Abstract
A small percentage of patients with asymptomatic carotid artery stenosis (ACAS) who are on optimal medical management do go on to develop ischemic stroke or transient ischemic attacks (IS/TIA). Several diagnostic tools have been studied to identify those patients who are at increased risk. However, most of these diagnostic tools are not available for routine clinical use or are resource intensive. We performed a retrospective study to assess the incremental value of external carotid artery stenosis progression (ECASP) along with internal carotid artery stenosis progression (ICASP) in predicting risk of ipsilateral IS/TIA in a cohort of patients with ACAS. We conducted a retrospective analysis of patients with ACAS who had at least two serial duplex ultrasounds (DUS) at our center. A total of 356 patients (712 carotid arteries) were included in the study (mean age 74.7±9 years, 49.2% male) with a mean follow-up of 60.7±32.7 months. In univariate analysis, concurrent progression of ICA and ECA stenosis on the same side arteries was associated with a very significant increased risk of ipsilateral IS/TIA (14.7% vs 4.6%, p<0.001). Also, multivariable regression analysis showed that concurrent ECA/ICA progression was an independent predictor of IS/TIA (OR=3.6, 95% CI 1.64–7.8; p=0.001). ECASP along with ICASP is significantly associated with increased risk of ipsilateral IS/TIA and provides incremental risk stratification over that provided by ICASP alone. The ECA is routinely evaluated in clinical practice, and it could serve as an additional marker for identifying higher risk patients with ACAS.
Keywords
Introduction
Risk of ischemic stroke and transient ischemic attack (IS/TIA) in patients with asymptomatic carotid artery stenosis (ACAS) treated with optimal medical management has declined significantly over the last two decades.1,2 However, many patients with ACAS do become symptomatic and suffer IS/TIA. Currently there is no reliable method to identify these high-risk patients, and clinical characteristics are generally not very helpful. In the patients with good medical management, the degree of internal carotid artery (ICA) stenosis has not been found to be uniformly predictive of future events.3,4 It is generally agreed that IS/TIA are related to the atheroembolic potential of the plaque rather than the anatomical severity of ICA stenosis. Thus, investigators have attempted to identify these ‘unstable plaques’ for risk stratification. Evidence of intraplaque hemorrhage on magnetic resonance imaging (MRI), echolucent plaque, ulcerated plaque, large plaque burden on duplex sonography (DUS), micro-emboli on transcutaneous Doppler, or silent infarct on brain imaging all identify active unstable plaques and have been shown to predict a higher risk of IS/TIA.5–9 Most of the diagnostic modalities and techniques mentioned above (apart from ICA stenosis progression) are either time or resource consuming and are not part of routine clinical practice. Increasing plaque burden represents an active atherosclerotic process (thus an unstable plaque), and it could predict the risk of IS/TIA. In line with this reasoning, some studies have found a significant correlation between progression of ICA stenosis and higher risk of neurologic events.10–13 The atheromatous plaque in the ICA is part of a larger plaque burden frequently involving the common carotid and the external carotid artery (ECA) at the carotid bifurcation. Thus, similar to ICA plaque, a progressive increase in ECA stenosis is reflective of increasing overall plaque/atheroma burden and an active atheromatous process. ECA velocity measurements are easy to perform and are routinely measured during DUS (though mostly ignored as not having much clinical significance). We hypothesized that the presence of concomitant ECA stenosis along with ICA stenosis is indicative of a larger plaque burden, and progression of ECA stenosis on DUS will add incremental risk stratification to that provided by progression of ICA stenosis alone.
We performed a retrospective study to assess the incremental value of ECA stenosis progression (ECASP) along with ICA stenosis progression (ICASP) in predicting the risk of ipsilateral IS/TIA in a cohort of patients with ACAS.
Method
Study design and patient selection
We conducted a retrospective longitudinal study enrolling patients who underwent carotid DUS at the vascular laboratory of the University of Kansas Medical Center from January 2001 to December 2014. Subjects were identified using hospital electronic medical records. The study was approved by the human subjects committee of the University of Kansas Medical Center. Patients who had at least two serial DUS were considered for the study. Primary outcomes assessed were progression of ICA stenosis, progression of ECA stenosis, and the occurrence of new IS/TIA on follow-up. Subjects with any history of atrial fibrillation/atrial flutter and IS/TIA in the 6 months preceding the first DUS and previous history of carotid artery revascularization were excluded from the study. Furthermore, subjects were censored from further follow-up if any carotid revascularization was performed for either IS/TIA or stenosis progression before the second DUS.
Data collection
All data were collected by the research team using manual chart review of the electronic medical records and DUS reports. The demographic and clinical characteristics of the patients were obtained at the time of their first DUS (baseline) and follow-up. Also, low-density lipoprotein (LDL) levels at the first visit and follow-up visit for each patient were averaged to obtain the final LDL values. The occurrence of any ipsilateral IS/TIA after the baseline DUS was recorded. ECASP and ICASP were evaluated by comparing respective systolic velocities of the follow-up DUS to the baseline DUS.
Duplex sonography
The DUS was performed and interpreted using the standard clinical protocols at the University of Kanas Medical Center. The vascular ultrasound lab is accredited by the Intersocietal Accreditation Commission. Our protocol mandates an assessment of the ECA at its origin and proximal segment. Transverse and longitudinal B-mode imaging is routinely performed to assess plaque at the bifurcation of the common carotid into the ICA and ECA. ECA velocity was measured as peak systolic velocity at the proximal segment of the ECA.
Grading of the ICA stenosis was done according to the Society of Radiologists in Ultrasound consensus for the diagnosis and stratification of ICA stenosis. 14 An ECA velocity of >1.25 m/sec was categorized as ≥50% stenosis. 15
ICASP was defined as an increase in stenosis severity by at least one grade or an increase in velocity by 25% from baseline. Since there is no accepted DUS criterion for grading ECASP, it was empirically defined as an increase in carotid artery peak systolic velocity by more than 25% compared to the baseline.
Statistical analysis
The prevalence of carotid artery stenosis was expressed as a percentage of the total number of carotid arteries studied. IBM SPSS (Version 24.0; IBM Corp., Armonk, NY, USA) was used for statistical analysis. Continuous data were presented as mean ± standard deviation (SD). The analyses involved comparing continuous and categorical data using Student’s t-test, chi-squared, and Fisher’s exact tests, as appropriate. A p-value <0.05 was considered statistically significant. Multivariable regression analysis was used to assess the association of risk factors, ICASP, and ECASP with the endpoint of ipsilateral IS/TIA.
Results
Baseline characteristics
A total of 356 patients met the inclusion criteria for the study and were enrolled. The mean follow-up duration was 60.7±32.7 months, and the mean age was 74.7±9 years; 49.2% of patients were male. The basic demographic and clinical characteristics of the overall group and of those with and without ipsilateral IS/TIA on follow-up are shown in Table 1.
Demographic and clinical characteristics of study subjects.
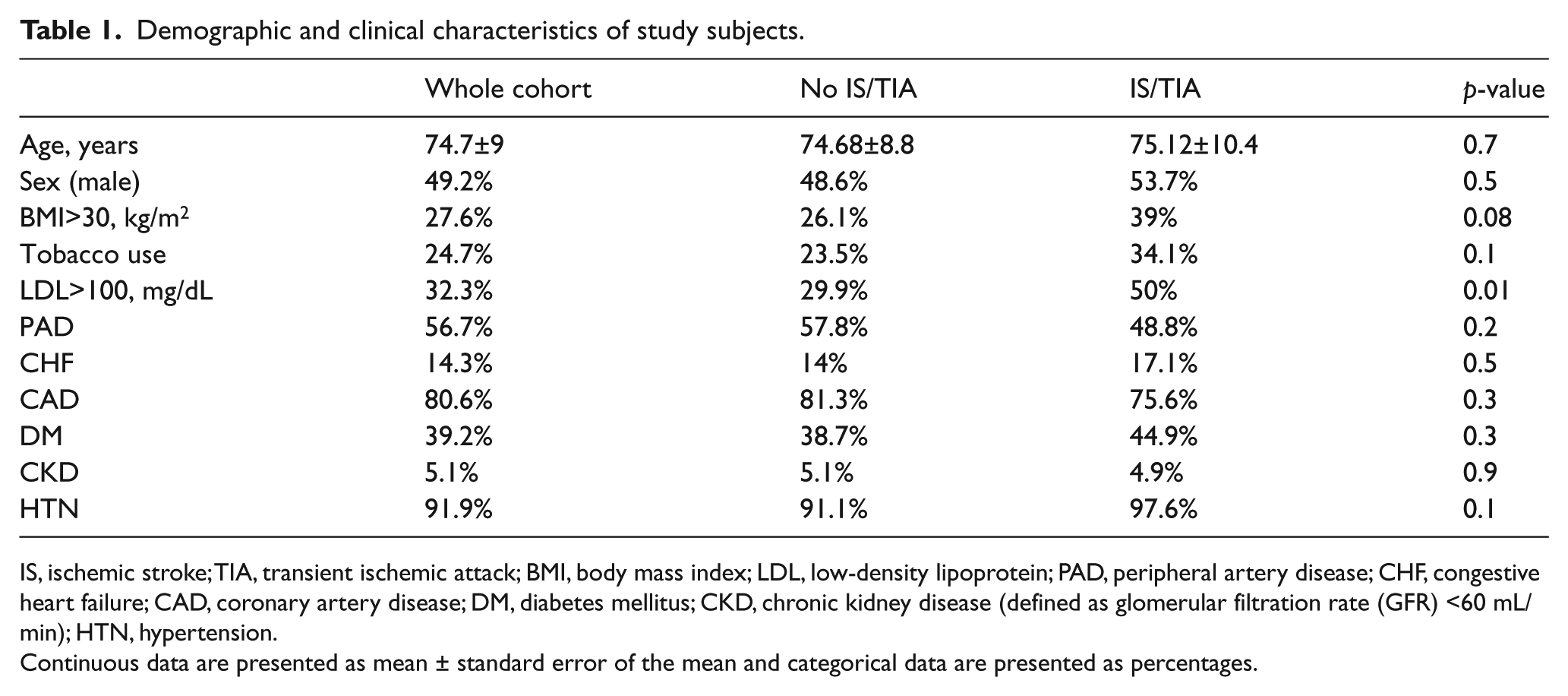
IS, ischemic stroke; TIA, transient ischemic attack; BMI, body mass index; LDL, low-density lipoprotein; PAD, peripheral artery disease; CHF, congestive heart failure; CAD, coronary artery disease; DM, diabetes mellitus; CKD, chronic kidney disease (defined as glomerular filtration rate (GFR) <60 mL/min); HTN, hypertension.
Continuous data are presented as mean ± standard error of the mean and categorical data are presented as percentages.
ICA and ECA stenosis
There were a total of 712 carotid arteries studied. At the time of the initial DUS, 1.4% (n=10) were 100% occluded, 3.8% (n=27) had ≥70% ICA stenosis, 32.2% (n=229) had 50–69% ICA stenosis, and 62.6% (n=446) had <50% ICA stenosis. Also at the initial DUS, 394 (55.3%) ECAs had <50% stenosis (Figure 1).
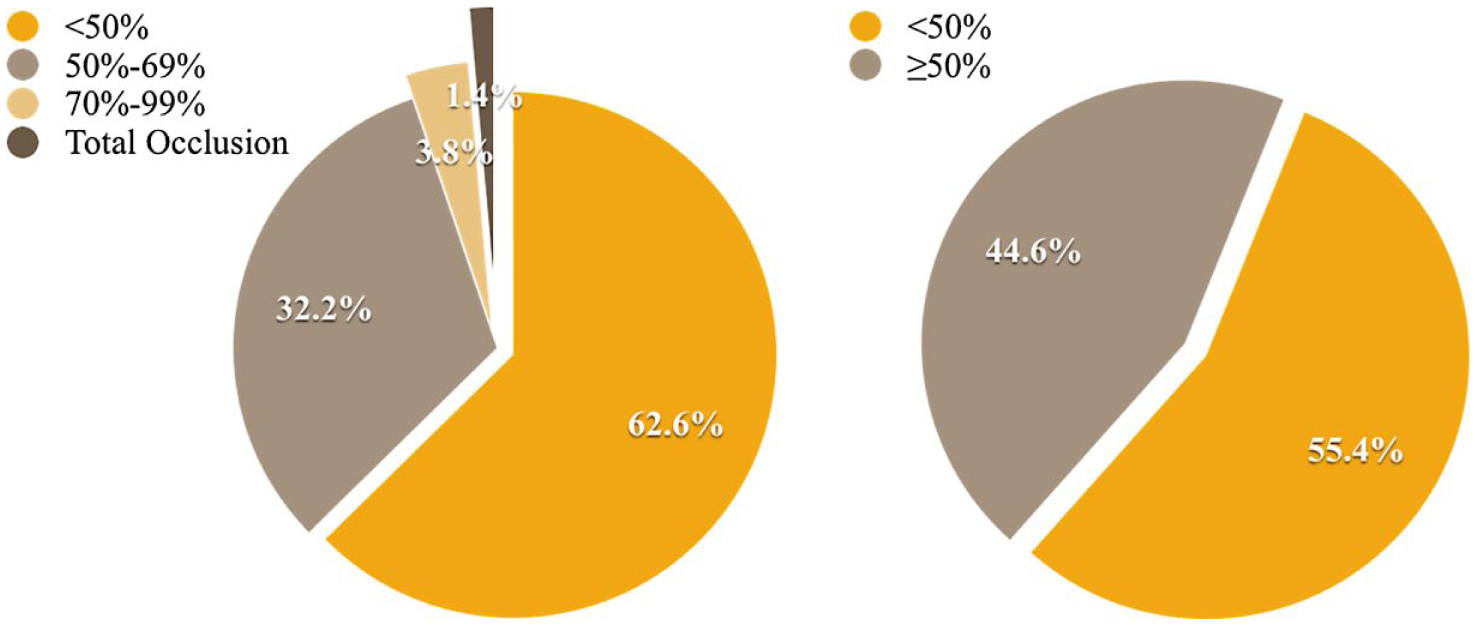
Distribution of baseline internal carotid artery stenosis (left panel); distribution of baseline external carotid artery stenosis (right panel).
Progression of ICA stenosis was seen in 223 (31.3%) arteries. Progression of ECA stenosis was seen in 209 (29.6%) arteries, with 114 arteries (16.1%) having progression without concomitant ICASP stenosis progression. Progression in both arteries (on the same side) was seen in 13.4% of external and internal carotid arteries.
Ischemic stroke/transient ischemic attacks
During the follow-up period, a total of 49 IS/TIA events were reported in the overall group.
Baseline ICA stenosis ≥50% or ECA stenosis ≥50% did not correlate with the risk of future ipsilateral IS/TIA in the follow-up period (Table 2).
The univariate analysis of baseline ICA/ECA stenosis and primary outcome (ipsilateral IS/TIA events).
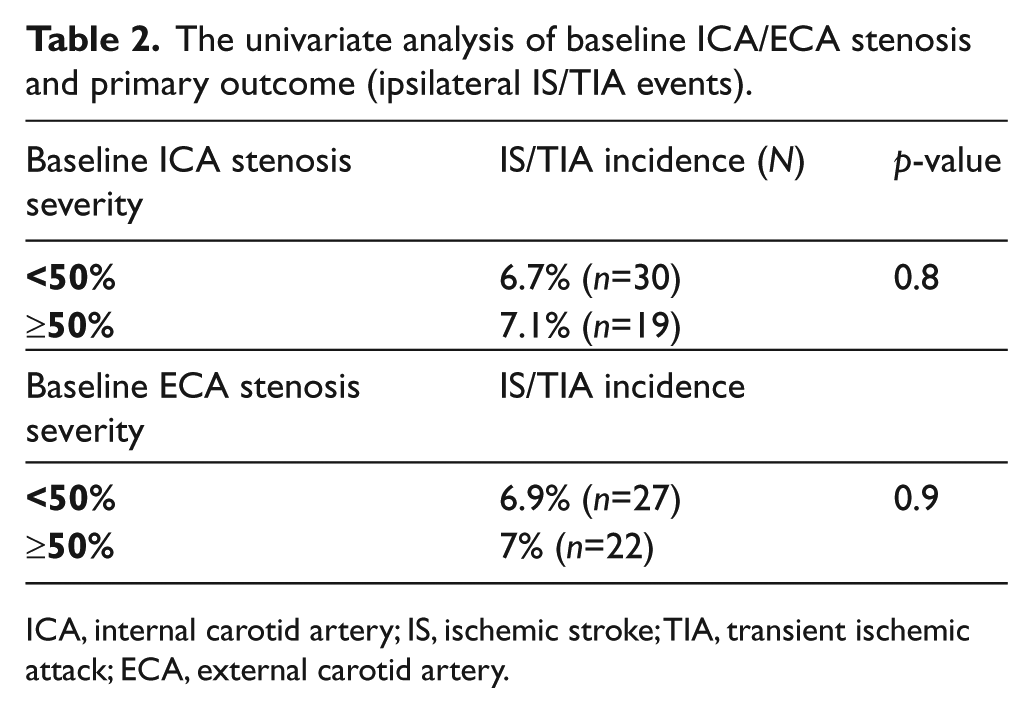
ICA, internal carotid artery; IS, ischemic stroke; TIA, transient ischemic attack; ECA, external carotid artery.
In univariate analysis, ICA progression alone was associated with a numerically but not statistically significant higher incidence of ipsilateral IS/TIA (8.7% vs 4.6%, p=0.08). Progression of ECA stenosis alone (without concurrent progression of ICA stenosis) was not associated with a significant increased risk of ipsilateral IS/TIA (5.3% vs 4.6%, p=0.7). However, concurrent progression of ICA and ECA stenosis in arteries on the same side was associated with a very significant increased risk of ipsilateral IS/TIA (14.7% vs 4.6%, p <0.001). The IS/TIA risk was lowest when either ECA or ICA velocities did not show any progression (Figure 2).
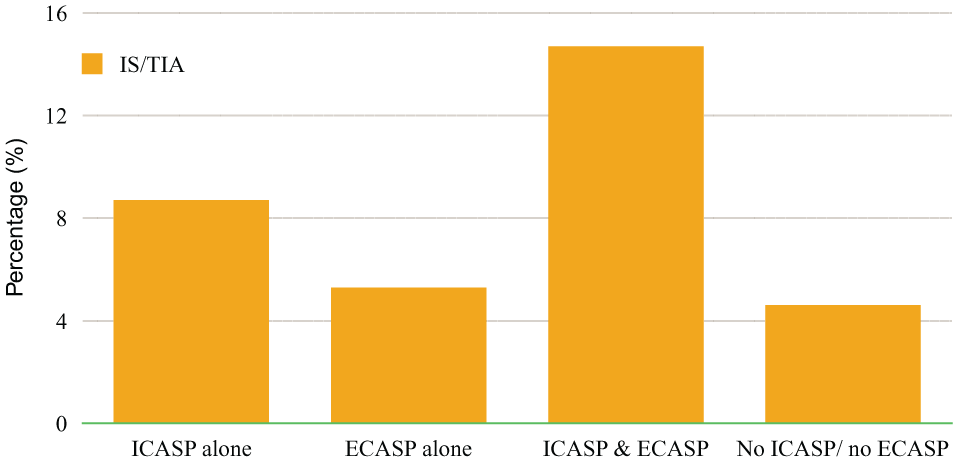
Unadjusted risk of ipsilateral ischemic stroke (IS)/transient ischemic attack (TIA) in patients with asymptomatic carotid artery stenosis according to the degree of external carotid artery stenosis progression (ECASP) and internal carotid artery stenosis progression (ICASP).
A multivariable regression analysis was performed after adjusting for age >70 years old, sex (male), LDL >70 mg/dL, baseline ICA stenosis >50%, baseline ECA stenosis >50%, ICASP alone, ECASP alone, and concurrent ICASP and ECASP. In multivariable analysis, LDL >70 mg/dL and concurrent ECASP/ICASP were found to be independent predictors of IS/TIA (Table 3).
Multivariable logistic regression analysis for ipsilateral IS/TIA in ACAS patients.
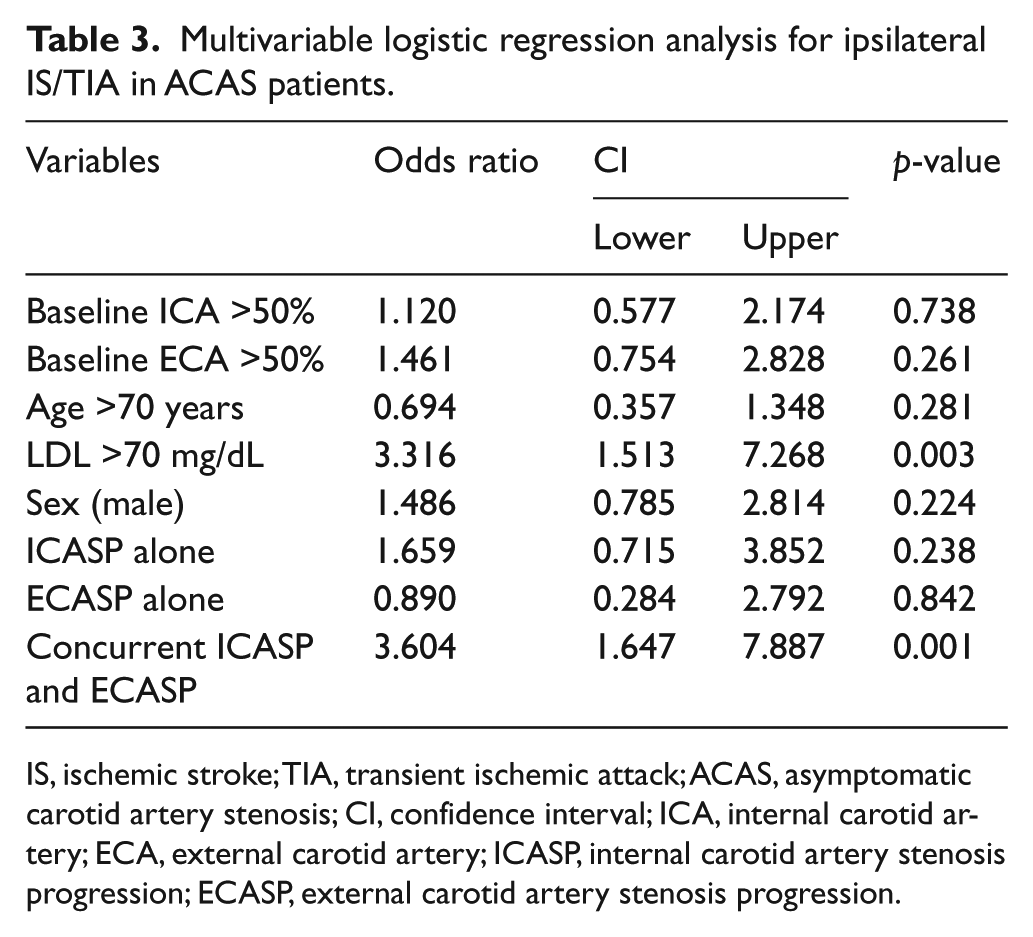
IS, ischemic stroke; TIA, transient ischemic attack; ACAS, asymptomatic carotid artery stenosis; CI, confidence interval; ICA, internal carotid artery; ECA, external carotid artery; ICASP, internal carotid artery stenosis progression; ECASP, external carotid artery stenosis progression.
Discussion
The main finding of this study is that progression of ECA stenosis along with a concomitant progression in ICA stenosis severity (as detected on serial DUS) is predictive of future ischemic neurologic events in patients with ACAS.
There is growing evidence that optimal medical therapy (OMT) using guideline-directed drug therapy and treatment of risk factors has significantly reduced the risk of ischemic neurologic events in patients with ACAS.1,2 With this demonstrated improvement in the natural history of ACAS, there is a need to reassess the approach to management of ACAS with more emphasis on OMT and less on revascularization for most patients. Although a large proportion of patients with ACAS will remain asymptomatic, a small percentage will go on to have IS/TIA. It is pivotal to identify the subset of patients who are at a greater risk of IS/TIA in the future. Ischemic neurologic events in this population are caused mostly by thromboembolism emanating from the carotid plaque rather than a consequence of reduced blood flow from severe stenosis. In fact, there is some evidence that stroke risk is lower in those with stable, near-occlusion stenosis compared to severe but not occlusive stenosis. 16 There are several studies that have attempted to help identify patients with ACAS who are at a higher risk of IS/TIA. These have mostly focused on methods to identify patients with higher thromboembolic potential and thus have assessed ways to detect plaque instability and embolic risk.7–9,17–27
Several morphologic plaque characteristics have been investigated to identify higher risk patients. Plaque characteristics such as presence of ulcerations and lipid rich content of plaque (identified by characteristics such as hypo-echoic plaques, intraplaque hemorrhage, etc.) have been shown to identify a higher risk group.5,8,17,21,25,26 However, most of these techniques, while providing prognostic information, are not feasible in routine clinical practice as most need dedicated software or advanced training. Other studies have looked at occult/subclinical embolism to identify higher risk patients. Detection of micro-emboli on trans-cranial Doppler and the finding of asymptomatic micro-infarcts on computed tomography (CT)/MRI of the brain have been studied to identify patients with ACAS who are at a higher risk of future IS/TIA.7,9,22,28–30 Trans-cranial Doppler to detect micro-embolic signals has been studied the most in this regard. Markus et al. published a meta-analysis including data from the largest of these studies (the Asymptomatic Carotid Emboli Study). 22 They found a sevenfold increase in the risk of ipsilateral stroke with the presence of spontaneous micro-embolization signals. Similarly, the ACRS (Asymptomatic Carotid Stenosis and Risk of Stroke) study found that those with brain CT evidence of silent embolic micro-infarcts had a threefold risk of late ipsilateral stroke (1% vs 3.6%, p=0.002). 9 However, even though these methods are helpful in risk stratifying ACAS patients, these technologies are not yet available in routine clinical practice and do place additional burden on resources.
Progression of plaque volume by measuring carotid plaque area has been shown to identify high-risk ACAS patients. Spence et al., using high-resolution ultrasound imaging with specialized protocol and software, showed that those with an increase in plaque area had a significantly higher 5-year risk of cardiovascular outcomes (stroke, myocardial infarction or vascular death) when compared to those with no progression or regression (15.7%, 9.4% and 7.6%, p=0.003). 25 However, this methodology is not yet feasible in routine practice. On the other hand, progressive worsening of ICA stenosis is easy to detect by routine sequential DUS. Similar to plaque area, progression in ICA stenosis severity is reflective of an active and unstable plaque. Several studies have found that progression of the ICA stenosis (increasing peak systolic velocities) is predictive of ipsilateral stroke. Sabeti et al. found that ICASP was associated with a twofold increase in the 3-year risk of stroke from 2.5% to 5% (OR 2.00, 95% CI 1.02–4.11). 13 Hirt conducted a retrospective review of data in 1469 patients from the deferred endarterectomy arm of the Asymptomatic Carotid Surgery (ACAS) Trial. 20 They found that the incidence of ipsilateral events was four times higher for a stenosis that had progressed by two categories than a stenosis that had remained unchanged and was seven times higher for a stenosis that had progressed by three categories. Useful as it is, ICASP alone is not sufficient to identify all patients at higher risk, and there continues to be a need for additional, easy to use, risk-detecting tools.
The ECA is routinely evaluated in clinical practice during carotid DUS, and its peak systolic velocity is also routinely reported. However, these findings are not usually considered in clinical decision making (as the ECA does not routinely supply the intracranial circulation) and are often ignored as not having much clinical significance.
We hypothesized that ECASP is also indicative of an active plaque and of increasing plaque burden in the carotid bifurcation region. This additional finding on routine carotid DUS may add incremental value to ICA velocity progression for identifying those with a higher thromboembolic risk in the future.
The study results demonstrate that the incidence of ICASP was 31.3%, which was consistent with the prior studies. 31 The incidence of ECASP was also similar at 29.6%, though not necessarily in the same patients who had ICA stenosis progression. In univariate analysis, the ICASP group had a higher incidence of IS/TIA than those without ICASP or ECASP, but the difference did not reach statistical significance. This was likely due to the small sample size. ECASP in univariate analysis did not show association with ipsilateral IS/TIA either. However, in multivariable regression analysis concurrent ECASP and ICASP were found to be significantly associated with a higher risk of future events. The most significant finding of the study was an almost threefold increase in the incidence of ipsilateral IS/TIA when the group with concurrent ICASP and ECASP was compared to those with no progression in either artery. In fact, over a mean follow-up period of almost 5 years, the incidence of ipsilateral IS/TIA was only 4.6% if there was neither ECASP or ICASP vs 14.7% when there is concurrent ECASP and ICASP.
Muluk et al. published their retrospective study on the natural history of ACAS using serial DUS. 10 The study demonstrated that in addition to baseline ICA stenosis severity, the presence of ECA stenosis of >50% also predicted future events. We did not find such an association for either baseline ICA or ECA stenosis. Our finding is well supported in the literature as both the ACAS trial and the ACST (Asymptomatic Carotid Endarterectomy Trial) did not find evidence that baseline stenosis severity predicted future stroke risk.3,4 These findings are likely related to the fact that with adequate risk factor control, a severe stenosis can have a stabilized plaque with less thromboembolic potential and hence a reduced risk of future IS/TIA, and a progressively increasing stenosis identifies a higher risk plaque.
To our knowledge, this prognostic significance of ECA stenosis progression has not been reported in published literature. Further, the risk of IS/TIA was extremely low in those who had neither ICASP or ECASP and thus further helps define a lower risk group for conservative management.
Study limitations
This is retrospective study and thus is subject to the usual limitations of such a study. The study was conducted in a large cardiovascular medicine practice where moderate carotid disease is followed with serial DUS as part of routine clinical practice. This limits selection bias, though we cannot exclude the possibility that the study may overestimate the sensitivity and correlation of ICASP or ECASP with IS/TIA, as those who are symptomatic may be more likely to get a repeat DUS done. We did not report the subtypes of strokes (embolic, lacunar, etc.), though we did only include subjects with a diagnosis of ischemic (non-hemorrhagic) stroke. We have reported the incidence of IS/TIA during the entire follow-up period after the duplex sonogram, but did not report interval time to event, which is a limitation of this study. Furthermore, the study numbers were relatively small.
Conclusion
The study results show that in patients with ACAS, ECASP with concomitant ICASP is significantly associated with risk of ipsilateral IS/TIA and provides incremental risk stratification over ICASP alone. Further, risk of future IS/TIA is extremely low in those without ECASP or ICASP, thus identifying a very low-risk group. ECA velocities are routinely available in every DUS done in conventional clinical practice and could serve as an additional marker for identifying higher risk patients with ACAS. If validated in larger studies, ECASP could be used along with other parameters to develop an IS/TIA risk prediction model in patients with ACAS.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
