Abstract
Although coronary obstruction due to atherosclerosis is the most common cause of myocardial ischemia, a significant proportion of patients have myocardial ischemia in the absence of obstructive epicardial coronary artery disease (CAD). This finding is more common among women and alternative causes can mediate myocardial ischemia. Abnormalities in vascular structure, alterations in coronary vasomotion and dysfunction of the coronary microcirculation can all cause ischemia in the absence of obstructive CAD due to atherosclerosis. In this review, we provide an update on three alternative causes of myocardial ischemia: spontaneous coronary artery dissection (SCAD), vasospastic angina (VSA) and coronary microvascular dysfunction (CMVD). We review pathophysiology, clinical presentation, diagnosis, treatment and outcomes related to these important clinical entities. There is increasing interest in better defining this patient population with use of advanced imaging and testing tools. Despite the increased associated risk with future cardiac events, evidence-based treatments for these diagnoses remain under-studied and poorly defined. These alternative diagnoses should be kept in mind when evaluating women with myocardial ischemia without obstructive CAD due to atherosclerosis.
Keywords
The University of Virginia School of Medicine designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™ per article. Physicians should claim only the credit commensurate with the extent of their participation in the activity. Learners are expected to read this article along with any references and supporting material as appropriate, and complete the online post-test questions with an 80% pass rate to receive credit. Post-test questions are accessed through the member portal on the Society for Vascular Medicine (SVM) website (www.vascularmed.org). Please note that CME credits are only available to members of the SVM. This activity expires two years after the publication date, on April 1, 2019.
Background
Sex-based differences in symptoms, treatment and outcomes related to ischemic heart disease are in part due to inherent differences in the pathobiology of vascular structure and function between men and women.1,2 Women are more likely to experience angina and present with acute myocardial infarction (AMI) in the absence of obstructive epicardial coronary artery disease (CAD) due to atherosclerosis.3–5 Furthermore, up to 10% of patients who present for diagnostic coronary angiography for evaluation of myocardial ischemia do not have evidence of obstructive CAD due to atherosclerosis or a ‘culprit lesion’. 6 In these patients, alternative causes of myocardial ischemia are responsible for the clinical presentation and should be considered.
The aim of this review is to provide an update on three clinical entities that can mediate myocardial ischemia and/or infarction in the absence of obstructive CAD due to atherosclerosis: spontaneous coronary artery dissection (SCAD), vasospastic angina (VSA), and coronary microvascular dysfunction (CMVD). It is important to keep each of these alternative causes of ischemia in mind when evaluating women who present with signs and symptoms of myocardial ischemia in the absence of obstructive epicardial CAD.
Spontaneous coronary artery dissection
Introduction
First reported in 1931 by Pretty, 7 spontaneous coronary artery dissection (SCAD) is now a well-known cause of AMI. SCAD is defined as spontaneous separation of the coronary arterial wall that is unrelated to trauma and atherosclerosis. 8 Once thought to be a rare diagnosis, SCAD is now diagnosed more frequently commensurate with advances in invasive coronary imaging, in particular optical coherence topography (OCT). Approximately 1500 cases of SCAD have been published, with the vast majority of reports occurring within the last 5 years.9–11 There remain questions about the true prevalence of SCAD given the limitations of angiography alone to make the diagnosis. In a Japanese study where OCT was routinely used, SCAD was diagnosed in 4% of all patients undergoing coronary angiography. 12
More than 90% of cases are reported in women. Age at presentation typically is from 44 to 55 years; however, it also has been described in older patients.13–15 In one series of women younger than 50 years of age, who presented with acute coronary syndromes, SCAD was diagnosed in 8.7% of the patients. 16 In a more contemporary series, the prevalence was as high as 20% among women younger than 50 years old.14,17 Furthermore, in a recent Japanese series, SCAD was the cause of myocardial ischemia in 35% of women younger than 50 years old. 18 SCAD is the most frequent cause of myocardial ischemia in pregnancy, accounting for 43% of all pregnancy-related myocardial ischemia. 19 Among women presenting with SCAD related to pregnancy, the age at presentation is much younger than in women with SCAD who are not pregnant.
Pathophysiology
There are two principal pathophysiologic mechanisms through which a spontaneous coronary artery dissection may occur. First, there can be bleeding within the vessel wall, which is thought to be secondary to spontaneous rupture in the vasa vasorum, leading to intramural hemorrhage and creation of a false lumen.11,20 Second, there can be frank disruption at the interface between the intima and the vessel lumen. This allows accumulation of blood within the vessel wall and creation of a false lumen with intramural hemorrhage (Figure 1). The final common pathway for myocardial ischemia and infarction is compression and occlusion of the true lumen by the false lumen. The presence of thrombi in the true and false lumen might also promote ischemia; however, this occurrence is less likely than compression of the true lumen. 21
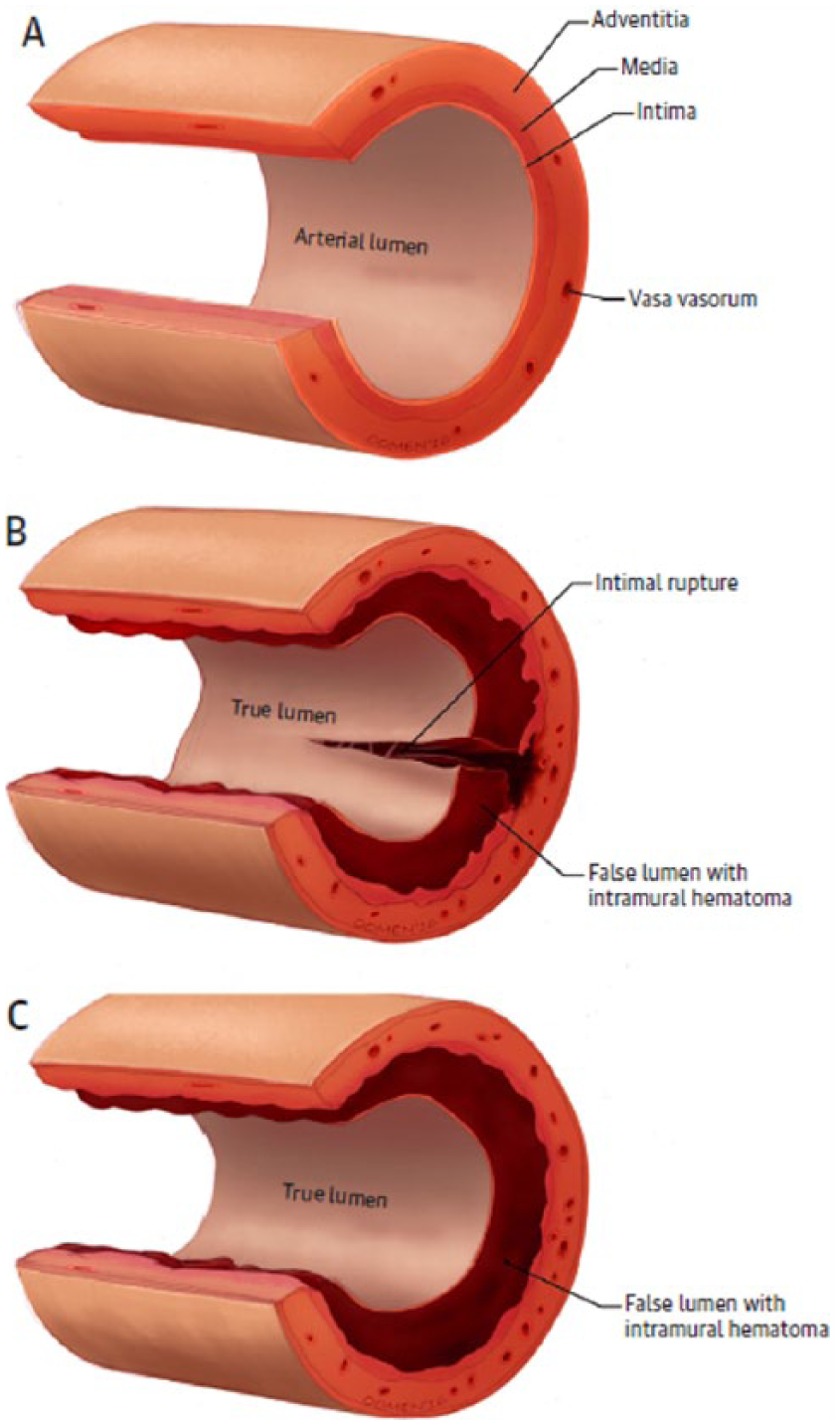
Pathophysiology of spontaneous coronary artery dissection (SCAD). (A) Normal coronary artery. (B) Intimal rupture initiating tear, with development of intramural hematoma. (C) Spontaneous bleeding into arterial wall, creating a false lumen filled with intramural hematoma. Reprinted from ref. 11, with permission from Elsevier. Copyright © 2016 American College of Cardiology Foundation.
Common triggers for SCAD include circumstances associated with increased cardiocirculatory shear stress, or blood pressure such as intense emotional trauma, isometric exercise, and activities which create a Valsalva-like response such as coughing, child labor or retching.22–24 Catecholamine surge may play a pathogenic role by increasing shear stress, leading to intimal tears or bleeding within the vasa vasorum. 13 More than half of the patients with SCAD have reported a trigger prior to the presentation. An emotional trigger was reported in 40%, and physical stress as a precipitating factor in 25% of patients. 13
The predisposing cause of SCAD is not completely understood, and most likely comprises multiple factors. There is an association of SCAD with fibromuscular dysplasia (FMD).25,26 FMD has been found in up to 80% of patients with SCAD.9,13 FMD can lead to dysplasia and disruption of the smooth muscle cells, fibroblasts and surrounding connective tissue of the vessel wall. Identifying features of coronary FMD is challenging, but coronary tortuosity, ectasia and dilated coronary artery segments have been observed among patients with suspected coronary FMD. 27
Pregnancy-associated SCAD can occur antepartum, or early or late in the post-partum period.19,28 Contrary to previous reports, pregnancy-related SCAD is thought to represent less than 5% of all SCAD cases. 29 Both estrogen and progesterone can play a part in weakening the vessel wall; elevated progesterone levels impair collagen synthesis leading to weakening of the arterial media. 30 In addition, changes in intravascular volume, and cardiac output may increase shear stress and predispose artery to intimal tears. 31 Likewise, exposure to exogenous estrogens and progesterone can lead to weakening of the arterial wall. 32 Hormone therapy including oral contraception and post-menopausal hormone replacement therapy has been shown to be associated with a twofold higher risk of recurrent myocardial ischemia with SCAD, compared with those not on hormones. 33
Clinical presentation and diagnosis
The majority of patients with SCAD and AMI have chest pain and elevated cardiac biomarkers.13,14 Some patients present with ventricular arrhythmias and cardiogenic shock.13,34 The presence of ST-segment elevations can vary. Compared with patients with myocardial ischemia caused by coronary atherosclerosis, patients with SCAD have lower levels of biomarker elevation and tend to have preserved left ventricular (LV) function. 18 In addition, the vast majority of patients with SCAD will have an improvement in LV function on follow-up.32,34
Although coronary angiography is the first step in the evaluation for patients with AMI, the diagnosis of SCAD by angiography alone can be challenging. More advanced coronary imaging tools, such as OCT and intravascular ultrasound (IVUS), enable more precise imaging of the arterial wall. 35 Both IVUS and OCT have allowed for a retrospective characterization of the various types of SCAD patterns seen on angiography. Nonetheless, the routine use of these tools comes with risk and increased cost, and they are not widely available. 11
The SCAD angiographic classification categorizes three types of coronary artery dissection (Figure 2). Type 1 dissections demonstrate arterial wall staining and the presence of multiple lumens. Type 2 dissections can be varied and subtle to diagnose on angiography. They tend to be diffuse with striking change in vessel caliber, and subcategorized depending on whether they are bordered by normal-appearing vessel segments (Type 2A) or extend to the distal tip of the artery (Type 2B). Type 3 dissections can mimic atherosclerosis and cannot be diagnosed on angiography. Type 2 SCAD is the most prevalent presentation (67.5%) followed by Type 1 SCAD (29.1%). 13 SCAD can affect any of the major or branch coronary arteries and involve proximal or distal segments. The left anterior descending artery and its branches are most commonly involved (45–61%), followed by the left circumflex and right coronary arteries.13,35,36 Multiple concurrent coronary artery dissections are uncommon (less than 10%). 36
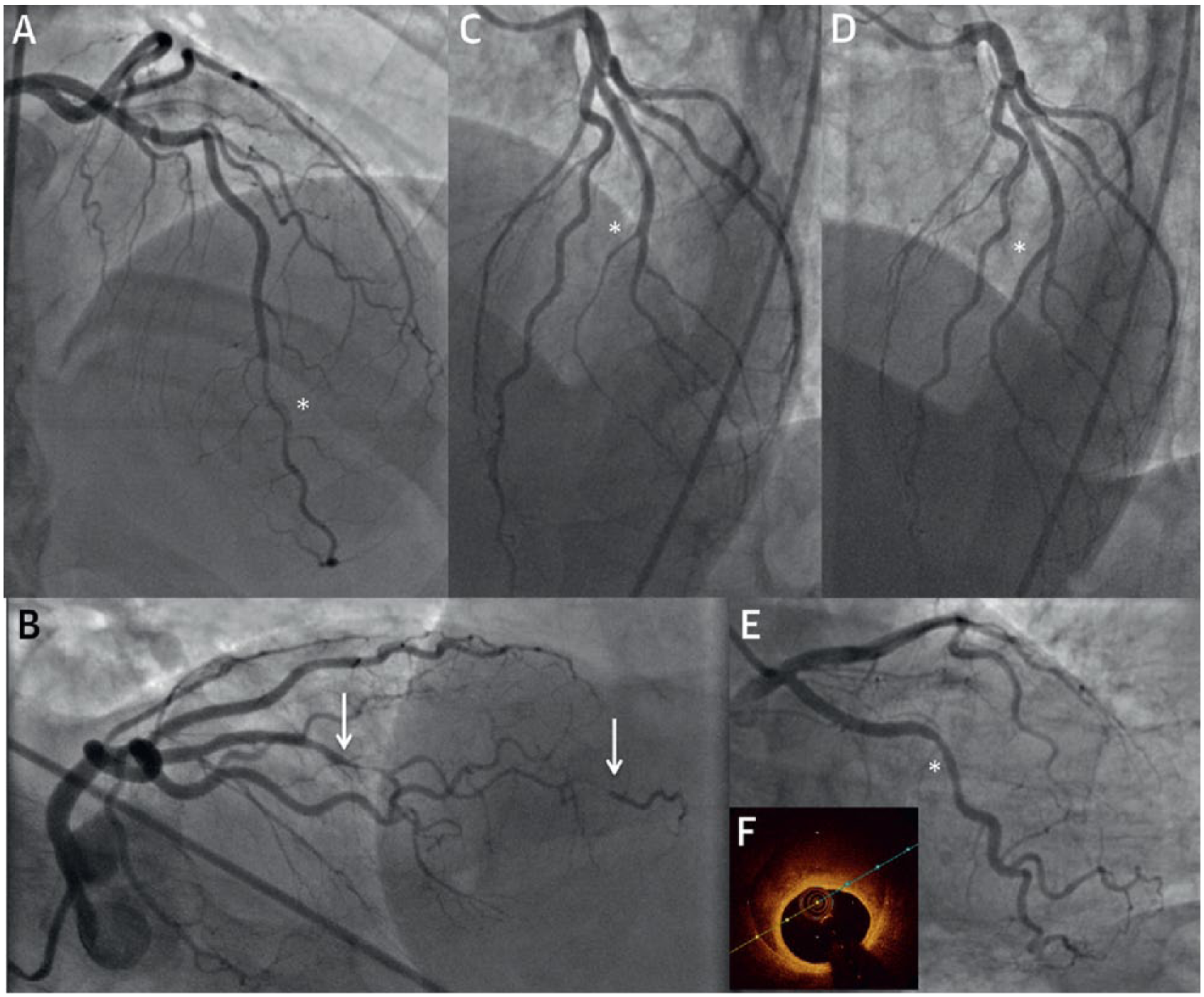
Angiographic classification of spontaneous coronary artery dissection (SCAD). (A) Type 1 dissections with arterial wall staining (*) and presence of multiple lumens. Type 2 dissections can be varied and more subtle to diagnose on angiography. They tend to be diffuse with striking change in vessel caliber. (B) Type 2A dissections (arrows) are surrounded by normal vessel segments (arrows). (C) Type 2B dissections or extend to the distal tip of the artery (*). (D) Healed segment from one year later (*). (E) Type 3 dissections mimic atherosclerotic dissection and cannot be diagnosed on angiography (*). (F) Optical coherence tomography showing intramural hematoma. Reprinted from ref. 11, with permission from Elsevier. Copyright © 2016 American College of Cardiology Foundation.
As mentioned above, OCT and IVUS are used to better understand and aid in the diagnosis of SCAD (Figure 3). 37 OCT provides superior spatial resolution compared to IVUS. OCT better visualizes intimal tears, false lumens, and the presence of intramural hematoma. 21 IVUS has better tissue penetrance and better shows the extent of intramural hematoma. There are risks associated with performing either OCT or IVUS in patients with SCAD. These risks include propagation of dissection and also causing vessel occlusion as a result of embolization. 11
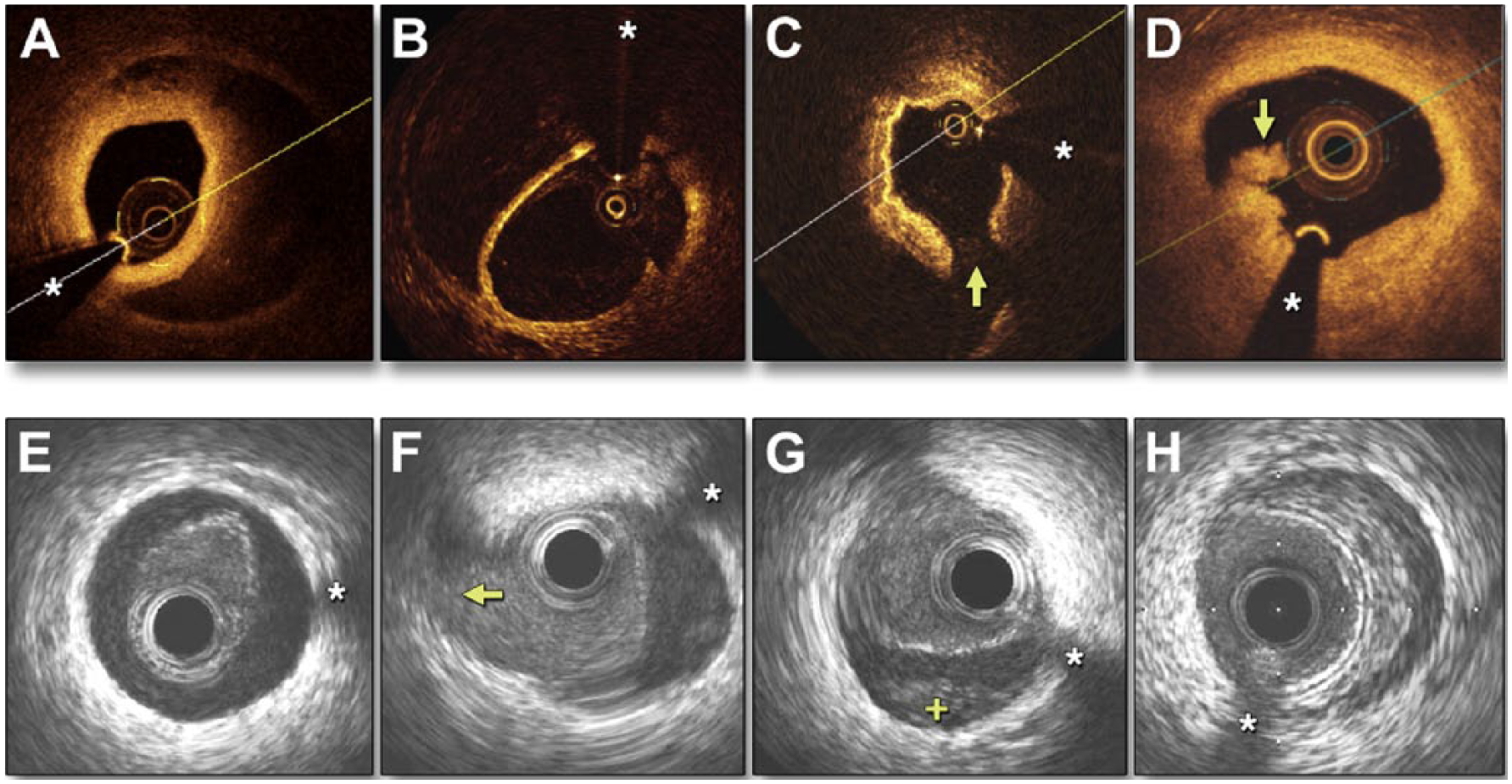
OCT and IVUS imaging in spontaneous coronary artery dissection (SCAD). Panels A–D show optical coherence tomography (OCT) images. (A) Double lumen with a thick intimomedial membrane. (B) Double lumen with a thin intimal membrane. (C) Intimal rupture (arrow). (D) Intracoronary thrombus protruding into the true lumen (arrow). Panels E–H show intravascular ultrasound (IVUS) images. (E) Elliptical, echogenic, true lumen fully detached from the outer vessel wall. (F) Double lumen with a side branch (arrow) emerging from the true lumen. (G) Double lumen with false lumen thrombosis (+). (H) Intramural hematoma. *Denotes wire artifact. Reprinted from ref. 37, with permission from Elsevier. Copyright © 2013 American College of Cardiology Foundation.
Treatment
Evidence-based therapies for SCAD are not well established and current recommendations are based mainly on expert opinion.8,38 Therapies typically used for the management of patients with acute coronary syndromes have not been evaluated among patients with SCAD.
The recommended mainstay of medical therapy is beta blockers, despite lack of a robust evidence base. These help to reduce myocardial workload and shear stress. In addition, beta blockers might lower the risk of ventricular arrhythmia in the setting of AMI following SCAD. The efficacy of antithrombotic therapies is unproven. 11 Theoretic benefit is preventing coronary thrombus formation and propagation, whereas potential risk is extension of intramural hematoma. In light of these relative benefits and risks it is reasonable to consider aspirin as a treatment option. 8 Similarly, P2Y12 inhibitors theoretically may be beneficial by reducing the risk of luminal thrombus. 39 By comparison, more potent anticoagulants, such as heparin and glycoprotein IIB/IIIA inhibitors, may increase the risk of vessel wall bleeding. In addition, thrombolytic therapy has also been associated with increased bleeding and worst outcomes in patients with SCAD. 40
The arterial injury usually heals over time in most patients who are managed medically and attempts at revascularization come at modest success rates.15,29 The vast majority of SCAD cases will heal in about a month. In a series when follow-up angiography was performed after 26 days, normalization of previously dissected arterial segments was reported in 100% of cases. 13
However, there are clinical scenarios which might require a more aggressive approach to reperfusion, including percutaneous coronary intervention (PCI) or surgical revascularization. In contemporary practice, about 3–10% of patients will require revascularization secondary to SCAD progression. 13 PCI comes with challenges, and historically has been associated with suboptimal success rates (64%). There is a reported high rate of dissection extension (57%), urgent coronary artery bypass (CABG) (12%) and stent thrombosis (6%).13,41 PCI is associated with the potential for propagating the dissection due to wire or catheter induced trauma 42 and also with the inability to obtain optimal stent apposition due to the presence of intramural hematoma. 43 In addition, a dissected distal vessel might be too small for PCI. OCT or IVUS assisted PCI helps to ensure wire position in the true lumen and optimize stent sizing.44,45
CABG can be considered for very high-risk SCAD patients with extensive myocardium at risk, such as those involving the left mainstem artery and also those who need rescue emergent CABG after failed PCI. Short-term outcomes are good; however, graft patency rates beyond the short-term are substandard, which may relate to re-establishment of competitive native coronary flow as the dissection heals.15,32
Outcomes
Overall in-hospital outcomes are favorable with a <5% mortality rate and a 5–10% rate of major adverse cardiac events (MACE).13,32,34 After discharge, MACE rates vary, but can be up to 10–20%. Approximately 15% of patients have recurrent SCAD at 2 years, 13 and about 25% of patients develop a recurrence in 4–5 years.32,34 Patients with pregnancy-related SCAD represent a higher-risk cohort and tend to have the worst prognosis, with a more proximal area of dissection and larger infarcts. 46
Patients with SCAD should be followed closely given the high recurrence rate. Patients should undergo screening for FMD in non-coronary beds including the cerebral, renal and iliac arteries.11,47 Patient on hormone replacement therapies should be advised against their continued use if possible. There is no proven role for genetic testing for connective tissue disorders. Patients should be informed about the SCAD community on Inspire, a social media driven website used to raise awareness and provide support for patients with SCAD. 48
In summary, owing to better awareness and advanced imaging tools, SCAD is recognized as an important and not infrequent cause of AMI in women. In general, a conservative approach to treatment is recommended, though at times percutaneous and surgical interventions are needed. Overall, patients with SCAD remain at risk for recurrent episodes.
Vasospastic angina
Introduction
VSA was previously referred to as Prinzmetal or variant angina due to variation from the classic description of Heberden’s angina where pain was provoked by increased cardiac work and relieved by rest or the administration of nitroglycerin. 49 Variant angina, as originally described by Myron Prinzmetal, was angina pectoris associated with transient ST-segment elevation that came on at rest or during ordinary activity but was not associated with exercise. 50 It is now considered part of a broader diagnosis characterized by episodes of rest angina which are caused by coronary artery vasospasm and respond to short-acting nitrate therapy.
The overall prevalence of VSA is unknown given the limited availability of testing to diagnose spasm. There is a predilection for VSA in Japanese compared to Caucasian populations.51,52 VSA due to epicardial spasm is more common among Japanese men compared to women. 53 There are reports of a higher prevalence of microvascular spasm among German women but higher rates of epicardial spasm among German men compared to women. 54 Among patients with documented epicardial spasm in response to maximal doses of acetylcholine (ACh), women are more likely to have a higher prevalence of microvascular spasm at submaximal doses of acetylcholine, suggesting contribution of the coronary microcirculation to symptoms and ischemia. This response is significantly less likely in men where epicardial spasm alone mediates myocardial ischemia. 55
Pathophysiology
Under normal circumstances, the epicardial coronary arteries contribute less than 10% to coronary resistance and to the regulation of myocardial perfusion. This is in part due to the low level of myogenic tone in epicardial arteries and the influence of the endothelium, which attenuates vasoconstrictor signals and enhances vasodilator capacity in these vessels. 56 However, when the endothelium is disrupted or damaged, impaired endothelial function combined with heightened vascular smooth muscle (VSMC) reactivity may lead to focal areas of intense constriction and coronary vasospasm. 57
VSMC hyperreactivity is thought to be the main substrate for coronary vasospasm. 58 Animal studies have shown that during spasm, the radial rearrangement of medial VSMC results in medial thickening and folding such that this creates a piston effect leading to constriction or narrowing of the vessel lumen. 59 The predisposition to hyperreactivity remains poorly understood but the rho-kinase pathway is a likely contributor. These kinases play an important part in regulating multiple cellular functions such as contraction, motility, proliferation and apoptosis. 60 Use of rho-kinase inhibitors markedly limits epicardial coronary spasm in porcine models. 61 This pathway remains a key potential target for treatment of coronary vasospasm.
In addition to VSMC hyperreactivity, alterations in pathways which affect endothelial-derived nitric oxide (NO) bioavailability play an important role in promoting coronary spasm. NO also suppresses the endothelial production of endothelin 1 and angiotensin II, which are potent vasoconstrictors and proliferators of smooth muscle cells.62–64 The presence of endothelial dysfunction, enhanced sympathetic nervous system activity, oxidative stress, inflammation and genetic factors are also thought to play a part in the pathophysiology of coronary vasospasm.65–68 These interactions are complex and incompletely understood.
Several triggers for spasm have been described. An increase in catecholamine surge, such as with intense physical exercise, exposure to emotional stress, or extreme cold, have all been described as triggers.69–71 Pharmacologic agents such as catecholamines (dopamine, epinephrine),72,73 parasympathomimetic agents (acetylcholine, methacholine), 74 ergonovine, 75 and cocaine 76 can trigger coronary vasospasm in predisposed patients. In addition, both smoking and alcohol have been associated with coronary spasm.77,78
Clinical presentation and diagnosis
Patients with VSA present with a chronic pattern of recurrent chest pain which can be indistinguishable from classical angina pectoris associated with obstructive CAD. There are certain associations which are more characteristic of VSA and these include episodes which occur predominantly at rest and in the midnight to early morning hours. 79 In addition, patients with VSA are often younger and exhibit fewer classic cardiovascular risk factors, and are more likely to have other vasospastic disorders, such as Raynaud phenomenon and migraine headache. 80 Also, hyperventilation can precipitate an attack of VSA. 81
Electrocardiographic ST-segment changes occur as a result of myocardial ischemia, and with complete occlusion can manifest as ST-segment elevations with reciprocal changes. Specific to coronary spasm, there may be development of a ‘monophasic curve’ with increasing amplitude of the ST-segment and widening of the R wave at the peak of the event and presence of a negative U wave (Figure 4).68,82 The distribution of the ST-segment changes usually reflect the artery feeding the myocardial territory involved, and multi-vessel spasm can precipitate ST-segment changes in multiple territories. The presence of ST-segment depression usually suggests less severe spasm, the presence of collateral vessels, or spasm in a small artery. 83 Patients can present with ventricular arrhythmias or bradyarrhythmias. Multi-vessel spasm may result in sustained lethal arrhythmias and sudden death. 84 Bradyarrhythmias are more likely to be associated with right coronary artery territory ischemia and may present as syncope due to high-grade block.
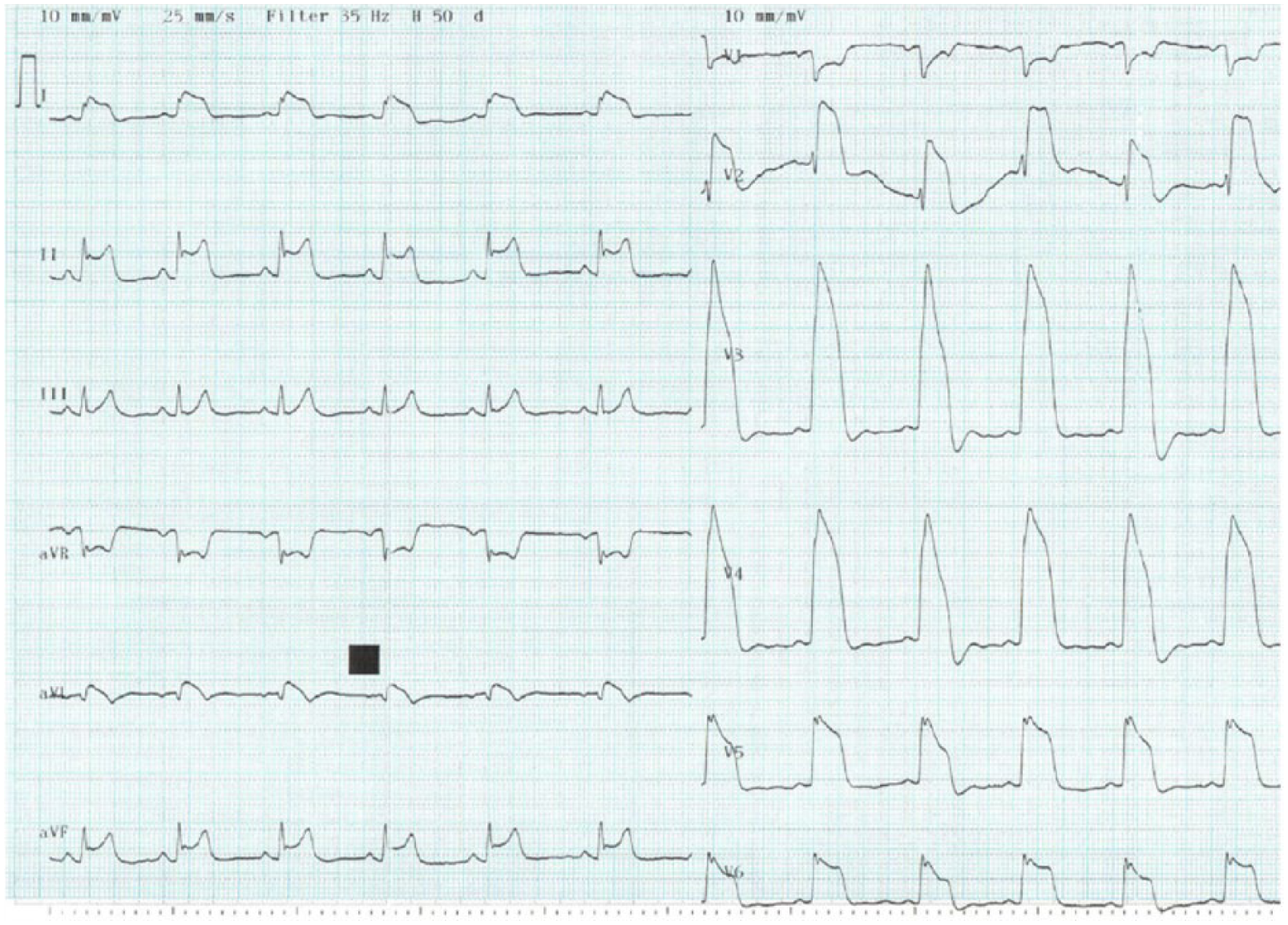
Example of a monophasic curve on electrocardiogram (ECG) during an attack of vasospastic angina. Reprinted from ref. 82, with permission from Elsevier. Copyright © 2015 Cardiological Society of India.
In 2015, the Coronary Vasomotion Disorders International Study Group (COVADIS) established a diagnostic criteria for VSA. 85 There are three core elements establishing the diagnosis of VSA, which include: (1) nitrate-responsive angina; (2) transient ischemic electrocardiographic changes; and (3) angiographic evidence of coronary artery spasm (Table 1). Provocative testing using pharmacologic agents, such as ergonovine and acetylcholine, was used in the past to diagnose VSA, but is employed much less frequently today.74,86–89 A positive test to these drugs is defined as reproduction of chest pain with ischemic ECG changes and >90% vasoconstriction on angiography. Various testing protocols using intracoronary and intravenous administration have been described and are beyond the scope of this review. 90
Diagnostic criteria for vasospastic angina.

ECG, electrocardiogram.
Reprinted with permission from Beltrame et al. (85).
Recommendations for testing differ by medical society consensus, with testing being a Class I indication from the Japanese Circulation Society 91 and more modest Class IIa and IIb recommendations from the European Heart Society and American Heart Association/American College of Cardiology, respectively (Table 2).92,93
Indications for provocative coronary spasm testing.

VSA, vasospastic angina; PCI, percutaneous coronary intervention.
Reprinted with permission from Beltrame et al. (85).
Neither acetylcholine nor ergonovine are approved by the US Food and Drug Administration for diagnosing coronary vasospasm and are only used in specialized experienced centers. Reported complication rates have varied. In the Multicenter Registry Study of the Japanese Coronary Spasm Association, the overall incidence of arrhythmic complications was 6.8%, and the incidence of ventricular tachycardia/fibrillation and bradyarrhythmias was 3.2% and 2.7%, respectively. 94
Treatment
Given the association of VSA, endothelial dysfunction95,96 and smoking, 77 patients should be encouraged to quit smoking. Nitrates and calcium channel blockers (CCB) are the mainstay of treatment. In the acute setting, sublingual nitroglycerin is a first-line agent. 63 Use of longer acting nitrates has not been shown to have prognostic benefit as a single agent to prevent recurrent episodes and their use should be limited to the treatment of more acute symptoms. 97 All classes of CCB can be used as first-line therapy for VSA. CCB inhibit inflow of calcium into the smooth muscle cells through voltage-sensitive calcium channels, thereby causing vasodilation. 98 They have been associated with improved survival in patients with VSA. 99 Other agents include fasudil, which is a rho-kinase inhibitor that inhibits acetylcholine induced spasm and has been used as a research agent but is not available for clinical use in the US. 100 Statins have also been shown to prevent coronary spasm by their effects on endothelial and VSMC function via the rho-kinase pathway. 101 The role of PCI for VSA is limited but can be considered in the presence of spasm with obstructive CAD, or for spasm refractory to medical therapy. 102
There are also drugs to avoid when VSA is suspected or confirmed. Non-selective beta blockers, such as propranolol, can worsen vasospasm by inhibiting beta-2 adrenergic mediated vasodilation, which might attenuate the effects of vasoconstrictive stimuli. 103 Similarly, aspirin should be used with caution and at low doses as it is an inhibitor of prostacyclin production at high doses, which can also promote vasospasm. 104
Outcomes
Long-term survival in patients with VSA is good. Five-year survival rate is estimated to be above 90%.105,106 Angina recurrence is common, with half the patients continuing to experience angina at 3 years. In 2013, the Japanese Coronary Spasm Association published a clinical risk score to predict outcomes in patients with VSA. It identified seven predictors of major adverse cardiac events (out of hospital arrest, history of smoking, resting angina, associated obstructive CAD, multivessel spasm, presence of ST-segment elevations and history of beta blocker use) and assigned an integer score to each proportional to their respective adjusted hazard ratio. Three risk strata were used to classify patients as low (score 0 to 2), intermediate (score 3 to 5) and high (score 6 or more) risk. The incidences of major adverse cardiovascular events in the low, intermediate and high-risk patients were 2.5%, 7.0%, and 13.0%, respectively. 97
Patients with VSA should be monitored for both symptomatic and asymptomatic episodes with ambulatory electrocardiographic monitoring, which would detect ST-segment changes. Persistence of asymptomatic episodes should lead to an intensification of therapy if tolerated.
In summary, VSA is an under-diagnosed cause of myocardial ischemia given the limitations related to testing for VSA in the acute setting. Presence of ischemic ECG changes, chest pain that is responsive to nitroglycerin, and the presence of spasm on angiography are core elements of the diagnosis. Japanese data show a high prevalence of VSA in Japanese men and testing is performed routinely among patients with objective evidence of ischemia in the absence of obstructive epicardial CAD due to atherosclerosis. Whether VSA is more common in women remains debated. Calcium channel blockers are effective drugs to prevent VSA. Overall, survival rates are above 90%; however, recurrence rates can be high.
Coronary microvascular dysfunction
Introduction
Coronary microvascular ischemia was first described in 1973 by Arbogast and Bourassa. They demonstrated objective evidence of transmyocardial lactate production in patients with ECG evidence of ischemia and angina in the absence of angiographic evidence of obstructive CAD due to atherosclerosis. 107 Since then, numerous studies have confirmed the presence of CMVD as a cause of angina and myocardial ischemia in the absence of obstructive CAD and is a finding which is thought to be more common among women.108–111
The prevalence of CMVD in the general population is not established, as testing for CMVD is usually not performed. In a registry of patients with objective evidence of ischemia and non-obstructive CAD by angiography, and who underwent invasive CMVD testing, the prevalence of CMVD was 30%. 112 In a larger cohort, abnormal measures of CMVD involving endothelial dependent or independent pathways were present in two-thirds of patients. 113 Ischemia without obstructive epicardial CAD is more common in women than men undergoing diagnostic coronary angiography and, within this subset of patients, more women are likely to have CMVD compared to men. 113 The reason for this predisposition is incompletely understood but may be due in part to a higher prevalence of luminal plaque erosion among women. Whether this leads to microembolization and subsequent development of CMVD remains debated. Hormonal influences, especially loss of estrogen, may mediate CMVD. Estrogen is a potent stimulator of NO,114,115 and whether cyclical changes in estrogens affect the microcirculation remains to be elucidated.
Pathophysiology
CMVD is likely a consequence of both altered structural and functional aspects of the microcirculation (Figure 5). 116 Functional abnormalities can result from either enhanced microvascular constriction or reduced dilation due to impaired endothelium dependent or independent release of vasoactive substances. 117 Impaired smooth muscle cell relaxation secondary to pathologic conditions such as hypertension can mediate non-endothelium dependent microvascular dysfunction. 118 Similarly, impaired endothelium dependent function may result in inadequate NO release and microvascular dysfunction. 119 Structural causes include physical obstruction (distal embolization), perivascular fibrosis, and rarefaction, as seen in response to conditions such as aortic stenosis and arterial hypertension. Extravascular compression in the clinical setting of hypertrophic cardiomyopathy also has been proposed as a mechanism for CMVD. 120 Predisposing conditions include the traditional risk factors for atherosclerosis such as diabetes, smoking, and hypertension; other factors such as 116 estrogen deficiency, 121 insulin resistance 122 and inflammation also are thought to play a role. 123
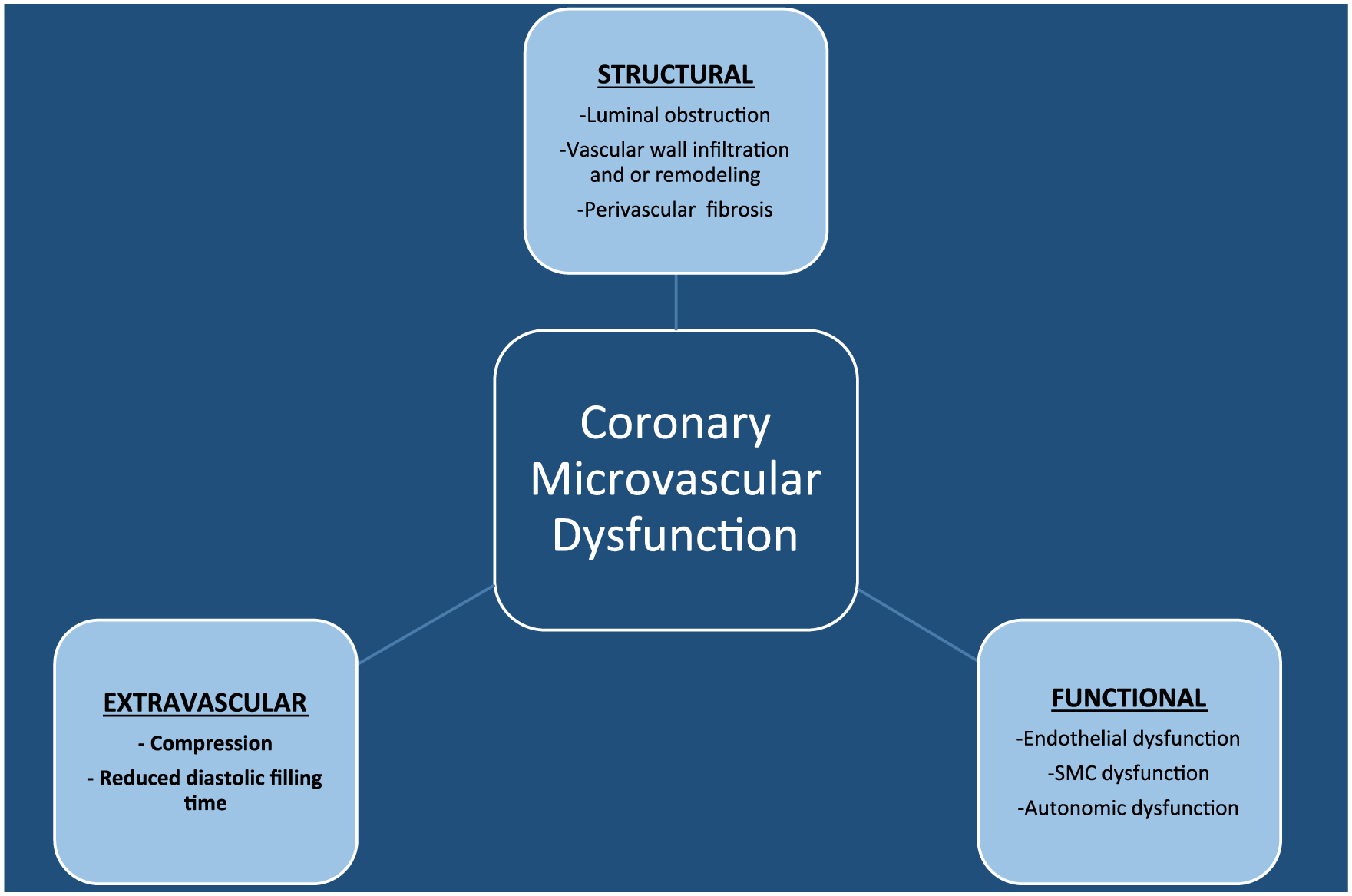
Causes of coronary microvascular dysfunction (CMVD). (SMC, smooth muscle dysfunction.) Reprinted from ref. 116, with permission. Copyright © 2014, Wolters Kluwer Health | Lippincott Williams & Wilkins.
Clinical presentation and diagnosis
Ischemia due to CMVD usually manifests as exertional angina similar to ischemia due to obstructive epicardial CAD from atherosclerosis. The presence of objective evidence of ischemia such as that found during stress testing and/or positive biomarkers in the absence of obstructive epicardial CAD due to atherosclerosis should prompt a work-up for CMVD.
Recently, there has been growing interest in myocardial infarction with non-obstructive coronary arteries (MINOCA). The diagnostic criteria for this includes clinical evidence of a myocardial infarction, a rise and fall of cardiac biomarkers above the 99th percentile, angiographic evidence of non-obstructive CAD (angiographic stenosis <50%), and absence of a specific diagnosis (Figure 6). 124 Recent efforts have focused on better defining this patient population, understanding differences in pathophysiology and evaluating outcomes. The 2015 European Society of Cardiology position paper on MINOCA, 124 provides a standard nomenclature, reviews specific clinical features and mechanisms of disease, and suggests an approach for its evaluation. There is clear need to increase dialogue and efforts towards better understanding this patient population in the hope of optimizing treatment and outcomes. CMVD is part of the differential diagnosis for MINOCA and should be considered in patients meeting these clinical criteria.
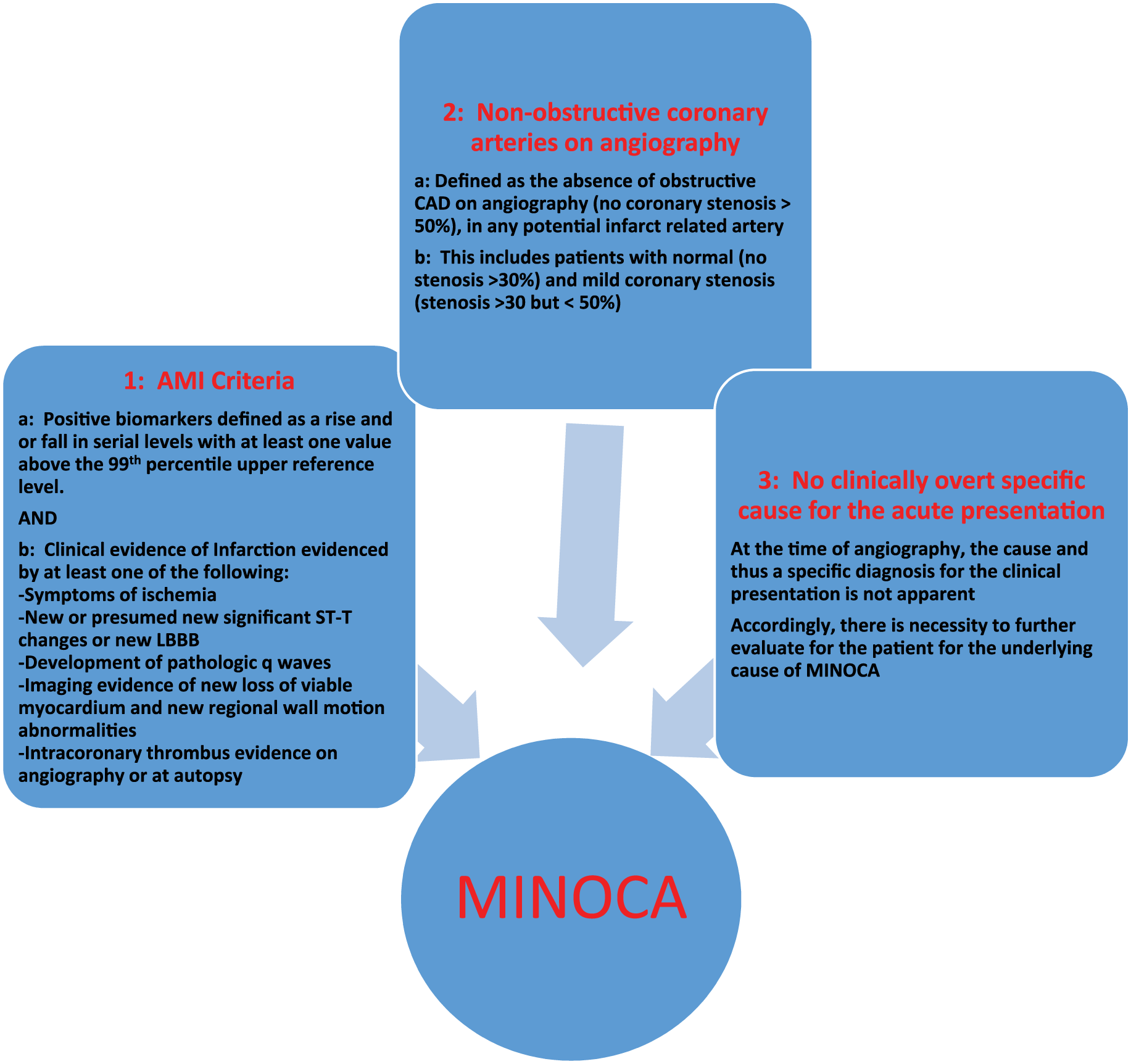
Diagnostic criteria for MINOCA. (AMI, acute myocardial infarction; LBBB, left bundle branch block; CAD, coronary artery disease; MINOCA, myocardial infarction with non-obstructive coronary arteries.) Adapted from ref. 124.
CMVD is clinically versatile and can be associated with other disease entities. A classification scheme was published in 2007 and grouped patients into four types: Type 1 patients have CMVD without CAD or myocardial dysfunction; Type 2 have CMVD in the presence of other myocardial diseases; Type 3 are patients with concomitant CMVD and epicardial CAD; and Type 4 is CMVD as a consequence of reperfusion (Figure 7) . 125 This was revised by Herrmann and colleagues and added post-cardiac transplant CMVD as Type 5. In addition, they highlighted modifiers to take into account mode of presentation (acute vs chronic), presence or absence of symptoms, and treatment status. 126
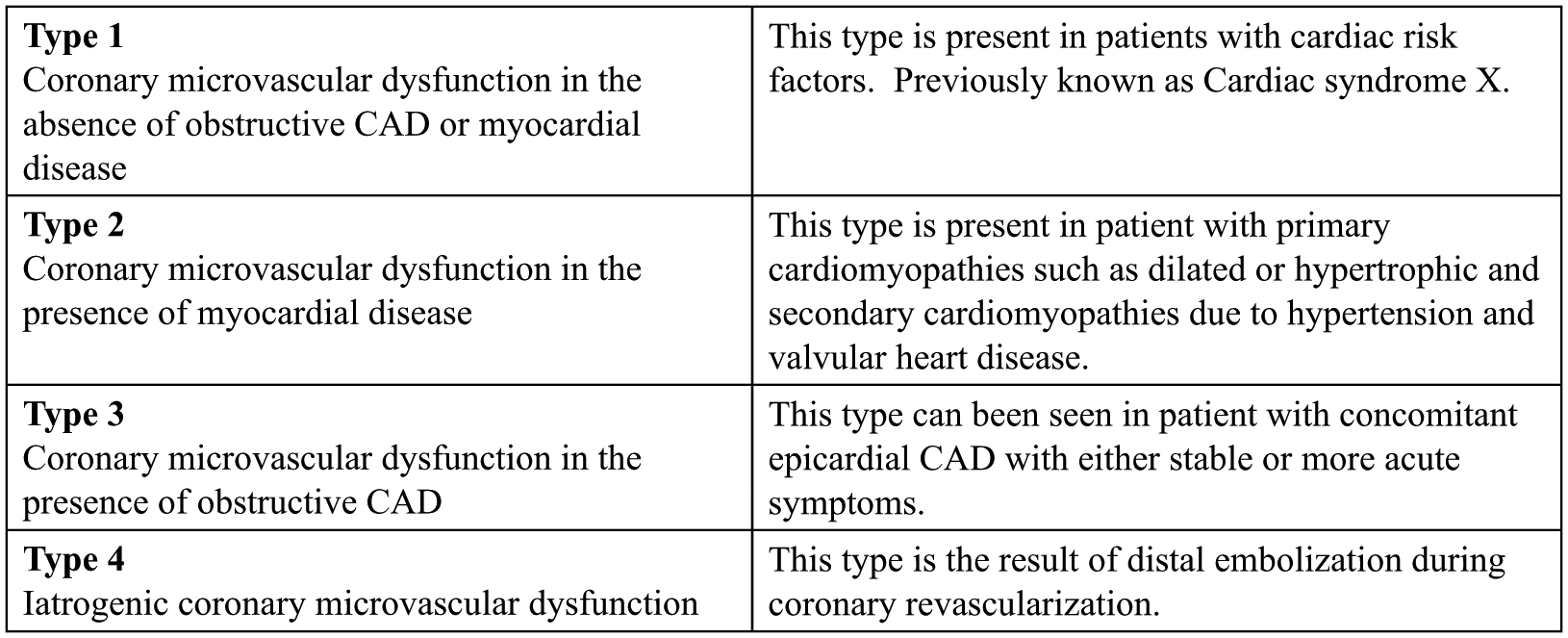
Clinical classification of coronary microvascular dysfunction (CMVD). CAD, coronary artery disease. From ref. 125, Copyright © 2007, Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Current methods of evaluation of the coronary microcirculation are based on quantification of coronary blood flow in response to vasoactive stimuli and angiographic evaluation of ‘myocardial blush’. All testing methods assess the functional status of the coronary microcirculation by indices of blood flow, since visualization of the microcirculation using current techniques is not possible. Non-invasive tests including positron emission tomography (PET), cardiac MRI (CMR) and several echo-based techniques and measurements have been validated and used to assess CMVD. PET uses radioisotopes to quantify myocardial blood flow at rest and in response to stimuli. Large cohort-based studies have found that abnormal PET-based coronary flow reserve is associated with an approximate threefold increase in annual cardiac event rates. 127 Similarly, CMR, along with spectroscopy, can be used to quantify cellular composition and assess myocardial perfusion reserves, and have confirmed the presence of CMVD among patients with angina without obstructive angiographic CAD.109,110 Despite the promise of CMR, it still remains an investigational tool for CMVD. Two-dimensional echo with contrast and Doppler have been used to estimate coronary flow reserve or myocardial blood flow using volumetric measurements during myocardial contrast echocardiography. Doppler echo coronary flow reserve correlates well with invasive Doppler flow wire measurement of coronary flow reserve. 128 It is an independent predictor of adverse outcomes, including death among patients with non-ischemic dilated cardiomyopathy, and those with suspected CAD and normal wall motion. 129
Despite the continued work with non-invasive tools, invasive testing remains the gold standard. Several invasive techniques are used to assess CMVD, and multiple reports support their safety. 130 Angiographic thrombolysis in myocardial infarction (TIMI) frame count and myocardial blush score are considered surrogate markers of microvascular perfusion in the setting of acute myocardial infarction and reperfusion; however, these are limited by their subjective, non-quantitative measurement of blood flow and flow velocities. 131 Invasive techniques using a coronary Doppler wire have been studied extensively.132,133 Flow velocity is measured in response to stimuli, which have known effects on endothelium dependent (acetylcholine) and independent pathways (adenosine). Another well-validated technique employs the use of a 0.014 inch pressure-temperature wire to calculate global microcirculatory resistance. Index of microcirculatory resistance (IMR) is calculated in the state of maximal hyperemia as the distal pressure multiplied by hyperemic transit time. IMR has been shown to be a reliable measure of overall microvascular resistance in animal and human studies and it offers more reproducible results compared to Doppler-based measures of coronary flow reserve. 134 An IMR of >25 has been shown to correlate with increased microvascular resistance (Figure 8).
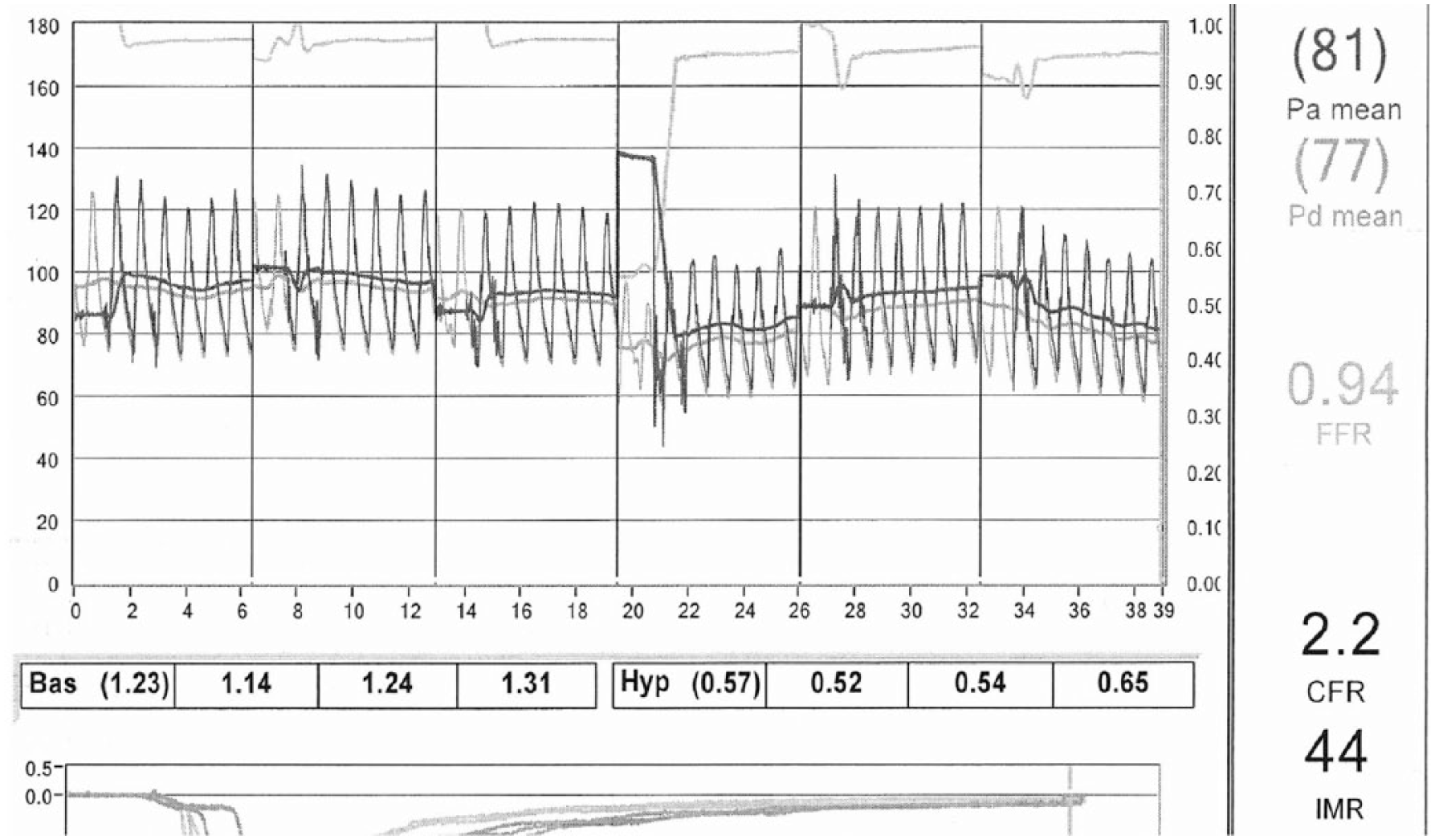
Index of microcirculatory resistance (IMR) measurement in a patient with angina with non-obstructive epicardial CAD. IMR is calculated as hyperemic transit time (HTT) × hyperemic distal pressure (Pd). (Pa, mean aortic pressure; Pd, mean distal pressure; FFR, fractional flow reserve; CFR, coronary flow reserve; Bas, baseline transit time; Hyp, hyperemic transit time.) Reprinted from ref. 116, with permission. Copyright © 2014, Wolters Kluwer Health | Lippincott Williams & Wilkins.
Treatment
There is scarce data to support evidence-based treatments for CMVD. In the presence of secondary causes, treatment centers on optimizing underlying pathology. For patients with Type 1 CMVD, treatment includes medications for symptom relief and optimization of cardiovascular (CV) risk factors. Small studies have shown decreased frequency of chest pain among patients with CMVD who are treated with beta blockers.135,136 Calcium channel blockers such as nifidipine and verapamil and nitrates have been evaluated in patients with CMVD and the results have been inconsistent.135,137 More recently, there is interest in the anti-anginal medication ranolazine for CMVD. Despite an unclear mechanism of effect, ranolazine, compared to placebo, relieves anginal episodes and increases exercise capacity among patients with CMVD. 138
Outcomes
Among patients with or without obstructive CAD, CMVD is an independent predictor of increased long-term risk of CV events.139–141 For example, among women enrolled in the Women’s Ischemia Syndrome Evaluation (WISE) study, those with abnormal coronary flow reserve were noted to have an increased risk of CV events over the 6-year follow-up period. 139 Similarly, abnormal endothelial dependent function was an independent predictor of adverse outcomes among WISE women. 142 In addition, CMVD, secondary to non-ischemic cardiomyopathy, 143 AMI, and as a direct result of reperfusion, is associated with adverse long-term outcomes. 144
In summary, there has been considerable evolution in our understanding of coronary physiology and the coronary microcirculation over the last two decades. CMVD is associated with a myriad of cardiac diseases. Type 1 CMVD, previously known as cardiac syndrome X, can be present in up to a third of patients with ischemia in the absence of obstructive epicardial CAD. A clinical classification can help characterize this heterogeneous group of patients. A growing interest in the use of non-invasive and invasive tools of assessment such as IMR can help make the diagnosis ad hoc at the time of diagnostic coronary angiography. Treatment options remain limited and center on symptom relief and risk factor modification for Type 1 CMVD and treatment of the underlying disorder for causes related to other cardiac pathology.
Conclusion
Women with stable or acute symptoms of ischemia are less likely to present with obstructive epicardial CAD due to atherosclerosis when compared to men. In these patients, alternative causes for myocardial ischemia can be mediated by changes in coronary vascular structure such as occurs in SCAD, abnormal vascular function predisposing to VSA, and both functional and structural alterations within the microcirculation resulting in CMVD. These alternative diagnoses should be part of the evaluation among this subset of patients. Continued efforts towards the use of advanced diagnostic tools will help lay the foundation for better understanding these patients and developing and testing targeted treatments for these important clinical entities.


