Abstract
An embolic event originating from thrombus on an otherwise un-diseased or minimally diseased proximal artery (Phantom Thrombus) is a rare but significant clinical challenge. All patients from a single center with an imaging defined luminal thrombus with a focal mural attachment site on an artery were evaluated retrospectively. We excluded all patients with underlying anatomic abnormalities of the vessel at the attachment site. Six patients with a mean age of 62.5 years were identified over a 2.5-year period. All patients had completed treatment for or had a current diagnosis of malignancy and none were on antiplatelets or other anticoagulants. Four thrombi originated in the aorta proximal to the renal arteries and one originated distal. One thrombus was found in the common carotid artery and one was in an arterialized vein graft. Mean follow-up was 22 months. None of the patients underwent removal or exclusion of the embolic source. With systemic anticoagulation, four of the phantom thrombi were resolved on imaging within 8 weeks, one resolved after 72 weeks. One phantom thrombus reoccurred after 6 months on reduced anticoagulant dosing. There was one acute and one death in follow-up (26 months). One patient required a partial foot amputation secondary to tissue necrosis from the initial thromboembolic event. Arterial thrombi forming on otherwise normal vessels are a distinct clinical entity. In patients with a phantom thrombus, a strategy of therapeutic anticoagulation for management of the embolic source seems to be safe and effective over both the short and intermediate-term.
Introduction
Embolization of thrombus from an arterial (non-cardiac) source is rare and can cause substantial morbidity and mortality. Treatment options that have been employed to avoid embolic complications include surgical extraction of the embolizing thrombus, endograft coverage of the lesion and prolonged systemic anticoagulation. The optimal treatment will prevent further embolization, eliminate the embolic source and minimize procedural risks to the patient.
As imaging techniques have improved, there are non-occlusive arterial thrombi with embolic potential that occur in vessels that have no evident underlying wall pathology. The thrombi themselves are not symptomatic and appear on imaging as ghost-like shadows floating in the arterial lumen; thus we have created the term, Phantom Thrombus, to designate these specific lesions. Prior investigations reporting treatment results often do not distinguish these unique lesions with embolizing thrombi associated with varying degrees of underlying vascular disease (e.g. ulcerated plaque or aneurysm). As a result, the specific therapeutic outcomes for the patients with phantom thrombi are difficult to determine.
Recent studies have suggested that phantom thrombi may be adequately treated with a regimen of anticoagulation alone.1,2 The purpose of this study is to report the results of a strategy of anticoagulation alone in a consecutive series of patients with phantom thrombi from a single academic practice.
Methods
We defined phantom thrombus as non-occlusive intraluminal mural-based thrombus in an arterial vessel that was without evidence of any aneurysmal, dissection or atherosclerotic change at the site of thrombus attachment. Consecutive patients presenting to the Vascular Surgery service at a single academic institution with an imaging study demonstrating findings potentially consistent with phantom thrombus were evaluated. Seven patients were reviewed, and one was excluded due to underlying atherosclerotic disease. A search of a deidentified database (Synthetic Derivative, Vanderbilt University) was also performed over the same time interval, but did not yield any additional patients with a finding of arterial thrombus in a non-diseased vessel. These patients were evaluated retrospectively under a specific Institutional Review Board-approved protocol (Vanderbilt University #151822). Descriptive statistics are used to characterize the group data.
Results
Demographics
During the period December 2014 to June 2017, a total of six patients with imaging findings consistent with phantom thrombus were identified and charts reviewed. Demographic data are shown in Table 1. This diagnosis was made in three men and three women (1:1), and the mean age of presentation was 62.5 years (range 51–79). All but one of the patients were smokers.
Patient demographics at presentation.
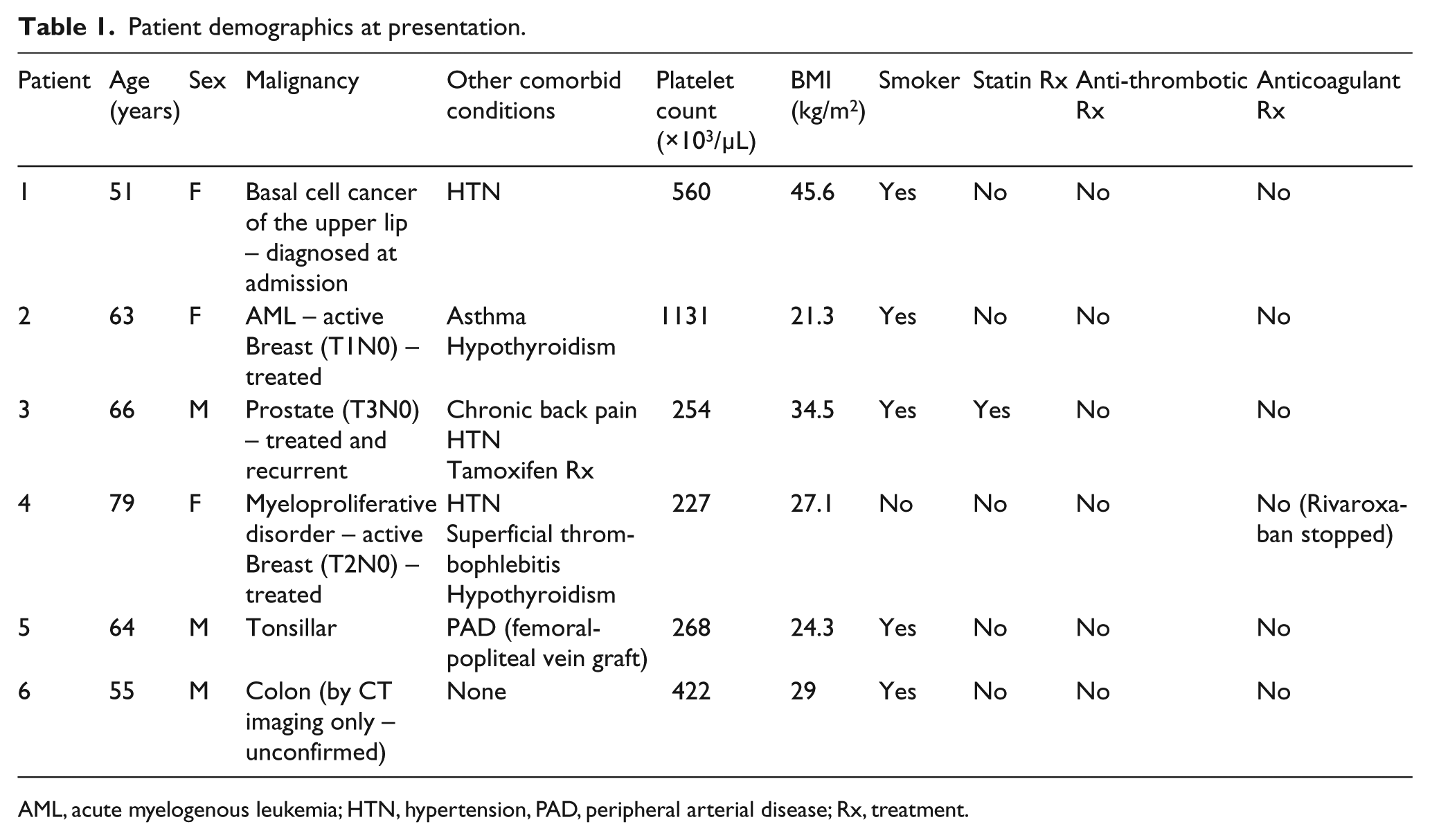
AML, acute myelogenous leukemia; CT: computed tomography; HTN, hypertension, PAD, peripheral arterial disease; Rx, treatment.
Three of the patients presented with high platelet concentrations. None of the patients were taking anticoagulants, aspirin or other platelet inhibitors at presentation, although one patient had been on rivaroxaban approximately 1 week prior to presentation for a diagnosis of saphenous vein superficial thrombophlebitis. Only one of the patients was taking an HMG-CoA Reductase inhibitor. One patient was taking Tamoxifen for gynecomastia.
All patients were diagnosed with a malignancy. Two patients were diagnosed during the index admission, one basal cell cancer of the face and one colon cancer (based on colonic thickening and retroperitoneal adenopathy on CT scan, but not confirmed by tissue diagnosis). The remaining patients all had a history of solid organ tumors and two of the patients also had blood dyscrasias.
Presentation and initial management
The origin of the mural thrombus, embolic event and initial management are listed in Table 2. The CT imaging from two of the patients are shown in Figure 1. All patients were treated on presentation to the vascular surgery service with systemic, continuous unfractionated heparin, and at discharge were transitioned to warfarin anticoagulation with a target INR of 2.0–3.0. Enoxaparin was used as a bridge, if the patient was discharged with an INR < 2.0.
Initial presentation.
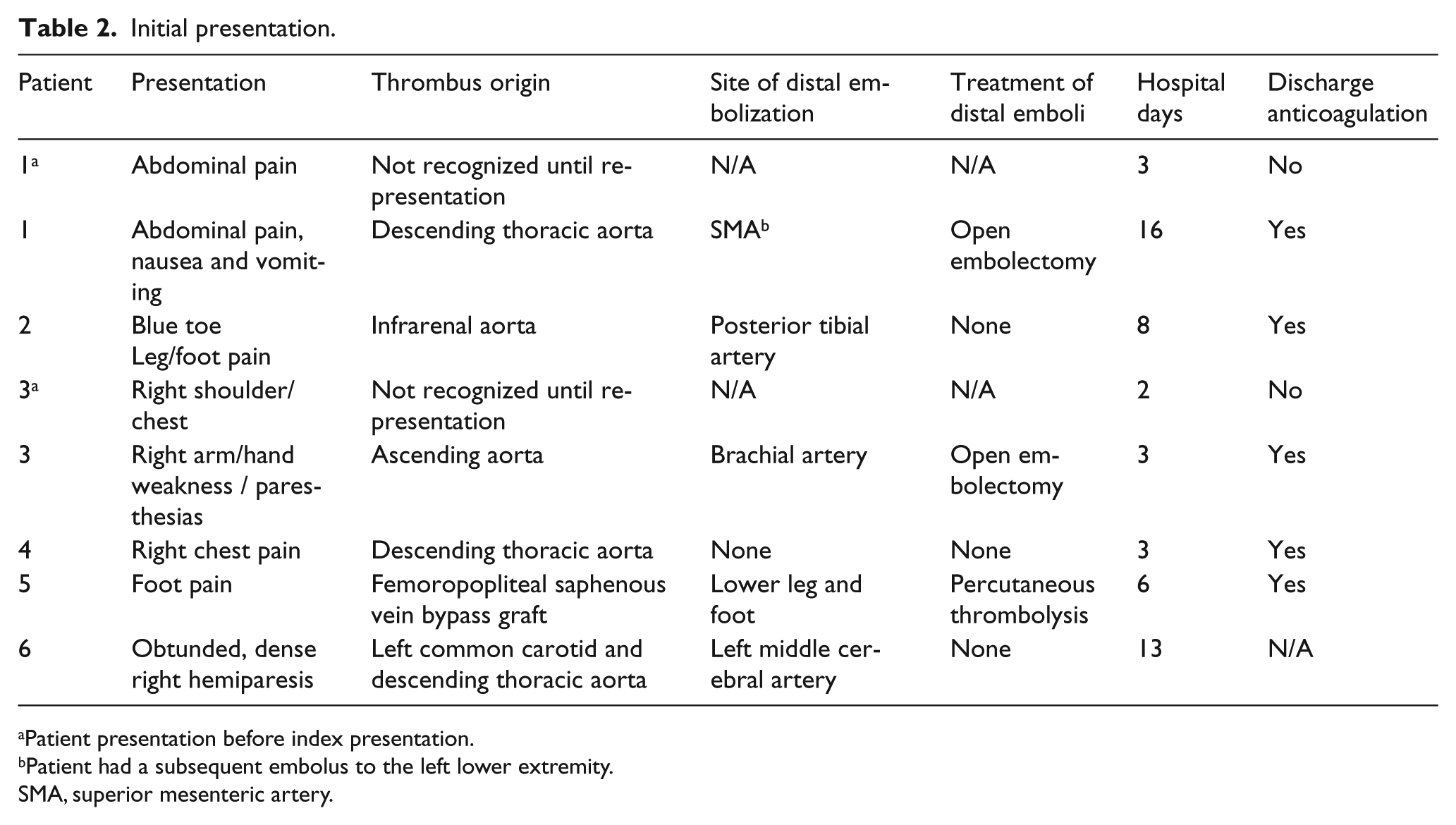
Patient presentation before index presentation.
Patient had a subsequent embolus to the left lower extremity.
N/A, not applicable; SMA, superior mesenteric artery.
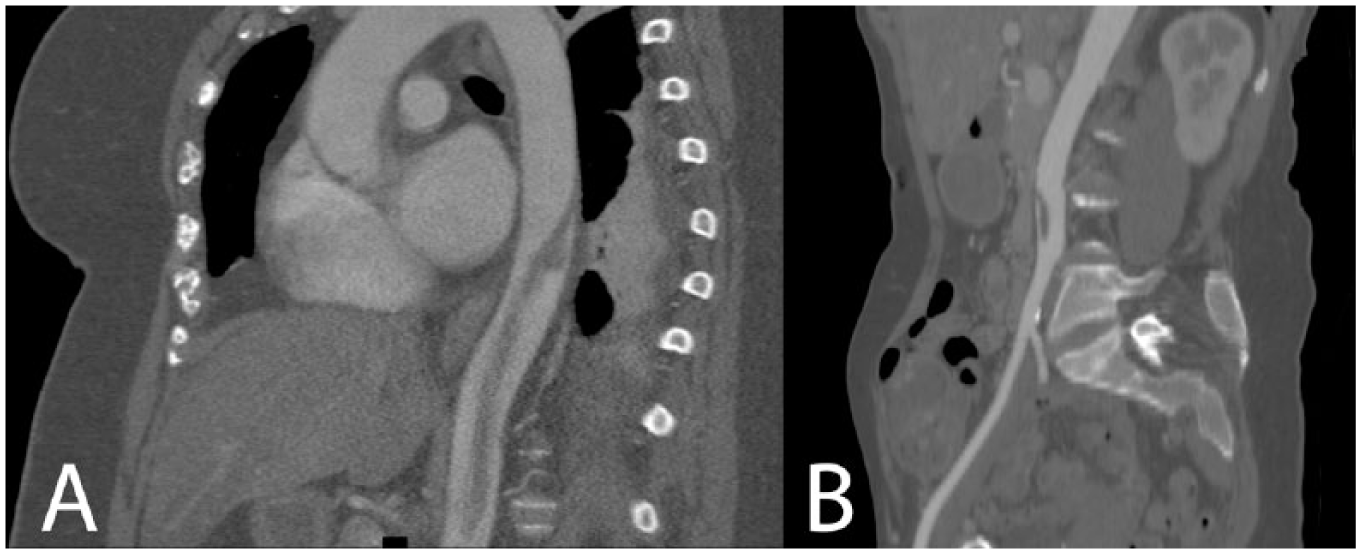
Reformatted sagittal CT images of phantom thrombi. (A) in the descending thoracic aorta and (B) in the infrarenal aorta. Note the lack of underlying arterial disease as well as the very narrow attachment point.
There were four procedures to treat embolic events in 3 patients. Two of these patients (#1 and 3) presented with non-specific abdominal or chest pain and CT imaging performed at that time demonstrated phantom thrombus. In both cases, the diagnosis went unrecognized by the radiologist and clinical staff and the patients were discharged. Patient number 1 re-presented after 14 days with worsening abdominal pain where a repeat CT scan demonstrated a new finding of superior mesenteric artery embolus. The patient underwent laparotomy and mesenteric embolectomy and was started in heparin infusion. Five days later, the patient had sub-therapeutic anticoagulant levels for over 24 hours and had a second embolic event to the left lower extremity. Subsequently, the patient was maintained on therapeutic anticoagulation and had no further embolic events. The second patient (#3) presented 4 days later with right arm pain and a brachial artery embolus, and underwent a successful brachial embolectomy.
Patient 5 uniquely developed a phantom thrombus in the mid-portion of a lower extremity venous bypass graft which embolized to the tibial arteries. Catheter-directed thrombolysis in the tibial arteries successfully restored the majority of pedal perfusion. Subsequent arteriograms after thrombus resolution did not demonstrate any intra-graft or anastomotic abnormalities.
No procedures were performed on the other three patients. Patient 4 had the phantom thrombus in the descending aorta identified incidentally on a CT scan for a non-specific acute short-interval of chest pain. No distal emboli were seen on imaging. The patient was started on systemic anticoagulation after the pain had resolved and never developed any clinical embolic phenomenon. Patient 2 who had a phantom thrombus in the infrarenal aorta had small distal pedal emboli that were not amenable to embolectomy. Patient 6 had a phantom thrombus in the left common carotid and a large middle cerebral artery embolus that presented late and was not a candidate for intervention; he died in the hospital due to a massive stroke.
None of the patients had mechanical exclusion or removal of the embolic or potentially embolic source. The patient undergoing catheter-directed thrombolytic therapy of the distal lower extremity emboli may have had accelerated dissolution of the proximal thrombus.
Follow-up
All 5 patients discharged from the initial hospitalization returned for follow-up (Table 3) at a mean of 22 months (Range 11 months to 27 months). Four patients were maintained on anticoagulation throughout follow-up, while one patient was maintained on aspirin alone after 5 months of warfarin therapy. Three of the four patients were maintained on long-term warfarin and one was switched to rivaroxaban after follow-up imaging demonstrated resolution of the phantom thrombus.
Follow-up.
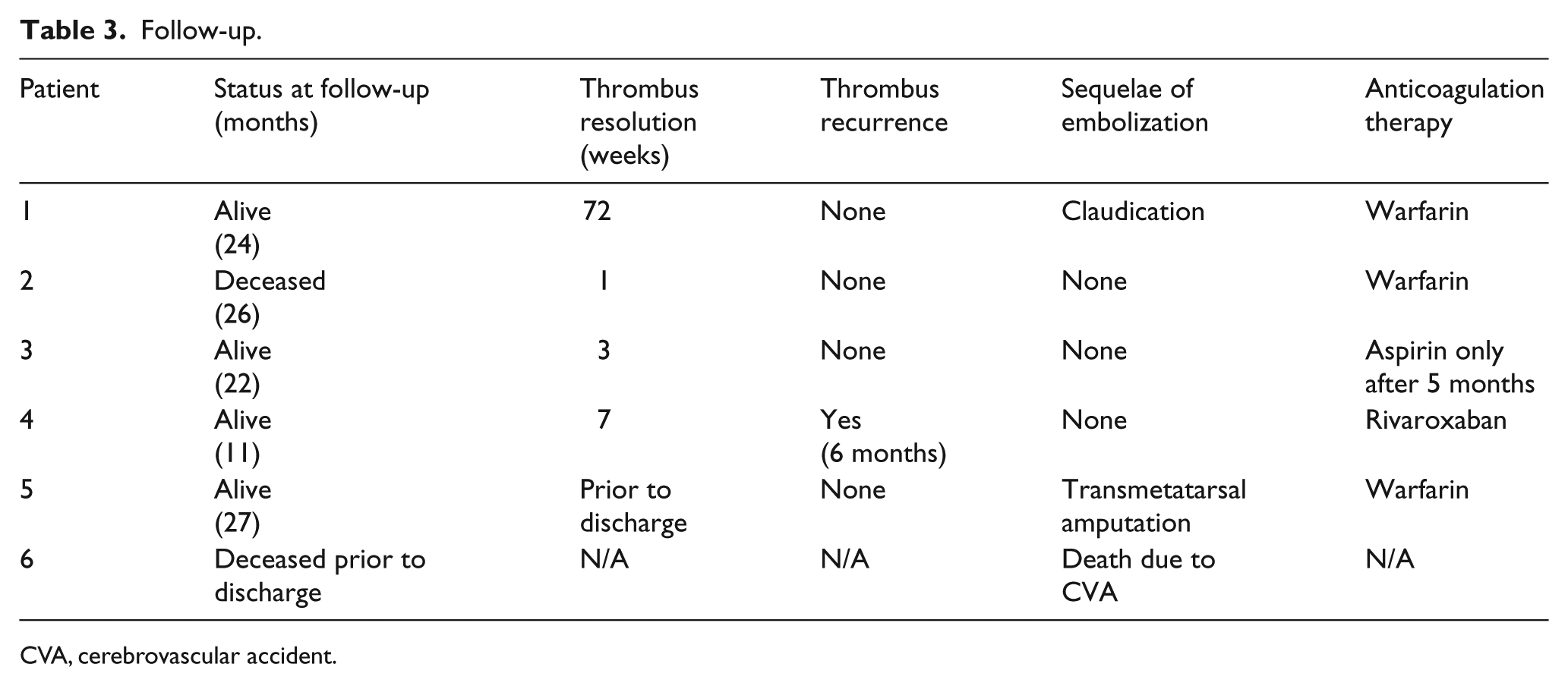
CVA, cerebrovascular accident; N/A: not applicable.
There were no episodes of recurrent embolization following discharge. Although most patients demonstrated resolution of the phantom thrombus on early follow-up imaging, one patient had slow but continued resolution that did not complete for 72 weeks.
One patient who was switched to rivaroxaban after initial resolution on warfarin was initially treated at 20 mg/day and was subsequently reduced to 10 mg/day by her hematologist. Planned imaging at 6 months following her initial event, demonstrated recurrence of the phantom thrombus at the same location. Therapeutic enoxaparin anticoagulation was initiated for 4 weeks with complete resolution of the thrombus. The patient then resumed rivaroxaban at 20 mg/day without recurrence.
In addition to the in-hospital death, there was one death in follow-up at 26 months attributed to pulmonary complications of acute myeloid leukemia.
Discussion
Peripheral thromboembolic events have decreased in frequency, but continue to result in significant mortality and disability. 3 Arterial thromboembolic events overwhelmingly originate from a cardiac source (> 85%).4,5 Other sources can include atherosclerotic plaque, ulcerated plaque, aneurysms or injured vascular wall. In this report, we focused on embolic sources from arterial mural thrombus which develops on otherwise normal vessels by diagnostic imaging (Figure 1).
The terminology used to describe these lesions is not well-defined or consistent throughout the literature. They have been described as ‘idiopathic’ or ‘atypical’ to distinguish these lesions from mural thrombi which occur secondary to intrinsic pathology of the artery, such as aneurysmal degeneration or atherosclerotic ulceration. The terms ‘primary’ or ‘spontaneous’ has been used to indicate that these are believed to be formed de novo and are not secondary to a more proximal source. Some use the term ‘mural’ thrombus while some use the term ‘mobile’ to describe the limited attachment and movement with the cardiac cycle. Many reports specify ‘aortic’ or ‘thoracic’. While these are common locations, there is no evidence to suggest that aortic thrombi have a different natural history than similar entities seen in other large vessels. To increase clarity and specificity in reporting, we are proposing use of a simplified term ‘phantom thrombus’ to refer to non-occlusive primary intraluminal thrombi occurring in otherwise locally non-diseased (without atherosclerosis, dissection or aneurysm) arteries.
We have elected to choose the term ‘phantom’, meaning ghost-like, to describe the thrombus, both because of its appearance in the vessel on CT imaging and because the primary lesion development occurs without symptoms in a location that bears no evident predisposition. In our series, we had three patients with phantom thrombus who presented with pain symptoms, but without a clinical diagnosis of embolism. Two of those did not receive anticoagulant therapy and subsequently returned with clinically evident embolic events. The third had resolution of the pain symptoms after imaging but prior to starting anticoagulation. We do not believe that any of the symptoms that prompted imaging were from the phantom thrombus itself, but were either due to small and/or unrecognized embolic events or unrelated phenomenon.
When underlying vascular disease exists at the site of thrombus formation, the natural history and management outcomes are likely to be different. Increased diagnostic specification of this entity from similar but distinct processes may allow us to better understand both the natural history and may help gain insight into the pathologic process. In this report, it is notable that all the patients with phantom thrombus had at least a history of a solid organ tumor within the past 5 years, and none of the patients was on aspirin or other antithrombotic agents. Other reports have also seen an association of arterial emboli with malignancy,6,7 but further investigation will be needed to develop a more clear understanding of the relationship of phantom thrombi with malignancy.
The natural history of phantom thrombus and the factors that may influence the clinical outcome are poorly understood. An autopsy study of nearly eleven thousand patients found 48 with mural aortic thrombus in normal caliber aortas. 8 Of these, 20 were described as not having generalized atherosclerosis on careful review of the findings reported. This report, like most reports of non-cardiac arterial thromboembolism, are mixed series including patients with and without underlying atherosclerotic disease.9–13 Therefore, the disease specific risks and benefits of the various therapeutic options are not well known. This is particularly true of the effects of anticoagulant therapy.
The earliest reports of arterial emboli from non-cardiac sources implicated heparin as the cause of the embolic transformation of arterial thrombi. The first of these was a 1958 publication of a series of cases with arterial embolic events that were attributed to ‘heparin effect on pre-existing, unsuspected mural aortic fibrin-platelet thrombi’. 14 Subsequent series also related the development of aortic thromboembolic complications to the use of heparin.15–17
A landmark series which undermined the theory that heparin promoted emboli was published in 1999. 18 It was one of the first descriptions of full anticoagulation resulting in regression and resolution of the primary thrombus without additional emboli. Others have published similar results in small series, although it unclear whether these are homogeneous embolic sources.2,7,19 Despite these more recent reports, primary therapy of the phantom lesion with heparin is not widely accepted. Open extraction of the thrombus directly or with balloon embolectomy catheters has been extensively reported.14–16,20–23 As endovascular procedures have become more common for aortic and peripheral vascular diseases, there have been many reports of covering these lesions with endografts.24–31 Higher rates of complications1,29 as well as occasional catastrophic results 32 have also been reported with this approach in patients with phantom thrombus.
Based on this single center experience, full therapeutic anticoagulation appears to be a safe and appropriate strategy for the treatment of phantom thrombi. This may be true even when the thrombus is slow to resolve. Further, our intermediate term results suggest good outcomes with longer-term anticoagulant therapy and anecdotally support that some patients have an underlying thrombophilia that may otherwise lead to recurrence. There have been some reports that anti-platelet agents alone may be adequate in these patients. 33
This is a relatively small group of patients, and care in generalizing the findings is appropriate. All surviving patients in this study received unfractionated heparin intravenously followed by warfarin oral anticoagulation until resolution of the phantom thrombus. There was substantial herterogeneity in the long-term treatment regimens. Further studies and reports are necessary to determine the optimal therapeutic regimen for safety and efficacy.
Conclusions
Modern imaging can provide an accurate diagnosis of a phantom thrombus which can be safely treated with therapeutic anticoagulation alone following appropriate intervention for embolic complications. Consideration of underlying malignancy or other thrombophilia should be made. Future studies and reports should distinguish the outcomes of phantom thrombi from other arterial embolic sources.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
