Abstract
Information on the association between arterial stiffness and cerebral small vessel disease (cSVD) is limited and confined to white and Asian populations. More regional information is needed to confirm this association in different ethnic groups. Using the Atahualpa Project cohort, we aimed to assess whether the aortic pulse wave velocity (PWV) is associated with the total cSVD score, as well as with each of the neuroimaging signatures of cSVD, in a population of Amerindians living in rural Ecuador. Atahualpa residents aged ⩾ 60 years were offered a brain magnetic resonance imaging scan (MRI) and aortic PWV determination. An ordinal logistic regression model, adjusted for demographics and cardiovascular risk factors, was constructed to predict the total cSVD score by levels of aortic PWV. The association between the neuroimaging signatures of cSVD and the aortic PWV was assessed by adjusted logistic regression models. Of 437 candidates, 303 (69%) underwent a brain MRI and aortic PWV determinations. The total cSVD score was 0 points in 65% of individuals, 1 point in 18%, 2 points in 11%, and 3–4 points in 6%. The mean aortic PWV was 10.4 ± 1.8 m/s, which increased from 9.8 ± 1.2 to 12.3 ± 1.8 m/s in individuals with a cSVD score of 0 and 3–4, respectively (p < 0.001). An ordinal logistic regression model showed significant association between the PWV and the cSVD score. A change of one unit of the aortic PWV increased the odds of having a higher total cSVD score by 1.73 (95% CI: 1.28–2.35; p < 0.001). In addition, individual neuroimaging signatures of cSVD, with the exception of lacunar infarcts, were associated with the aortic PWV. This study shows a significant association between the aortic PWV and total cSVD score and most of its individual components in older Amerindians.
Keywords
Introduction
Owing to increased life expectancy and changes in lifestyles, the burden of cerebrovascular diseases is on the rise in Latin American countries, not only in urban centers but also in remote rural settings.1,2 Epidemiological data suggest that cerebral small vessel disease (cSVD) is one of the most common pathogenetic mechanisms underlying stroke (ischemic and hemorrhagic) in the region. 3 However, information from rural areas is limited because of the paucity of population-based studies using MRI and other sophisticated diagnostic tools, which would allow the recognition of silent neuroimaging signatures of cSVD and mechanisms implicated in their pathogenesis.4–6 More regional information is needed for the implementation of intervention strategies in apparently healthy individuals at risk, before catastrophic events occur.
Arterial stiffness is an important determinant of systemic and intracranial atherosclerosis,7,8 and has also been associated with cSVD in several,9–13 but not all studies. 14 Some studies have been confined to stroke patients, or have only assessed the association between arterial stiffness and isolated neuroimaging signatures of cSVD. In addition, this association has been assessed in white and Asian populations, but there is no information from other races/ethnic groups. Using the Atahualpa Project cohort, 15 we aimed to assess whether the aortic pulse wave velocity (PWV), used as a surrogate of arterial stiffness, was associated with each of the neuroimaging signatures of cSVD as well as with the total cSVD score. The latter was developed to address the combined effect of these neuroimaging signatures, as a better predictor of the cSVD burden in a given subject.16,17 The present study was conducted in community-dwelling older adults living in rural Ecuador.
Methods
Study population
Atahualpa is a rural Ecuadorian village that achieves several requisites to be considered an optimal setting for the practice of epidemiological studies. 15 Inhabitants do not migrate and are homogeneous regarding race/ethnicity (Amerindians), living conditions, and dietary habits; almost all men belong to the blue-collar class and most women are homemakers. These consistencies reduce the risk of unexpected confounders at the time of analyses.
Study design
Atahualpa residents aged ⩾ 60 years identified during door-to-door surveys were offered a brain magnetic resonance imaging scan (MRI) and aortic PWV determination, and those who signed the informed consent form and had no contraindications for the practice of these exams were enrolled. Using a population-based cross-sectional design, we assessed the association between the aortic PWV and the total cSVD score and each of its components (as the dependent variables), after adjusting for relevant confounders (see below). The institutional review board of Hospital-Clínica Kennedy, Guayaquil, Ecuador (FWA 00006867) approved the protocol and the informed consent.
Neuroimaging protocol
MRIs were performed with a Philips Intera 1.5T (Philips Medical Systems, The Netherlands). As previously described, MRIs were read by a neurologist and a neuroradiologist blinded to clinical information; kappa coefficients for interrater agreement were high for the lesions of interest (0.90 for white matter hyperintensities (WMH), 0.76 for deep cerebral microbleeds (CMB), 0.90 for lacunar infarctions (LI), and 0.83 for the presence of > 10 enlarged basal ganglia perivascular spaces (PVS)), and discrepancies were resolved by consensus. 18 Exams were reviewed following research standards for cSVD proposed by Wardlaw et al. 19 In particular, WMH of presumed vascular origin were defined as lesions appearing hyperintense on T2-weighted images that remained bright on fluid attenuated inversion recovery (without cavitation) and graded according to the modified Fazekas scale, 20 CMB were identified and rated according to the microbleed anatomical rating scale, 21 LI were defined as fluid-filled cavities measuring 3–15 mm located in the territory of a perforating arteriole, 19 and enlarged PVS were defined as small (< 3 mm) structures of cerebrospinal fluid (CSF) intensity located in the basal ganglia that followed the orientation of perforating arteries. 22
The total SVD score was calculated according to previous descriptions.16,17 In brief, each neuroimaging signature of cSVD was given 1 point if present, with a minimum score of 0 and a maximum score of 4. Points were assigned to WMH if they were graded as moderate-to-severe, to CMB and LI (respectively) if there was at least one lesion located deep in the brain (including the subcortical white matter, basal ganglia, thalamus or brainstem), and to enlarged basal ganglia PVS if there were > 10 of these lesions (Figure 1).
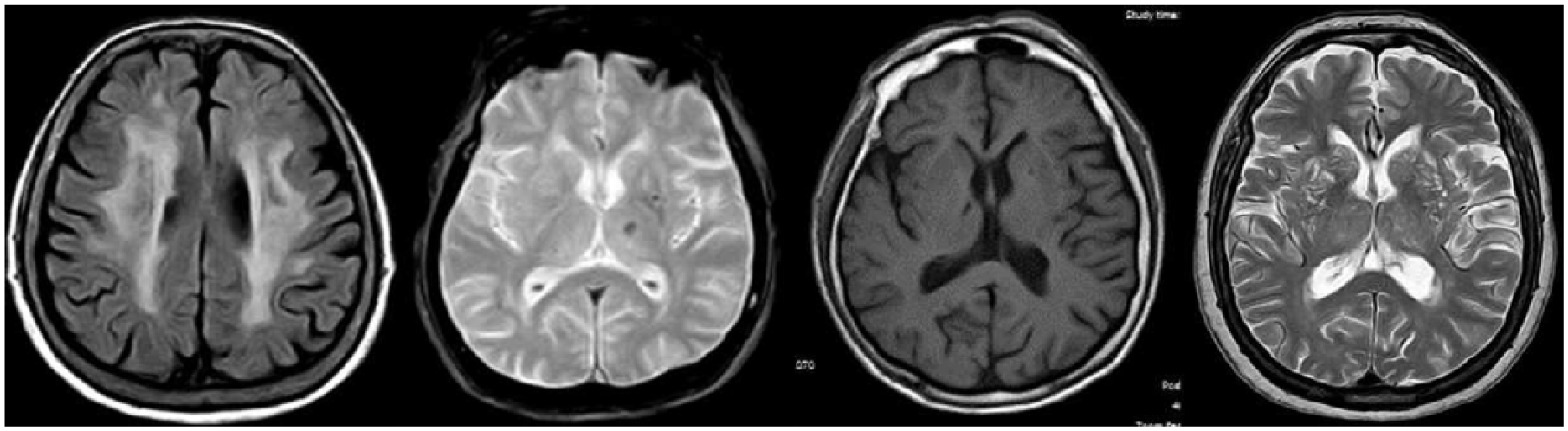
Neuroimaging signatures of cerebral small vessel disease. Left to right: white matter hyperintensities of presumed vascular origin (fluid-attenuated inversion recovery: TR 9000, TE 120, TI 2500); deep cerebral microbleeds (gradient-echo: TR 686, TE 23); lacunar infarct in basal ganglia (T1-weighted: TR 594, TE 15); and > 10 enlarged basal ganglia periventricular spaces (T2-weighted: TR 4500, TE 100).
Arterial stiffness evaluation
Exams were performed with individuals resting in the supine position at comfortable room temperature levels. Participants were instructed to avoid caffeine-containing products, nicotine, and alcohol for 24 hours before the test. Arterial stiffness was evaluated by the Mobil-O-Graph NG (IEM, Stolberg, Germany) device. This device estimates the aortic PWV based on the oscillometric detection of the brachial pressure wave with a single cuff. The Mobil-O-Graph NG does not need operator expertise and has demonstrated good repeatability for PWV assessment and a higher repeatability than devices measuring carotid-femoral PWV in elderly high-risk populations. 23
Clinical covariates investigated
Demographics and cardiovascular risk factors were selected as potential confounding variables. These confounders were assessed through interviews and procedures previously described in the Atahualpa Project. In brief, we used the American Heart Association (AHA) criteria to assess smoking status, physical activity, diet, the body mass index, blood pressure, fasting glucose, and total cholesterol blood levels. 24 The AHA stratified each of these risk factors (or cardiovascular health metrics) in ideal, intermediate or poor, according to well-defined cut-offs, including: (1) smoking status: ideal (never or quit > 1 year), intermediate (quit ⩽ 1 year), and poor (current smoker); (2) body mass index: ideal (< 25 kg/m2), intermediate (25 to < 30 kg/m2), and poor (⩾ 30 kg/m2); (3) physical activity: ideal (⩾ 150 minutes/week moderate intensity or ⩾ 75 minutes/week vigorous intensity or equivalent combination), intermediate (1–149 minutes/week moderate intensity or 1–74 minutes/week vigorous intensity or equivalent combination), and poor (no moderate or vigorous activity); (4) diet: ideal (four to five healthy components), intermediate (two to three healthy components), and poor (none or one healthy component), based on five health dietary components (⩾ 4.5 cups fruits and vegetables/day, ⩾ two 3.5-oz servings fish/week, ⩾ three 1-oz equivalent servings fiber-rich whole grains/day, < 1500 mg sodium/day, and ⩽ 450 kcal sugar-sweetened beverages/week); (5) total cholesterol: ideal (untreated and < 200 mg/dL), intermediate (treated to < 200 mg/dL or 200–239 mg/dL), and poor (⩾ 240 mg/dL); (6) blood pressure: ideal (untreated and < 120/< 80 mmHg), intermediate (treated to < 120/< 80 mmHg or 120–139/80–89 mmHg), and poor (⩾ 140/90 mmHg); and (7) fasting glucose: ideal (untreated and < 100 mg/dL), intermediate (treated to < 100 mg/dL or 100–125 mg/dL), and poor (⩾ 126 mg/dL). For this study, each metric in the poor range was considered a cardiovascular risk factor.
Statistical analysis
Data analyses were carried out using Stata Statistical Software: Release 15 (StataCorp LLC, College Station, TX, USA). In univariate analyses, continuous variables were compared by linear models and categorical variables by the chi-squared or Fisher’s exact tests as appropriate. In order to predict the total cSVD score (as an ordinal variable) by levels of the aortic PWV, we first constructed an ordinal logistic regression model adjusted for demographics and cardiovascular risk factors. In a second step, the association between the presence of each of the neuroimaging signatures of cSVD (as the dependent variables) by levels of the aortic PWV was assessed by fitting logistic regression models adjusted for the same confounders.
Results
Of 437 community-dwelling Atahualpa residents aged ⩾ 60 years identified during door-to-door surveys, 303 (69%) underwent brain MRI and adequate aortic PWV measurements. Reasons for not obtaining these studies included refusal to consent (n = 68), severe disability (n = 15), contraindications for the practice of MRI (n = 9), and an impossibility to perform aortic PWV measurements due to poor collaboration (n = 29); 13 additional persons had died or emigrated between the survey and the invitation.
The mean age of the 303 participants was 70.3 ± 7.8 years (median age: 69 years; age range: 60–95 years), 178 (59%) were women, and 233 (77%) had primary school education only. A body mass index ⩾ 30 kg/m2 was noticed in 69 (23%) persons, blood pressure ⩾ 140/90 mmHg in 132 (44%), fasting glucose ⩾ 126 mg/dL in 91 (30%), and total cholesterol levels ⩾ 240 mg/dL in 40 (13%). Six (2%) participants were current smokers, 14 (5%) had a poor diet, and 22 (7%) had poor physical activity. The mean value of the aortic PWV was 10.4 ± 1.8 m/s (median aortic PWV: 9.9 m/s). Moderate-to-severe WMH were noticed in 60 individuals (20%), deep CMB in 22 (7%), LI in 29 (10%), and > 10 PVS in 74 (24%). The total cSVD score was 0 points in 196 individuals (65%), 1 point in 55 (18%), 2 points in 34 (11%), 3 points in 10 (3%), and 4 points in eight (3%).
Characteristics of participants across the categories of the total cSVD score are summarized in Table 1. It was noted that univariate analyses showed individuals with higher total cSVD scores were older (p < 0.001), had worse physical activity (p = 0.005), and more often had blood pressure levels ⩾ 140/90 mmHg (p = 0.001). The aortic PWV increased together with the total cSVD score (p < 0.001). Also, in univariate analyses, mean (± SD) values of the aortic PWV (m/s) were significantly higher among persons with moderate-to-severe WMH when compared with those with none-to-mild WMH (12 ± 2.2 vs 10 ± 1.4; p < 0.001), with deep CMB when compared with those without CMB (11.4 ± 2.1 vs 10.3 ± 1.8; p = 0.007), with LI when compared with those without LI (11.6 ± 1.8 vs 10.3 ± 1.8; p < 0.001), and with ⩽ 10 enlarged basal ganglia PVS when compared with those with > 10 enlarged PVS (11.8 ± 2.1 vs 9.9 ± 1.5; p < 0.001).
Characteristics of Atahualpa residents aged ⩾ 60 years across categories of the total cSVD score (univariate analyses).
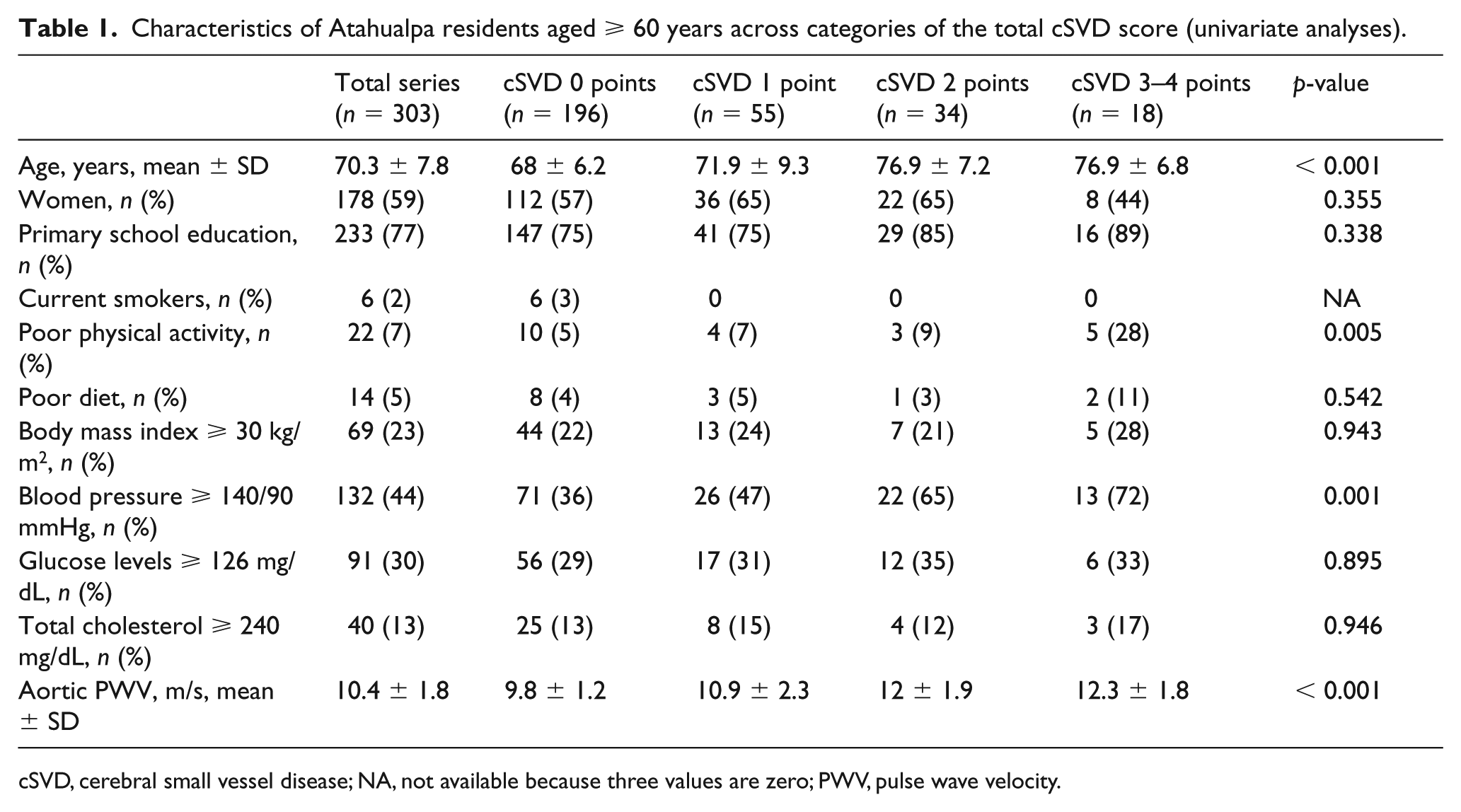
cSVD, cerebral small vessel disease; NA, not available because three values are zero; PWV, pulse wave velocity.
An ordinal logistic regression model, fitted to predict the total cSVD score by levels of the aortic PWV, showed a significant association between severity of the cSVD score and the aortic PWV, after adjusting for demographics and cardiovascular risk factors. In this model, a change of one unit (m/s) of the aortic PWV significantly increased the odds of having a higher total cSVD score by 1.73 (95% CI: 1.28–2.35; p < 0.001). None of the confounders remained independently significant in this multivariate model (Table 2).
Ordinal logistic regression model adjusted for demographics and cardiovascular risk factors, showing the independent association between the aortic PWV and the total cSVD score. a
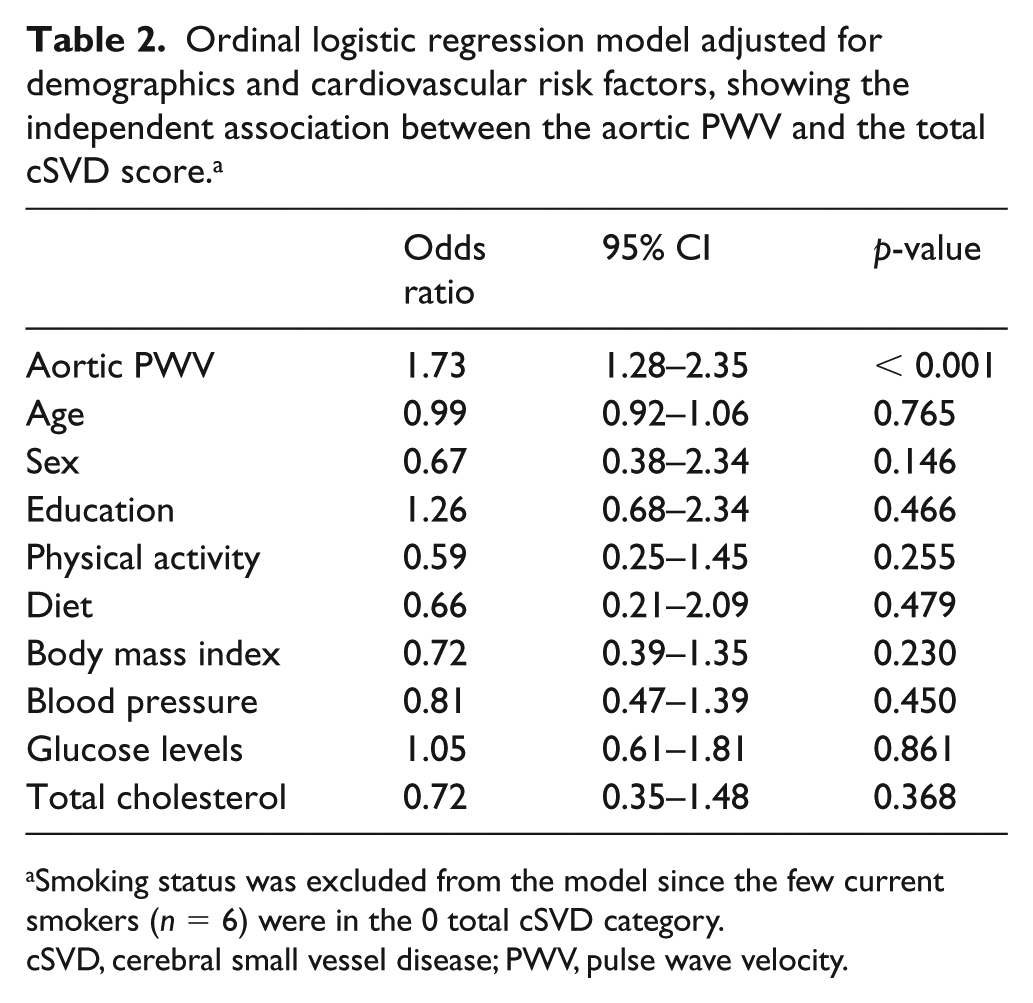
Smoking status was excluded from the model since the few current smokers (n = 6) were in the 0 total cSVD category.
cSVD, cerebral small vessel disease; PWV, pulse wave velocity.
Next, logistic regression models were fitted to predict the presence of each of the neuroimaging signatures of the cSVD score by levels of the aortic PWV. The models showed a significant association between the aortic PWV and moderate-to-severe WMH (odds ratio (OR): 0.59; 95% CI: 0.41–0.87; p = 0.007), deep CMB (OR: 0.57; 95% CI: 0.34–0.96; p = 0.034), and > 10 enlarged basal ganglia PVS (OR: 0.56; 95% CI: 0.39–0.80; p = 0.002), after adjusting for demographics and cardiovascular risk factors. In contrast, the independent association between the aortic PWV and LI was not significant (OR: 0.72; 95% CI: 0.46–1.11; p = 0.133).
Discussion
In this study, involving older adults living in a rural Ecuadorian village, 35% have at least one neuroimaging signature of cSVD, a burden that provides support to the concept that this condition is frequent in the region. The high prevalence makes this population suitable for studying the effect of aortic PWV on the total cSVD score, as well as on the different neuroimaging signatures of cSVD.
In the present study, the total cSVD score, as well as most of the individual neuroimaging signatures of cSVD, were associated with the aortic PWV, after adjustment for relevant confounders. The only exception was the lack of association with LI. This could be related to the fact that LI may not be related to cSVD in a sizable proportion of cases, but to different pathogenetic mechanisms, such as cardiogenic brain embolism, or to artery-to-artery emboli originating in plaques located in the carotid arteries, reinforcing the old concept of the ‘fallacy of the lacunar hypothesis’. 25 Such discrepancies on risk factors associated with LI and the other neuroimaging signatures of cSVD have been previously reported in the population of Atahualpa.26,27
Literature on the relationship between arterial stiffness and the total cSVD score is limited, and most available studies have not uniformly compared the role of all the independent neuroimaging signatures of cSVD at the population level. In 1460 individuals enrolled in the Rotterdam Scan Study, 9 the aortic PWV correlated with severity of WMH but not with the presence of LI. In that study, only the subset of individuals with uncontrolled hypertension showed a significant association between the aortic PWV and deep CMB, and a borderline association between the aortic PWV and LI.
In a study conducted in 782 hypertensive individuals from Spain, the association between the carotid-femoral PWV and the total cSVD score was investigated. 10 In this study, the total cSVD score and the PWV were significantly associated. Of the individual neuroimaging signatures of cSVD, a significant association was found between the PWV and deep LI and basal ganglia enlarged PVS, but the association with WMH was borderline and there was no association with deep CMB. Therefore, the results of the Spanish study are similar to the present study in the association between PWV and the total cSVD score, but differed in the association with individual neuroimaging signatures of cSVD.
In a Korean study of 255 patients with stroke or TIAs – where 22% of the population had CMB – the association between the brachial–ankle PWV and CMB (irrespective of whether they were located deep in the brain or in the cerebral cortex) was highly significant (as found in the present study); this study did not consider associations with the total cSVD score or with other neuroimaging signatures of cSVD. 11 Yet in a Japanese study of 240 stroke-free individuals, the brachial–ankle PWV was associated with WMH, but other signatures of cSVD were not investigated. 12 In a smaller Dutch series (167 hypertensive patients), the aortic PWV was associated with WMH and LI but not with CMB. 13 As noted, differences in study design and the characteristics of participants probably accounted for the observed variability in the association between arterial stiffness and neuroimaging signatures of cSVD.
According to the Rotterdam Scan Study, 9 the association between arterial stiffness and cSVD is apparently not mediated by traditional cardiovascular risk factors, which suggests the presence of other (hidden) factors mediating this relationship, such as inflammatory or genetically determined markers. Similar findings were noted in the population of Atahualpa (Table 2). While pathogenetic mechanisms involved in the relationship between arterial stiffness and cSVD are not totally understood, it is possible that diffuse microvascular damage related to aging and several vascular risk factors contribute to arteriolosclerosis of small vessels. We have previously demonstrated association between neuroimaging signatures of cSVD and kidney dysfunction 28 and damage of retinal vessels 29 in the population of Atahualpa, findings that are in line with the occurrence of a disseminated vascular damage accounting for the observed associations; such damage might be mediated by arterial stiffness-induced decreased elasticity of small arteries. 30
Study strengths and limitations
The strengths of our study include the population-based design with unbiased selection of participants, the systematic evaluation of participants with well-defined neuroimaging protocols, the use of the aortic PWV as the most reliable surrogate of arterial stiffness, 31 and the homogeneous characteristics of the study population. Our results may not be generalizable to other populations or ethnic groups and this is a limitation of the present study. Another limitation is the fact that we did not investigate the presence of large vessel occlusive disease in the study population. In addition, we cannot rule out the presence of a hidden confounder that might explain our findings, and the cross-sectional design precludes the assessment of causation.
Conclusion
The present study shows a strong association between the aortic PWV and the total cSVD score and most of its components (with the exception of LI) in a population of older Amerindians. Arterial stiffness determination may be useful for identifying candidates for MRI in the search for neuroimaging signatures of cSVD, allowing the recognition of apparently healthy individuals at risk for developing cerebrovascular events. Further longitudinal studies are needed to assess causality and whether the control of arterial stiffness (if possible) may reduce the burden of cSVD in the follow-up.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: study support was provided by Universidad Espíritu Santo–Ecuador. The sponsor had no role in the design of the Study, or in the collection or analysis of data.
