Abstract
Loss to follow-up (LTF) has been associated with worse outcomes after procedures. We sought to identify differences in lower extremity peripheral vascular intervention (PVI) patients with and without LTF, and to determine if LTF impacted survival. Patients in the PVI registry of the Vascular Quality Initiative (VQI) were included (n = 39,342), where t-test and chi-squared analysis were used to compare those with and without LTF. Multivariable logistic regression was used to identify factors associated with LTF while Cox regression analysis was applied to compare survival among those with and without LTF. The overall 1-year follow-up rate was 91.6%. LTF patients were more often male, Hispanic, of black race, and had a higher rate of diabetes, coronary artery disease, congestive heart failure, and dialysis. LTF patients had a higher prevalence of critical limb ischemia, underwent popliteal or distal intervention, and were intervened upon urgently. There was also a higher rate of postoperative complications, and a lower rate of technical success for LTF patients. After controlling for center effects, the independent variables associated with LTF included male sex, age, diabetes, dialysis dependence, ASA class 3 or greater, as well as complications requiring admission. Preoperative aspirin, preadmission home living status, prior carotid intervention, and discharge aspirin were protective against LTF. Adjusted survival analysis showed decreased survival in LTF, with those returning face-to-face surviving longer than those with phone follow-up. Efforts should be focused on understanding these differences to improve follow-up rates and help improve overall survival.
Keywords
Introduction
Peripheral vascular interventions (PVI) have far outpaced lower extremity bypass for the treatment of peripheral artery disease (PAD). 1 PAD remains a major public health burden, affecting 8 million people over 40 years of age in the United States, and 12–20% of Americans above the age of 65.2,3 Yet, while peripheral interventions continue to burgeon, and new advances in endovascular technology seemingly appear every day, the optimal management of patients following endovascular intervention is not well defined. Specifically, the role of appropriate and timely follow-up has not been established. Indeed, the recent Society for Vascular Surgery guidelines on the management of PAD recommend baseline and 3–6 month follow-up visits, but admit that the evidence for said recommendation is low. 4 The follow up of patients after other vascular procedures, such as endovascular aneurysm repair (EVAR), has been published recently, espousing the importance of in-person follow-up and surveillance imaging, to reap the benefits of close surveillance on mortality. 5 The authors go on to stress the importance of office-based outpatient follow-up, particularly in those patient populations that they discovered were more likely to be lost to follow-up. We sought to establish whether loss to follow-up (LTF) at 1 year was associated with worse outcomes in the national Vascular Quality Initiative (VQI).
Methods
Deriving the patient cohort
Patients entered from January 2010 to January 2017 in the PVI module of the VQI were considered. PVI was defined as endovascular procedures from the aorta distally which were performed to improve circulation to the limb. Participation in the VQI mandates that 1-year follow-up (between 9 and 15 months) is performed and recorded in a follow-up form, in accordance with previous studies. 6 For ease of analysis, only the first chronologically occurring PVI was included as the index procedure for the patient. Owing to possible issues of survivorship bias, those patients who died before 9 months from the procedure were excluded from the denominator, as they were considered ineligible for follow-up. Follow-up was further categorized as face-to-face, phone follow-up, no follow-up possible, and other source. Face-to-face and phone contact were both considered valid means of follow-up. The follow-up form consists of demographics, medications, ambulatory, functional, and symptom status. Ankle–brachial index, patency, and events such as amputation and reintervention are also recorded.
Statistical analysis
Patients were separated into those lost to follow-up and those with follow-up. Student’s t-test and chi-squared analysis were performed to compare continuous and categorical variables thought to be relevant to the study question. Multivariable logistic regression was then performed to identify variables associated with LTF, where a forward selection approach was undertaken using p = 0.25 as the cut-off for inclusion for all variables reaching a p-value of 0.10 on univariate analysis. Complications included cardiac, pulmonary, renal, access-related issues, bleeding, thrombosis, stroke, amputation, and death. Any complications incurred that required readmission to the hospital were defined as complications requiring admission. Death was ascertained by the following methods: when a patient was due for follow-up, data were entered regarding mortality by the center. For all other patients, the Social Security Death Index was queried. Cox regression analysis was used to evaluate survival by follow-up status adjusting for other variables. The statistical threshold for significance for all analyses was p < 0.05. All analyses were performed using Stata statistical software, release 12 (StataCorp LP, College Station, TX, USA). Because this study qualified as a Quality Improvement Project, it did not meet the requirements for formal review by the University of Pennsylvania Institutional Review Board.
Results
There were 74,144 patients identified as having PVI procedures during that time period, with 39,342 having follow-up forms. Of the patients with follow-up forms, the 1-year follow-up rate was 91.6%. Overall, the average age of the cohort was 67.9 years, and there were 60.0% males, 5.3% of Hispanic ethnicity, and 18.7% of nonwhite race. Patients lost to follow-up were more often male (62.2% vs 59.8%, p < 0.001), of nonwhite race (20.9% vs 18.5%, p = 0.013), hypertensive (90.0% vs 87.5%, p < 0.001), diabetic (54.4% vs 47.2%, p < 0.001), congestive heart failure (CHF) patients (26.4% vs 15.5%, p < 0.001), dialysis-dependent (12.6% vs 6.4%, p < 0.001), ASA class 3 or higher (87.4% vs 77.1%, p < 0.001), and critical limb ischemia patients (49.3% vs 44.7%, p < 0.001). LTF patients were less likely to be current or former smokers (74.7% vs 80.8%, p < 0.001), less likely to have a preadmission home living status (92.5% vs 96.9%, p < 0.001), history of prior carotid endarterectomy (CEA)/carotid artery stent (CAS) (18.4% vs 23.5%, p < 0.001), and were less likely to be compliant with preoperative aspirin or statin. Further details regarding demographic differences are given in Table 1.
Demographics/preoperative characteristics.
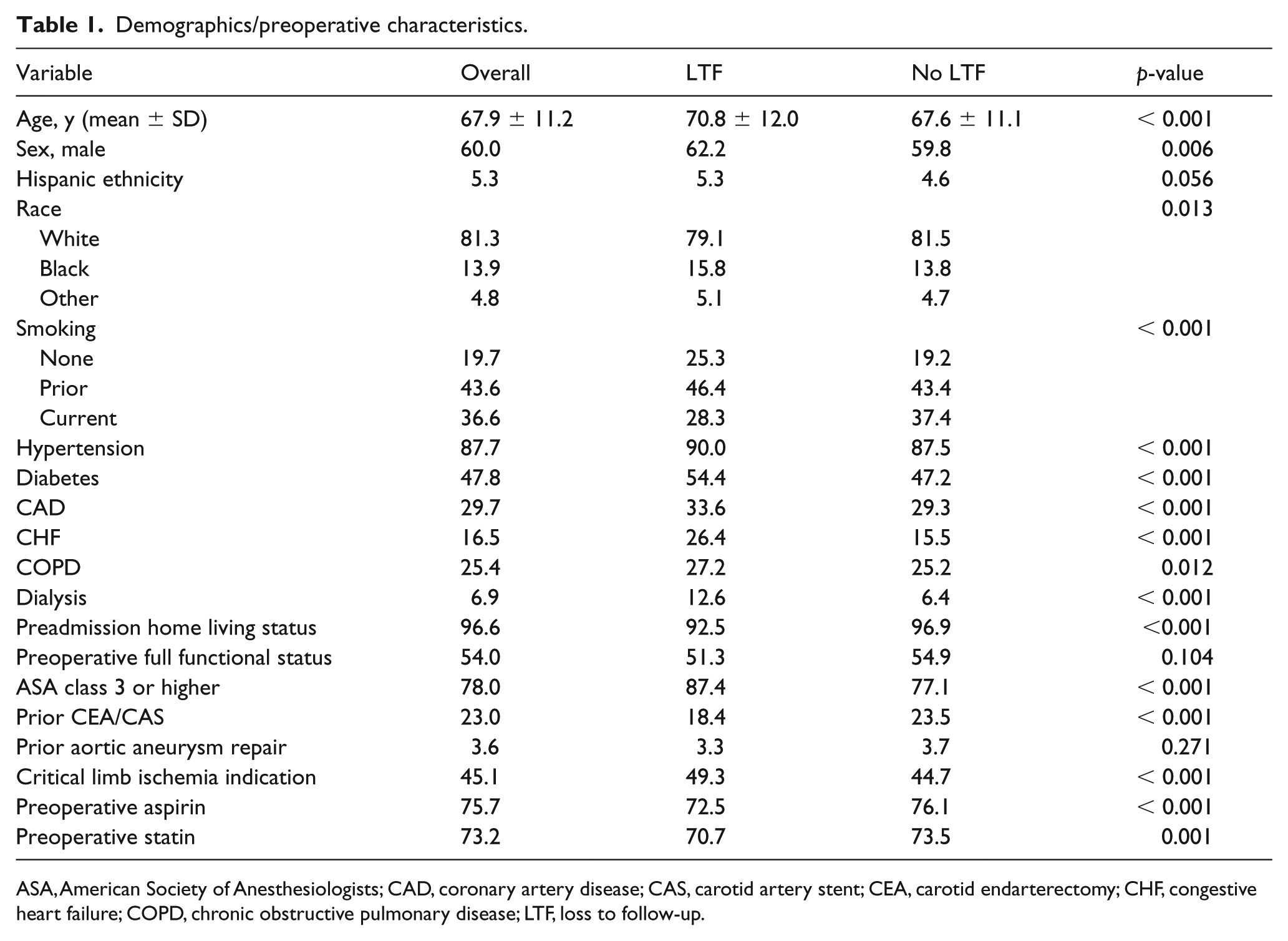
ASA, American Society of Anesthesiologists; CAD, coronary artery disease; CAS, carotid artery stent; CEA, carotid endarterectomy; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; LTF, loss to follow-up.
Regarding operative variables, LTF patients were more likely to be operated on in an urgent manner (21.9% vs 14.0%, p < 0.001), had a greater fluoroscopy duration (23.6 vs 17.7 minutes, p < 0.001), smaller sheath size (5.9 vs 6.2, p < 0.001), fewer access sites (1.16 vs 1.19, p < 0.001), fewer arteries treated (1.43 vs 1.67, p < 0.001), less technical success (67.7% vs 99.3%, p < 0.001), and more popliteal and infrapopliteal artery treatment (34.2% vs 21.9%, p < 0.001). Details regarding intraoperative variables are provided in Table 2.
Operative characteristics.
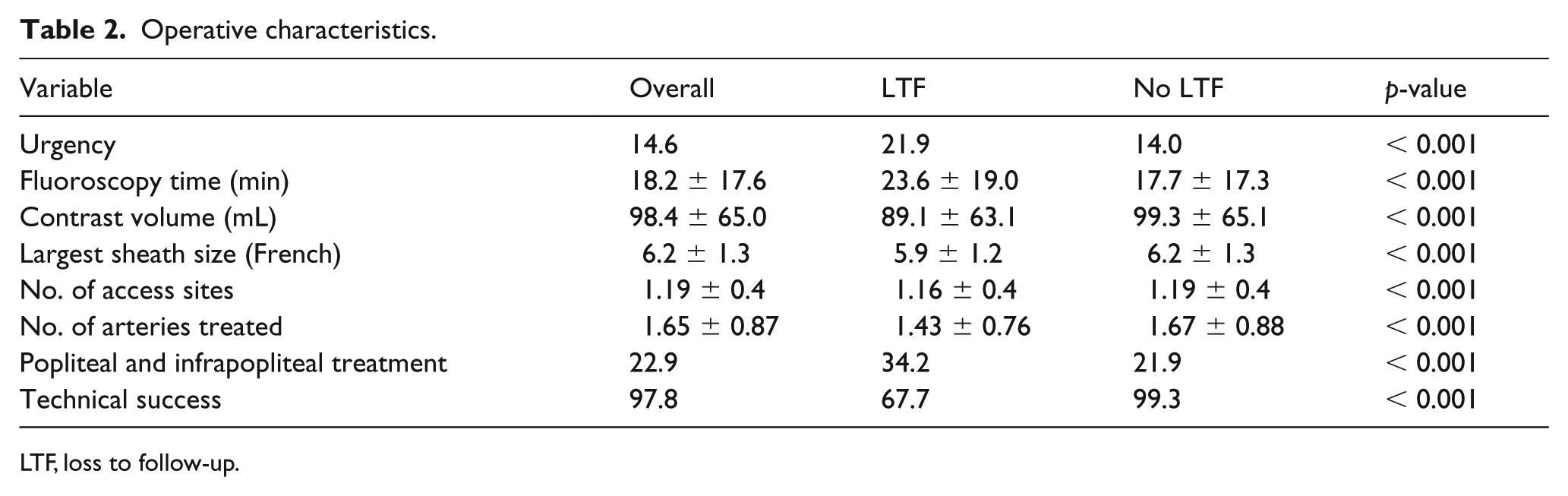
LTF, loss to follow-up.
The postoperative differences between the LTF and no LTF groups are provided in Table 3. Patients lost to follow-up were more likely to have postoperative complications requiring admission (0.09% vs 0.02%, p = 0.014), but less likely to be discharged on aspirin (77.2% vs 81.8%, p < 0.001), P2Y12 antagonist (48.6% vs 68.9%, p < 0.001), or statin (72.8% vs 75.5%, p = 0.001).
Postoperative variables.
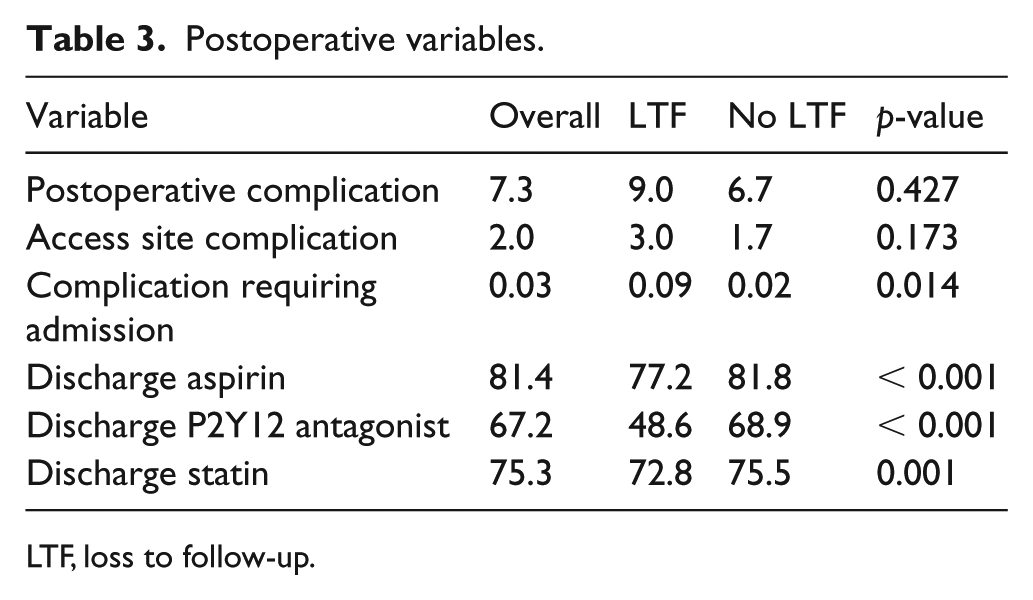
LTF, loss to follow-up.
Overall survival in the entire cohort was 87.6% (90.6% vs 54.2%, no LTF vs LTF, p < 0.001). The independent predictors for LTF after adjusting for center effects are given in Table 4. Age, dialysis dependence, ASA class 3 or higher, fluoroscopy time, number of access sites, infrapopliteal treatment, and complication requiring admission were significantly associated with LTF. Female sex, home preadmission living status, prior CEA/CAS, number of arteries treated, and discharge aspirin were protective against LTF.
Multivariable logistic regression for LTF. *
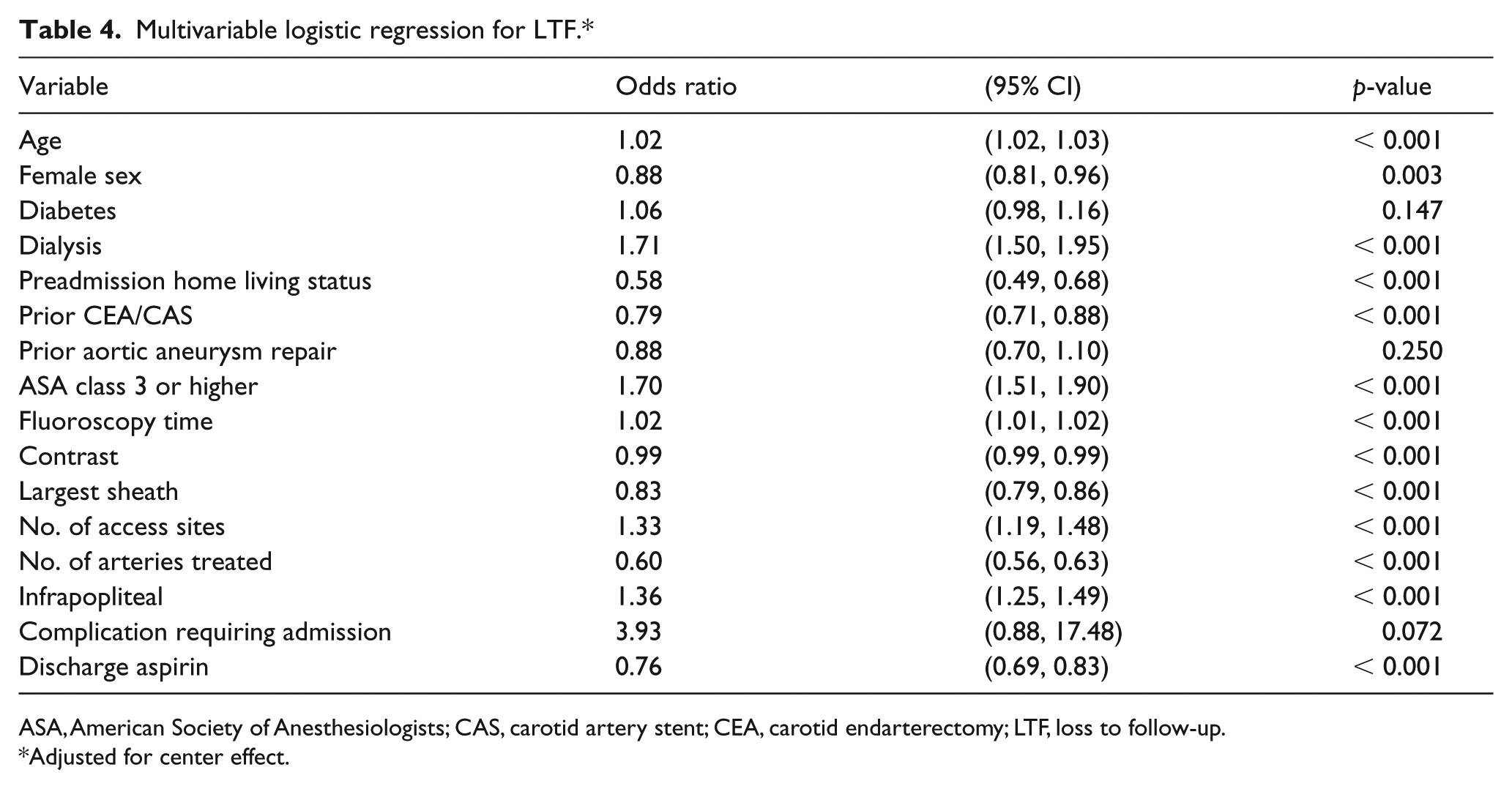
ASA, American Society of Anesthesiologists; CAS, carotid artery stent; CEA, carotid endarterectomy; LTF, loss to follow-up.
Adjusted for center effect.
Cox regression analysis revealed that LTF increased the risk of long-term mortality, even after adjusting for other variables (hazard ratio = 6.56, 95% CI 6.16, 6.99; p < 0.001) (Table 5). Over an interval of 8 years, those patients who were seen in follow-up had a significantly greater survival compared to those lost to follow-up (83.5% vs 43.2%, p < 0.001) (Figure 1). Face-to-face follow-up was associated with greater survival compared to phone follow-up (Figure 2).
Multivariable Cox regression for mortality.
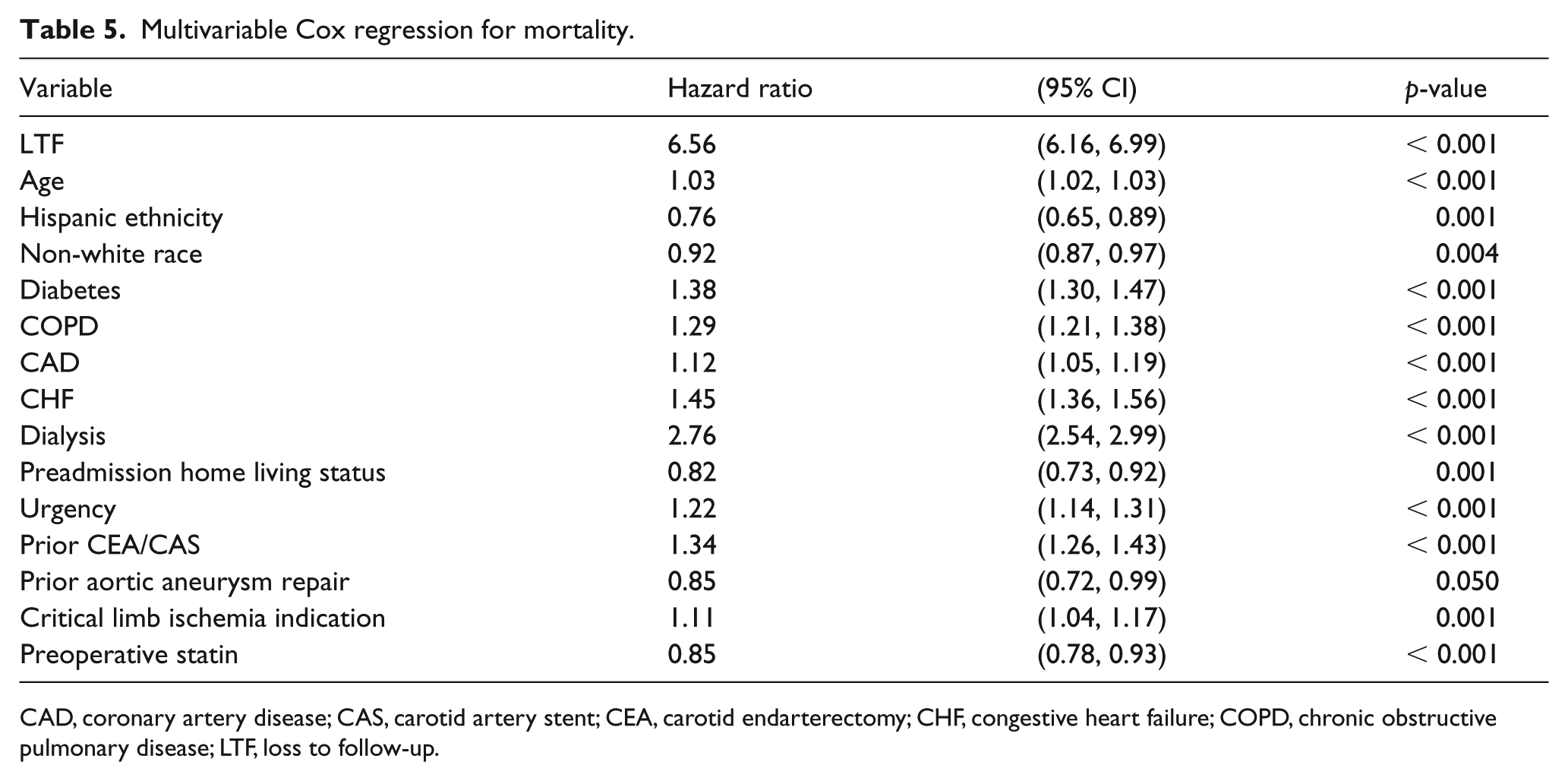
CAD, coronary artery disease; CAS, carotid artery stent; CEA, carotid endarterectomy; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; LTF, loss to follow-up.
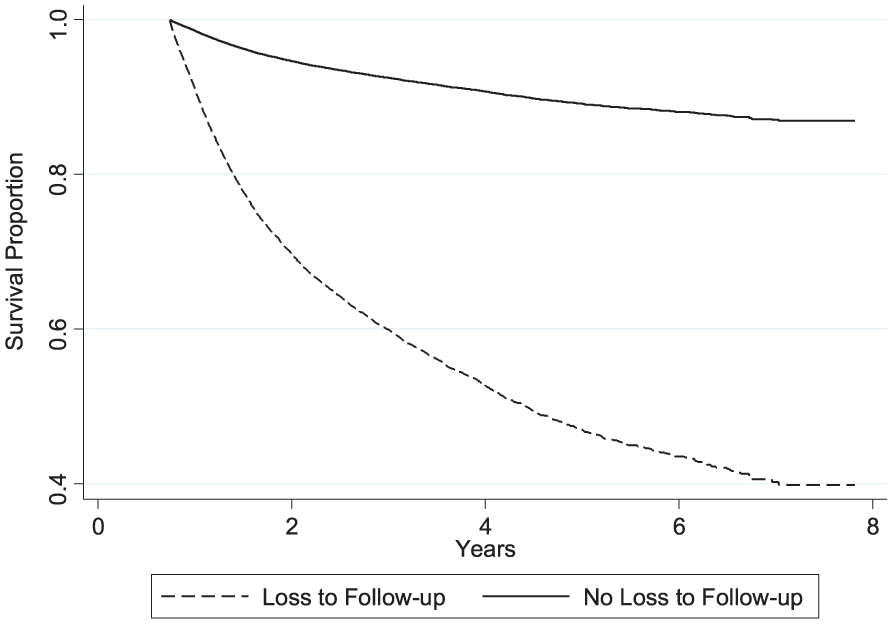
Adjusted survival curves, by follow-up.
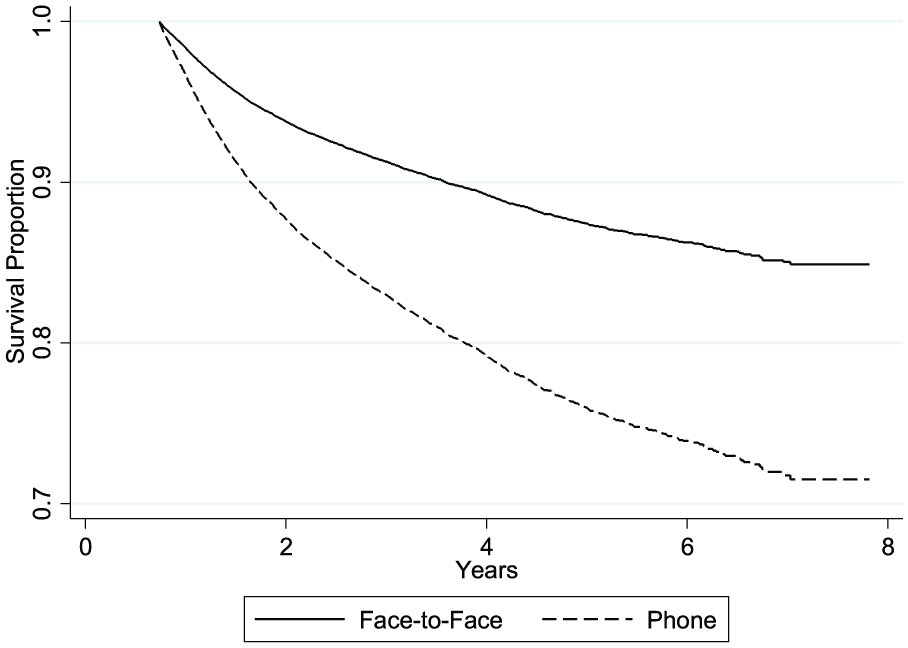
Adjusted survival curves, face-to-face versus phone follow-up.
Discussion
Endovascular interventions have superseded lower extremity bypass in the treatment of PAD. 1 Yet, while the importance of regular follow-up after lower extremity bypass procedures has been well established and accepted,7,8 the utility of follow-up after endovascular interventions is less well known. The purpose of this study was to compare the patient populations undergoing PVI in the VQI who were lost to follow-up and not lost to follow-up, and to determine if follow-up influenced long-term survival. Those who were lost to follow-up in this study had important demographic differences including a higher prevalence of nonwhite race, diabetes, and urgent presentations, perhaps reflective of disparities in access to care. These findings are reminiscent of those from a study utilizing a large California database of patients undergoing PVI, which found that Hispanic and black individuals had a significantly decreased amputation-free survival and a higher reintervention rate compared to non-Hispanic white patients. 9 In another study using the Nationwide Inpatient Sample of patients admitted for PAD, black and Hispanic patients were more likely to undergo amputation, and less likely to undergo revascularization procedures. 10 In another study utilizing the national VQI registry, black patients were found to present with more advanced vascular disease, again underscoring the contributing role of race to lack of access to care. 11 Urgent presentations and a propensity for postoperative complications could be related to less access to preventative care as well. Urgent presentation has also been shown to be associated with increased mortality in a large NSQIP study. 12
Age, diabetes, and dialysis dependence were all associated with an increased risk of LTF. In addition, as expected, home preadmission status was protective against LTF, pointing to a possible adverse effect of frailty on follow-up compliance. 13 Females were less likely to be lost to follow-up, congruent with prior studies showing that males exhibited a higher no-show rate for outpatient clinic appointments. 14 Additionally, prior carotid intervention and discharge aspirin were protective against LTF. Patients may be more inclined to follow up if they already have an established relationship with their vascular specialist. In addition, appropriate discharge with antiplatelets may indicate that high performing centers achieving best medical therapy, are also most successful in having the patients return for a clinic visit. Indeed, Judelson et al. found that center-specific attributes were highly influential in impacting LTF status. 6 The modality of follow-up ultimately was influential on the long-term survival of these patients, with face-to-face follow-up rendering a more protective effect against mortality compared to phone follow-up. Face-to-face follow-up was similarly found to be protective against mortality in a study evaluating the impact of follow-up on EVAR patients. 5 Hicks et al. found that in-person follow-up was associated with a higher rate of follow-up imaging, thus protecting patients against long-term mortality. Detailed discussion about the information gleaned from the arteriogram itself (appearance, multifocality, long-length lesions) could be used as armamentarium with which to counsel the patient to adhere to best medical therapy or other beneficial behaviors, such as exercise therapy. While phone follow-up could serve some of these purposes, it was not as efficacious as in-person follow-up in this particular cohort, similar to findings noted in the EVAR study. LTF, in contrast, was associated with worse survival. Taken together, the results of our study suggest that contact with a provider is important after PVI, and has dramatic effects on survival.
Patients lost to follow-up were less likely to be compliant with a statin preoperatively and were less likely to be discharged on a statin. Further, preoperative statin use was protective against mortality. Thus, our data corroborate data from earlier randomized clinical trials highlighting the protective effect statins have against cardiac morbidity as well as carotid or peripheral vascular disease-related progression. 15 The association of these survival benefits with follow-up can be gleaned from studies highlighting the impact of optimal medical therapy on survival of vascular patients. 16 De Martino et al. demonstrated the beneficial effect of discharge aspirin and statin on 5-year survival in the national VQI dataset. 17 Recent data also elegantly highlighted the differential beneficial effect of high-dose statins on limb salvage and survival in a Veteran’s Affairs PAD cohort. 18 The importance of antiplatelet and statin medications in the PAD population is also highlighted in the most recent 2016 American Heart Association/American College of Cardiology guidelines on the management of patients with lower extremity PAD, where the strength of recommendation was class I-A. 19 Continued smoking cessation also has survival benefits, 20 with the outpatient setting serving as another opportunity to convince the patient of continued behavioral modifications to improve cardiovascular survival, 21 again supported by the AHA/ACC guidelines. 19 These data are congruent with our findings that follow-up care was associated with improved survival in this PAD population.
While the focus of this study was to determine if follow-up had any associated survival benefit for PAD patients, there is growing evidence that PVI follow-up is important to maintain assisted primary patency of the intervention. Indeed, the recent Society for Vascular Surgery published guidelines on follow-up after vascular surgery arterial procedures acknowledged that there was as yet no universal protocol for surveillance studies after endovascular interventions for PAD, and that the quality of the evidence for such recommendations was low. 4 Nonetheless, threshold duplex criteria for diagnosis of post-angioplasty or in-stent stenosis has been published previously8,22–24 with much of the focus on femoropopliteal lesion restenosis. The authors of the guideline went on to recommend that clinical examination, ABI and duplex examination be performed within 1 month of intervention to establish a baseline, with 3–6 month follow-up consisting of clinical examination and ABI, lending support to the notion that close follow-up of patients after endovascular intervention improves assisted primary patency rates. In summary, follow-up of PVI patients derives both patency as well as survival benefit for the patient, and underscores the importance of appropriate scripting and discharge instructions after these procedures. It also highlights the need for vascular interventionists to provide comprehensive care to these patients that extends beyond those required for the procedure.
Study limitations and strengths
We note important limitations of this study. For ease of analysis, and because patients could have undergone multiple interventions in any given year, the first chronologically occurring intervention was considered the index procedure. There was a significant portion of our original sample size that was excluded due to the lack of a follow-up form. Because we do not have reason to suspect that those without follow-up forms differed in a systematic way from those who did have them, we do not feel this impacted the results of our analysis. Patients may have undergone the index procedure and followed up with another physician, and thus could have been misclassified as being lost to follow-up. While this may be possible, these patients did not derive the same survival benefit as those patients that followed up with the provider who performed the intervention, and thus the conclusions of the study remain the same. Because the patient data are deidentified at the national level, it is not possible to link data from other facilities, or to glean if patients followed up at another center. This could have led to an underestimation of the overall follow-up rate, and an overestimation of the effect of LTF on survival. Owing to the large number of patients included, while mean sheath size and number of access sites were statistically significantly different, these numeric differences were small and may not be clinically significant. As the VQI is a data registry, it is subject to the same limitations of any registry where errors with data entry can occur. However, the VQI is a well-vetted registry that undergoes regular review to ensure that variables and help text are continually updated. There may be unmeasured confounders that exist; however, the VQI contains an exhaustive number of variables that are relevant to models of cardiovascular outcomes. While the Social Security Death Index (SSDI) is a commonly used method for ascertainment of death, as of 2014 there is a lag in data entry into the system; thus, patients from this study could be misclassified as being alive. Again, since we have no reason to believe that this occurred differentially for the follow-up versus no follow-up patients, we believe the conclusions remain the same. The dataset is inclusive of patients in the VQI and is thus not necessarily generalizable to all patients undergoing PVI; however, the VQI includes data from 18 regions, across 46 states and Canada, and is representative of current vascular practice.
We also note important strengths of this paper. It is the first study to examine PAD patients and their follow-up patterns post-procedure. This study reaffirms the importance of follow-up care as it has demonstrated survival benefits. The VQI encompasses procedures which are performed across the United States and includes centers in Canada; it spans multispecialty providers including vascular surgeons, cardiologists, and interventional radiologists, ensuring that the data are current and broadly representative of PVIs today.
Conclusions
Patients who follow up after PVI differ markedly from those who are lost to follow-up, and, importantly, exhibited increased survival. The best survival benefit was conferred to those who followed up in person with their operating physician. The findings from this study underscore the importance of dedicated follow-up of this subset of PAD patients. Further study should be dedicated to understanding these differences.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
