Abstract
The objective of this study was to determine how postoperative skin perfusion pressure (SPP) as a measure of blood flow after revascularization affects limb prognosis in patients with critical limb ischemia (CLI). We retrospectively reviewed 223 consecutive bypass surgery cases performed in 192 patients with CLI during a 10-year period. SPP was measured 1–2 weeks before and after the procedure. An SPP of 40 mmHg was set as the cut-off value for revascularization. Patients were grouped according to their postoperative SPPs, and amputation-free survival (AFS) was analyzed. An SPP of ≥ 40 mmHg was recovered in 75% of the patients, but no significant difference was found between this group and the group that did not reach 40 mmHg. On the other hand, the values increased by ≥ 20 mmHg from the preoperative values in 70% of the patients. This group had a significantly better AFS than the group that did not increase by 20 mmHg. Logistic regression analysis revealed that (1) a preoperative SPP of < 20 mmHg and (2) a high serum albumin level (> 3.0 g/dL) were significant factors in increasing SPP by 20 mmHg. These results showed that an increase in SPP of ≥ 20 mmHg after bypass surgery was associated with better limb prognosis.
Keywords
Introduction
Skin perfusion pressure (SPP) is a non-invasive tool for predicting the natural course of wound healing in patients with critical limb ischemia (CLI).1–5 Brownrigg reported the superiority of SPP to ankle–brachial index (ABI) for predicting wound healing. 4 This can be explained by the high medial calcification rate of lower limb arteries in CLI patients, which could falsely elevate ABI. 6 For the past two decades, we have adopted SPP to evaluate lower limb blood flow for patients with tissue loss or gangrene.
SPP has been evaluated as a tool to assess limb perfusion, whereby a threshold value may prompt revascularization when tissue perfusion is insufficient.2,5 While some studies report a threshold SPP of 30 mmHg for poor prognosis, 5 others have reported 40 mmHg as the predictor.2,7,8 From these results, we set an SPP of 40 mmHg as an indicator of ischemia. Values of < 40 mmHg would identify candidates for revascularization consisting of bypass surgery or endovascular treatment (EVT).
On the other hand, only a few studies have investigated whether this threshold is sufficient to expect wound recovery specifically after revascularization.9,10 For example, when an SPP of 40 mmHg was used as a revascularization threshold, whether restoring this value when performing intervention enables limb recovery and has prognostic ability remains to be answered. The procedures and contexts that affect recovery, especially in terms of SPP, remain unknown.
Here, we aimed to discover the correlation between postoperative SPP results (absolute values and increase after the intervention) and long-term prognosis (amputation-free survival (AFS)) in patients with CLI. In doing so, we also aimed to identify factors that can be used to predict these SPP changes.
Materials and methods
This retrospective cohort study adhered to the Declaration of Helsinki, and written informed consent was obtained from each participant before the surgery. This included the agreement to use their data throughout their treatment and follow-up for CLI. This study, which used the aforementioned retrospective data, was approved by the research ethics committee of our institution (Chairperson Nobuhiko Haga; protocol number: 3316-(2); date of approval: March 14, 2016).
All the patients who underwent bypass surgeries for CLI between 2005 and 2016 were enrolled. Patients with symptoms of aneurysmal or iatrogenic origin were excluded from the study. Reinterventions involving a bypassed graft or inserted stent were also excluded.
Generally, we perform revascularization for all patients with CLI due to peripheral artery disease who visit our hospital. CLI was defined as: (1) local status of Rutherford class ≥ 4 11 and (2) accompanied by ischemia. For the diagnosis of ischemia, SPP of the dorsal and plantar portions of the foot were measured preoperatively in each patient by using a LASERDOPP PV2000 (Vasamedics, St Paul, MN, USA) or PAD 4000 (Kaneka Corporation, Tokyo, Japan). In short, SPP was obtained by inflating a cuff over the skin at the measurement point above the systolic blood pressure to cease the blood flow. The cuff was then slowly deflated until an optical sensor detected the return of blood flow by using the laser Doppler technology. The examination was usually performed in the afternoon after approximately 30 minutes’ rest. In case of an open wound at the point of measurement, we measured the SPP at a viable skin location just proximal to the wound. Further details of the measurements have been explained previously.12–14 The lower value of the two measurement points was determined as the SPP of the foot, and an SPP of < 40 mmHg was used as the basis for diagnosis of ischemia and a criterion for surgery. 2
Generally, bypass surgery is selected whenever possible for patients with CLI. The target artery for each anastomosis was decided according to the status of the artery, such as calcification and stenosis. We took angiosome into account only if there were multiple arteries for anastomosis with equal status. We did not perform bypass surgery in patients who were considered intolerant of the operation because of their medical comorbidities, had no recipient artery for bypass, had an infection or gangrene that invaded the anastomosis site, had no usable vein available for grafting (< 2.5 mm in diameter), or refused to undergo the operation. If we considered the patients unfit for bypass surgery, we would then offer EVT if it was possible. However, because of this policy, if we included the patients undergoing EVT in this study, it was possible that the results would show the inferiority of EVT due to referral and selection bias. Therefore, in this study, we have excluded the patients who underwent EVT.
The recorded preoperative characteristics included age, sex, hypertension, diabetes, dyslipidemia, renal failure, smoking status, serum albumin values, ejection fraction measured using transthoracic echocardiography, history of coronary artery disease (CAD) or cerebrovascular disease (CVD), and ambulatory status at 1 month before admission. Patients were considered hypertensive if they were under medication or if their blood pressure was > 140/90 mmHg and as diabetic if they were under medication for diabetes or if their hemoglobin A1c level was > 7.0% with or without medication. 15 Current smokers were defined as those who had been smoking within 6 months of admission. Patients were considered ambulatory if they could walk with or without a cane. As reported by Taylor, non-ambulatory patients were further classified as ‘non-ambulatory transfer’ if they could move into a wheelchair without assistance, while the remaining patients were classified as ‘non-ambulatory bedridden’. 16 The index wound was characterized according to the Wound, Ischemia, and foot Infection (WIfI) criteria. 17
Intraoperative data included whether surgery revascularized direct flow to the ankle (i.e. ‘straight line to the ankle’). ‘Distal’ bypass was defined as bypass to the crural arteries or below. ‘Long’ bypass was defined as a distal bypass with inflow from the femoral arteries or above. The region of revascularization was categorized as iliac, femoropopliteal, or inferior to the knee. For example, distal bypass from the common femoral artery to the posterior tibial artery was recorded as revascularization for two regions (i.e. femoropopliteal and below-the-knee).
Our general rule for wound treatment was to irrigate the wound with saline, but the treatment after this differed before and after the intervention. Preoperatively, we would use iodine ointment to treat or prevent infection. Postoperatively, we would use basic fibroblast growth factor (bFGF) spray to promote granulation presuming that the blood flow to the wound has increased after revascularization. Negative wound pressure therapy would be used if necessary.
The patients were followed up 1–2 months after surgery to evaluate the wound and graft patency. All the patients underwent antiplatelet therapy, but prostaglandins and statins were used only when indicated within our national health insurance. In addition, postoperative SPP was obtained 1–2 weeks after surgery, and the effects of preoperative and intraoperative data on postoperative SPP were analyzed. This time point for the postoperative SPP recording was set according to the report by Saucy. 18 In addition, 1–2 weeks after surgery was when the patients’ pain was stable and, for most of the patients, just before leaving the hospital. Just like the preoperative SPP, the postoperative SPP was still the lower value of the two measurement points and not the value of the same preoperative measurement point. We assumed that the prognosis of the limb was related to the foot as a whole. We validated whether the intervention recovered the SPP of 40 mmHg.
We also evaluated the increase in SPP after the intervention. We determined the optimal cut-off value of SPP increase by using two methods. The first was by using the Youden index (sensitivity + specificity − 1) of the receiver operating characteristic (ROC) curve, and calculated area under the curve (AUC). To obtain the ROC curve, we needed to choose a time point which was of clinical interest.
19
We chose 1 year after the intervention as the time point, considering the severity of CLI. The direct estimation method was used to account for censored patients before 1 year after intervention.
20
As the second method, Harrell’s concordance index (C-index) was used.21,22 We calculated the C-index at several cut-off values and searched for a cut-off with a higher C-index. The resulting
All data were analyzed using the JMP® Pro software, Version 12 (SAS Institute Inc., Cary, NC, USA), and
For survival analysis regarding postoperative SPP, we excluded patients whose graft was occluded during the follow-up period because SPP could have been altered after graft occlusion. The Kaplan–Meier method was used to evaluate survival. Intergroup comparisons were performed using univariate analysis with the log-rank test. The primary end point of this study was AFS. Regarding secondary end points, we calculated limb salvage and survival rates. Limb salvage was defined as no amputation above the ankle.
Results
Within the study period, 419 bypass surgeries were performed. Of these, surgeries that were performed for patients without CLI symptoms, additional surgeries for bypassed graft or inserted stent, and performed for patients without preoperative and postoperative SPP data were excluded, as explained in the Methods section. This left 223 bypass surgeries performed in 192 patients. The preoperative baseline characteristics and intraoperative factors are shown in Table 1.
Preoperative and intraoperative characteristics of the patients (
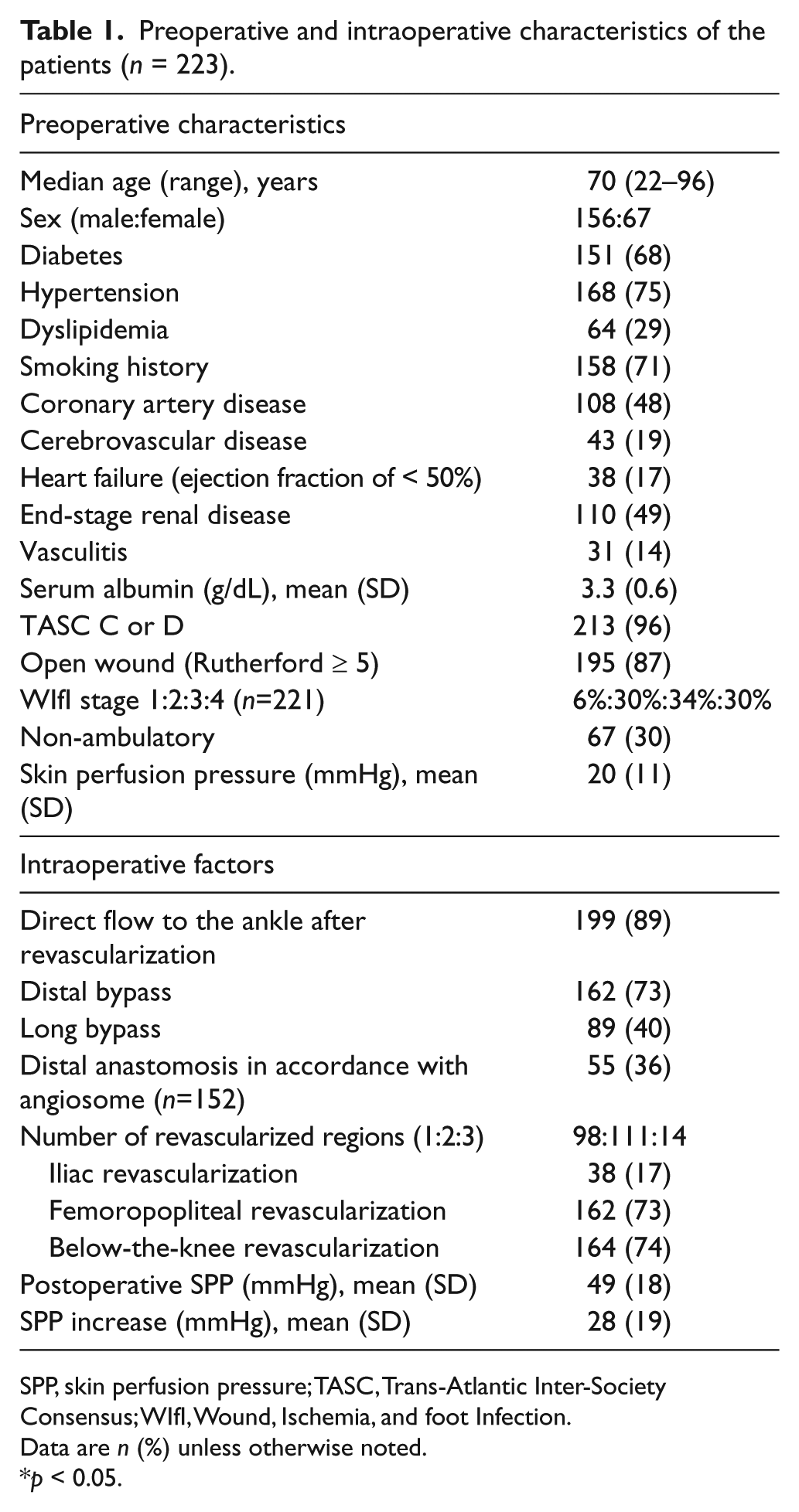
SPP, skin perfusion pressure; TASC, Trans-Atlantic Inter-Society Consensus; WIfI, Wound, Ischemia, and foot Infection.
Data are
Factors related to postoperative SPP
To analyze the correlation between prognosis and postoperative SPP, we restricted the materials to procedures performed for Trans-Atlantic Inter-Society Consensus II (TASC II) C or D lesions with confirmed graft patency. This left 189 procedures in 163 patients to be followed up for a median of 2.4 years (range, 0–11.6 years). During this period, 15 major amputations and 45 deaths occurred.
For prognostic analysis, we grouped the patients in two ways: (1) by whether or not the postoperative SPP recovered 40 mmHg, which was the cut-off value used as the indication for revascularization; (2) according to the increase in SPP after revascularization. We adopted two methods to calculate the optimal cut-off value. The Youden index showed an increase of 20 mmHg as optimal and the ROC curve showed an AUC of 63% (
Prognosis and postoperative SPP
There were 141 bypass surgeries (75%) in which the SPP was ≥ 40 mmHg after revascularization. However, when this group was compared to the group that did not reach 40 mmHg, AFS did not exhibit any significant change (Figure 1A). Meanwhile, 132 bypass surgeries (70%) resulted in an increase in the SPP value by at least 20 mmHg from its preoperative value. This group had a significantly better prognosis than the group that had not increased its SPP by 20 mmHg (Figure 1B).
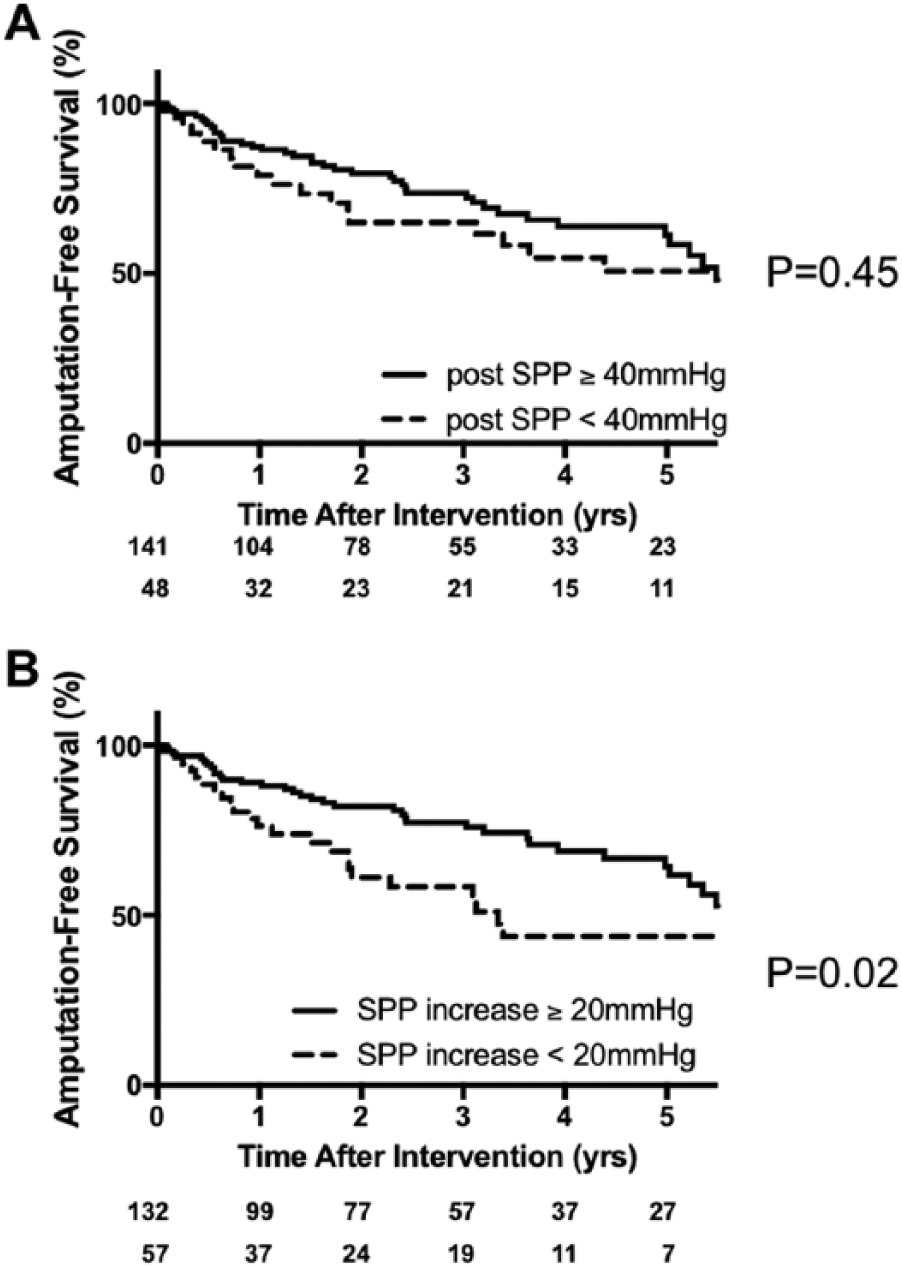
Amputation-free survival rates after bypass surgery grouped according to the presence or absence of (A) a postoperative SPP of ≥ 40 mmHg and (B) an increase in postoperative SPP of ≥ 20 mmHg.
The survival curves regarding the secondary end points are shown in Figure 2 and Figure 3. Both the limb salvage rate and overall survival rates showed no significant differences in both cut-off SPPs. However, the SPP increase tended to be more associated with overall survival than with limb salvage rate, especially after a few years (Figures 2B and 3B).
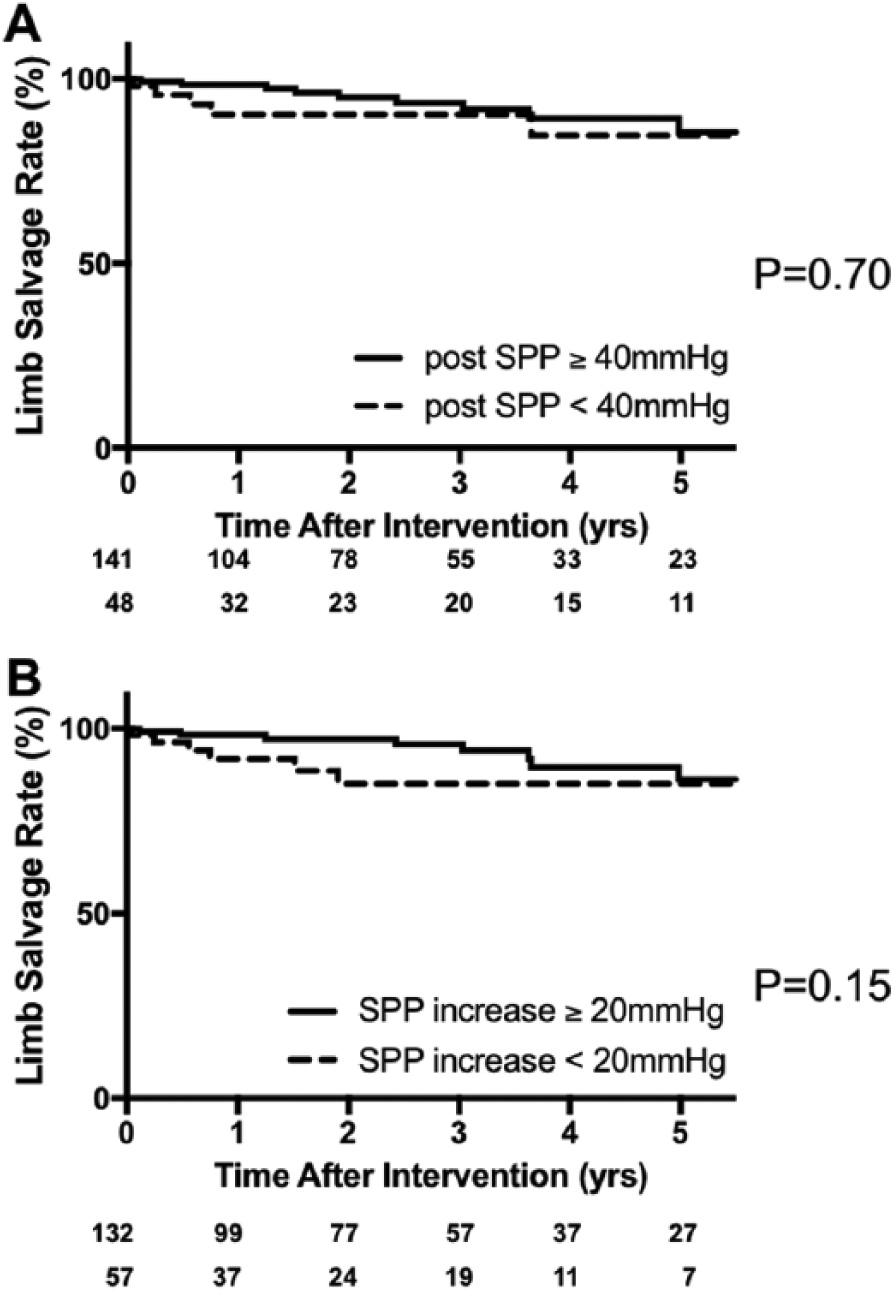
Limb salvage rates after bypass surgery grouped according to the presence or absence of (A) a postoperative SPP of ≥ 40 mmHg and (B) an increase in postoperative SPP of ≥ 20 mmHg.
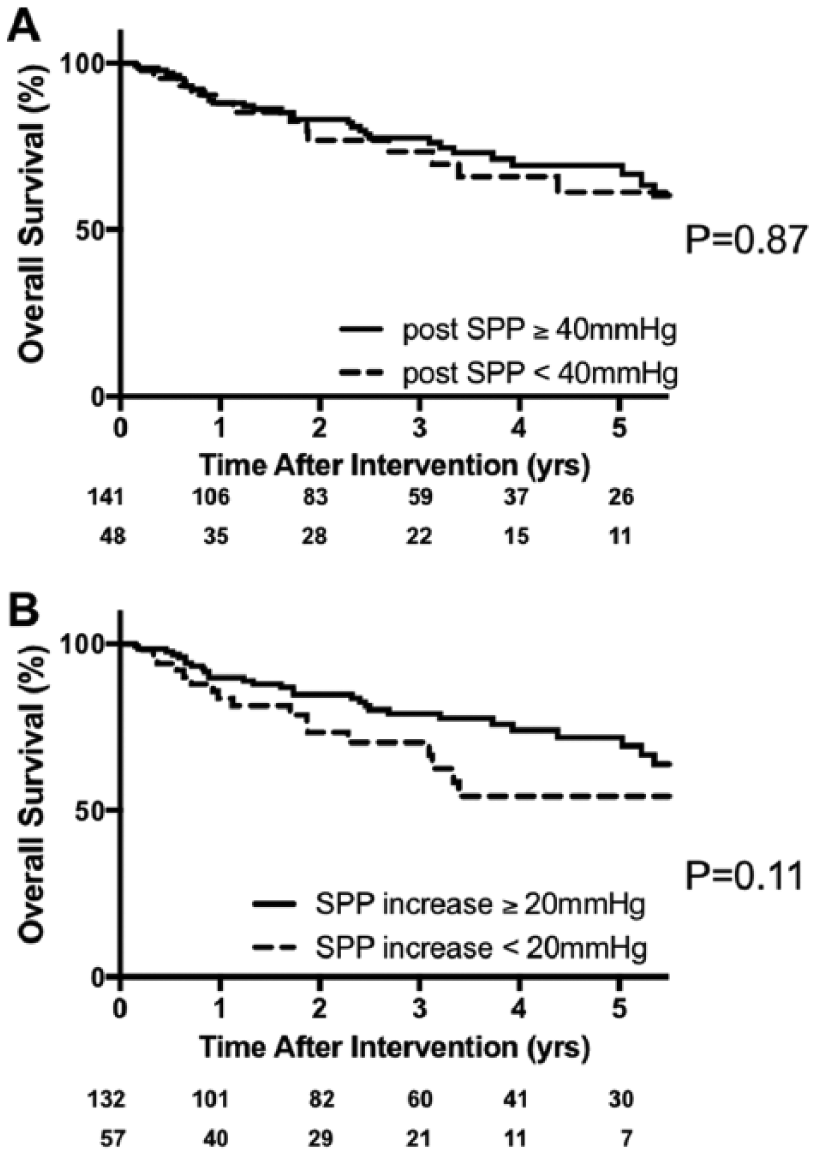
Overall survival rates after bypass surgery grouped according to the presence or absence of (A) a postoperative SPP of ≥ 40 mmHg and (B) an increase in postoperative SPP of ≥ 20 mmHg.
Factors that affected the postoperative increase in SPP
As an increase in SPP of 20 mmHg had a better association with AFS, we then looked at the factors that could affect the increase in SPP after revascularization. Therefore, we reviewed the whole cohort and analyzed 223 cases that included both preoperative and postoperative SPPs, which enabled us to calculate the SPP increase after the procedure. After the univariate analysis, three factors exhibited significance (Table 2). These three factors for predicting a good increase in SPP after intervention included: (1) a preoperative SPP < 20 mmHg; (2) a high serum albumin level (≥ 3.0 g/dL); and (3) femoropopliteal revascularization. These factors were subsequently assessed using a multivariate analysis, which indicated preoperative SPPs and serum albumin levels as significant (Table 3). Femoropopliteal revascularization was not significant but showed a high tendency (odds ratio, 1.8;
Univariate analysis of increase in the SPP value ≥ 20 mmHg or more.
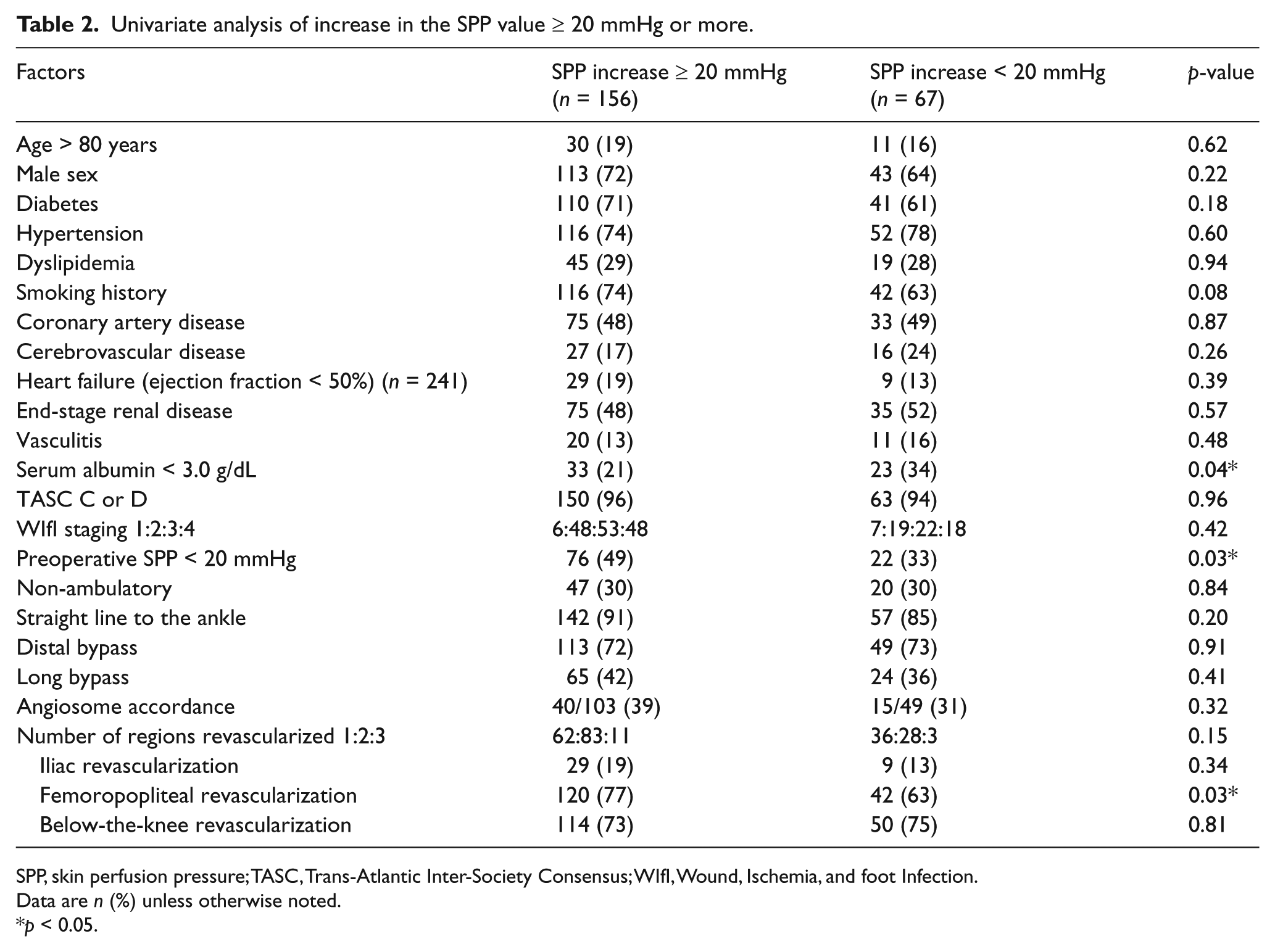
SPP, skin perfusion pressure; TASC, Trans-Atlantic Inter-Society Consensus; WIfI, Wound, Ischemia, and foot Infection.
Data are
Multivariate analysis of increase in the SPP value of ≥ 20 mmHg.
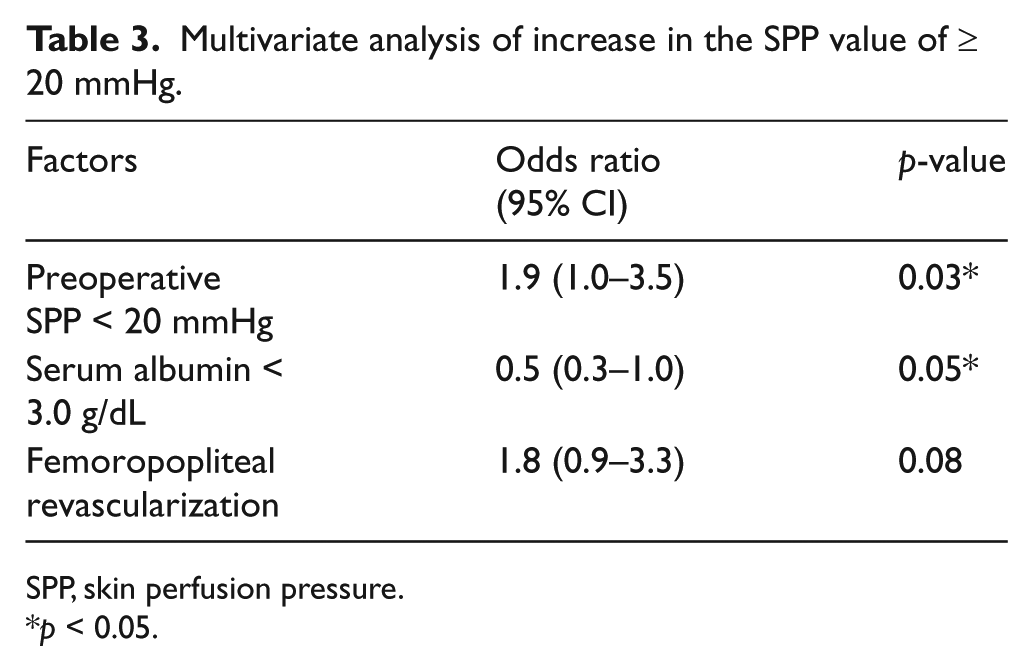
SPP, skin perfusion pressure.
Discussion
Since first presenting the SPP values of CLI patients during their natural course of recovery, we have used an SPP of < 40 mmHg as an indicator for revascularization. 2 We have deduced that restoring the SPP to 40 mmHg would significantly heal the wound and produce a better prognosis. Although some studies have set a certain SPP as an immediate indicator of successful revascularization,23–27 only a few have examined the actual prognosis according to the postoperative SPP.9,10
SPP has been known to measure ischemia in the presence of calcified arteries in CLI patients in which ABI may show an abnormally high value. 5 The non-inferiority of SPP over other non-invasive modalities has been reported. SPP showed good correlation with toe pressure, 28 but we prefer SPP because it can also examine ischemia at any level of the limb in addition to the SPP of the toe. Another non-invasive test is transcutaneous oxygen pressure (TcPO2). We use TcPO2 measurement as a supplemental test to SPP measurement if needed because it is relatively time-consuming as compared with SPP.
In this study, we found that an increase in, rather than a certain postoperative SPP may be more predictive of prognosis (AFS). We used AFS as the primary end point, so we also evaluated the amputation rate (limb salvage rate) and death rate (survival rate). Although both end points showed no significance due to the low number of these events in each group, this AFS seemed to be influenced more by the difference in overall survival than that of limb salvage rate, especially after several years. We considered two explanations for these results.
First is that this increase in SPP was a result of the preoperative general status rather than the cause of poor prognosis, and SPP increase revealed the systemic circulatory resistance that could affect survival due to cardiovascular events. The result that hypoalbuminemia was associated with SPP increase also supports this theory.
The second theory is that the increase in SPP affected limb prognosis. For example, a patient with CLI whose SPP value has recovered from 10 mmHg to 30 mmHg (an increase of 20 mmHg) has a higher chance of obtaining a better limb prognosis than a patient whose SPP has recovered from 30 mmHg to 40 mmHg. This result may be explained by the fact that ulcer formation and its healing mechanism do not rely on the degree of ischemia alone, as explained in WIfI staging. 17 As some patients develop an ulcer at an SPP of 30 mmHg and some patients do not exhibit any symptoms even with an SPP of 10 mmHg, ulcer healing may be better achieved by increasing blood flow from its preoperative value. That is, patients with CLI whose SPPs are just below the cut-off point may have multiple factors beyond ischemia that may affect wound healing. Therefore, for these patients, a slight increase in SPP achieved by incomplete procedures is insufficient to determine prognosis or guarantee repair. Whichever the reason, from our results, the increases in SPP were associated more with AFS than with the absolute values and therefore predictive of limb prognosis.
Upon considering these results, we then investigated how we could achieve a greater increase in SPP. Several factors affected the increase. The first factor, a preoperative SPP of < 20 mmHg, indicates that patients with severe ischemia are more likely to recover blood flow by revascularization alone. The second factor, serum albumin level, reflects the presence of malnutrition, a known risk factor for limb prognosis after bypass surgery.29–31 Finally, the last factor, though not significant (
Limitations
This study has several limitations. As our strategy for CLI was based on bypass surgery performed first, EVT was excluded from this study. Inclusion of our EVT patients would have led to referral and selection bias, and we assumed that this would produce inferior results for EVT. Data from cases where EVT was the first option strategy would be needed to see the degree of SPP recovery after EVT. Whether SPP recovery after EVT affects prognosis remains to be solved.
Another limitation is that the factors that increased postoperative SPP are patient related and not solved by intervention. However, these factors may enable us to estimate surgical results and limb prognosis.
Conclusion
A ≥ 20 mmHg rise in SPP was associated with improved amputation-free survival among CLI patients revascularized by bypass surgery. Patients with severe ischemia and higher serum albumin levels are more likely to achieve this goal.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
