Abstract
Pseudoxanthoma elasticum (PXE) is a rare disorder characterized by skin, eye, and cardiovascular lesions due to ectopic mineralization and fragmentation of elastic fibers of connective tissues. We present an atypical case of PXE with diffuse vascular calcification and negligible skin and eye lesions. The patient was a 37-year-old man suffering from severe bilateral arterial calcifications in superficial femoral and posterior tibial arteries. Eye fundoscopy and skin examination were first considered normal. This phenotype suggested first the diagnosis of Arterial Calcification due to Deficiency of CD73 (ACDC) characterized by mutations in NT5E gene. However, we found two variants in ABCC6 gene, and no variant in NT5E. Skin reexamination revealed few lateral skin papules confined to the scalp. Phenotypic overlap was described in vascular calcification disorders, between GACI and PXE phenotypes, and we discuss here expansion of this overlap, including ACDC phenotype. Identification of these expanding and overlapping phenotypes was enabled by genetic screening of the corresponding genes, in a systematic approach. We propose to create a calcification next generation sequencing (NGS) panel with NT5E, GGCX, ENPP1, and ABCC6 genes to improve the molecular diagnosis of vascular calcification.
Introduction
Pseudoxanthoma elasticum (PXE; OMIM 264800) is a rare disorder caused by mutations in ABCC6 gene and characterized by ectopic mineralization of connective tissues. Primary manifestations are skin lesions (yellowish papules in flexural sites and sides of the neck) and ocular signs (retinal peau d’orange and angioid streaks). In our French cohort of 306 cases, peripheral vascular symptoms are present in 42% of cases, including intermittent ischemic claudication with calcified arterial plaques, and more rarely strokes. 1 Arterial calcification disorders are a handful of recessive conditions with various levels of severity, ranging from neonatal heart failure in GACI (Generalized arterial calcification of infancy; OMIM 208000) due to ENPP1 gene defects, 2 extensive and severe lower-limb arterial calcifications in ACDC (Arterial Calcification due to Deficiency of CD73; OMIM 211800) caused by NT5E gene defects, 3 to inconsistent vascular lesions in PXE-like disorder (OMIM 610842) due to GGCX defects and PXE. We present a case with clinical presentation evoking ACDC, with biallelic ABCC6 mutations confirming PXE diagnosis. This case highlights the diagnostic issues in the field of arterial calcification disorders.
Clinical presentation and genetic analyses
In 2012, a 37-year-old man was referred to the Vascular Medicine Department of the Georges Pompidou European Hospital (HEGP, Paris, France), because of intermittent claudication of his left leg after a 200-meter walk, evolving for 3 months. He presented unusual calf pain during professional sports training. Cardiovascular risk factors were absent. He had a personal history of high-level sport (marathon and jumping). He suffered from highly recurrent ureteral colic with calcic renal lithiasis. His mother and mother’s relatives also presented ureteral colic.
Skin examination revealed no skin lesion at first sight. In the left leg, pulses were decreased and systolic pressure index was less than 0.9 (normal range 0.9–1.3). In the right leg, pulses were present and systolic pressure index was higher than 1.2.
Investigations included positron emission tomography–computed tomography (PET CT), CT angiography (CTA) of lower limbs and thoracic aorta, magnetic resonance angiography (MRA) of head and supra aortic arteries, ultrasound of heart, and thoracic, renal and lower limb arteries, Holter electrocardiography, and eye fundoscopy. CTA disclosed extensive arterial calcifications in both superficial femoral and posterior tibial arteries (Figure 1). Stenoses were present in the posterior tibial arteries, at 70% and 60% in the right and left leg, respectively. Cardiac imaging revealed no cardiac calcification, normal valve and non-atheromatous thoracic aorta. Eye fundoscopy was normal. Laboratory analyses revealed normal plasma levels of calcium, phosphorous, creatinine, osteocalcin and parathyroid hormone (PTH). Familial atheroma, paraneoplasic, and infectious aetiologies were excluded.
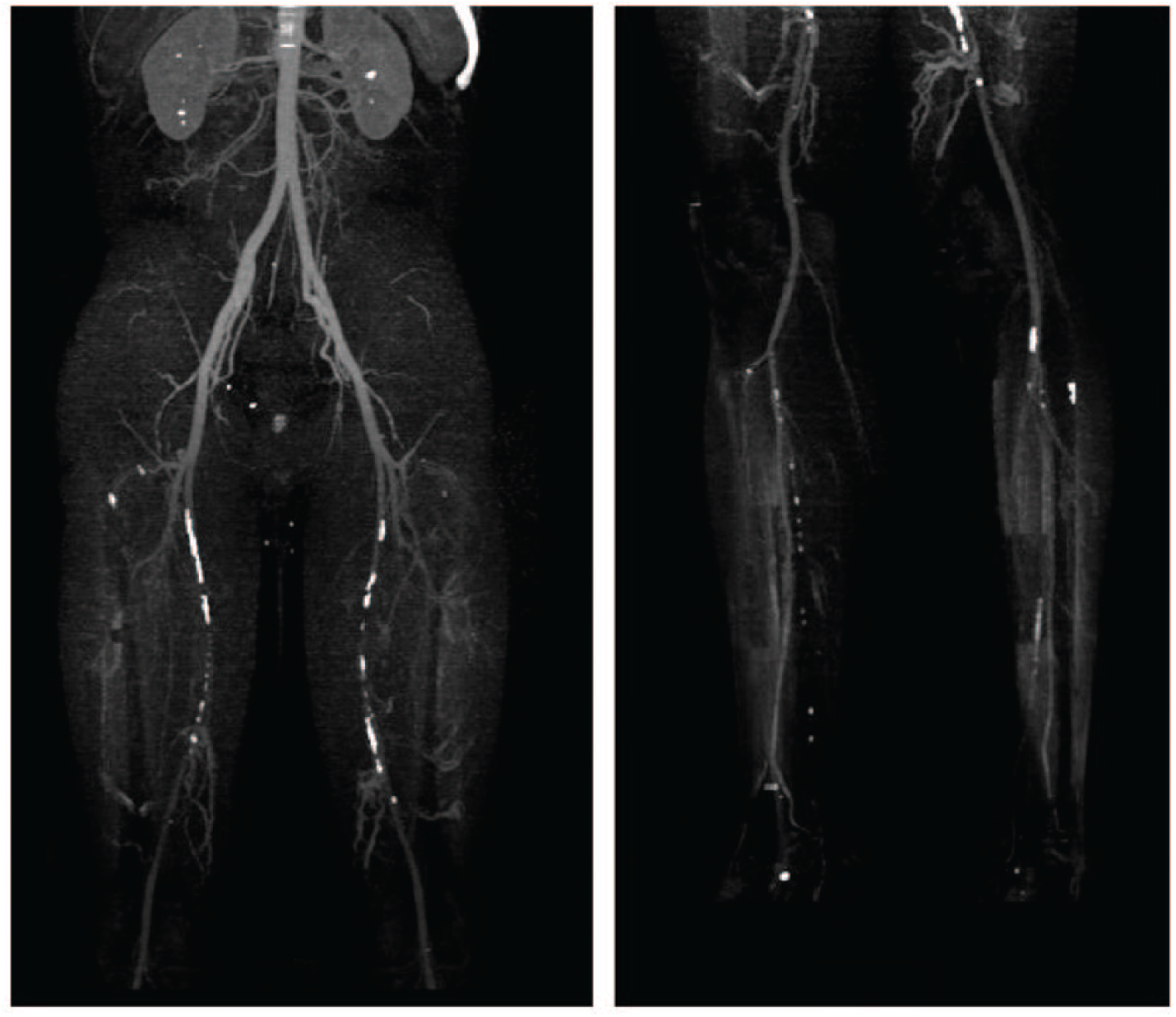
CT angiogram presented arterial calcifications of both superficial femoral arteries (left) and posterior tibial arteries (right).
ACDC was suggested first, because of preeminent obliterating arteriopathy of the lower limbs, which can sometimes be associated with articular calcifications of the hands and feet joints.3–6 After genetic counseling and informed consent in the Genetics Department of the HEGP, NT5E and ABCC6 genes were analyzed by Sanger sequencing, and two heterozygous frameshift variants were found in ABCC6: Ile1000TrpfsTer60 (corresponding to the recurrent deletion of exons 23–29) and Asp1368GlufsTer35 in exon 29, confirming the final diagnosis of PXE (Figure 2).
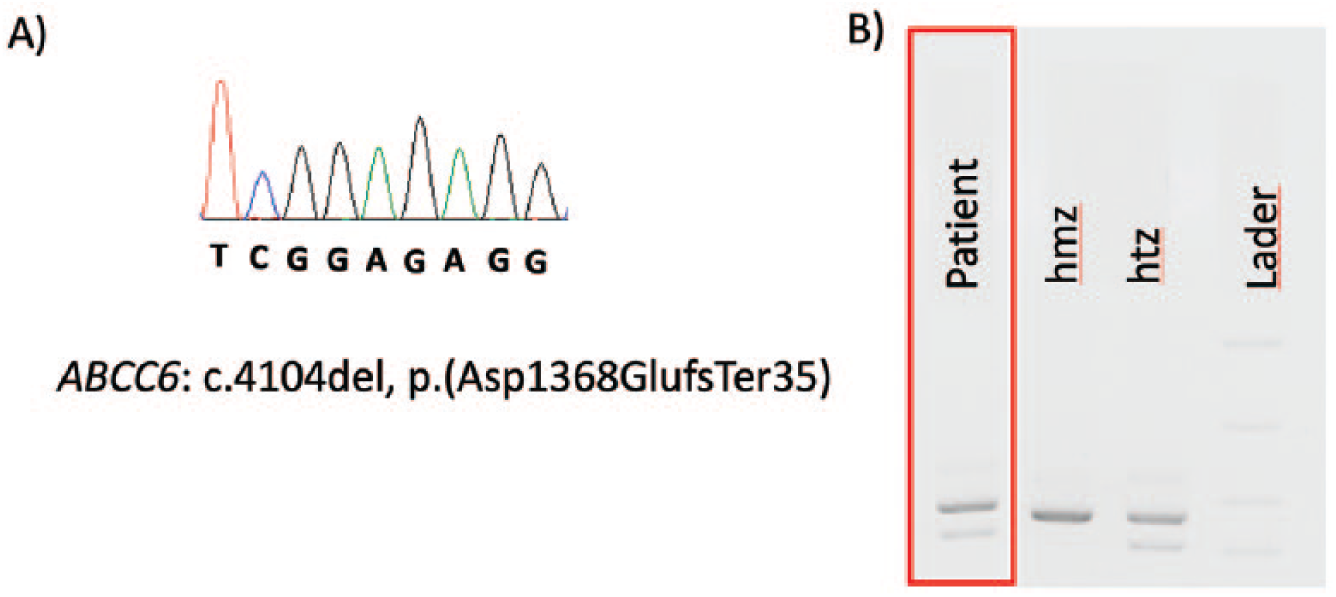
Detection of 2 ABCC6 pathogenic variants in the proband. A. Sanger sequencing showing a pathogenic variant in exon 29: electrophoregram of ABCC6 exon 29 revealing the deletion at the hemizygous state. B. Agarose gel showing the exon 23-29 deletion of ABCC6 gene (NM_001171.5) in the proband, for the patient (surrounded in red) and controls (hmz, homozygous; htz, heterozygous).
Skin reexamination of the patient revealed few fine papules on the left side of the scalp (Figure 3), with otherwise normal flexural sites.
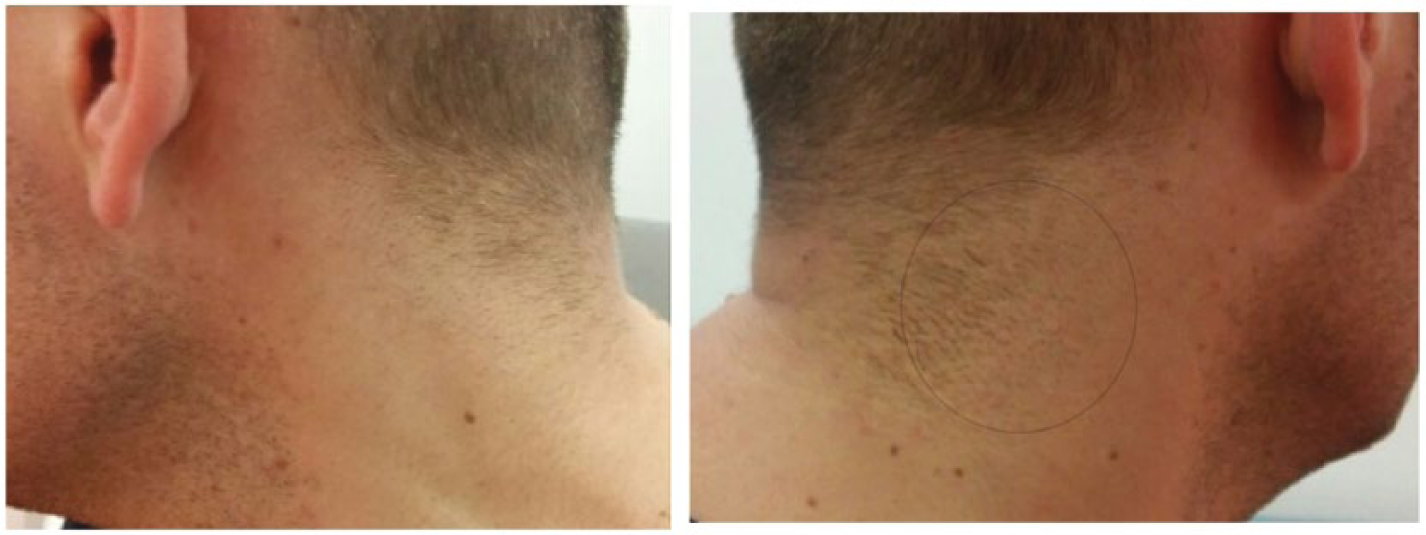
Pictures of both sides of the patient’s neck. Faint papules (circle) are present in the right side, within the lowest part of the scalp.
Discussion
St Hilaire et al. published in 2011 nine cases with ACDC. 3 To date, 13 cases were described with this condition. 4 Patients suffer from intermittent claudication, and irregular articular pain on wrists and ankles. Calcifications affect lower limb arteries, small joints, and in rare cases infrarenal aorta and upper limb arteries. 4 Compared to PXE, skin and eye manifestations are not present and arterial calcifications are severe and extensive.
Conversely, the diagnosis of PXE is mainly based on skin and/or eye lesions, because they are present in about 90% of cases, whereas vascular complications occur in less than half of cases. 1 In our experience, this presentation was never found in PXE: amongst the 306 French cases cohort, no PXE patient with molecularly proven ABCC6 mutations had neither skin lesion nor eye lesion. 1 Thus, in the case described here, because skin lesions were faint and arterial lesions were extensive and selectively located in the lower limbs, we first evoked the diagnosis of ACDC before that of PXE.
Indeed, this case raises the notion of blurring limits between ACDC and PXE phenotypes. In the context of arterial calcification disorders, an overlap between GACI and PXE phenotypes has been described previously. Some cases of GACI were reported without ENPP1 variant but biallelic ABCC6 variants, and some others with biallelic ENPP1 variants and adult-onset typical PXE manifestations.2,7
The association between PXE and nephrolithiasis has been described in few cases. In the French cohort, Legrand et al reported 25 (13%) cases upon 194 cases with nephrolithiasis. This possible association should be considered by clinicians while evaluating a PXE patient.
Arterial calcification disorders are the consequence of an imbalance in the metabolic pathway of inorganic pyrophosphate (PPi), a physiological inhibitor of ectopic tissue mineralization (Figure 4). ABCC6 encodes an ATP-binding cassette protein of unknown precise function although its inactivation reduces plasma levels of PPi. 8 ENPP1 encodes an enzyme that converts ATP to AMP and PPi. The loss of ENPP1 results then in a decrease of AMP, PPi, and adenosine extracellular concentration. NT5E encodes CD73, an enzyme converting AMP into adenosine and inorganic phosphate (Pi). The tissue non-specific alkaline phosphatase (TNAP) degrades PPi. TNAP activity is upregulated in CD73 deficiency, decreasing extracellular adenosine and PPi levels, and provoking premature arterial ectopic calcification 9 . All these pathologies characterized by deregulation of the homeostasis of PPi present arterial calcifications but are considered to lead to specific symptoms, as previously depicted.
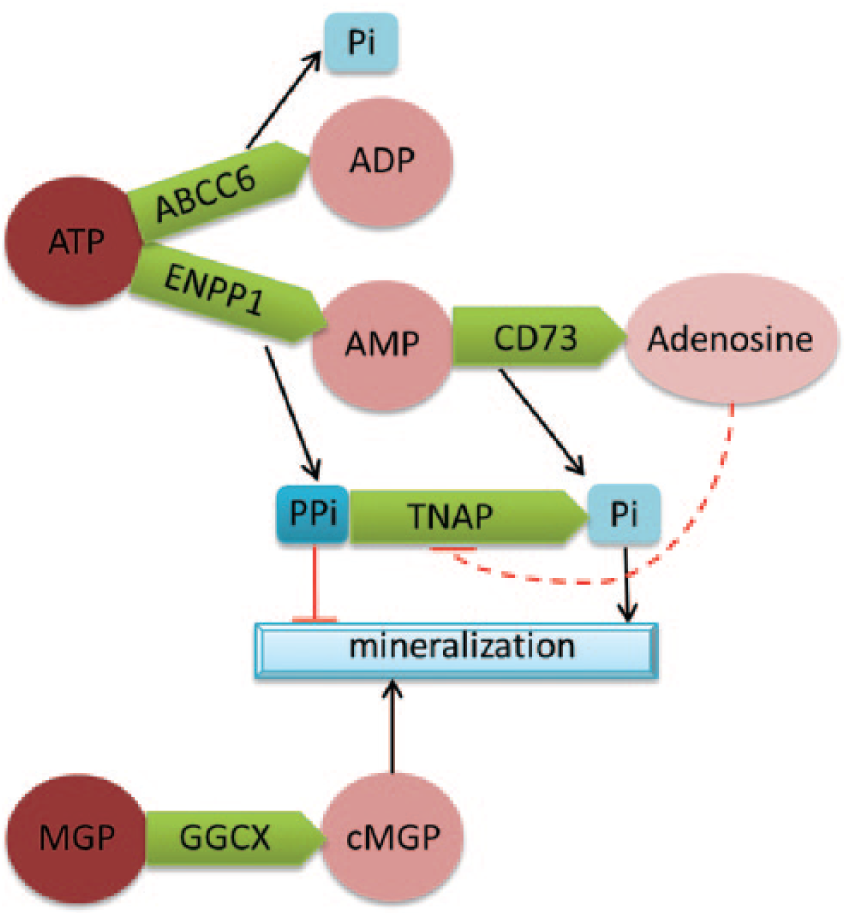
Diagram of the PPi pathway and the corresponding enzymes and related arterial calcification disorders. ATP, Adenosine tri phosphate. ADP, Adenosine di phosphate. AMP, Adenosine mono phosphate. MGP, Matrix gla protein. cMGP, carboxyl matrix gla protein. Pi, inorganic phsophate. PPi, inorganic pyrophosphate. TNAP, tissue non-specific alkaline phosphatase.
Conclusion
Increasing knowledge about overlapping phenotypes should lead the medical community to consider these disorders as an entity of ectopic calcification syndromes. We propose to develop a calcification next generation sequencing (NGS) panel including NT5E, GGCX, ENPP1, and ABCC6 genes to test patients with calcified vascular lesions that would be more specific than larger panels such as the connective tissue NGS panel proposed by the National Center for Biotechnology Information (NCBI) 10 and that would be more appropriate for patients referred to vascular medicine centers.
Footnotes
Acknowledgements
We thank the patient for his kind collaboration.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Anne Legrand is a recipient of a PhD fellowship from the Region Ile de France and has also been supported by the ‘DHU PARC – Pathologies Arterielles Communes’ funded by Assistance Publique – Hôpitaux de Paris.
