Abstract

Keywords
The clinical significance of external carotid artery (ECA) plaques remains unclear, whereas the risks of asymptomatic internal carotid artery (ICA) stenosis are well established. 1 Recently, Kim et al. found that isolated ECA plaques are an independent predictor of all-cause mortality. 2 Presuming that ECA plaques are associated with an increased cardiovascular death rate, which is mainly caused by coronary artery disease (CAD) and stroke, 3 and the latter is associated with the presence of intracranial stenosis (IS),4,5 we hypothesized that ECA plaques and IS should be related. Accordingly, we aimed to examine whether ECA plaques, diagnosed by extracranial carotid duplex ultrasound (CDU), are associated with asymptomatic IS in patients with advanced CAD.
The study involved 389 patients with three vessels/left stem CAD, which was confirmed using angiography in our hospital from March 2010 to December 2014. Subjects with atrial fibrillation/flutter, heart valve disease, a myocardial infarction (MI) within the last 6 weeks, ventricular aneurysm, neurological focal symptoms, or decompensated chronic diseases were excluded. All patients underwent a detailed cardiovascular risk and physical assessment and vascular examination by CDU as well as transcranial colour-coded duplex sonography (TCCS). The study was approved by the Regional Biomedical Research Ethics Committee of Vilnius, Lithuania. Informed consent was obtained from each patient.
All the sonographic examinations were performed by a single experienced investigator (JV) using a standard ultrasound system (GE Healthcare, Tokyo, Japan). CDU was performed to evaluate the atherosclerotic lesions of the common carotid artery (CCA), ICA and ECA. The plaque was defined as having a local intima-media thickness (IMT) > 1.5 mm or > 50% of the surrounding IMT value. 6 The obstructive ICA lesions were classified in accordance with the criteria of the Society of Radiologists in Ultrasound, based on the North American Symptomatic Carotid Endarterectomy Trial (NASCET). 7 After a careful review of the presence/absence of plaque in the longitudinal and transversal B-mode views of the proximal ECA, all the ECA were divided into two categories (with/without plaque). IS of the internal carotid (C1 and C5 segments), middle cerebral, anterior cerebral, posterior cerebral, vertebral, and basilar artery was diagnosed by standard TCCS examination, which followed the protocol and criteria established in previous studies.8,9
The data analysis was conducted using the Statistical Package for the Social Sciences (Version 17; SPSS Inc., Chicago, IL, USA) and Statistica (Version 10 for Windows; StatSoft, Inc., Tulsa, OK, USA). The logistic regression analysis was performed and the models were evaluated by the Hosmer–Lemeshow goodness-of-fit test and the area under the receiver operator curve (ROC). The differences among the areas under the curve (AUC) were tested with a non-parametric test using a StAR (statistical analysis of ROC curves) online tool. 10 The significance level was set at p < 0.05.
The mean age of the entire study cohort (27% female, 44% smokers and 3% patients with a prior history of stroke/TIA) was 66.7 ± 9.1 (range 39–88) years. Patients with ECA plaques were significantly older (68.0 ± 9 vs 64.4 ± 9.1 years, p < 0.001) but had a lower frequency of dyslipidaemia (88.1% vs 95.9%, p = 0.01). The distribution of the other risk factors was without significant difference.
Overall, ECA plaques were found in 244 (62.7%) patients, ICA and/or CCA plaques in 385 (98.9%) patients. At least one high grade ICA stenosis (⩾ 70%) was diagnosed in 39/389 (10%) patients. Among the patients with a sufficient acoustic window for TCCS (n = 346), IS was diagnosed in over three-quarters (n = 187, 83.1%) of patients with ECA plaques (n = 225).
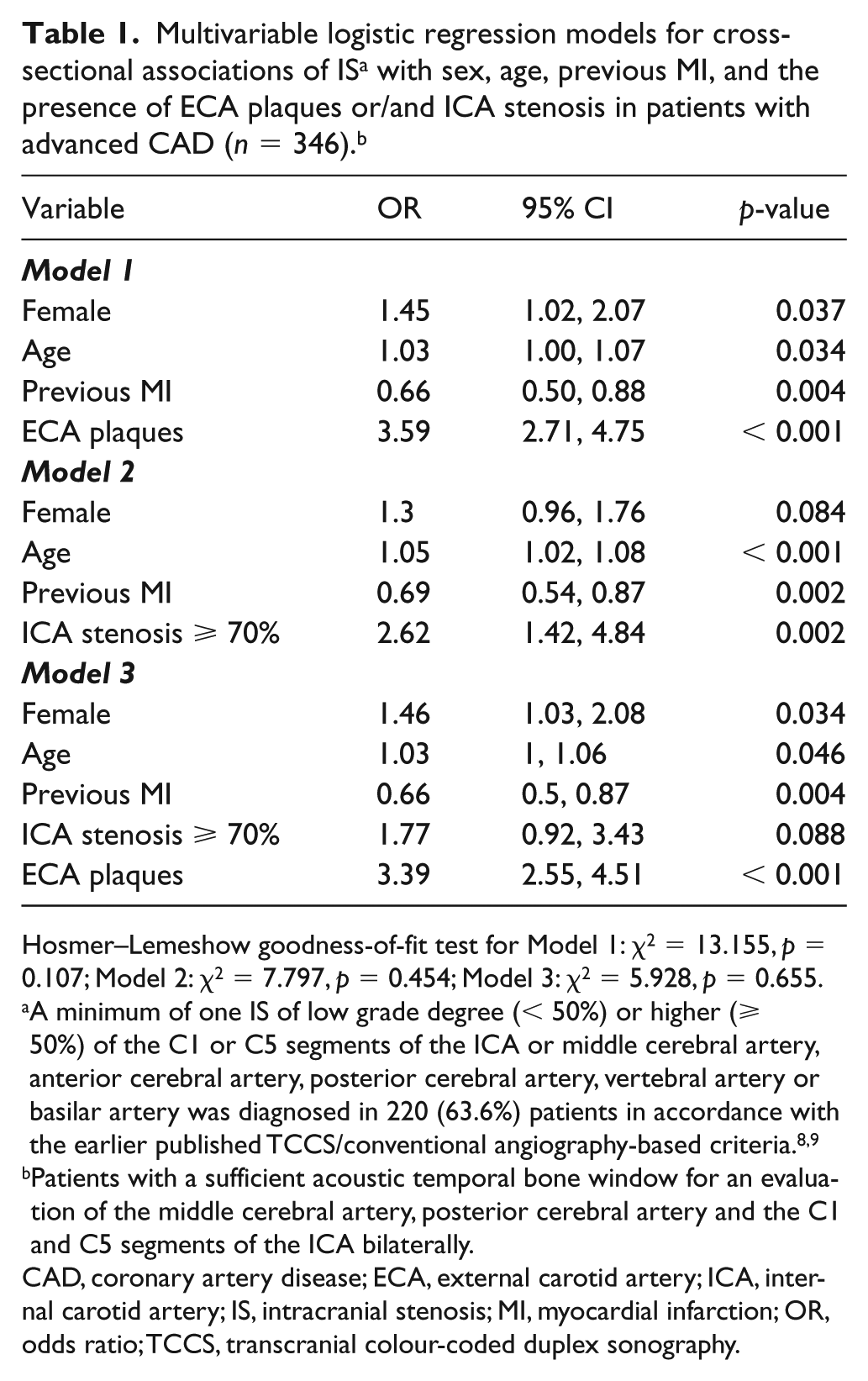
Three multivariable logistic regression models were constructed to examine which factors are associated with IS. Sex, age, previous MI, and one of the following were included as independent variables: the presence of ECA plaques (Model 1), ICA stenosis ⩾ 70% (Model 2) or both (Model 3). All the models demonstrated adequate calibration (Table 1) and good discrimination. Model 1 yielded 84% sensitivity and 73% specificity (AUC 0.84, 95% CI 0.79–0.88, p < 0.001); Model 2, 73% sensitivity and 61% specificity (AUC 0.71, 95% CI 0.66–0.77, p < 0.001); and Model 3, 83% sensitivity and 75% specificity (AUC 0.84, 95% CI 0.80–0.89, p < 0.001). Models 1 and 3 performed significantly better than Model 2, but Model 3 did not perform significantly better than Model 1.
Hosmer–Lemeshow goodness-of-fit test for Model 1: χ2 = 13.155, p = 0.107; Model 2: χ2 = 7.797, p = 0.454; Model 3: χ2 = 5.928, p = 0.655.
A minimum of one IS of low grade degree (< 50%) or higher (⩾ 50%) of the C1 or C5 segments of the ICA or middle cerebral artery, anterior cerebral artery, posterior cerebral artery, vertebral artery or basilar artery was diagnosed in 220 (63.6%) patients in accordance with the earlier published TCCS/conventional angiography-based criteria.8,9
Patients with a sufficient acoustic temporal bone window for an evaluation of the middle cerebral artery, posterior cerebral artery and the C1 and C5 segments of the ICA bilaterally.
CAD, coronary artery disease; ECA, external carotid artery; ICA, internal carotid artery; IS, intracranial stenosis; MI, myocardial infarction; OR, odds ratio; TCCS, transcranial colour-coded duplex sonography.
In Model 1, the presence of ECA plaques was associated with 3.6-fold greater odds of IS (95% CI 2.71–4.75, p < 0.001). While in Model 2, ICA stenosis ⩾ 70% was also associated with greater odds of IS (OR 2.62, 95% CI 1.42–4.84, p = 0.002), Model 3, which combined both vascular variables, demonstrated that this association does not remain significant if the presence of ECA plaques is accounted for. Model 3 revealed that the presence of ECA plaques, but not ICA stenosis, was associated with the presence of IS.
To the best of our knowledge, the relationship between ECA plaques and IS has not yet been studied. We demonstrated that, in advanced CAD patients, the presence of ECA plaques is associated with an over threefold higher IS prevalence. The detection of ECA plaques may suggest considering further intracranial vessel diagnostic tests, where the detection of IS may modify preventive strategies and treatment decisions. However, longitudinal studies are still needed in order to translate this finding into a clinical policy.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.