Abstract
The introduction of electronic cigarettes has led to widespread discussion on the cardiovascular risks compared to conventional smoking. We therefore conducted a randomized cross-over study of the acute use of three tobacco products, including a control group using a nicotine-free liquid. Fifteen active smokers were studied during and after smoking either a cigarette or an electronic cigarette with or without nicotine (eGo-T CE4 vaporizer). Subjects were blinded to the nicotine content of the electronic cigarette and were followed up for 2 hours after smoking a cigarette or vaping an electronic cigarette. Peripheral and central blood pressures as well as parameters of arterial stiffness were measured by a Mobil-O-Graph® device. The peripheral systolic blood pressure rose significantly for approximately 45 minutes after vaping nicotine-containing liquid (p<0.05) and for approximately 15 minutes after smoking a conventional cigarette (p<0.01), whereas nicotine-free liquids did not lead to significant changes during the first hour of follow-up. Likewise, heart rate remained elevated approximately 45 minutes after vaping an electronic cigarette with nicotine-containing liquid and over the first 30 minutes after smoking a cigarette in contrast to controls. Elevation of pulse wave velocity was independent from mean arterial pressure as well as heart rate in the electronic cigarette and cigarette groups. In this first of its kind trial, we observed changes in peripheral and central blood pressure and also in pulse wave velocity after smoking a cigarette as well as after vaping a nicotine-containing electronic cigarette. These findings may be associated with an increased long-term cardiovascular risk.
Keywords
Introduction
Tobacco use is considered to be one of the leading causes of preventable death and disease in Western Europe and the United States. 1 In this context, the working group of Shaw published data indicating that each smoked cigarette shortens life by 11 minutes. 2 Recent data differentiate smoking from other types of nicotine products. In addition to cigarettes and traditional tobacco products, the electronic cigarette (e-cigarette) has become more popular. In addition to rising attention and distribution in print and internet media, lifetime and 30-day prevalence use of e-cigarettes is also increasing. Current data show there are 13 million users of e-cigarettes all over the world. 3
On the one hand, publications postulate that vaping is less harmful than smoking.4–6 On the other, publications describe the health impact of e-cigarettes or cast a scrutinizing view on them.7–10 In addition, e-cigarettes are discussed as a tool for smoking cessation, 11 although long-term efficacy data are still missing. 12
In general, smoking is accepted as one of the most significant risk factors for cardiovascular events.13,14 Apart from the importance of nicotine as a risk factor, smoking is one of the most modifiable ones. 15
Several trials have evaluated the chronic effect of smoking on cardiovascular events. Based on the guidelines of the European Society of Hypertension, as well as the European Society of Cardiology, measurement of arterial stiffness and endothelial dysfunction may be used as an assessment of subclinical end organ damage as an indication of developing cardiovascular disease. 16
We therefore aimed to determine the acute effects of vaping nicotine-containing or nicotine-free liquids versus cigarette smoking on both peripheral and central blood pressures including arterial stiffness.
Methods
Study cohort and design
This single-center pilot study included 15 young, active, traditional cigarette smokers. The trial was designed as a cross-over study of the acute use of three tobacco products. The subjects were blinded to the nicotine content of the e-cigarette.
The participants were randomized to one of the three study groups during the first visit by drawing pieces of paper from a closed envelope (Figure 1). The envelope contained three pieces of numbered paper (one to three); participants’ order was moved by drawing three times. The numbers denoted e-cigarette with nicotine, e-cigarette without nicotine, or cigarette.
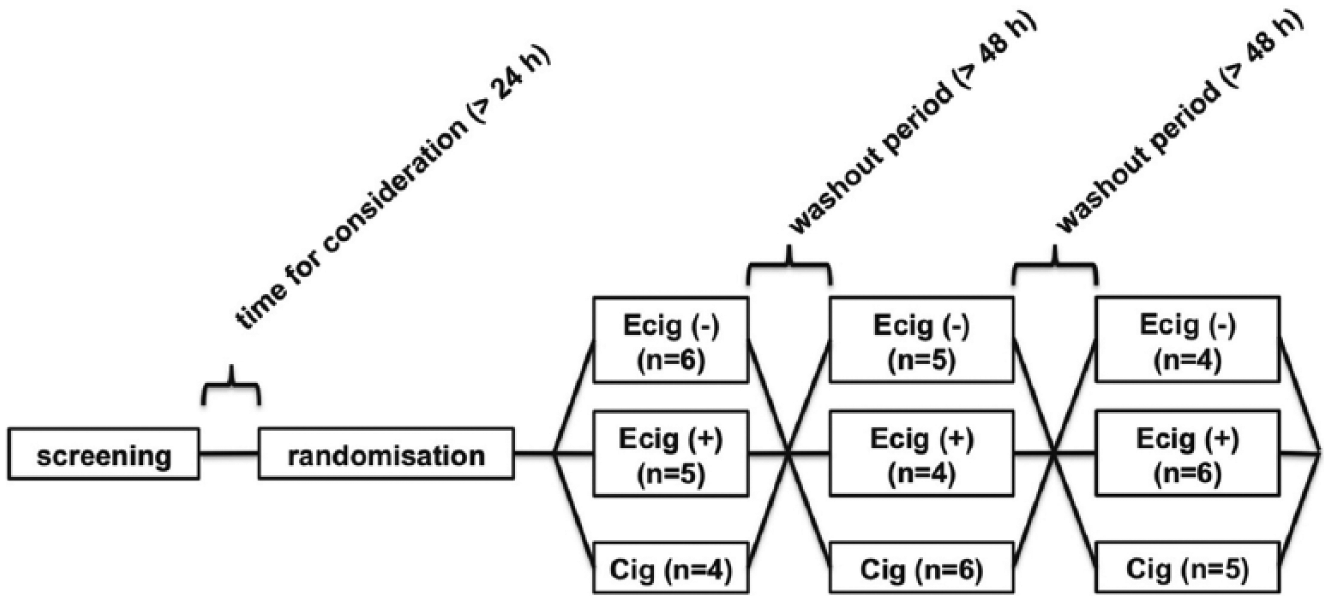
Study design.
The participants were recruited from amongst students from the University of Luebeck. During screening, all participants were checked for exclusion criteria: (i) non-smoking or non-vaping; (ii) mental disorders; (iii) cardiovascular diseases; (iv) thyroid disease; (v) diabetes; (vi) abnormalities during physical examination; (vii) hypertension and/or (viii) elevated cholesterol or triglycerides. Furthermore, pregnancy excluded female participants. Participants were excluded if previously enrolled in any other kind of study and if they declared being strict non-smokers before they were given a written informed consent form. The study had the permission of the local ethics committee and was registered on German Register Clinical Trials (DRKS) (DRKS00012919).
In accordance with previous studies using arterial stiffness,17,18 alcohol and/or smoking cigarettes were not allowed 24 hours prior to the measurements. The smoking-free phase was tested by Micro+™ Smokerlyzer (Bedfont Scientific Ltd, Maidstone, UK) with a cut-off of 6 ppm CO. This phase was necessary to show the acute effects of each intervention. Furthermore, an elapse of 48 hours was scheduled between each test day to avoid any acute interaction between devices.
The three different study groups were the following: (1) smoking a cigarette and inhaling into the lungs (Cig) (Philip & Morris, New York, USA); (2) vaping an e-cigarette with nicotine (ECig (+)) (DIPSE, eGo-T CE4 vaporizer (third generation), SSR Produkt GmbH & Co KG, Oldenburg, Germany, 3.3 volts, 1.5 ohms and 7.26 watts; 24 mg/mL nicotine, 55% propylene glycol and 35% glycerin, tobacco flavor); and (3) vaping an e-cigarette without nicotine (ECig (–)) (0 mg/mL nicotine, 55% propylene glycol and 35% glycerin, tobacco flavor). Both investigators and participants were blinded (i.e. both were unaware of the kind of liquid used). The participants were asked to smoke the cigarette and inhale into the lungs. During the study, smokers who were inexperienced in the use of e-cigarettes were introduced to vaping and trained to use an e-cigarette by an experienced e-cigarette user. All participants had to vape the e-cigarette with a minimum of one puff every 30 seconds for 10 puffs, in accordance with different publications. 19 Every puff had to last for 4 seconds for this study. Each participant had to fulfill all three conditions to complete the study and to be analyzed.
Generally, measurements were started at least 30 minutes before vaping or smoking. Blood pressure were measured by a Mobil-O-Graph® (I.E.M., Stollberg, Germany)20–22 every 5 minutes and with a conventional blood pressure monitor (Omron MIT Elite Plus®; Omron, Kyoto, Japan) every 15 minutes. Measurements discontinued not less than 2 hours after the application. The three measurements were taken around the same time of day to avoid change due to circadian rhythms.
Measurement of peripheral and central blood pressures and arterial stiffness
The blood pressure measurements with additional parameters were performed with the validated Mobil-O-Graph (software version HMS CS 4.2; I.E.M.), which allows recording of central pressures and arterial stiffness parameters as transmitted.20,21,23 Briefly, the Mobil-O-Graph uses the oscillometric technique with a standard blood pressure cuff at the arteria brachialis. 20 Central systolic blood pressure was determined from brachial waveforms, recorded with the cuff at the level of diastolic blood pressure and processed with the ARCSolver transfer function. Using the derived central waveforms, pulse waveform analysis is performed and pulse wave velocity (PWV), augmentation index (AIx) and augmentation pressure are estimated.21,22 Measurements started 30 minutes before smoking or vaping in a sitting position. Therefore, the mean values of baseline were calculated from three measurements every 5 minutes directly before smoking or vaping. These mean values were used as references for statistical analyses.
Statistical analysis
Statistical analyses were performed with IBM SPSS statistical software, version 23 (IBM Corp., Armonk, NY, USA), graphs were edited with SigmaPlot 8.0 (SYSTAT Software Inc., San Jose, CA, USA) and prepared for publication with CorelDRAW 11.0 (Corel Inc., Mountain View, CA, USA). Baseline mean values were taken for statistical references of blood pressure as well as arterial stiffness. During the observation period of 2 hours, measurements were performed in intervals of 5 minutes. Mean values were calculated of three measuring points to form 15-minute intervals for statistical analyses. The investigator who analyzed the vascular data was blinded to the order of the devices, as well as the study intervention.
Before further analyses of the blood pressure, peripheral as well as central hemodynamics were analyzed for normal distribution by Kolmogorov–Smirnov tests. Because of the cross-over design, we calculated a two-way repeated measures analysis of variance (ANOVA) evaluating for an interaction between time and type of tobacco product used. Accordingly we did post hoc tests (Bonferroni) by G*Power if there was an interaction. In addition, the data were analyzed via paired Student’s t-tests corrected for multiple testing and the Wilcoxon test where appropriate to compare individual time points within the three test settings. ANOVA was used to analyze the differences at the different time points between the three different groups, respectively. Where applicable a multivariate analysis of variance (MANOVA) was performed correcting for age, mean arterial pressure (MAP), heart rate (HR) and sex. If not stated otherwise, all data are expressed as mean ± SD. A p-value of < 0.05 was defined as statistically significant.
Results
Baseline characteristics
Baseline characteristics for all subjects are presented in Table 1. The participants did not show any significant clinical differences between the initial three groups formed by the randomization to the different order (data not shown). The participants were all smokers without a history of vaping or dual use. All participants finished the trial. The number of participants in each group is presented in Figure 1.
Baseline characteristics (n=15).
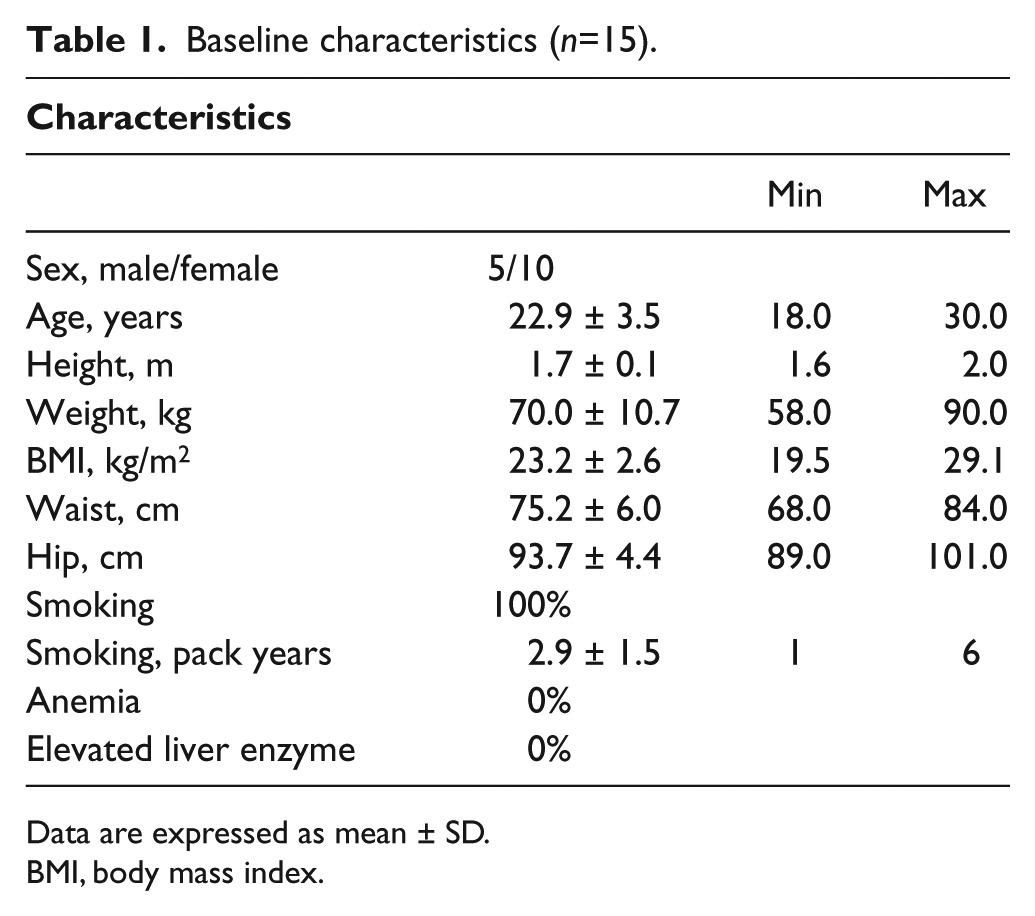
Data are expressed as mean ± SD.
BMI, body mass index.
Cigarettes and e-cigarettes with nicotine increased SBP (Mobil-O-Graph)
The two-way repeated measures ANOVA showed a significant difference between the three study groups for the type of study group (p < 0.05), which remained statistically significant after post hoc testing. Using additional statistics, the systolic blood pressure (SBP) was increased significantly within the Cig and ECig (+) groups by more than + 3% (p < 0.05; Figure 2) and the three groups differed significantly from each other (p < 0.05; Figure 2). There were no significant changes of SBP within ECig (–) during follow-up (p > 0.05; Figure 2).
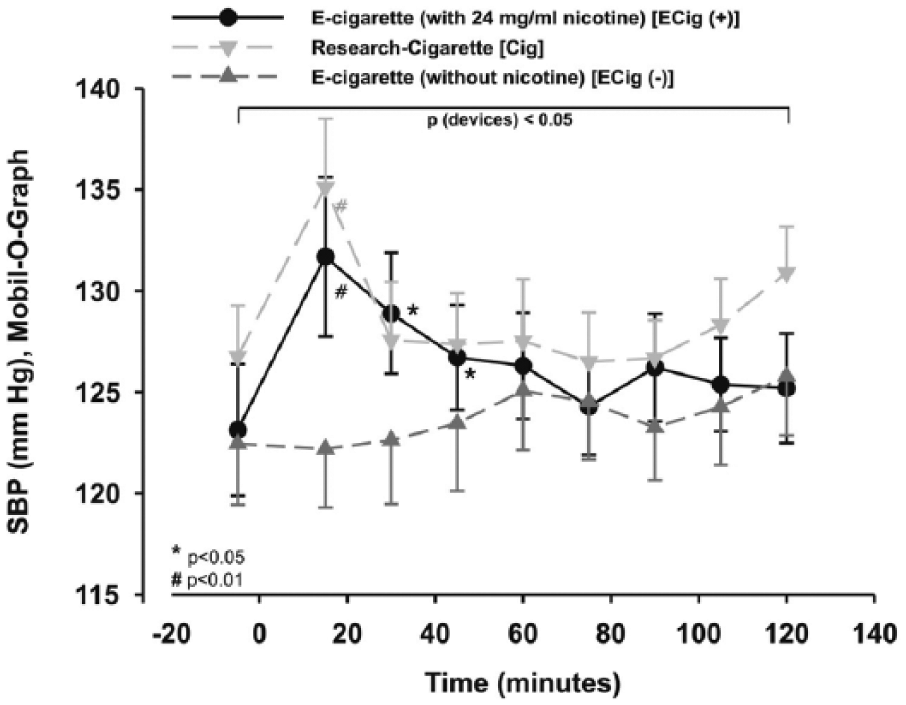
Peripheral SBP was increased significantly within Cig and ECig (+). Two-way measured ANOVA showed a significance between the devices (p < 0.05).
The three groups showed significant differences for peripheral diastolic blood pressure (DBP) from each other within the two-way repeated measures ANOVA for the type of study group, which remained statistically significant after post hoc testing (p < 0.05; Figure 3), too. Furthermore, DBP was significantly increased by more than 5% in the Cig arm using additional statistics (p < 0.05; Figure 3). In contrast to these findings, in ECig (–), DBP decreased by more than 4%, significantly after 30 minutes (p < 0.05; Figure 3).
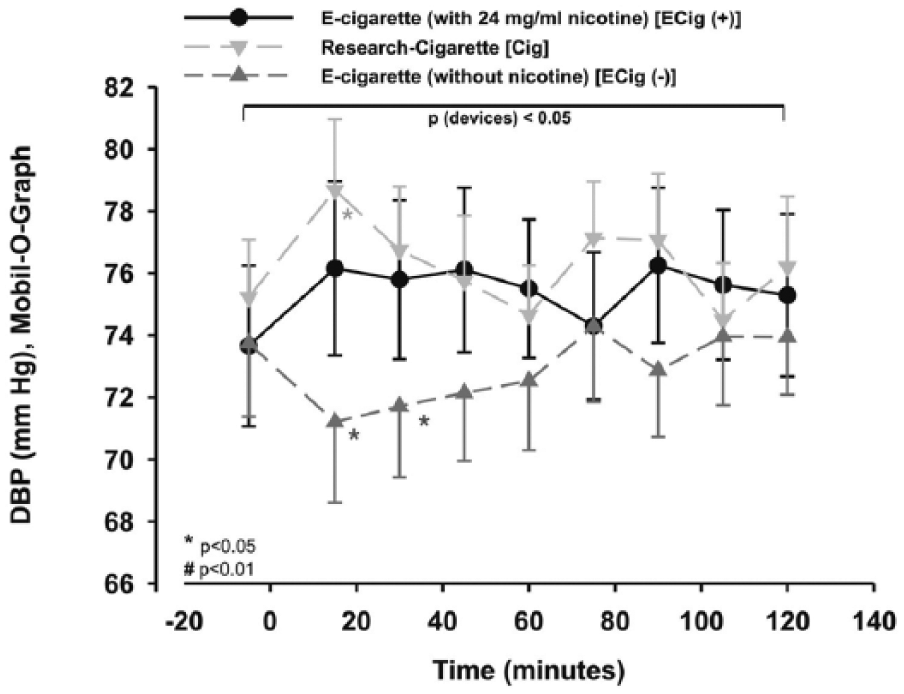
Peripheral DBP was changed significantly for Cig and ECig (-). Two-way measured ANOVA showed a significance between the devices (p < 0.05), which remains after post hoc testing.
No significant difference for the peripheral pulse pressure (PP) was found between the three groups within the two-way repeated measures ANOVA (p > 0.05; Figure 4), although PP showed a similar result to the SBP, with an increase at the first measuring points in the Cig and ECig (+) arms after smoking or vaping (Figure 4) using additional statistics.
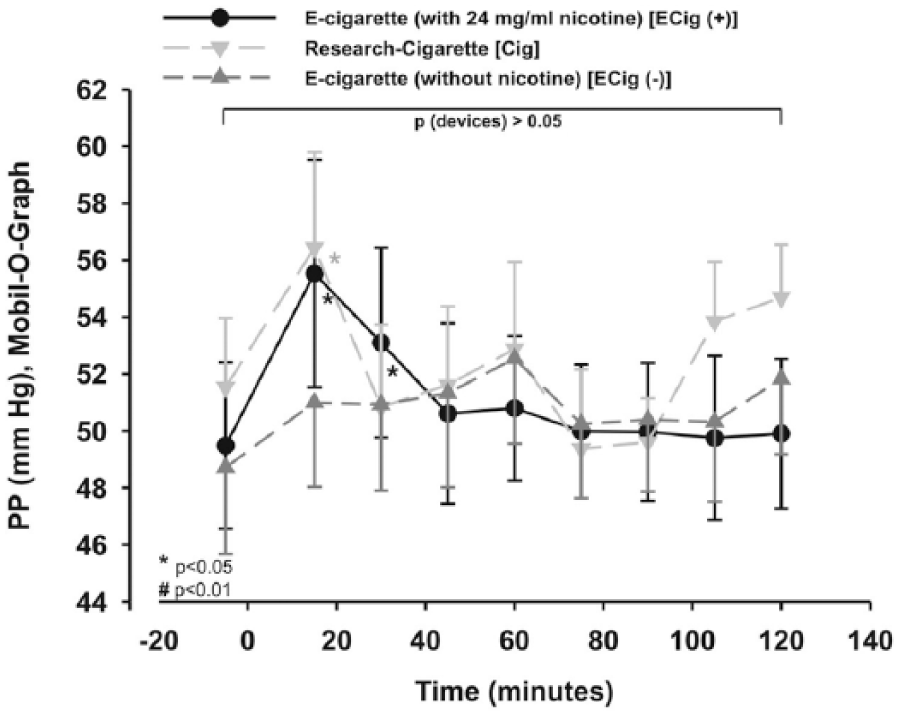
Peripheral PP was increased significantly within Cig and ECig (+) using additional statistics. Two-way measured ANOVA did not show a significance between the devices (p < 0.05), which remains after post hoc testing.
HR was significantly elevated by smoking and vaping with nicotine
The HR of the three groups differed significantly for the type of study group as well as for time within the two-way repeated measures ANOVA (p < 0.05; Figure 5). Using additional statistics, there was a significant effect after vaping over 45 minutes (+12%; p < 0.05; Figure 5). HR was increased by more than 8% during the first 30 minutes after smoking a cigarette (p < 0.05; Figure 5).
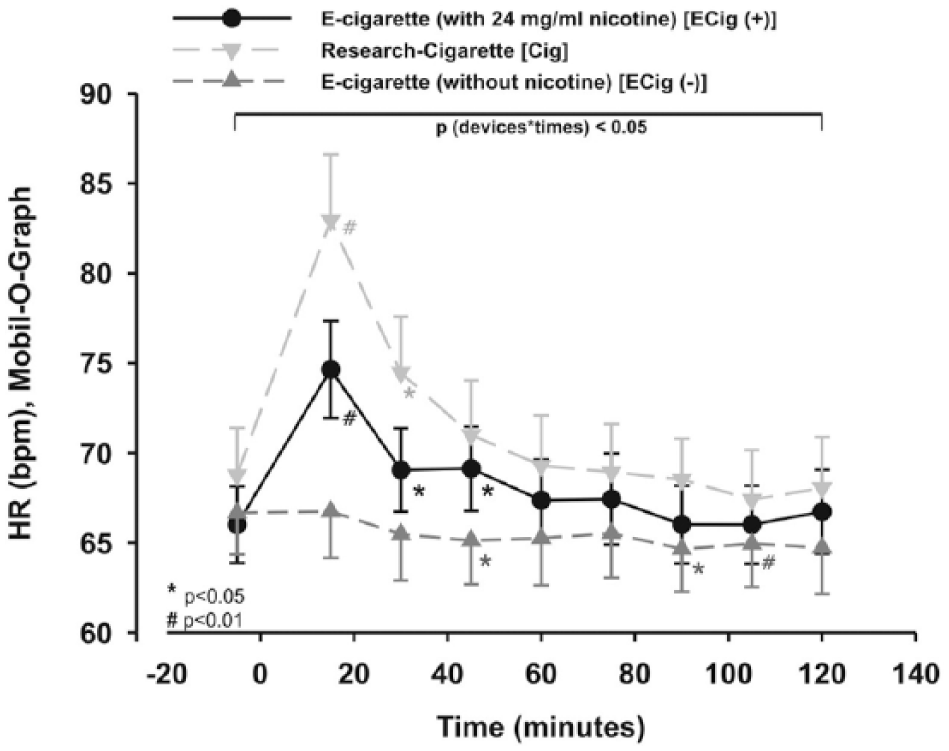
HR was increased within Cig and ECig (+). Two-way measured ANOVA showed a significance between the devices and time (p < 0.05), which remains after post hoc testing.
Vaping with nicotine changed central hemodynamics
The two-way repeated measures ANOVA showed a trend without reaching significance between the different study groups (p=0.053) within central systolic blood pressures (cSBP). The cSBP increased after vaping with ECig (+) and smoking a cigarette, showing a trend without reaching significance (p=0.088/p=0.084; Figure 6) using additional statistics. ECig (–) did not show any significant changes of cSBP during the 2 hours of observation after vaping (p > 0.05; Figure 6).
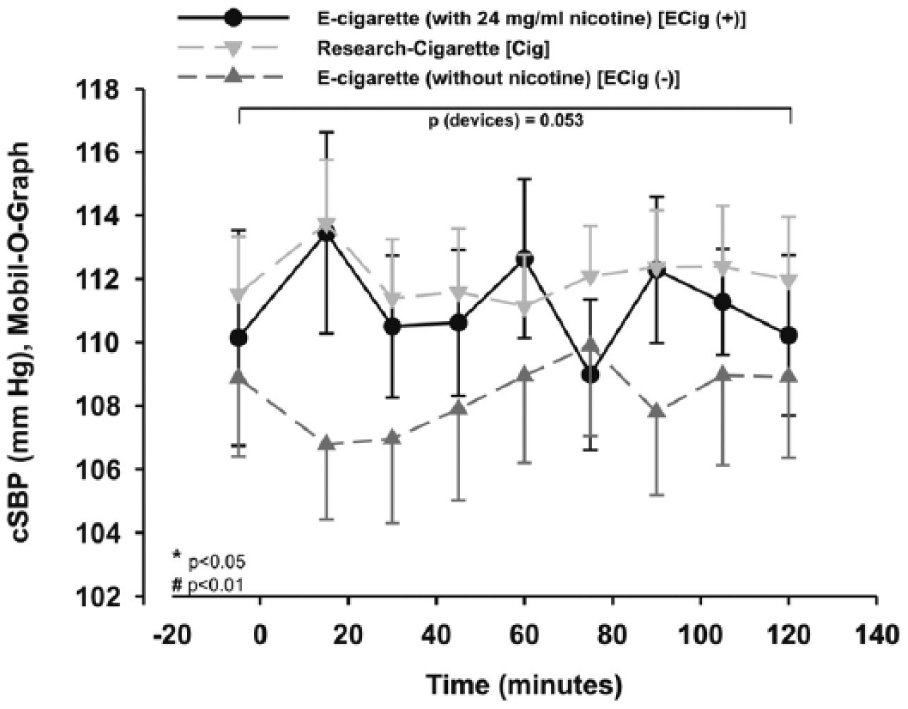
cSBP was not change significantly in the three different groups. Two-way measured ANOVA showed a trend without reaching significance between the devices (p=0.053).
The two-way repeated measures ANOVA for central diastolic blood pressure (cDBP) showed a significant difference between the different study groups as well as for time (p < 0.05). Using additional statistics, the cDBP of ECig (–) was significantly decreased during the first 30 minutes after smoking or vaping (p < 0.01 and p=0.005; Figure 7), and showed a trend of increasing within the Cig group without reaching significance within the first 15 minutes (p=0.064; Figure 7). However, there was no significant difference at any time point in the ECig (+) group (p > 0.05; Figure 7).
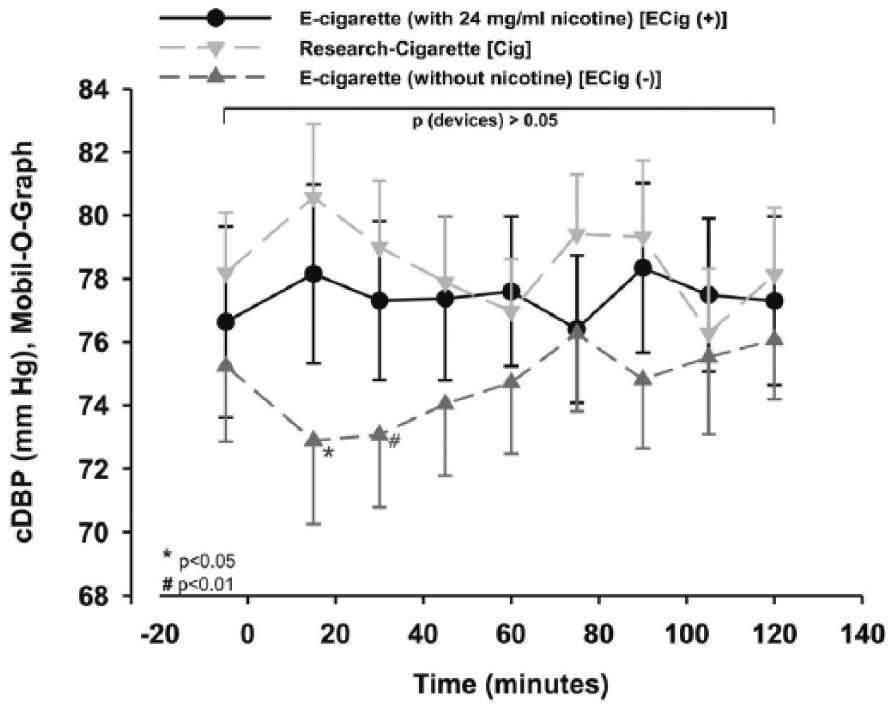
cDBP was decreased within ECig (-) using additional statistics. Two-way measured ANOVA showed a significant difference between the devices (p < 0.05).
All three groups differed significantly from each other, as analyzed by the two-way repeated measures ANOVA (p < 0.05) for AIx, which was adjusted for a HR of 75 beats per minute (AIx75). In more detail, using additional statistics, the AIx75 increased in the ECig (+) significantly 15 minutes after vaping (p=0.001), 30 minutes after vaping (p < 0.05), 60 minutes after vaping (p < 0.05), 75 minutes after vaping (p < 0.05) and 90 minutes after vaping (p < 0.05), and showed a trend to an increase without reaching significance 45 minutes after vaping (p=0.064; Figure 8). The Cig group showed a significant increase after the first (p < 0.01) and the third 15-minute interval (p < 0.05; Figure 8). There was no significance in the ECig (–) arm (p > 0.05; Figure 8).
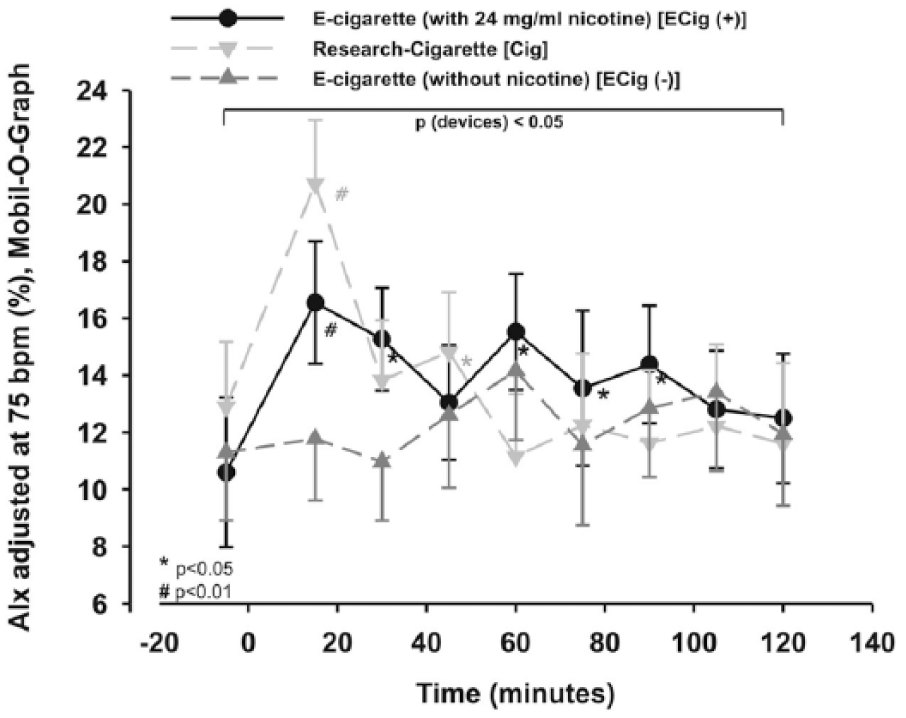
AIx adjusted for a heart rate of 75 bpm (AIx75). Two-way measured ANOVA showed a significance between the devices (p < 0.05), which remains after post hoc testing.
The PWV showed a significant difference between the study arms within the two-way repeated measures ANOVA (p < 0.01). In addition, the PWV showed a significant alteration after 15 minutes for ECig (+) (p < 0.05; Figure 9) and Cig (p < 0.01; Figure 9), whereas no significant change occurred within the ECig (–) arm (p > 0.05; Figure 9). ECig (–) was lowered compared to both of the other groups, showing a lowering trend without reaching significance for the first 15 minutes (p=0.086; Figure 9). After calculating multivariate analysis of PWV changes during the 15-minute intervals, changes remained significant in relation to blood pressure or HR (Box test: p > 0.05; Levene’s test: p < 0.05; multivariate tests (Wilks’ lambda): p < 0.05) for the ECig (+) and Cig arms.
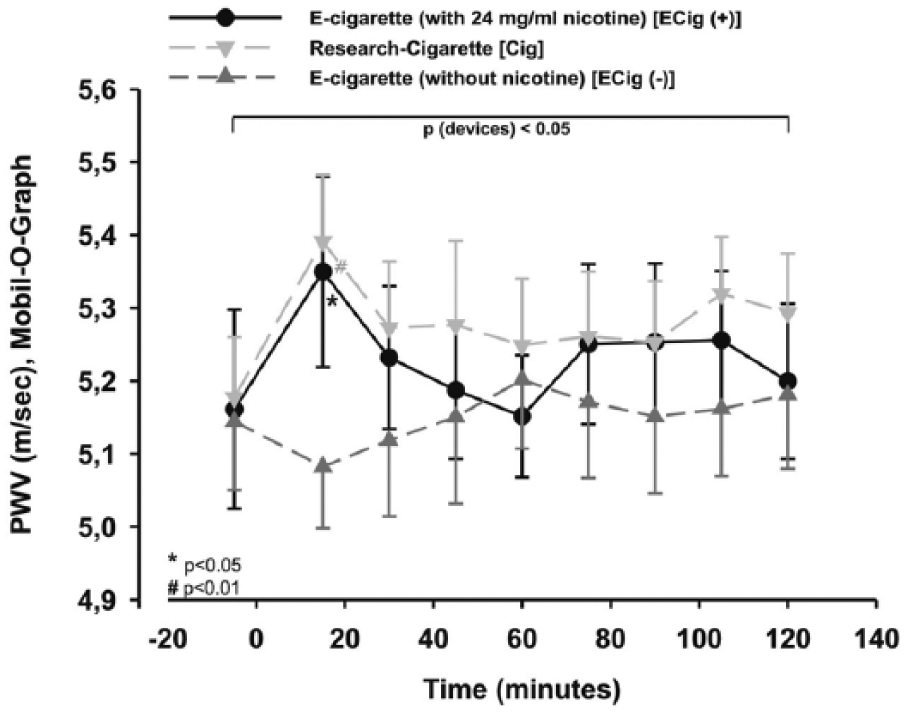
PWV was increased significantly within Cig and ECig (+). Two-way measured ANOVA showed a significance between the devices (p < 0.05), which remains after post hoc testing.
Discussion
In addition to published studies, this is the first study that has observed the effects of using cigarettes, e-cigarettes with nicotine, or e-cigarettes without nicotine – the latter used as a control group. In the present study, we demonstrate acute changes in peripheral and central blood pressure for a short period of time after vaping a liquid with nicotine or smoking conventional cigarettes. This is in line with previous data on cigarettes and e-cigarettes published by, for example, Vlachopoulos et al.24–28
The temporary lowering of DBP can be explained by a relaxation caused by the use of a device or cigarette which supports the finger–mouth coupling (i.e. there will be a short-term relaxation in the group with the nicotine-free liquid). Furthermore, this relaxation inhibits the expected increase in the other two groups. The expected increase is based on the data published by the working group of Mahmud and Feely. 29 The acute increase in SBP and arterial stiffness, especially for AIx75, could be explained by different mechanisms. These mechanisms could be triggered by an increase in circulating and local catecholamines and by nicotine. As already published, nicotine stimulates sympathetic ganglia and therefore increases sympathetic neuronal discharge-impaired nitric oxide production in the central nervous system.29–31
The pathogenesis of coronary heart disease and, consecutively, cardiovascular events, are triggered by a stiffening of the arteries.22,32 Besides the arterial stiffness, central blood pressures seem to be more important compared to peripheral blood pressures.18,33,34
Recently published trials have demonstrated discrepant results of the effects induced by vaping e-cigarettes.25,26 The working group of Szoltysek-Boldys did not show any significant effect of vaping or smoking on SBP, DBP or HR in a study on 15 healthy smokers. 25 However, they showed an increase of the Stiffness Index and Reflection Index. In contrast to this study, Vlachopoulos and colleagues analyzed a group of smokers. The authors showed that vaping an e-cigarette with nicotine-containing liquid had an impact on central hemodynamics as well as on peripheral blood pressure. 26 As previously shown,27,28,35 both cigarettes and e-cigarettes with a nicotine-containing liquid led to higher SBP, HR, PWV and adjusted AIx75, although the DBP decreased after vaping e-cigarettes with a nicotine-free liquid in our study.
In contrast to the published trials, there were trends and changes within all three different groups within the second half of the observation phase. The changes within the second hour ultimately remained unclear and could not be attributed to the effects of nicotine. This is due, among other things, to the physiological effect of nicotine on the body. One explanation could be that the subjects became restless during observation and looked forward to the end of the test. A passive and therefore secondhand exposure seemed unlikely due to air conditioning. In addition, each of the participants was examined individually and therefore distraction from another participant seemed unlikely.
Study limitations
Unlike the recently published trial, 26 this study was limited by consisting of three different study arms but the number of individuals tested was still limited. As a further limitation, the intensity of vaping the e-cigarette could not be standardized for the depth of breath of each puff. However, to avoid differences being too large, the frequency of puffs was defined. Furthermore, the e-cigarette without nicotine formally contained flavoring agents with the taste of tobacco and propylene was used. Both the liquid with and the liquid without nicotine did not contain the same number of additives as the cigarette. This should be considered in further studies. Another aspect, which has to been taken into account, was the fact that the concentration of nicotine (24 mg/mL) was quite high. However, we have chosen this concentration to illustrate expected effects.
Conclusion
This is the first trial to evaluate the acute changes in peripheral and central hemodynamics as well as PWV using an e-cigarette without nicotine as a control group. The increased parameters within the nicotine-containing devices might be a link to an increased cardiovascular risk, which is well known for cigarettes. Future trials should focus on the chronic effects of vaping nicotine-containing or nicotine-free liquids on peripheral and central blood pressures, as well as on arterial stiffness.
Since no endothelial dysfunction or sex differences have been described for the three different arms in the literature, it would be important for future trials to address these items. The number of laboratory values should be extended to include, for example, catecholamines.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was totally financed by Medizinische Klinik III of the Universitaetsklinikum Schleswig-Holstein (UKSH). The UKSH did not influence the interpretation or study design. However, as listed on the title page, some of the authors are employees of the Medizinische Klinik III of the UKSH.
