Abstract
A Dacron wrap aortoplasty has been described as an adjunct to reduction ascending aortoplasty for the repair of Type A aortic dissections. We report a case of an uncomplicated hemiarch repair with wrap aortoplasty of the distal anastomosis which resulted in severe acute hemolysis. Despite only minimal focal graft deformation on imaging, the patient was found to have a flow gradient across the distal anastomosis, which was reduced by > 50% following release of the outer graft wrap. To our knowledge, only 29 additional cases of hemolytic anemia following aortic dissection repair have been described in the English literature. The reported mechanisms included aortic graft stenosis (50%), graft kinking (23%), external compression of the graft (20%), and a folded elephant trunk appendage (7%). The mean onset of hemolysis following aortic dissection repair occurred 32.2 ± 44.4 months after surgery, with only 16.7% of cases occurring within 2 weeks. This review details the clinical, laboratory, and imaging findings suggestive of mechanical hemolysis following aortic surgery.
Introduction
Type A aortic dissections are surgical emergencies that occur in three to four out of 100,000 patients annually. 1 If left untreated, mortality in the initial 24–48 hours is approximately 2% per hour. 2 While the most common surgical approach for Stanford Type A dissections involves replacement of the dissected aorta with a synthetic graft, adjunct maneuvers have been described, including a secondary wrap around the aortoplasty.3,4 A recent study found the operative mortality associated with acute Type A aortic dissection repair across 12 experienced referral centers to be 26%. 5 Hemolytic anemia secondary to aortoplasty is a relatively rare complication that has been previously reported. To our knowledge, 29 cases have been described in the English literature.6–28 We report the 30th case and review the literature for clinical, laboratory, and imaging findings indicative of mechanical hemolysis following aortoplasty.
Case report
A 57-year-old African American male with a history of hypertension and obstructive sleep apnea was found to have a 5.3 cm aortic root on transthoracic echocardiogram during evaluation for a right hemispheric cerebrovascular accident the previous year. A contrast-enhanced computerized tomography (CT) scan revealed a Type A aortic dissection originating at the sinotubular junction, extending to the distal arch with involvement of 1–2 cm of the proximal innominate artery. The patient underwent hemiarch repair of the ascending aorta with resuspension of the native aortic valve using a 26 mm Hemashield (Maquet; Rastatt, Germany) tube graft with wrap aortoplasty of the distal suture line for hemostatic purposes. The wrap aortoplasty was fashioned using another section of 26 mm woven polyester graft opened longitudinally, positioned across the distal anastomosis, and approximated using interrupted 3-0 polypropylene sutures (Figure 1A). Antegrade cerebral perfusion was utilized during deep hypothermic circulatory arrest. The patient recovered well and was discharged home on postoperative day 6.
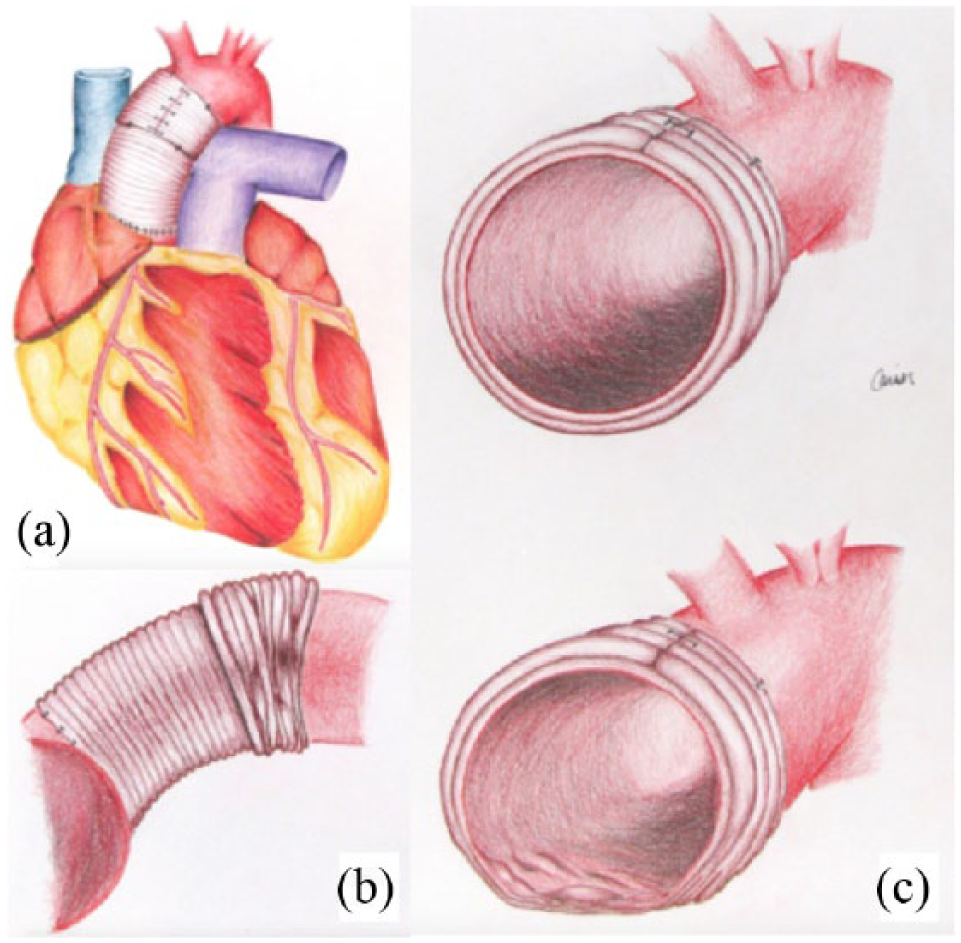
Schematic representation of the (a) wrap aortoplasty, (b) subsequent inferior wrap migration, and (c) subtle graft deformation leading to hemolysis.
Three days after discharge, the patient re-presented with shortness of breath, dizziness, and progressively darkening urine. Workup revealed a hemoglobin of 5 g/dL, lactate dehydrogenase (LDH) of 3116 U/L, haptoglobin < 4 mg/dL, total bilirubin of 2.4 mg/dL, and unconjugated bilirubin of 0.72 mg/dL, all suggestive of hemolysis. A peripheral blood smear showed marked schistocytes, marked polychromasia and few dacrocytes. Further investigations revealed a negative direct antiglobulin test and ADAMSTS13 activity of 90%. His creatine phosphokinase (CPK) on admission was 960 U/L and his creatinine rose to 3.9 mg/dL over the next 4 days despite aggressive hydration. Given his acute kidney injury, a chest magnetic resonance imaging (MRI) scan without contrast was obtained that revealed minimal focal kinking of the ascending aortic graft (Figure 2). The aortic graft was widely patent with a turbulent jet at the level of the distal anastomosis, suggesting a mild degree of narrowing. Transthoracic echocardiography demonstrated an intact aortic valve without evidence of regurgitation and no ascending aortic gradient. Additionally, an exhaustive evaluation was undertaken to ascertain the etiology of the hemolysis, including assays for red cell dyscrasias and hemoglobinopathies, all of which were negative (Table 1).
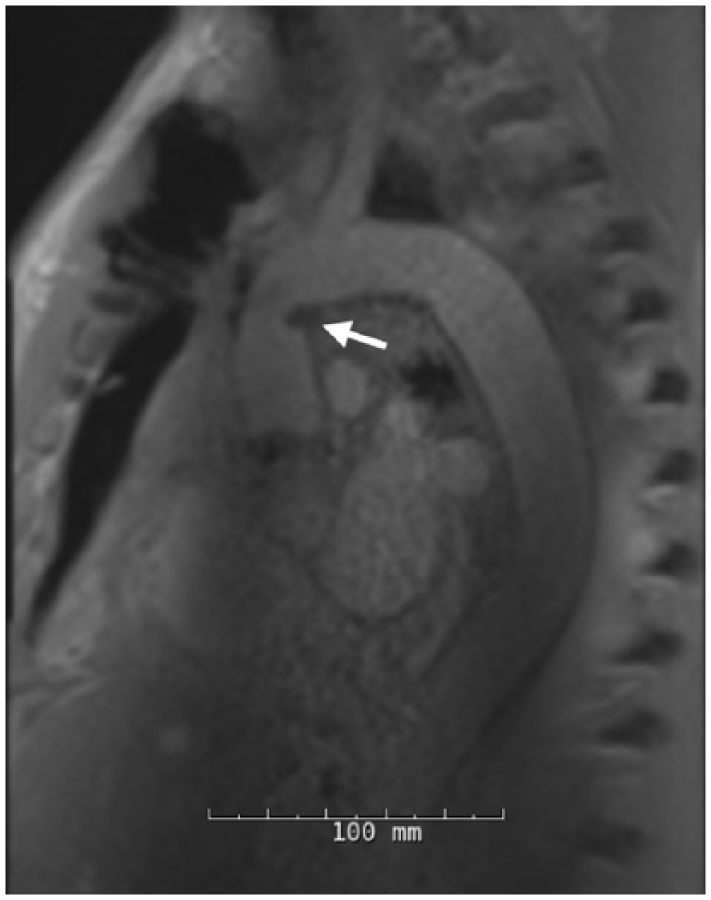
A non-contrast MRI demonstrating focal kinking of the distal hemiarch anastomosis.
Summary of hematology, imaging, and microbiology studies utilized during the workup of hemolytic anemia following aortoplasty.

Denotes studies that were not utilized in the current case workup.
ANA, anti-nuclear antibody; ANCA, anti-neutrophilic cytoplasmic antibody; AP, anteroposterior; CPK, creatinine phosphokinase; CT, computed tomography; G6PD, glucose-6-phosphate-dehydrogenase; LAT, lateral; LDH, lactate dehydrogenase; MRA, magnetic resonance angiography; MRI, magnetic resonance imaging; PA, posteroanterior; TEE, transesophageal echocardiogram; TTE, transthoracic echocardiogram.
Considering the patient’s rapid clinical decline, the presence of a turbulent jet identified at the distal anastomosis site, and the absence of an alternative etiology for hemolysis, the decision was made to re-explore the patient on postoperative day 13. Externally, the graft appeared normal without obvious kinking. Intraoperative flow measurements using transesophageal echocardiography (TEE) and direct aortic puncture indicated elevated velocities and an approximate gradient of 8 mmHg across the distal anastomosis. The Dacron wrap was removed by cutting the interrupted 3-0 polypropylene sutures. The graft had migrated slightly along the underside of the aorta, resulting in intralumenal irregularities and some external constriction (Figures 1b and 1c). Serial Doppler velocity and direct pressure measurements were taken and immediately revealed a reduction in the gradient across the distal anastomosis, with a mean gradient of only 3–4 mmHg. There were no other areas of elevated velocities, turbulent flow patterns, or elevated gradients identifiable. Intraoperative LDH and CPK levels were 3070 U/L and 1025 U/L, respectively. Serial urine samples demonstrated rapid pigment clearing. The patient did well postoperatively and was discharged home 13 days following his revision with improving renal function, stable hemoglobin, and down-trending LDH and CPK (1210 U/L and 430 U/L, respectively). Seven months later, his LDH was 336 U/L, hemoglobin 13.3 g/dL, and creatinine 1.12 mg/dL.
Systematic review
A systematic review of the PubMed, Medline and EMBASE databases was performed in August 2017 using Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines with a PRISMA checklist. The search was performed using combinations of the following MESH search terms: Anemia, hemolytic; Aneurysm, dissecting/surgery; Aorta/pathology; Aorta/surgery; Aorta, thoracic/surgery; Blood vessel prosthesis; Graft occlusion, vascular/complication; Hemolysis; Prosthesis failure. All references within included studies were cross-referenced for potential inclusion if omitted from the initial search. The inclusion criteria for full-text review were case reports, review papers, and peer-reviewed publications. Exclusion criteria included non-English language articles. The literature search yielded 23 peer-reviewed case reports and review papers which collectively reported 29 unique cases of hemolysis following aortoplasty for Type A dissection (Table 2).6–28
Summary of reported cases of hemolytic anemia following aortic dissection repair.
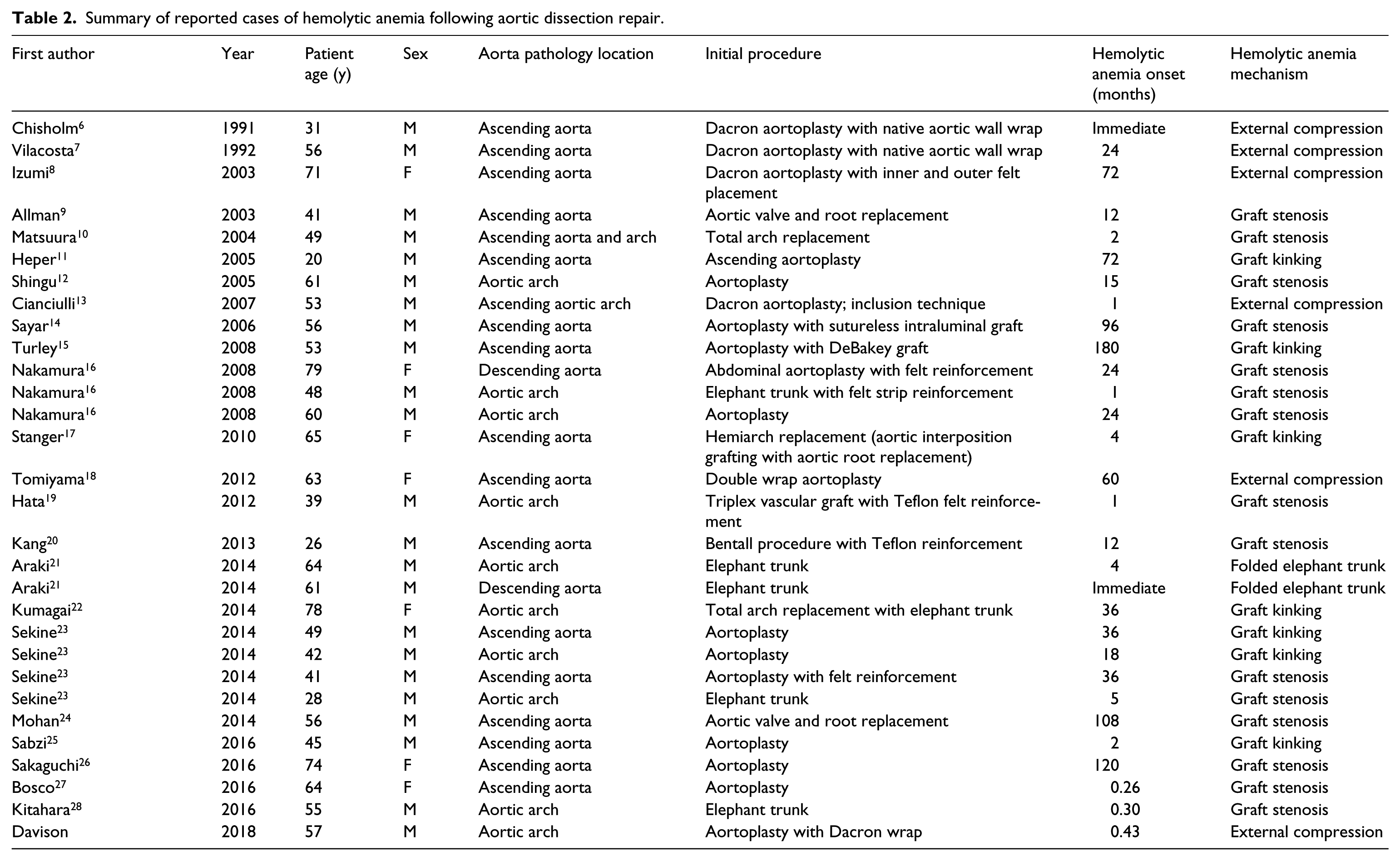
Including the current case report, 30 complications of hemolysis have been described in the medical literature. The average age of the affected patients was 52.8 years ±14.9 years and 77% (n=23) were male. The mechanisms of hemolysis consisted of graft stenosis (50%), Dacron graft kinking (23%), external graft compression (20%), and a folded elephant trunk (7%). The onset of hemolysis ranged from the immediate postoperative period to 180 months following aortoplasty, with the average onset being 32.2 ± 44.4 months.
Discussion
The conventional treatment for Stanford Type A aortic dissections is replacement of the dissected aorta with or without aortic valve repair or replacement. A Dacron wrap aortoplasty at the distal anastomosis for hemostatic purposes, while not a standard component of Type A dissection repair, has been described as an adjunct to reduction ascending aortoplasty 3 and for rapid hemostasis of suture lines following aortic replacement. 4 A similar technique was recently described for Type A dissection repair with root involvement as it serves as structural reinforcement, mitigates intraoperative bleeding, and may assist in maintaining aortic root geometry. 29 In the current case, the wrap aortoplasty was performed prophylactically in order to facilitate hemostasis. There were no issues with the surgical technique in this particular case. We have utilized this procedure without severe postoperative hemolysis in over 100 cases to date. While we feel it is a useful technique, many surgeons do not use it, stating that it may lead to additional tension at the suture lines, peri-prosthetic hematomas, or pseudoaneurysm formation.6,13 Epiaortic ultrasound, TEE, or direct aortic pressure measurements to evaluate flow characteristics during the index operation may be useful adjuncts to avoid this complication in future instances when a wrap aortoplasty is employed. In retrospect, tacking the wrap circumferentially may have prevented wrap migration in the current case.
To date, 30 cases of hemolytic anemia secondary to aortoplasty have been reported in the English literature (Table 2).6–28 The reported mechanisms included aortic graft stenosis (50%), graft kinking (23%), external compression of the graft (20%), and a folded elephant trunk appendage (7%). Graft stenosis was largely attributed to the inversion of Teflon strips; however, other cases involving implant narrowing secondary to graft dehiscence or dissection flap obstruction at the distal anastomosis have been reported.9,24 Cases of external graft compression mostly resulted from suture line hemorrhage and adjacent pseudoaneurysm formation. To our knowledge, the current case of hemolysis due to external graft compression from a constricting outer Dacron wrap aortoplasty is the first in the literature. While Tomiyama et al. also reported hemolysis complications with the use of a Dacron wrap aortoplasty, their case involved blood flow into an outer false lumen with subsequent narrowing of the true lumen. 18
Most commonly, hemolysis after Type A aortic dissection repair occurs in a delayed fashion, often months to years after surgery (Table 2). A review of the literature revealed that the mean onset of hemolysis following aortic dissection repair occurred 32.2 ± 44.4 months after surgery, with only 16.7% of cases occurring within 2 weeks. The causes for this variability in presentation remain unknown. When comparing mechanisms between acute-onset hemolysis (within 2 weeks postoperatively) and more delayed presentations, no incidences of hemolytic anemia associated with synthetic graft kinking were noted in the acute presentation group. This is notable as graft kinking was reported as the causal mechanism in 23% of all the cases reported in the literature. Additionally, there were no reports of aortic valve regurgitation in the acute-onset cases, which was a finding commonly observed with delayed presentations. It is possible that in the early-onset cases, there was not enough time for sufficient aortic root dilation to result in a regurgitant aortic valve.
The characteristics and degree of aortic valve intervention during the primary dissection repair were considered as possible contributors to the incidence and time course of hemolysis, as valvular alterations may alter the flow mechanics through the repaired aorta. A review of the previously published cases revealed that only 10 (33%) contained comments pertaining to the primary aortic valve intervention. Of these cases, 40% involved resuspension of the native aortic valve, 40% utilized mechanical valve replacement, 10% involved bio-prosthetic valve replacement, and 10% used a composite graft replacement. Considering the paucity of information available, general patterns of hemolysis relating to aortic valve manipulation could not be discerned.
There is no single test that is diagnostic for hemolytic anemia; instead, a combination of clinical and laboratory findings are often necessary to make the diagnosis (Table 1). Hemolysis secondary to mechanical factors related to graft kinking presents as extracorpuscular, non-immune, intravascular hemolysis similar to that described following prosthetic valve replacement, except often more acute and severe. Common laboratory tests used to characterize hemolysis and suggest a probable cause include serum LDH, peripheral blood smear, reticulocyte index (RI), total bilirubin, direct and indirect bilirubin, and direct antiglobulin test (Coombs test). Although there is limited evidence demonstrating quantitative predictive value for many of these parameters, abnormalities across multiple tests, and in some cases the degree of abnormality, can suggest an etiology as well as severity of disease.
LDH is a non-specific marker of cell turnover and tissue destruction that can be abnormally elevated in a variety of pathologies. It is ubiquitously elevated in all forms of hemolysis, mostly comprised of LDH-1 and LDH-2 isozymes, and can be useful in trending treatment response. 30 In some cases, degree of elevation may be useful in distinguishing extravascular versus intravascular etiologies of hemolysis, as more significant elevations are generally seen in the latter. In the current review, reported serum LDH values ranged widely from modest elevations of 299 IU/L to values as high as 4892 IU/L.8,23
Another biomarker, serum haptoglobin is significantly decreased in both intra- and extravascular hemolysis. Prior studies have estimated a sensitivity and specificity of 83% and 96%, respectively, of haptoglobin levels ⩽ 25 mg/dL in discriminating hemolytic disorders from non-hemolytic disease. 31 Though less frequently reported than LDH in the literature, haptoglobin levels were significantly below this threshold in all but one case, which paradoxically noted a serum haptoglobin of 158 mg/dL. 23 Importantly, serum haptoglobin concentrations can be confounded in a variety of scenarios such as hepatic cirrhosis, hemodilution or inflammatory states. 32
Reticulocyte response is evaluated to determine the adequacy of bone marrow response to anemia, and in cases of isolated mechanical hemolysis secondary to aortic graft complications (without concomitant marrow pathology) an appropriate compensatory increase is expected. This would be indicated by a calculated RI [(reticulocyte count × (patient’s hematocrit/normal hematocrit))/hematocrit-dependent maturation factor] greater than 2%. Reticulocyte counts reported in this series of cases ranged from 2.6% to 45%.16,23
The presence of schistocytes on a peripheral blood smear is a characteristic finding of mechanical hemolysis as well as pathologies that result in microangiopathic hemolytic anemia (thrombotic thrombocytopenic purpura (TTP), hemolytic uremic syndrome (HUS), disseminated intravascular coagulation (DIC), pre-eclampsia/eclampsia, and malignant hypertension). In a healthy individual, these fragmented cells represent < 0.5% of identifiable structures. An absence of other abnormal cell morphologies (spherocytes, hypochromic/microcytic erythrocytes, sickle cells, etc.) is helpful in ruling out other causes of anemia. Findings reported in the literature range from rare schistocytes to severe fragmentation, with damaged erythrocytes representing 6% of the population on a peripheral blood smear.24,28 There is no defined schistocyte count that is diagnostic of mechanical hemolysis. 30
Elevation in indirect bilirubin is also frequently reported in cases of mechanical hemolysis. Various urinary findings such as hematuria, hemoglobinuria, hemosiderinuria, and bilirubinuria have also been described in patients with intravascular hemolysis. Urinary findings were not often reported in the cases reviewed; however, some patients presented with hematuria and/or hemoglobinuria.14,17,18,24,25 The Coombs test represents an essential component of the workup in hemolytic anemia in order to rule out immune-mediated hemolysis.
We examined the literature to compare trends in cases of acute hemolysis and delayed presentations. We found no significant difference in average serum hemoglobin (7 g/dL vs 7.46 g/dL, p=0.672) or LDH (2182 U/L vs 2133 U/L, p=0.940), the two most consistently reported biomarkers of hemolysis, between acute and delayed presentations of hemolysis following aortic repair. There were subjective differences reported in parameters showing increased average reticulocytes (5% vs 8.2%) and total bilirubin (2.1 mg/dL vs 3.6 mg/dL) in delayed presentations; however, these biomarkers were inconsistently reported and are likely not statistically significant. There is insufficient data to draw conclusions about serum haptoglobin, creatinine, or proportion of schistocytes with regard to acute versus delayed-onset hemolysis.
Various imaging modalities have been used to characterize aortic graft complications in cases of suspected mechanical hemolysis, such as: transthoracic echocardiogram (TTE) and TEE with flow Doppler evaluation, CT with or without three-dimensional construction, computed tomography angiography (CTA), MRI, magnetic resonance angiography (MRA), and conventional angiography. Limitations of these studies include difficulty in evaluation of the distal aspect of the aortic arch with TEE 7 or lack of sufficient detail with TTE/TEE and standard thoracic CT to characterize more subtle graft stenosis related to inversion of a segment of felt. Some authors have suggested that the most important imaging finding suggestive of mechanical hemolysis secondary to a graft complication is a ‘mosaic pattern’ on echocardiography representative of turbulent blood flow. 26 This finding, when paired with a typical pattern of laboratory values, should increase suspicion for mechanical hemolysis secondary to graft complications and may prompt reoperation to correct a probable defect.
In the current case, the only radiographic indication for re-exploration was a mild focal Dacron graft constriction without significant flow voids on chest MRI (Figure 2). This is different than many of the previously reported cases of hemolysis resulting from aortic valve regurgitation, peri-prosthetic leak, felt inversion, or pseudoaneurysm formation. These findings are often more readily demonstrated with chest CT, TTE, or TEE than mild graft kinking.7,13 Considering the minimally invasive nature of these imaging modalities, CT, MRI, TTE, and TEE should be routinely used in the workup of patients with suspected mechanical hemolysis following aortic surgery.
Chisholm et al. reported a case of postoperative hemolysis in which their diagnostic workup of a hemodynamically unstable patient 8 hours following aortoplasty was inconclusive. In response to the patient’s rapid clinical deterioration, an emergency cardiac catheterization was performed, revealing a 40 mmHg flow gradient at the level of the graft prompting reoperation. 6 In the current case, intraoperative flow measurements revealed elevated velocities and a pressure gradient across the distal anastomosis suggesting that a preoperative cardiac catheterization may have revealed similar findings. However, the authors decided against preoperative percutaneous catheterization as it was felt that surgical re-exploration was ultimately necessary and the most expeditious approach.
Synthetic aortic graft kinking is not a postoperative complication exclusive to the ascending aorta. Fransen et al. examined the frequency of aortic stent graft kinking following endovascular abdominal aortic aneurysm repair in 4613 patients from the EUROSTAR registry. 33 The authors identified 170 (3.7%) patients with graft kinking with a mean follow-up of 21 months. It was noted that the presence of abdominal aortic graft kinking was most often associated with both proximal and distal type I endoleaks, type III endoleaks, graft stenosis, graft limb thrombosis, and graft migration. To our knowledge, no cases of hemolytic anemia secondary to abdominal or descending aortic graft constriction or kinking have been reported. It remains unclear why hemolytic anemia has been reported from constriction of ascending aortic grafts but not an abdominal aortic graft. One theory is based on differences in the mechanical environment between the ascending and abdominal aorta. Specifically, the differing flow velocities, with the abdominal aorta having lower velocities than that of the ascending aorta. Perhaps aortic graft hemolysis is instigated by a combination of excessive shear forces in the arch and any luminal abnormalities. 34
To complicate the matter, disseminated intravascular coagulation (DIC) has also been reported in association with endovascular aneurysm repair (EVAR).35–38 Intravascular hemolysis is seen with DIC, which could present similarly to mechanical hemolysis secondary to graft complications (dyspnea, fatigue, jaundice or pallor, flow murmurs, hematuria/hemoglobinuria). A link has been suggested between the development of DIC and the presence of an endoleak after EVAR. Of the various classifications of endoleaks reported, the most common is a type II endoleak, with an incidence of 10.2% to 22%.39,40 Despite its frequency, there has only been a single reported case of DIC with a type II endoleak 38 and rare reports of DIC associated with type I35–37 and type III 37 endoleaks.
DIC and intravascular mechanical hemolysis can both result in microangiopathic hemolytic anemia and produce similar laboratory findings including anemia, increased LDH, decreased haptoglobin, reticulocytosis, increased bilirubin, and a negative Coombs test. The presence of schistocytes is also a characteristic of hemolysis secondary to DIC; however, the prominence of schistocytes may be lower in DIC, typically between 0.5% and 1% 30 compared to the reported prevalence of 6% fragmented red blood cells on a peripheral smear in a case of hemolysis due to graft stenosis following elephant trunk repair of the aortic arch. 28 DIC can be differentiated from hemolytic anemia secondary to graft kinking or stenosis by characteristic laboratory abnormalities such as thrombocytopenia, elevated coagulation times (prothrombin time (PT), activated partial thromboplastin time (aPTT)), decreased fibrinogen, and increased fibrin degradation products and d-dimers which are not typical of isolated mechanical hemolysis. 41
Conclusion
Hemolytic anemia secondary to aortoplasty for the treatment of Type A aortic dissection is a relatively rare complication. While hemolysis typically occurs months following aortoplasty, a certain percentage of patients experience hemolysis within 2 weeks of surgery and tend to deteriorate rapidly. Therefore, it is important to recognize the characteristic clinical, radiographic, and laboratory findings associated with postoperative hemolysis so that the diagnosis can be rapidly ascertained and managed appropriately. While no single finding is diagnostic, clinicians should instead consider the constellation of findings when evaluating these patients.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
