Abstract
The impact of heart failure (HF) on long-term survival in patients with critical limb ischemia (CLI) has not been well described. Outcomes stratified by left ventricular ejection fraction (EF) are also unknown. A single center retrospective chart review was performed for patients who underwent treatment for CLI from 2006 to 2013. Baseline demographics, procedural data and outcomes were analyzed. HF diagnosis was based on appropriate signs and symptoms as well as results of non-invasive testing. Among 381 CLI patients, 120 (31%) had a history of HF and 261 (69%) had no history of heart failure (no-HF). Within the HF group, 74 (62%) had HF with preserved ejection fraction (HFpEF) and 46 (38%) had HF with reduced ejection fraction (HFrEF). The average EF for those with no-HF, HFpEF and HFrEF were 59±13% vs 56±9% vs 30±9%, respectively. The likelihood of having concomitant coronary artery disease (CAD) was lowest in the no-HF group (43%), higher in the HFpEF group (70%) and highest in the HFrEF group (83%) (p=0.001). Five-year survival was on average twofold higher in the no-HF group (43%) compared to both the HFpEF (19%, p=0.001) and HFrEF groups (24%, p=0.001). Long-term survival rates did not differ between the two HF groups (p=0.50). There was no difference in 5-year freedom from major amputation or freedom from major adverse limb events between the no-HF, HFpEF and HFrEF groups, respectively. Overall, the combination of CLI and HF is associated with poor 5-year survival, independent of the degree of left ventricular systolic dysfunction.
Keywords
Introduction
Critical limb ischemia (CLI) is the most severe form of peripheral artery disease (PAD), encompassing ischemic rest pain, minor and major tissue loss or gangrene. This diagnosis is associated with greater than 50% mortality at 5 years.1–3 The prognosis for limb salvage is poor without revascularization as major amputation rates approach 50% and serve as an independent predictor of mortality.4–7 In addition to advanced age and non-ambulatory status, many studies have also identified other co-morbidities as independent risk factors for increased mortality and/or major amputation in CLI patients, including heart failure (HF), end-stage renal disease (ESRD), coronary artery disease (CAD), and diabetes mellitus (DM).8–16
Cardiac co-morbidities are common in CLI patients, and the prevalence of HF has been reported to be 10–40% in this population.8–16 Although HF has been identified as an important risk factor for increased mortality and/or amputation, most studies failed to stratify HF patients by left ventricular ejection fraction (EF)10,12,13,17 or limited their evaluation to those with left ventricular systolic dysfunction.11,14,15 Inadequate systemic perfusion as a consequence of HF may pose a threat to CLI patients by putting them at a higher risk for embolism. Thus, it may be theorized that patients with concomitant diagnoses of CLI and HF have worse limb outcomes compared to non-HF CLI patients. However, long-term outcomes for patients with CLI and HF have not been well reported.
This study aims to compare the long-term (5-year) outcomes in patients with CLI and concomitant HF with preserved or reduced EF to those without a diagnosis of HF. We hypothesize that patients with HF, and to a greater extent those with a reduced rather than preserved EF, will have significantly higher mortality rates and worse outcomes than those without HF. Identifying high-risk patients and optimizing medical management of co-morbidities is critical to improving outcomes in this challenging patient population.18–21
Methods
Study design
This retrospective, single-center study utilized data from the University of California Davis Peripheral Artery Disease (UCD-PAD) Registry which comprises all patients with clinically diagnosed PAD who underwent diagnostic angiography and/or therapeutic endovascular intervention at the UC Davis Medical Center between 1 June 2006 and 1 May 2013. 22 The study protocol was approved by the Institutional Review Board of the UC Davis Medical Center.
Study population and data collection
Demographics, clinical history, laboratory results, and procedural data were collected through pre-procedure clinical notes, inpatient documentation, and angiography review available on the Electronic Medical Record (EMR). Patient outcomes were determined by review of post-procedural clinic visits as well as EMR documentation of subsequent hospitalizations and discharge summaries. Echocardiograms within 2 years of initial presentation were reviewed in order to determine baseline EF. Direct chart documentation and the Social Security Death Index were used to confirm mortality.
Data definitions
CLI was defined as advanced PAD with Rutherford category 4–6 disease, including ischemic rest pain, minor tissue loss and major tissue loss or gangrene, respectively. A history of HF was defined by prior presentation with appropriate signs and symptoms of congestive heart failure, including pulmonary edema and evidence of systemic volume overload, and by results of two-dimensional echocardiography. Patients with HF were further stratified by EF; HF with preserved ejection fraction (HFpEF) was defined as a baseline EF≥40% and HF with reduced ejection fraction (HFrEF) was defined as a baseline EF<40%. This cut-off was chosen based on the definitions adopted by the American College of Cardiology and American Heart Association (ACC/AHA) ‘Guideline for the Management of Heart Failure’. 23
Outcomes
The primary end-point was 5-year overall survival. Secondary end-points consisted of freedom from major adverse cardiovascular or cerebrovascular events (MACCE) (defined as death, non-fatal myocardial infarction and non-fatal stroke), amputation-free survival (AFS), freedom from amputation (FFA) and freedom from major adverse limb events (MALE) (defined as the need for major amputation or bypass surgery).
Statistical analysis
Baseline patient characteristics are presented as mean with standard deviation for continuous variables and percentages for categorical variables. Continuous variables were compared with one-way analysis of variance to determine overall statistical difference and pairwise t-tests with the Bonferroni correction to determine between-group differences among characteristics that met overall l statistical significance. Categorical values were analyzed using chi-squared or Fisher’s exact test. Unadjusted hazard ratios (HR) were calculated for primary and secondary outcomes. Cox proportional hazards models were created using age, male sex, smoking status, aspirin use, statin use, history of HF, DM, CAD and ESRD as covariates. All analyses were performed using STATA software (Release 13; StataCorp LP, College Station, TX, USA). HR are provided with 95% confidence intervals (CI). For all tests, a p-value < 0.05 was considered significant.
Results
A total of 1400 patients who underwent 2000 procedures were included in the registry, of which 381 with a diagnosis of CLI were analyzed for this study. Approximately one-third of patients had a history of HF (n=120; 31%), of which 74 patients (62%) had HFpEF and 46 (38%) had HFrEF.
Patient demographics, co-morbidities, medication use and baseline characteristics are reported in Table 1. The average EFs within the three groups (no-HF, HFpEF and HFrEF) were 59±13%, 56±9% and 30±9%, respectively. There was no difference in body mass index (BMI) between the three groups, but there were fewer males in the HFpEF group. While the majority of patients in the HFpEF cohort were female, most patients in the HFrEF group were males. This higher prevalence of women with HFpEF has also been described in other cohorts of patients with HF. Patients in the no-HF group were more likely to be younger and active smokers compared to the two HF groups.
Patient demographics, co-morbidities, medication use and baseline characteristics within the total cohort.
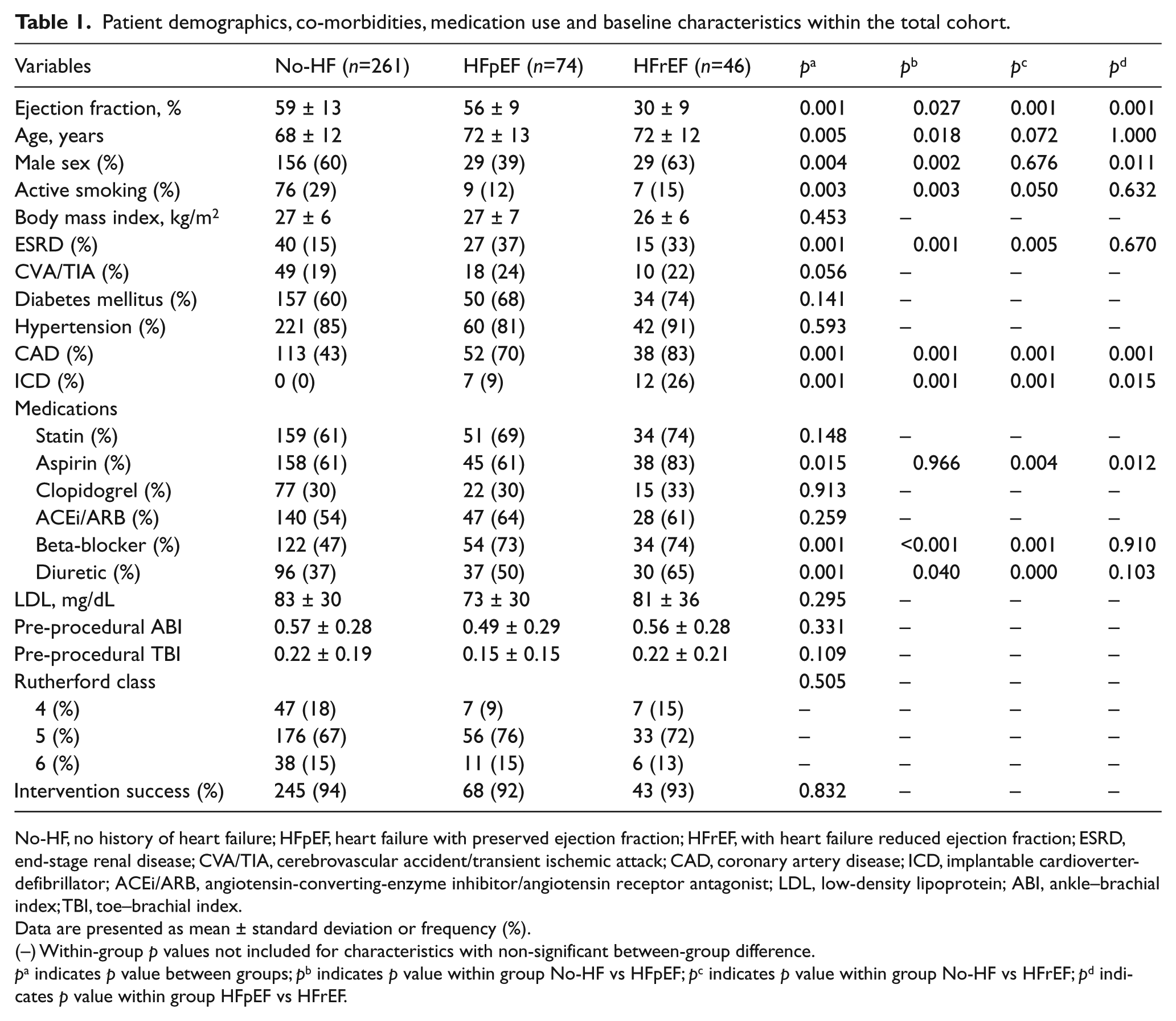
No-HF, no history of heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, with heart failure reduced ejection fraction; ESRD, end-stage renal disease; CVA/TIA, cerebrovascular accident/transient ischemic attack; CAD, coronary artery disease; ICD, implantable cardioverter-defibrillator; ACEi/ARB, angiotensin-converting-enzyme inhibitor/angiotensin receptor antagonist; LDL, low-density lipoprotein; ABI, ankle–brachial index; TBI, toe–brachial index.
Data are presented as mean ± standard deviation or frequency (%).
(–) Within-group p values not included for characteristics with non-significant between-group difference.
pa indicates p value between groups; pb indicates p value within group No-HF vs HFpEF; pc indicates p value within group No-HF vs HFrEF; pd indicates p value within group HFpEF vs HFrEF.
There was no difference in the history of cerebrovascular accident/transient ischemic attack, DM, hypertension or low-density lipoprotein levels between the three groups. The likelihood of having concomitant CAD was lowest in the no-HF group (113/261, 43%), higher in the HFpEF group (52/74, 70%) and highest in the HFrEF group (38/46, 83%) (p=0.001). The no-HF group was less likely to have ESRD compared to both HF groups (15% no-HF vs 37% HFpEF vs 33% HFrEF, p=0.001). The HFrEF group was more likely to have an implantable cardioverter-defibrillator (ICD) (26% vs 9%, p=0.015) compared to the HFpEF group. Finally, there was no difference in Rutherford class, pre-procedural ankle–brachial or toe–brachial indices, or interventional success between the three groups.
There was no difference in use of statins, clopidogrel, or angiotensin-converting-enzyme inhibitors or angiotensin receptor blockers (ACEi/ARB) between the three groups. Patients in the no-HF group were less likely to be prescribed beta-blockers (47% no-HF vs 73% HFpEF vs 74% HFrEF, p=0.001) or diuretics (37% no-HF vs 50% HFpEF vs 65% HFrEF, p=0.001). Compared to both the no-HF and the HFpEF groups, the HFrEF group had higher rates of aspirin use (61% no-HF vs 61% HFpEF vs 83% HFrEF, p=0.015).
Primary and secondary outcomes are reported in Tables 2A and 2B. The mean follow-up was 1.7 years, which is likely secondary to the high mortality in this patient population. Five-year survival (Figure 1) was on average twofold higher in the no-HF group (43%) compared to both the HFpEF group (19%, HR 2.47, CI 1.66–3.67, p=0.001) and the HFrEF group (24%, HR 2.74, CI 1.79–4.10, p=0.001). There was no difference in 5-year survival between the two HF groups (HR 0.85, CI 0.52–1.38, p=0.499). The 5-year freedom from MACCE (Figure 2) was also on average twofold higher in the no-HF group (44%) compared to both the HFpEF group (22%, HR 1.85, CI 1.27–2.71, p=0.001) and the HFrEF group (20%, HR 2.70, CI 1.80–4.06, p=0.001), with no difference between the two HF groups (HR 0.65, CI 0.40–1.03, p=0.067). The 5-year AFS (Figure 3) was also higher in the no-HF group (36%) compared to both the HFpEF group (16%, HR 1.88, CI 1.31–2.70, p=0.001) and the HFrEF group (15%, HR 2.33, CI 1.57–3.46, p=0.001), with no difference between the two HF groups (HR 0.79, CI 0.50–1.24, p=0.307). However, there was no difference in 5-year FFA (77% vs 78% vs 63%, p=0.667) (Figure 4) or MALE (64% vs 64% vs 58%, p=0.555) (Figure 5) between the no-HF, HFpEF, and HFrEF groups, respectively.
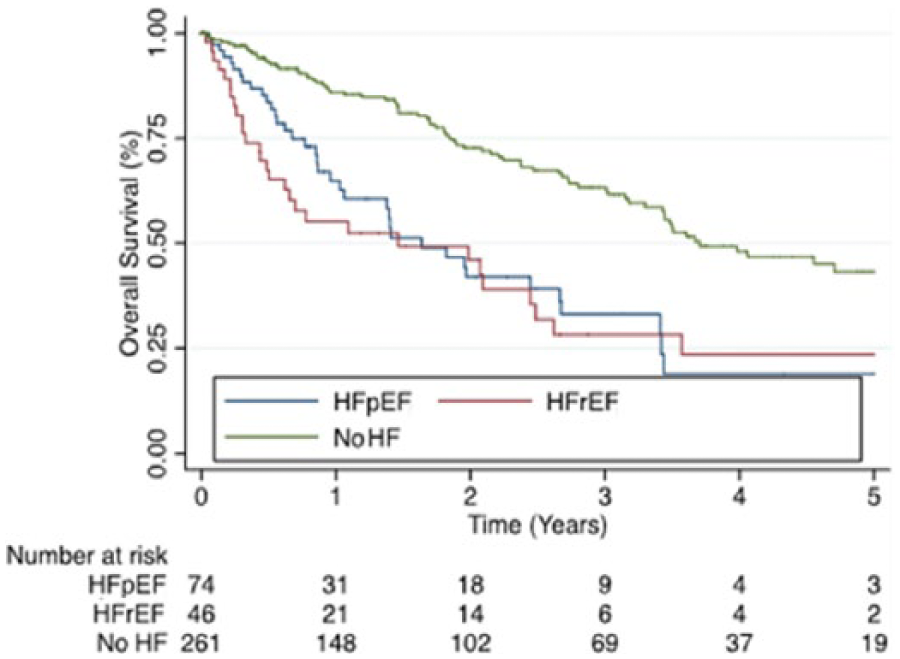
Kaplan–Meier survival curves comparing those with no-HF vs HFpEF vs HFrEF in terms of overall survival. (No-HF, no prior history of heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.)
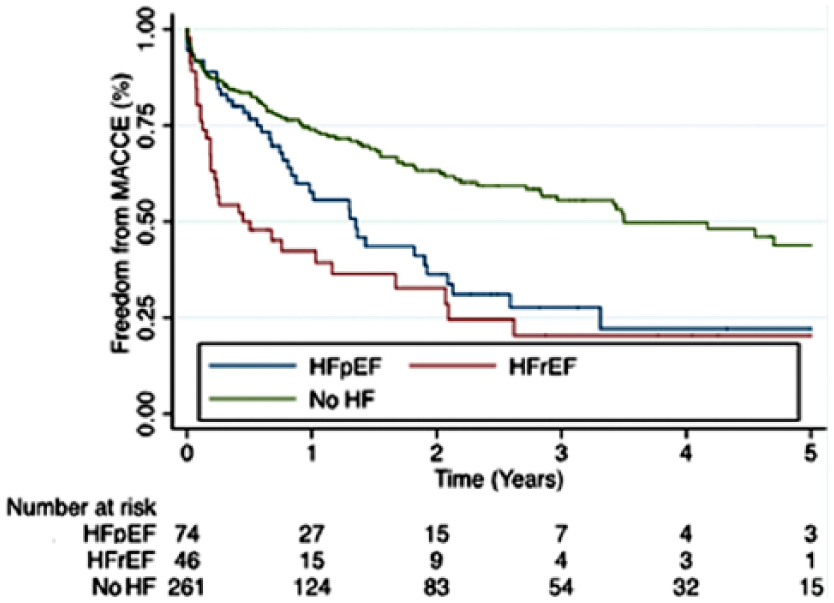
Kaplan–Meier survival curves comparing those with no-HF vs HFpEF vs HFrEF in terms of freedom from MACCE. (No-HF, no prior history of heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; MACCE, major adverse cardiovascular/cerebrovascular events.)
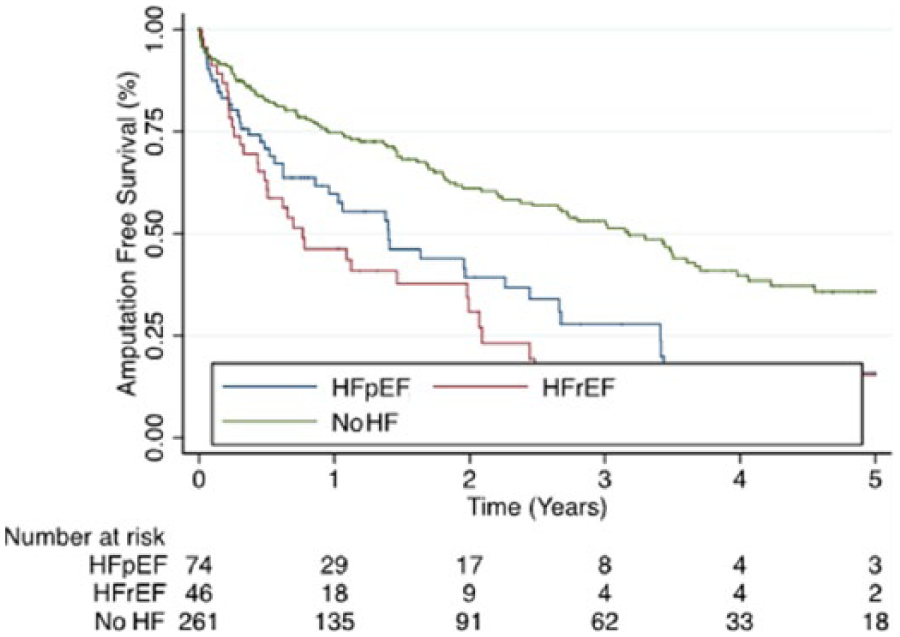
Kaplan–Meier survival curves comparing those with no-HF vs HFpEF vs HFrEF in terms of amputation-free survival. (No-HF, no prior history of heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.)
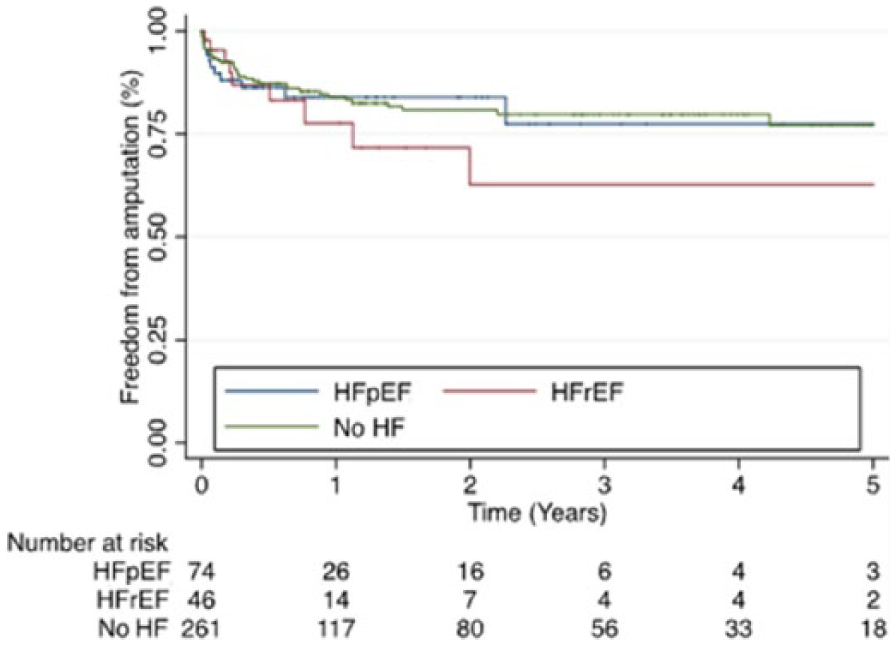
Kaplan–Meier survival curves comparing those with no-HF vs HFpEF vs HFrEF in terms of freedom from amputation. (No-HF, no prior history of heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.)
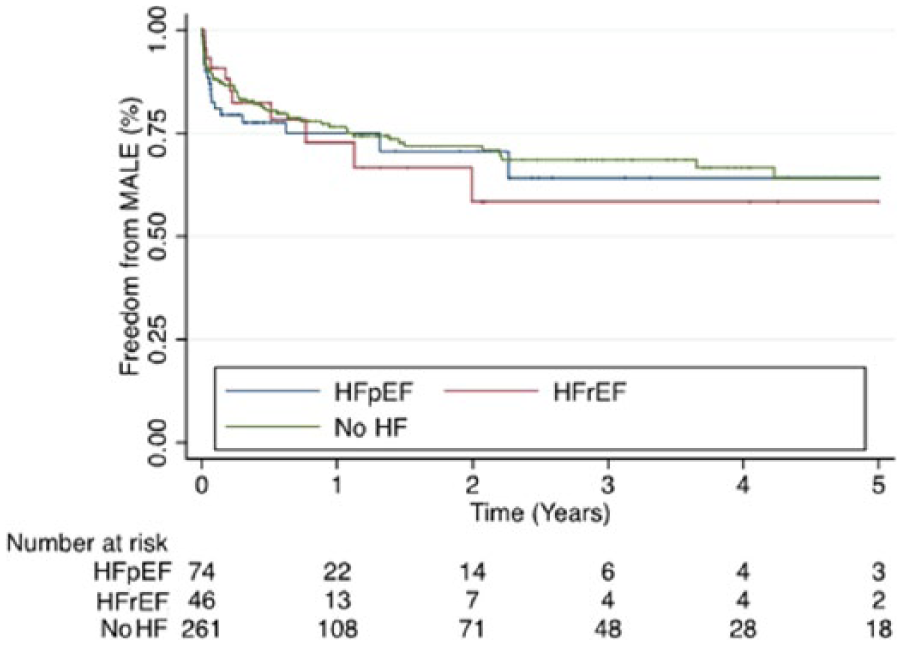
Kaplan–Meier survival curves comparing those with no-HF vs HFpEF vs HFrEF in terms of freedom from MALE. (No-HF, no prior history of heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; MALE, major adverse limb events.)
Five-year outcome rates and adjusted hazard ratios: (A) within-group comparison; (B) between-group comparison.
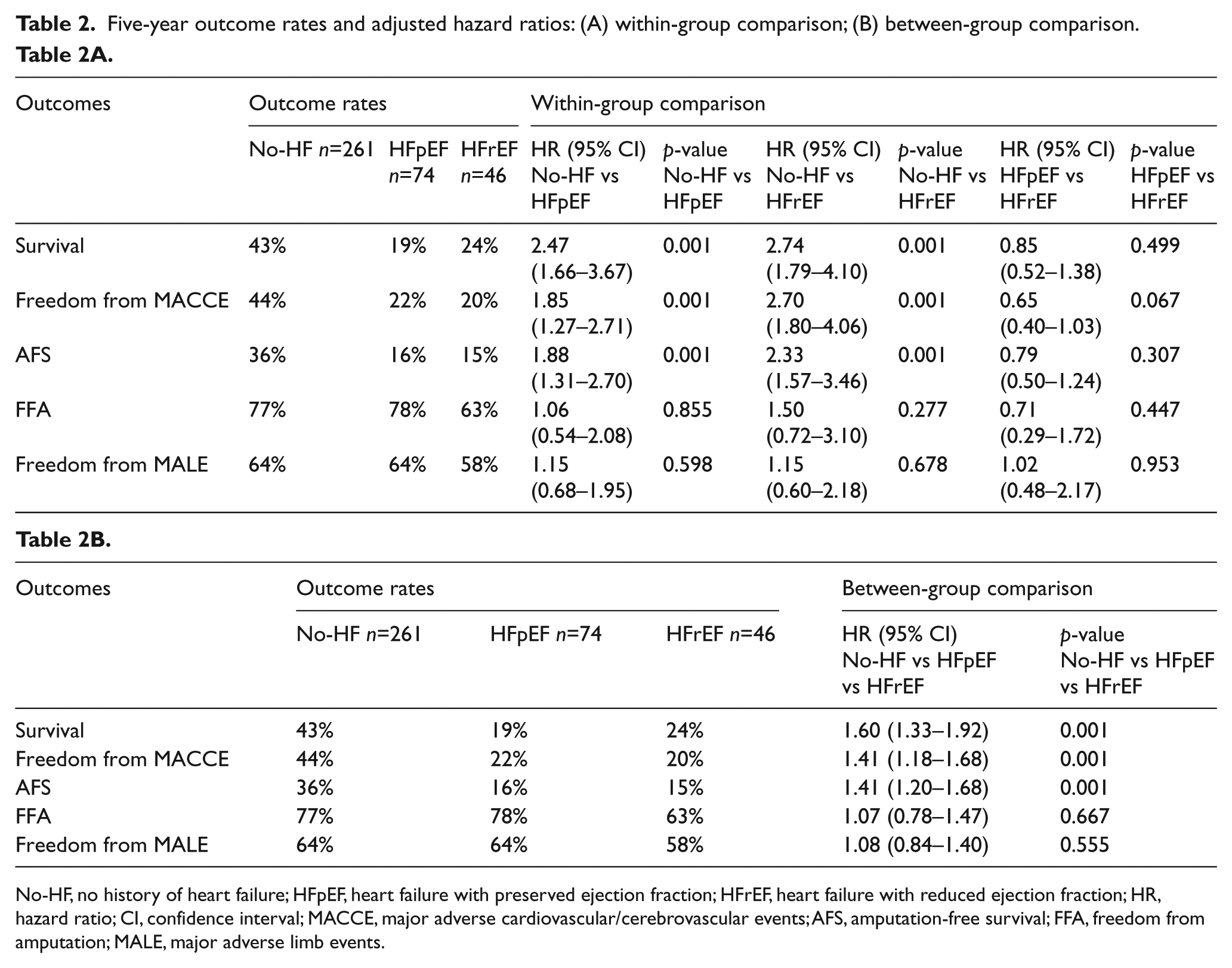
No-HF, no history of heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; HR, hazard ratio; CI, confidence interval; MACCE, major adverse cardiovascular/cerebrovascular events; AFS, amputation-free survival; FFA, freedom from amputation; MALE, major adverse limb events.
Multivariate analysis revealed that advanced age (HR 1.05, CI 1.03–1.06, p=0.001), a history of HF (HR 1.33, CI 1.09–1.64, p=0.006), CAD (HR 1.58, CI 1.07–2.32, p=0.021) and ESRD (HR 2.28, CI 1.55–3.35, p=0.001) were independent risk factors for 5-year mortality when adjusted for other co-morbidities (Table 3).
Multivariate analysis of risk of mortality with unadjusted and adjusted hazard ratios.
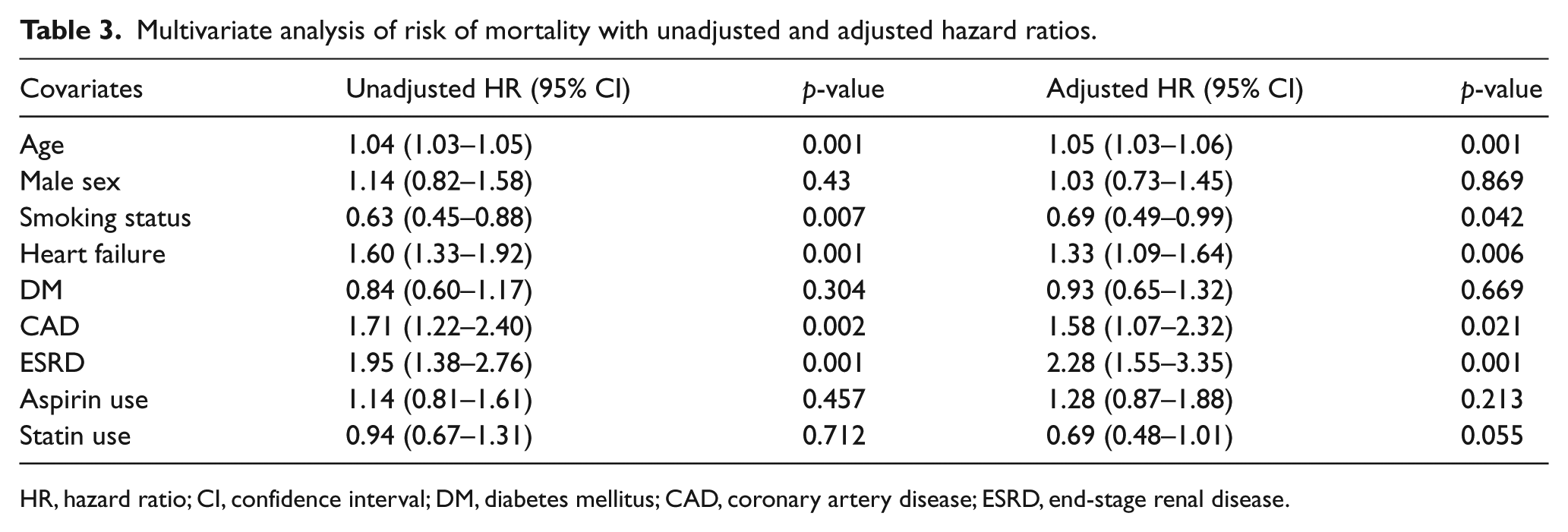
HR, hazard ratio; CI, confidence interval; DM, diabetes mellitus; CAD, coronary artery disease; ESRD, end-stage renal disease.
Discussion
In our cohort of patients who presented with CLI, the concomitant diagnosis of heart failure was associated with nearly twofold higher mortality and MACCE than in those without heart failure. Patients with HFrEF and HFpEF had similarly poor outcomes. While these rates are based on a relatively small cohort of patients, it suggests that these dismal outcomes for CLI patients with heart failure were independent of the presence of left ventricular systolic dysfunction. Interestingly, the diagnosis of heart failure was not associated with greater FFA or MALE (i.e. need for amputation or bypass surgery at 5-years post-intervention), suggesting that adverse outcomes in PAD patients with heart failure are driven largely by cardiovascular/cerebrovascular events rather than limb-related events.
Within the general heart failure population, studies comparing survival between those with HFpEF and HFrEF have been inconsistent.24–27 However, two large meta-analyses revealed significantly lower mortality in patients with HFpEF compared to those with HFrEF.28,29 Thus, the equally poor outcomes in our HFpEF and HFrEF groups were unexpected, especially given the higher prevalence of CAD in those with HFrEF. In addition to the restricted sample size, possible explanations for the similar outcomes between the heart failure groups include lower aspirin (61% vs 83%, p=0.012) and ICD use (9% vs 26%, p=0.015) in the HFpEF group compared to those with HFrEF. Notably, the rate of ICD implantation in the HFrEF group was only 26%, which is considerably lower than anticipated given the current ACCF/AHA heart failure guideline recommendation for ICD implantation for primary prevention in those with an EF ≤35%. 23 Increased compliance with this recommendation may improve outcomes in the HFrEF population. The higher than expected mortality in the HFpEF group, which cannot fully be explained by less favorable baseline characteristics, demonstrates the importance of recognizing both those with preserved and reduced ejection fraction and a concomitant diagnosis of CLI as high-risk patients.
Previous studies evaluating the impact of heart failure on short-term mortality and limb outcomes in CLI patients have demonstrated that heart failure is associated with increased mortality.13,17,30–33 However, mortality data extending beyond 1-year post-intervention is scarce and many have focused only on reduced ejection fraction as a risk factor for increased mortality.11,14,15 The study by Meltzer and colleagues is the only other to our knowledge that directly compared the outcomes of PAD patients with heart failure based on ejection fraction. 16 They similarly reported reduced 1- and 2-year survival in patients with a diagnosis of heart failure (75.3±3.4% 1-year and 64.3±4.5% 2-year survival) compared to those without a diagnosis of heart failure (89.4±1.3% 1-year and 82.3±1.8% 2-year survival rates), with no significant survival difference between the two heart failure groups stratified by ejection fraction (p<0.001). 16 These survival rates were higher across all groups compared to our cohort, which may be related to the higher Rutherford class and increased prevalence of concomitant ESRD in our cohort. Our study extends these findings to a longer follow-up period of 5 years, indicating that the overall trend toward worse survival in heart failure patients extends well beyond the early post-intervention period. Furthermore, survival remains equally poor between the two heart failure groups stratified by ejection fraction, even at 5 years post-intervention.
Optimizing heart failure management will be critical in improving outcomes in this patient population. Previous studies have revealed that increased compliance with guideline-directed medical therapy (GDMT) can reduce mortality and decrease hospitalizations in heart failure patients.34,35 In our study, overall medication adherence was suboptimal, illustrating the need for strategies focused on enhanced application of GDMT. Within the HFrEF group, improving compliance with beta-blockers, ACEi/ARB, hydralazine, nitrates and aldosterone receptor antagonists should be prioritized as it would likely improve outcomes. Additionally, the introduction of the 2016 ACC/AHA/Heart Failure Society of America update on pharmacological therapy in heart failure offers novel medical therapies, including angiotensin receptor-neprilysin inhibitors and ivabradine.36–39 Incorporation of these newer agents where indicated may provide an opportunity to reduce mortality in patients with concomitant CLI and HFrEF. Our findings also emphasize that patients with HFpEF comprise a high-risk population, yet optimization of heart failure management in this group continues to pose a challenge and new therapies to improve mortality have yet to emerge. 40 Blood pressure control remains a mainstay of treatment for patients with HFpEF. 23 ACEi/ARB are appropriate anti-hypertensive agents, given their role as GDMT for secondary prevention of PAD.41–43 Additional investigations will be required to quantify the benefit of compliance with GDMT on outcomes in CLI patients with heart failure.
Data from our cohort also suggest that cardiovascular and cerebrovascular events, rather than limb-related complications, are linked to increased mortality in the heart failure group. Thus, cardiovascular risk factor modification is also essential in these patients. The current ACC/AHA guidelines recommend the use of antiplatelet therapy, statins, ACEi, and smoking cessation for secondary prevention and cardiovascular risk reduction in patients with PAD.41–43 In our cohort, aspirin utilization was highest in the HFrEF group (83%), which is likely related to the higher rates of CAD in this population. Active smoking was highest in the no-HF group (29%), which may be related to less smoking cessation education targeted toward this population. Statin and ACEi/ARB use were similar between the three groups and averaged 60–70%; these rates are clearly suboptimal and increasing compliance with these medications will be important in improving outcomes. A study from our PAD registry demonstrated that patients treated with all four GDMT had lower rates of MACCE, MALE, amputation and death despite higher rates of baseline co-morbidities, including heart failure, compared to those managed with fewer than four GDMT. 44 Thus, increasing the utilization of all four GDMT in this population will be critical in lowering the cardiovascular risk and mortality. In our cohort specifically, focus should be aimed at increasing the use of aspirin in the no-HF and HFpEF groups, emphasizing smoking cessation in the no-HF group and improving compliance with statins and ACEi/ARB in all groups.
This study outlines the importance of identifying heart failure as a major risk factor for mortality in CLI patients, as it has been largely under-emphasized historically. Several scoring systems have been created to determine risk factors associated with increased mortality in CLI patients undergoing revascularization. The PREVENT III risk scale,45,46 BASIL risk model, 47 CRAB risk model, 48 Finnvasc risk scale 49 and, most recently, the ERICVA model 50 have all been developed to predict mortality in CLI patients undergoing revascularization, with advanced age, presence of tissue loss, and a history of renal insufficiency/failure, DM and CAD among the common risk factors, but, surprisingly, heart failure was not included as a risk factor in these models. Soga and colleagues established a risk scoring system to determine 2-year life expectancy in CLI patients, which allocates points based on the degree of left ventricular systolic dysfunction but does not include heart failure patients with an EF ≥50%. 14 Our study emphasizes that the diagnosis of heart failure, irrespective of ejection fraction, should be considered to more accurately identify high-risk CLI patients. Risk factor modification and increased compliance with heart failure therapy optimization is crucial to improve outcomes in this patient population.
Limitations
This is a retrospective, observational cohort study that is subject to confounding variables and cannot infer causality, but only demonstrates associations. The results in this study are based on a relatively small sample size. The modest cohort size, especially within the heart failure subgroups, limits the ability to detect smaller differences between the groups. Additionally, much of the clinical data are gathered through review of the EMR, which has its own inherent limitations. Of note, many cases of death were identified using the social security death index, which does not provide details regarding cause of death. Through retrospective analysis, it was not always possible to assess the etiology of study endpoints, such as contributors to MALE or MACCE. Another limitation of the study is the inability to identifying patients with new-onset or worsening heart failure within the registry as a large number of patients within the registry did not follow up for their routine care at UC Davis and the diagnosis of heart failure was based on co-morbidities identified during the initial visit only. Defining worsening heart failure becomes subjective and certain markers such as BNP, pulmonary edema, weight gain, etc., may be subject to confounders. Utilizing the number of hospital admissions for heart failure would also be unreliable as many patients sought acute health care needs outside of the UC Davis system. The inability to quantify functional outcomes, such as return to ambulation, wound healing and exercise tolerance, in these patients is another limitation of this retrospective study.
Additionally, medication usage in the heart failure population was lower than expected, which cannot be fully explained by the data; it is possible this cohort of patients was non-compliant with medications, which may confound study outcomes. It may also be that systematic under-treatment with ACEi was related to a clinical focus on the patients’ symptoms of PAD. Consistent with this, other studies of patients with PAD have shown low prevalence of ACEi prescriptions despite a Class IIa indication in the setting of PAD, and a Class I indication for heart failure.51,52 Finally, this is a single-center study and our results may not be generalizable to CLI patients with heart failure in other institutions and regions.
Conclusions
Our study reveals that for patients with CLI, concomitant heart failure is associated with extremely poor 5-year survival. The equally poor survival in heart failure patients with both preserved and reduced ejection fractions highlights that left ventricular systolic dysfunction alone is inadequate for predicting adverse outcomes. Cardiovascular risk reduction and optimization of heart failure management in patients with CLI is needed to improve outcomes in this high-risk population.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John Laird is a consultant for Boston Scientific, Covidien, Abbott, Bard, and Medtronic and Ehrin Armstrong is a consultant for Abbott Vascular and Spectranetics; however, these authors report no conflicts of interest related to this study. Stephen W Waldo receives unrelated investigator initiated research grants from Abiomed Incorporated, Cardiovascular Systems Incorporated and Merck Pharmaceuticals.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
