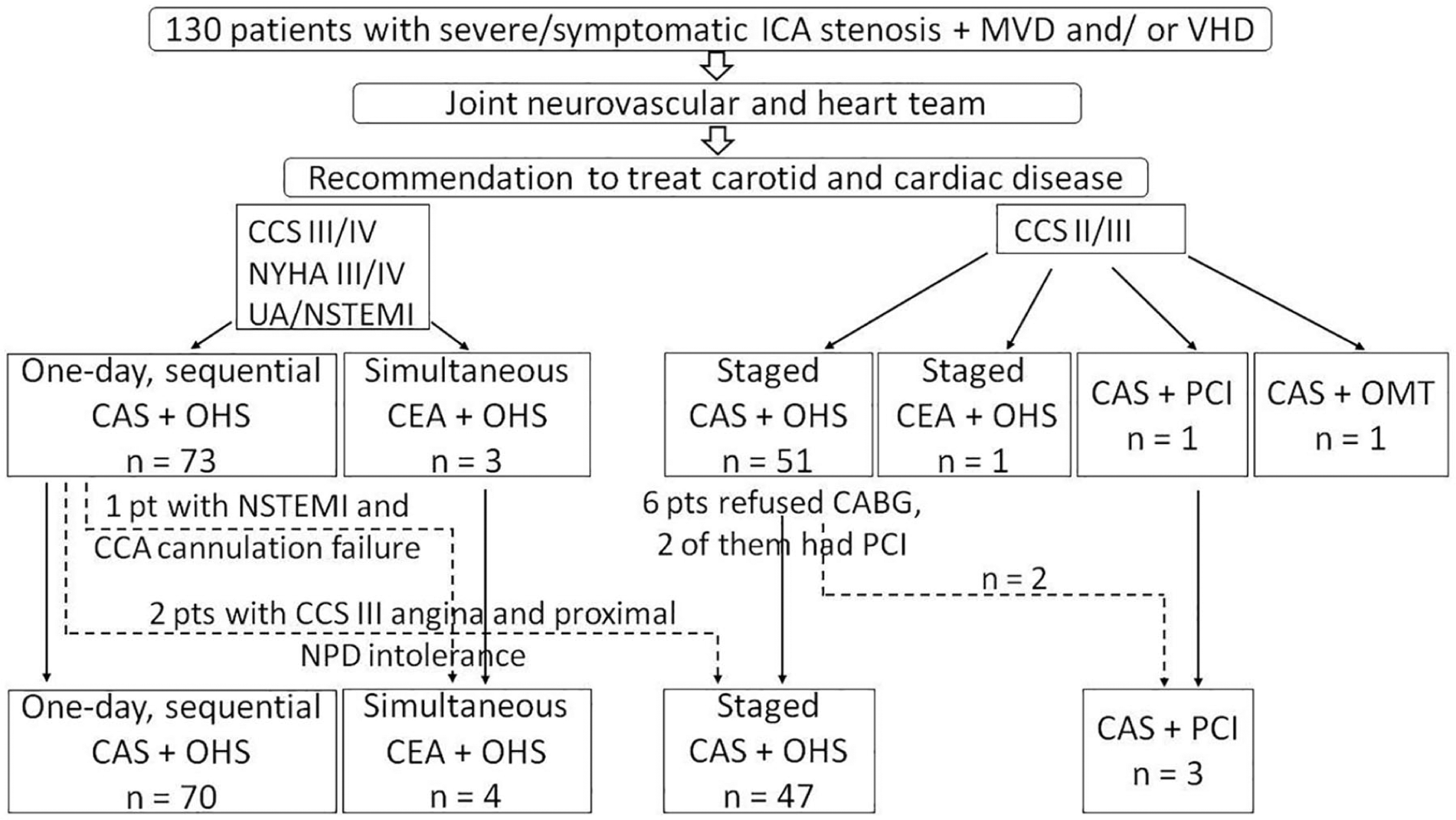
Management of patients with severe carotid and cardiac disease.
One patient with NSTEMI initially accepted for one-day, sequential CAS + CABG was converted to simultaneous CEA + CABG due to common carotid artery cannulation failure. Two other patients with CCS III angina had transient neurological symptoms associated with proximal neuroprotection device (NPD) intolerance that resolved completely after the procedure; computed tomography (CT) showed no abnormalities, but owing to the decision of the neurologist and anaesthetist the cardiac surgery had to be delayed. Those patients (n=3) were not included in further analysis as they were allocated to CAS + delayed cardiac surgery.
The study had the approval of the local ethics committee and each patient gave informed written consent.
Patient evaluation
Patients were eligible if angiographically confirmed ICA stenosis was > 50% in symptomatic disease and ⩾ 80% in asymptomatic disease. Neurological status was assessed by an independent neurologist prior to and after the procedure, and at discharge. ICA stenosis was defined as neurologically symptomatic in cases of ipsilateral stroke or TIA in the preceding 6 months. 15 Patients with asymptomatic ICA lesions (⩾ 80%) were included in the presence of at least one of the following: impaired cerebral flow on transcranial Doppler examination; silent stroke ipsilateral to the ICA lesion on CT scan; soft, ulcerated plaque of high embolic potential or contralateral occlusion. ICA stenosis was initially evaluated on carotid Doppler ultrasound, using combined Bluth and NASCET (North American Symptomatic Carotid Endarterectomy Trial) criteria,16,17 and then confirmed on angiography. Surgical risk was estimated with the EuroSCORE II calculator. 18
One-day, sequential procedure
CAS was always performed in the first stage and it was immediately followed by cardiac surgery (one-day, sequential, ‘hybrid’ procedure). All CAS procedures were done under local anaesthesia via femoral access, using a 6–9 F (French) introducer sheath. NPD use during CAS was mandatory. CAS was done according to the ‘tailored’-CAS approach, which is used for all CAS procedures in the centre. According to the ‘tailored’-CAS algorithm, proximal NPD and closed-cell or mesh-covered stents are indicated for soft, ulcerated, symptomatic/string-sign ICA lesions (Figure 2) and distal NPD is used for non-severe or asymptomatic stenoses.19–21 CAS procedural success was defined as a residual stenosis ⩽ 30% after stent placement. Immediately after CAS, patients were transferred directly to the operating room, or both procedures were performed in the hybrid room. Surgical procedures included isolated CABG/valve surgery or complex procedures.
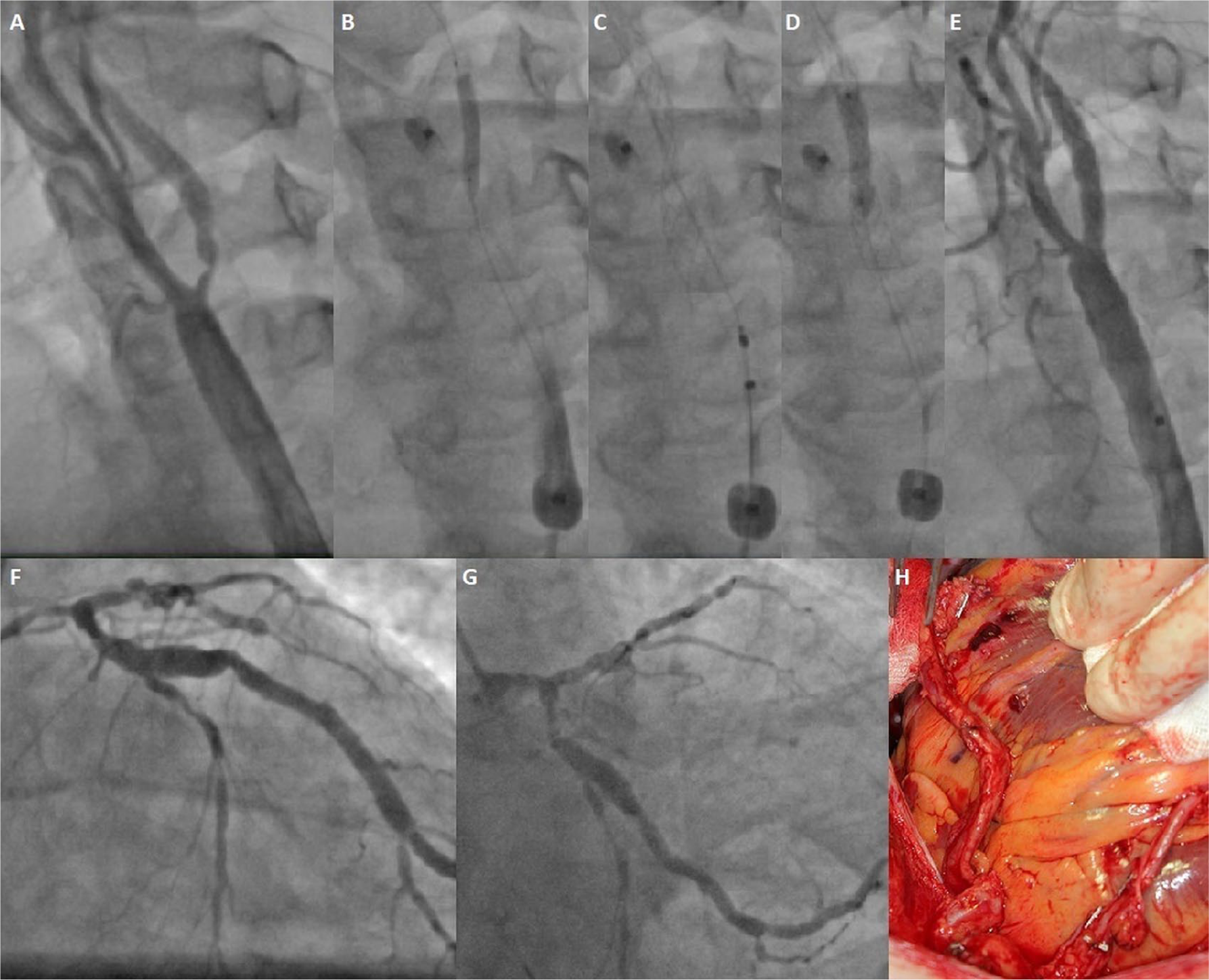
One-day, sequential CAS (‘tailored’ approach) and CABG. (A) Symptomatic (transient aphasia 2 weeks before) 90% LICA stenosis by ulcerated plaque in a patient with NSTEMI; (B) proximal NPD (Mo.Ma) placement and lesion predilatation; (C) mesh-covered stent Roadsaver 7.0 × 25 mm implantation; (D) stent postdilatation; (E) final result after CAS; (F) coronary angiography: critical left main and LAD stenosis; (G) diffuse disease in circumflex artery; (H) implantation of left IMA to marginal branch and right IMA to LAD.
Pharmacological treatment regimen
Patients were on acetylsalicylic acid (ASA) 75–100 mg daily for at least 5 days before CAS or received loading dose (300 mg) a day before procedure. During CAS, unfractionated heparin (UFH) of 100 IU/kg was administered intravenously to maintain activated clotting time (ACT) of ⩾ 250 ms and was then continued during surgical procedure under ACT control of ⩾ 400–450 ms. In the end of surgery protamine was given. Clopidogrel (loading dose of 300 mg) was administered in sixth postoperative hour, after extubation or via nasogastric tube, when major surgical bleedings were excluded and thoracic drainage was decreasing. Dual antiplatelet therapy (DAPT) was continued for at least a month at standard doses of ASA and clopidogrel.
Endpoints and follow-up
Patients were evaluated for adverse events during their in-hospital stay and at a month after hybrid procedures. Stroke, death, and MI were defined as major adverse cardiac and cerebrovascular events (MACCE). TIA was defined as focal brain or retinal ischaemic symptoms lasting no longer than 24 hours in the absence of CT changes and was considered a minor neurological event. Bleeding requiring rethoracotomy, acute renal failure with the need for haemodialysis, respiratory failure, systemic infection, and local complications of femoral puncture were defined as non-MACCE. Before discharge, each patient had carotid Doppler ultrasound and echocardiography. A month after procedures, an on-site follow-up visit and carotid Doppler ultrasound were performed.
Statistical analysis
Continuous variables were tested for normal distribution using the Kolmogorov–Smirnov test. The variables with normal distribution were expressed as means ± SD, and variables with skewed distribution as medians and IQR. Categorical data were expressed as a proportion and percentages. The univariate logistic regression model was used to find significant predictors for MACCE and non-MACCE outcomes. The statistical analysis was performed using the IBM SPSS version 23 statistical package (IBM Corp., Armonk, NY, USA). The significance level was set at α = 0.05.
Results
Patients included in the registry were of advanced age with many comorbidities. The overall characteristics of the population is shown in Table 1.
Baseline characteristics of 70 patients who underwent one-day, sequential CAS + cardiac surgery.
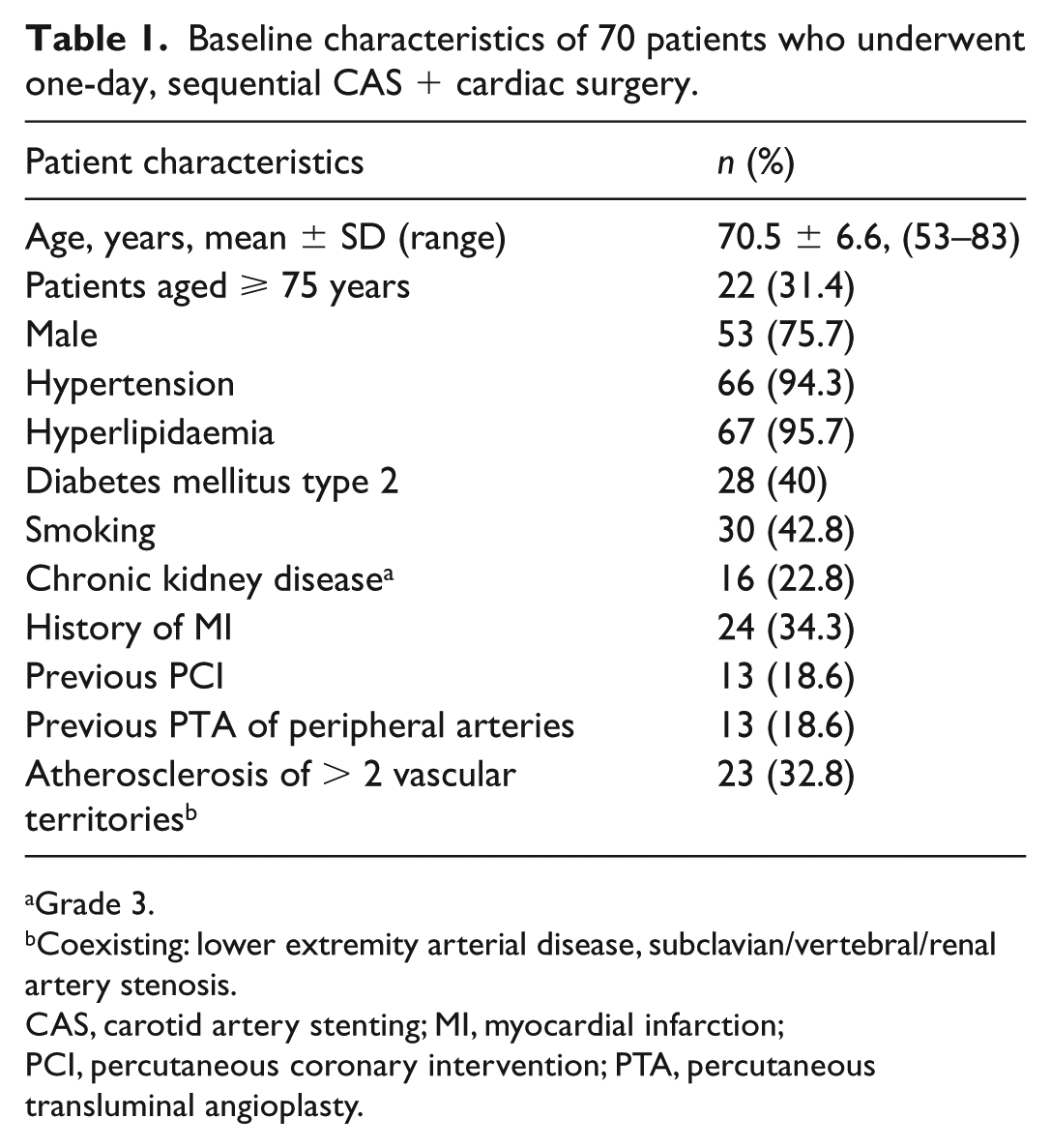
Grade 3.
Coexisting: lower extremity arterial disease, subclavian/vertebral/renal artery stenosis.
CAS, carotid artery stenting; MI, myocardial infarction; PCI, percutaneous coronary intervention; PTA, percutaneous transluminal angioplasty.
The majority of patients had isolated coronary artery disease (88.6%) and CCS class III angina symptoms (57.1%). CCS IV/unstable angina (UA) was present in 28.6% and there were 10% of patients with NSTEMI. Patients with VHD were in NYHA/CCS class III or had recent pulmonary oedema (in the preceding 2 weeks). Data on the symptoms and severity of carotid and cardiac disease are shown in Table 2.
Symptoms and severity of carotid and cardiac disease.
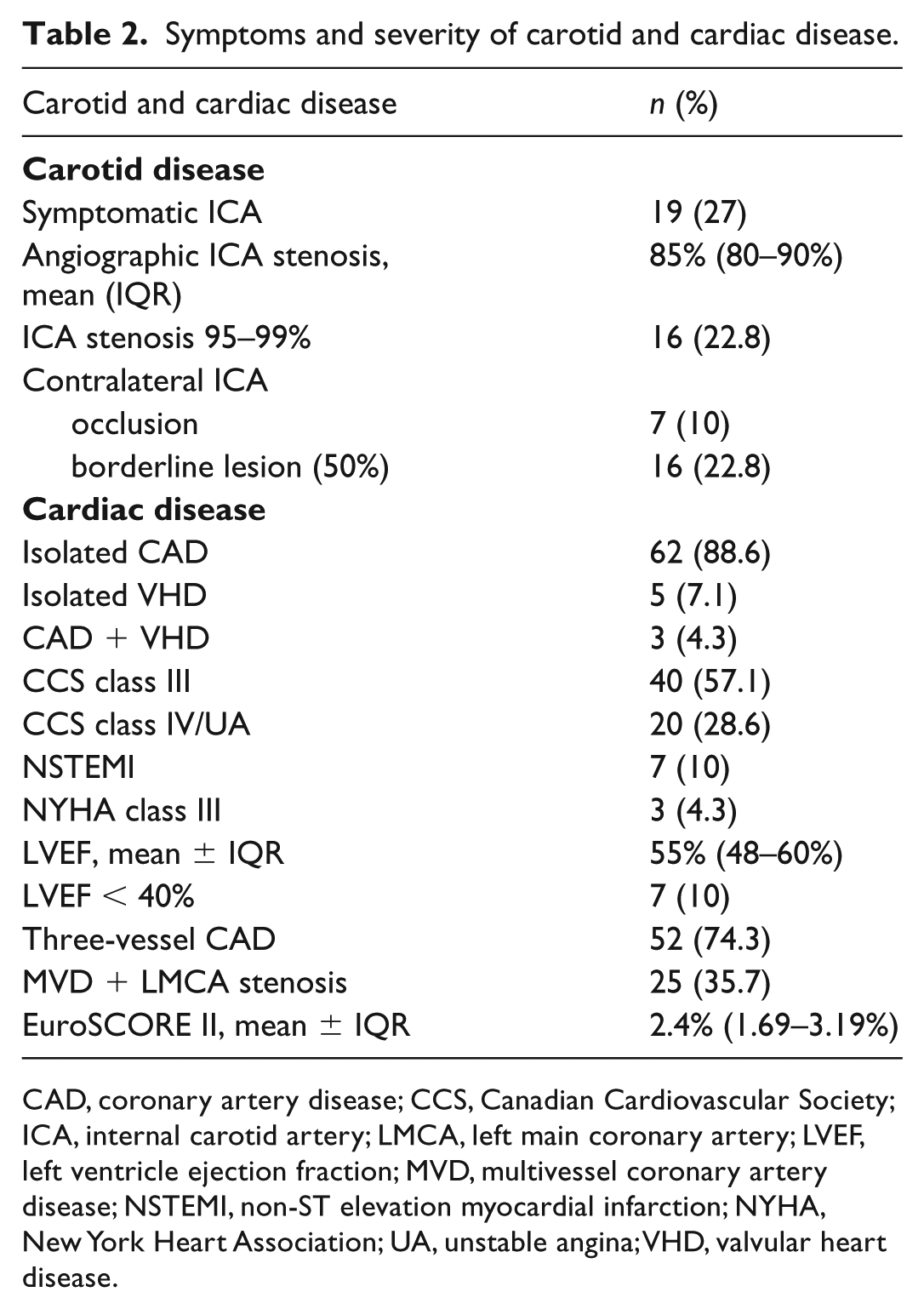
CAD, coronary artery disease; CCS, Canadian Cardiovascular Society; ICA, internal carotid artery; LMCA, left main coronary artery; LVEF, left ventricle ejection fraction; MVD, multivessel coronary artery disease; NSTEMI, non-ST elevation myocardial infarction; NYHA, New York Heart Association; UA, unstable angina; VHD, valvular heart disease.
All CAS procedures were technically successful. Proximal NPD was used in 44.3% of procedures. Closed-cell stents were implanted in 75.7% of patients and mesh-covered stents in 18.6% of patients. Others received open cell stents. Details on NPD and stent types are shown in Table 3. A femoral artery closure device was used in 10 (14.3%) CAS procedures.
Types of NPD devices and stents used in the study population of 70 patients.

CAS, carotid artery stenting; NPD, neuroprotection device.
Consistent with a typical referral profile to our centre, the majority of patients underwent isolated CABG (88.6%). Other cardiac surgery procedures included isolated valve surgery or complex procedures. All patients, after one-day, sequential CAS + cardiac surgery, were transferred to the intensive care unit (ICU), where the median stay was 2 (IQR 1–4) days. For data on surgical procedure and postoperative stay see Table 4.
Surgical procedure and postoperative care.
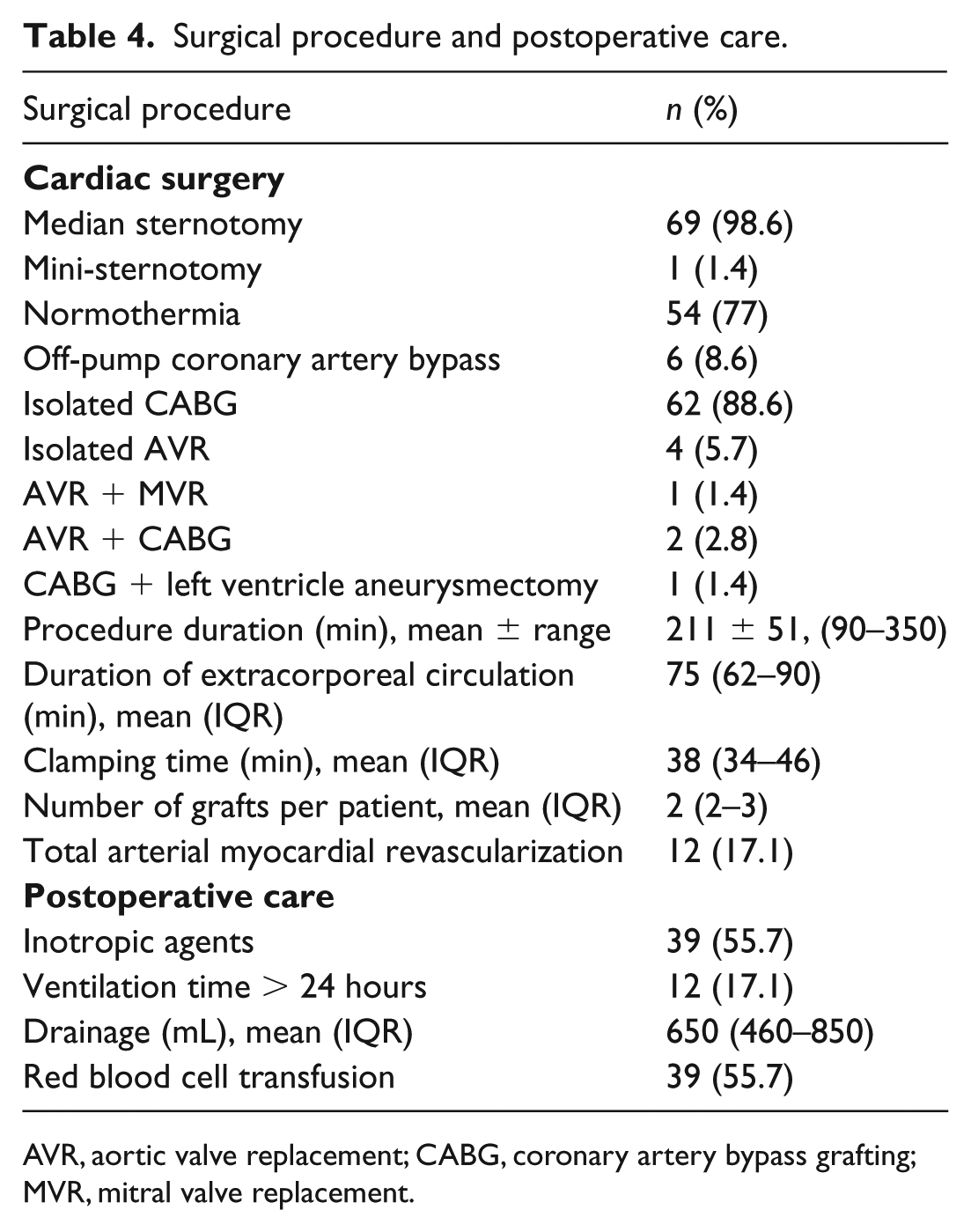
AVR, aortic valve replacement; CABG, coronary artery bypass grafting; MVR, mitral valve replacement.
Endpoints
No MACCE occurred at the CAS stage. All MACCE were related to cardiac surgery and occurred in high surgical risk patients. There were three (4.3%) MACCE: two deaths and one MI in the perioperative period. MI, in a patient after incomplete surgical revascularization, was successfully treated with right coronary artery percutaneous coronary intervention (PCI). One in-hospital death was due to multiorgan failure in a patient with a EuroSCORE II of 6.58%, who underwent CABG and left ventricle aneurysmectomy. Another patient died following isolated aortic valve replacement due to failure to wean from extracorporeal circulation. Neither strokes nor TIAs were observed. For up to 30 days, no further MACCE occurred.
There were 19 (27%) non-MACCE complications. Three (4.3%) non-MACCE complications were related to CAS and were associated with femoral artery puncture: two (2.8%) pseudoaneurysms, managed by local compression/percutaneous thrombin injection, and one (1.4%) acute right limb ischaemia on the second postoperative day, treated with embolectomy. A further 16 (22.8%) non-MACCE complications were associated with cardiac surgery. They included: bleeding requiring rethoracotomy, 6 (8.6%); systemic infection, 6 (8.6%); acute renal failure with the need for haemodialysis, 1 (1.4%); and respiratory failure, 3 (4.3%).
All carotid stents were patent with laminar flow, on carotid Doppler ultrasound, performed at discharge and at the 30-day follow-up. Neither in-stent thrombosis nor restenosis were observed.
On univariate logistic regression analysis, a non-CABG/complex procedure was a predictor of all (MACCE and non-MACCE) complications (odds ratio (OR) 6.944, 95% CI 1.454–33.176, p = 0.015). Other risk factors for all complications were prolonged (> 24 h) ICU stay (OR 1.412, 95% CI 1.098–1.815, p = 0.007) and NSTEMI (OR 5.128, 95% CI 1.018–25.824, p = 0.047). A non-CABG/complex procedure was the only predictor of MACCE (OR 20.333, 95% CI 1.599–258.521, p = 0.02), while the others were borderline significant: EuroSCORE II (OR 1.978, 95% CI 0.993–3.940, p = 0.052) and extracorporeal circulation duration (OR 1.057, 95% CI 0.990–1.129, p = 0.099). Risk factors for non-MACCE were prolonged ICU stay (OR 1.467, 95% CI 1.121–1.920, p = 0.005) and NSTEMI (OR 5.667, 95% CI 1.117–28.739, p = 0.036).
Discussion
Since CAS might be considered as an alternative to CEA according to the current guidelines, 15 the idea to combine the endovascular and surgical approach has grown. Staged CAS and delayed cardiac surgery has already been reported to have slightly lower MAE rate than staged CEA and cardiac surgery. 22 The strategy of one-day, sequential, ‘hybrid’ CAS followed by immediate cardiac surgery has a low perioperative complication rate, according to the already published registries.13,14,23–26 The main findings of the current study are that: one-day, sequential CAS + cardiac surgery was associated with a relatively low periprocedural MACCE rate of 4.3% with all MACCE related to the surgical stage only, there were no periprocedural neurological events, and no severe bleeding or thrombotic complications.
All CAS procedures in this registry were performed according to the tailored-CAS algorithm. Proximal NPD was used in 44.3% of procedures. Those strategies were reported to have a low MAE rate of about 2% (including 1.2–1.7% strokes).19–21,27 Mesh-covered stents were implanted in 18.6% of patients and there were no cerebrovascular events associated with the implantation of this stent type in registries.28,29 With the use of all those methods during CAS there were no periprocedural and 30-day neurological events in the current study population. In some previous studies, same day CAS + cardiac surgery was free from strokes as well,23,24 whereas others noted an incidence of 1–4.5% of contralateral13,14 and 0.75–2% of any strokes.25,26 Surgical, simultaneous CEA + CABG, according to the literature data, still carries a 3% risk of perioperative ipsilateral stroke. 9
The majority of patients included in this registry had neurologically asymptomatic ICA stenosis. Both the ACAS (Asymptomatic Carotid Atherosclerosis Study) and ACST (Asymptomatic Carotid Surgery Trial) randomized trials have already shown the benefit of carotid revascularization (CEA) versus optimal medical therapy in neurologically asymptomatic patients, if periprocedural risk of death/stroke is < 3%.30,31 CAS according to the ‘tailored’ algorithm was reported to have a 30-day death/stroke rate of 1.2% in asymptomatic patients. 21 Cardiopulmonary bypass is associated with blood pressure drops, which in the presence of subtotal, soft, ulcerated carotid lesion may significantly increase the risk of perioperative stroke (cerebral hypoperfusion/embolism). If ICA stenosis is left untreated, the risk of perioperative stroke in asymptomatic patients grows with stenosis severity and is highest among those with subtotal or bilateral stenosis.2,3,32 The benefit of ‘prophylactic’ ICA revascularization preceding cardiac surgery in asymptomatic patients is still not well proven, and according to the current guidelines it might be considered in selected cases only. 15 Owing to some experts’ opinions, however, it is reasonable to perform carotid revascularization in asymptomatic ICA stenosis ⩾ 80%, not only for perioperative stroke prevention, but also for long-term prognosis, and experienced centres should prefer CAS staged with CABG because of the lower MI rate. 33 One randomized trial showed a 7.7% ipsilateral stroke rate among patients with asymptomatic ICA stenosis who underwent cardiac surgery first and then CEA, versus 0% in the group who had CEA preceding cardiac surgery, p = 0.008. 34 Another randomized study – simultaneous CEA + CABG versus isolated CABG – reported a high 30-day ipsilateral stroke rate in both groups (12.7% vs 5%). This trial, however, included a small population of elderly patients with many comorbidities and was previously terminated due to low recruitment and, according to the authors’ considerations, the data might be insufficient to justify isolated CABG. 35 In the current registry, one-day, sequential CAS + cardiac surgery performed in patients with asymptomatic ICA stenosis was free from neurological events and previous registries13,14,23–26 also reported favourable outcomes. It seems that this group may benefit from ‘prophylactic’ carotid revascularization by CAS preceding cardiac surgery, but that still needs further evaluation.
The overall outcome of 4.3% in the present study shows that one-day, sequential CAS + cardiac surgery might be a safe treatment strategy. All events were due to the high surgical risk profile of the patients. Our data are consistent with those previously published showing MACCE of 2.2–4.5%.13,14,23,25,26 One study, including a high surgical risk population (redo and complex cardiac surgery), reported a higher MACCE rate of 9.2% (postoperative deaths, no ipsilateral strokes and MIs). 24 Complex cardiac surgery procedures, which are of higher risk than isolated CABG, 25 were also performed as a part of the one-day, sequential strategy in the current registry. Both deaths in the study population occurred after complex/non-CABG surgery, which was a risk factor for MACCE and non-MACCE on univariate analysis in this registry. A subpopulation of 68 patients, treated with hybrid CAS + CABG, in the multicentre FRIENDS (Finalised Research In ENDovascular Strategies) registry, showed a relatively high periprocedural outcome (MI 4.4%, stroke 5.9%). The authors indicate, however, that this strategy might be an option for severely diseased patients with unstable carotid and/or coronary symptoms and should be performed by experienced teams. 36 Recently published meta-analysis showed a 30-day death/stroke/MI rate of 6.5% for one-day, sequential CAS and cardiac surgery. 37
Cardiopulmonary bypass requires a higher ACT than CAS. The combination of DAPT with UFH, directly after CAS, during cardiac surgery, can potentially increase surgical and intracranial bleeding risk (haemorrhagic transformation of preexisting ischaemic brain lesion or cerebral hyperperfusion). Intracranial bleeding is rare (0.7%), but often a fatal complication of CAS. 19 The strategy to perform one-day, sequential CAS and cardiac surgery on ASA and UFH only, with administration of clopidogrel in the sixth postoperative hour (early postoperative DAPT), seems to be safe. In this registry there were no severe bleeding or thrombotic complications (no carotid stent thrombosis observed). Reoperation due to surgical bleeding was done in 8.6% of cases, which occurred in the beginning of the study, and compared to the literature data, reporting 2.4–6% reexploration rate after isolated cardiac surgery, 38 is slightly elevated, but still acceptable. If CAS is staged with (delayed) cardiac surgery, patients require DAPT for a month after CAS. The delay of cardiac revascularization carries a potential risk of cardiac complications in the waiting period.10,12,22
So far, there is one small randomized trial comparing simultaneous CAS + CABG versus CEA + CABG, which showed MACCE in the surgical group only. 39 When compared with the published data of simultaneous CEA + CABG, 9 one-day, sequential CAS + cardiac surgery in the current and previous studies13,14,23–26,39 seems to have a lower MACCE rate (especially neurological events).
Limitations
This is a non-randomized, single-centre registry. All CAS procedures were performed in the high-volume endovascular centre by experienced operators, which might have influenced the outcome.
Conclusion
Patients with severe, concomitant carotid and cardiac disease require cautious assessment and a multidisciplinary approach. One-day, sequential CAS and cardiac surgery in this registry was relatively safe and did not result in neurological complications at CAS and cardiac surgery stage, in patients with both symptomatic and asymptomatic carotid disease. All complications occurred in high surgical risk patients after cardiac surgery. Non-CABG/complex procedures, prolonged ICU stay, and NSTEMI seem to be risk factors for all MACCE and non-MACCE complications. This patient series indicates that CAS immediately followed by cardiac surgery could be considered as a treatment option for patients with severe carotid and cardiac disease requiring urgent cardiac surgery. One-day, sequential CAS and cardiac surgery might constitute a promising alternative to the conventional surgical methods. This strategy needs the cooperation of two teams (endovascular and surgical) and should be performed in high-volume cardiovascular centres. The one-day, sequential CAS and cardiac surgery approach might be applicable to other populations and health systems; however, it requires further verification in multicentre registries.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
