Abstract
The toe–brachial index (TBI) is used as an adjunct to the ankle–brachial index (ABI) for non-invasive lower limb vascular screening. With increasing evidence suggesting limitations of the ABI for diagnosis of vascular complications, particularly in specific populations including diabetes cohorts, the TBI is being used more widely. The aim of this review was to determine the sensitivity and specificity of the TBI for detecting peripheral artery disease (PAD) in populations at risk of this disease. A database search was conducted to identify current work relating to the sensitivity and specificity of toe–brachial indices up to July 2015. Only studies using valid diagnostic imaging as a reference standard were included. The QUADAS-2 tool was used to critically appraise included articles. Seven studies met the inclusion criteria. Sensitivity of the TBI for PAD was reported in all seven studies and ranged from 45% to 100%; specificity was reported by five studies only and ranged from 16% to 100%. In conclusion, this review suggests that the TBI has variable diagnostic accuracy for the presence of PAD in specific populations at risk of developing the disease. There was a notable lack of large-scale diagnostic accuracy studies determining the diagnostic accuracy of the TBI in detecting PAD in different at-risk cohorts. However, standardised normal values need to be established for the TBI to conclusively determine the diagnostic accuracy of this test.
Keywords
Introduction
Traditionally, the ankle–brachial index (ABI) has been used as a large vessel screening tool for clinical assessment of peripheral artery disease (PAD). 1 The ABI has been shown to be a sensitive and specific measure of detecting PAD in the general population. 2 However, there is increasing evidence to suggest that in specific populations there is a decrease in the diagnostic accuracy of the test.3,4 Medial arterial calcification (MAC), a stiffening of the arterial wall most commonly in infragenicular arteries used for the calculation of the ABI, 5 is prevalent in the diabetic population, particularly in men and in older age groups, and is thought to reduce the diagnostic accuracy of the ABI. 3 Although MAC artificially inflates the ABI, this cannot always be detected during routine clinical assessment as co-existent PAD may result in the ABI ratio presenting as normal or even low despite its presence. 6
Assessment of the small vessels within the foot and distal extremities also presents an issue for clinicians, as an ABI is not sensitive to occlusions and arterial disease below the ankle. 7 Current international guidelines recommend the toe–brachial index (TBI) as an alternate screening method for PAD in the presence of an elevated ABI;8,9 however, the evidence base for the use of the TBI as a stand-alone diagnostic test remains low. The TBI is a ratio of the systolic toe pressure divided by the highest systolic brachial pressure. Systolic toe pressure can be performed by placing an appropriately sized occlusive pneumatic cuff (between 15 and 25 mm) around the base of the proximal great or second toe, and a photoplethysmography (PPG) probe affixed to the distal pulp of the toe with adhesive tape (Figure 1). A continuous wave Doppler probe may also be used on the digital arteries in lieu of a PPG probe. Once a steady signal is obtained, the occlusive cuff is inflated by a sphygmomanometer 20 mmHg above the last visual PPG waveform. The occlusive cuff is then slowly deflated, with the pressure reading recorded when a consistent waveform returns.10,11 Normal values for the TBI are universally lower than the ABI, with normal being considered between >0.6 and >0.75;12–15 recent research suggests that in normal populations the mean TBI is between 0.94 and 0.98. 16
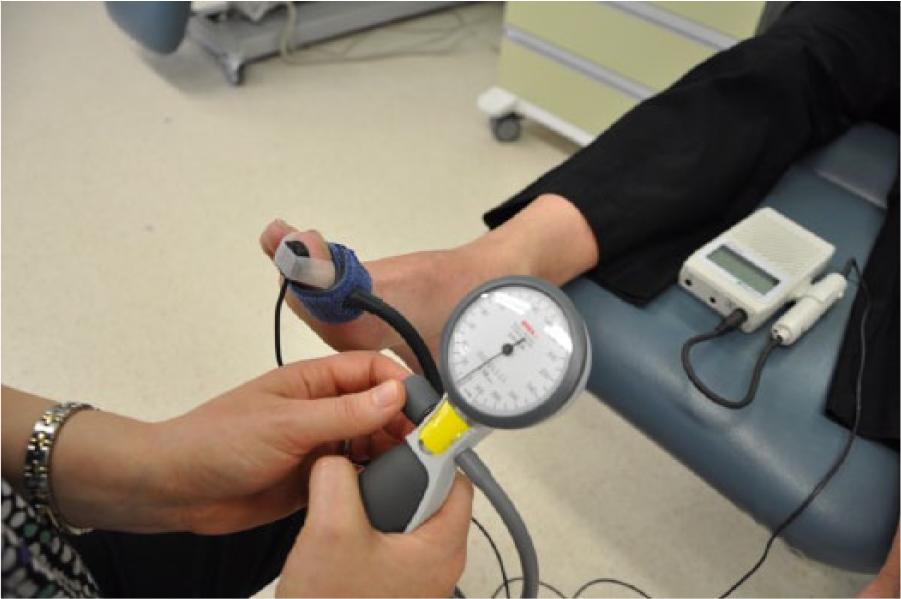
A systolic toe pressure being measured using the manual hand-held photoplethysmography (PPG) method. Continuous wave Doppler can also be used on the digital arteries in lieu of a PPG probe.
Accurate measurement of systolic toe pressure is dependent on a number of factors, including the control of ambient temperature. Similar to the ABI, strict control of patient factors needs to be undertaken to ensure test accuracy. Patients need to avoid smoking immediately prior to testing and lie completely flat with the legs and feet at the same level as the heart. In addition, the TBI is affected by ambient temperature and room temperature needs to be maintained between 23 and 25°C. 17 Unlike the ABI, the TBI is also affected by Raynaud’s disease or scleroderma and the measurements lack utility in these populations. 12 When pre-measurement protocols are adhered to, the TBI can be performed reliably in clinical environments with both automated and manual devices. 18 The measurement has also been shown to be an accurate indicator of PAD in populations prone to MAC including those with diabetes-related PAD, sensorimotor neuropathy, and patients undergoing haemodialysis for end stage renal failure.4,19,20 However, there is currently no consensus on the diagnostic accuracy of this test for identifying PAD across populations at risk of the disease.
The aim of this paper is to systematically review the evidence evaluating diagnostic accuracy of the TBI in detecting PAD in at-risk populations.
Materials and methods
Search strategy
A database search was conducted by the primary researcher (PT) up to July 2015 using Ovid Medline (1946–2015), CINAHL Plus (1982–2015), AMED (Ovid), Web of Science, Scopus and Embase.
Search terms were derived (Table 1) and truncated versions using wild card symbols were included to help broaden the search. No language restrictions were used. Reference lists of suitable articles were also hand searched for suitable work.
Search terms.

Inclusion and exclusion criteria
Original articles that diagnosed PAD using valid diagnostic imaging as a reference standard were included. Studies that used symptoms as a primary indicator of the severity of PAD, or, where PAD was diagnosed by ABI, TBI or Doppler waveform analysis alone were excluded. Studies that included participants with vasospastic disorders were not included as this is known to affect the accuracy of toe pressure measurements. 12
Study selection and data extraction
Literature searching was undertaken by a single reviewer (PT) who independently searched each database using the search terms and retrieved abstracts. Abstracts were then reviewed independently by two reviewers (PT and VC) and relevant articles were assessed according to the selection criteria. If any difference of opinion arose, the study in question was referred to a third party. Articles considered relevant were then obtained in full text. Reference lists of retrieved articles were searched for further potentially relevant studies. Data on sensitivity and specificity of the TBI in detecting PAD along with reference standards, room temperature, pre-rest time and demographic data were extracted by two researchers (PT and VC), independently, with disagreements resolved by a third researcher (DS). In cases where journal articles contained insufficient information, attempts were made to contact authors to obtain missing details.
Methodological quality was assessed using the Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) tool for systematic reviews. 21
Results
A total of 939 articles were retrieved for abstract review (Figure 2). Of these, 922 were excluded for lack of relevance. Seventeen articles in total were deemed relevant and full text versions were acquired. One study was excluded 22 as it was determining inter and intra tester reliability alone, and not diagnostic accuracy. One study 12 was excluded as it compared ankle–toe pressures rather than the TBI. Five studies23–27 were excluded as they did not diagnose PAD using diagnostic imaging for the reference standard. One study was excluded 28 as it reported correlation only and data examining sensitivity and specificity in this group were reported in another included study. 4 One study was excluded as it examined patients with vasospastic disorders 24 and one other study was excluded as it diagnosed calcification and not PAD. 29 Seven studies met all inclusion criteria for this review.4,20,30–34
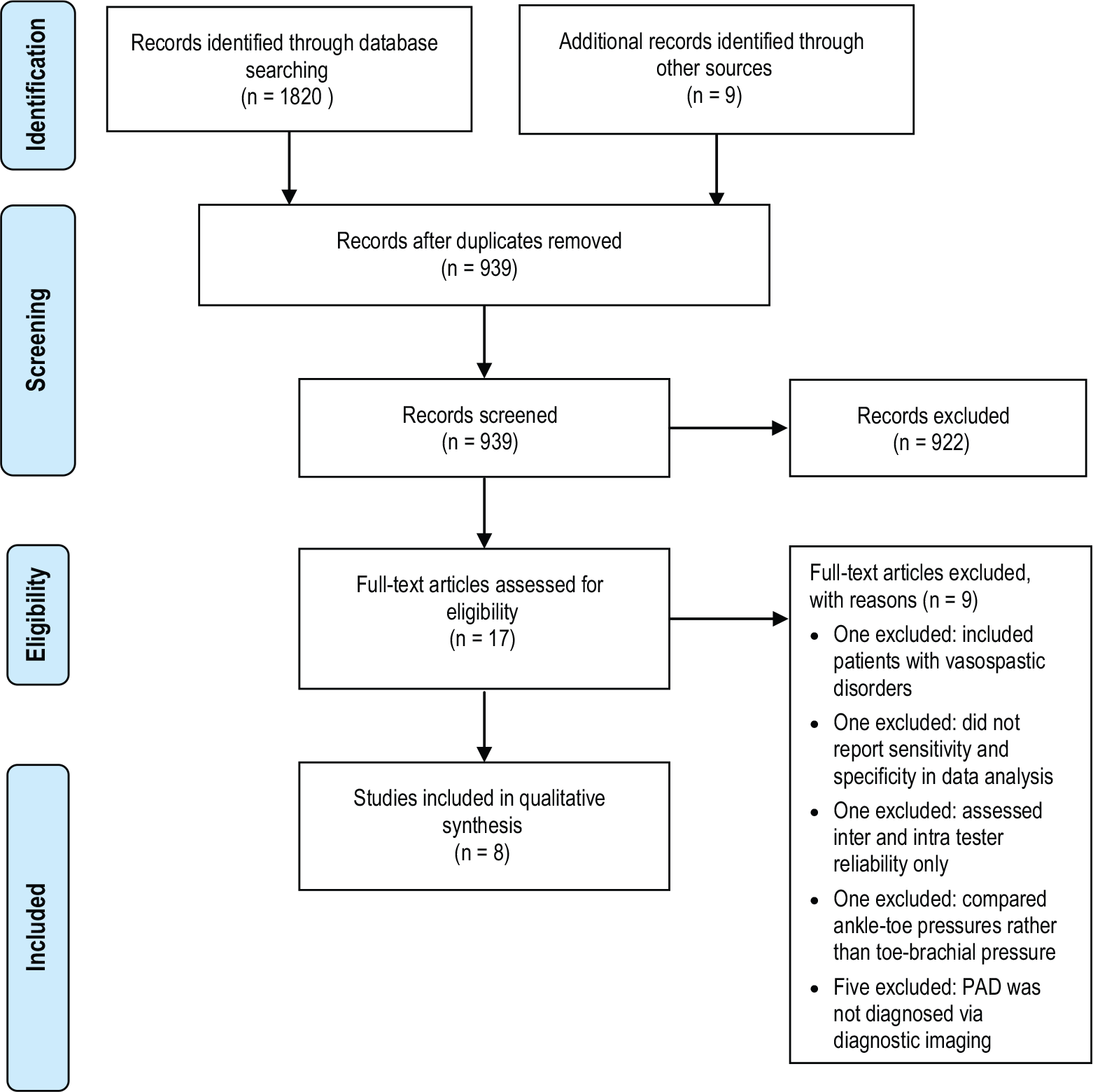
Search strategy. (PAD, peripheral artery disease.)
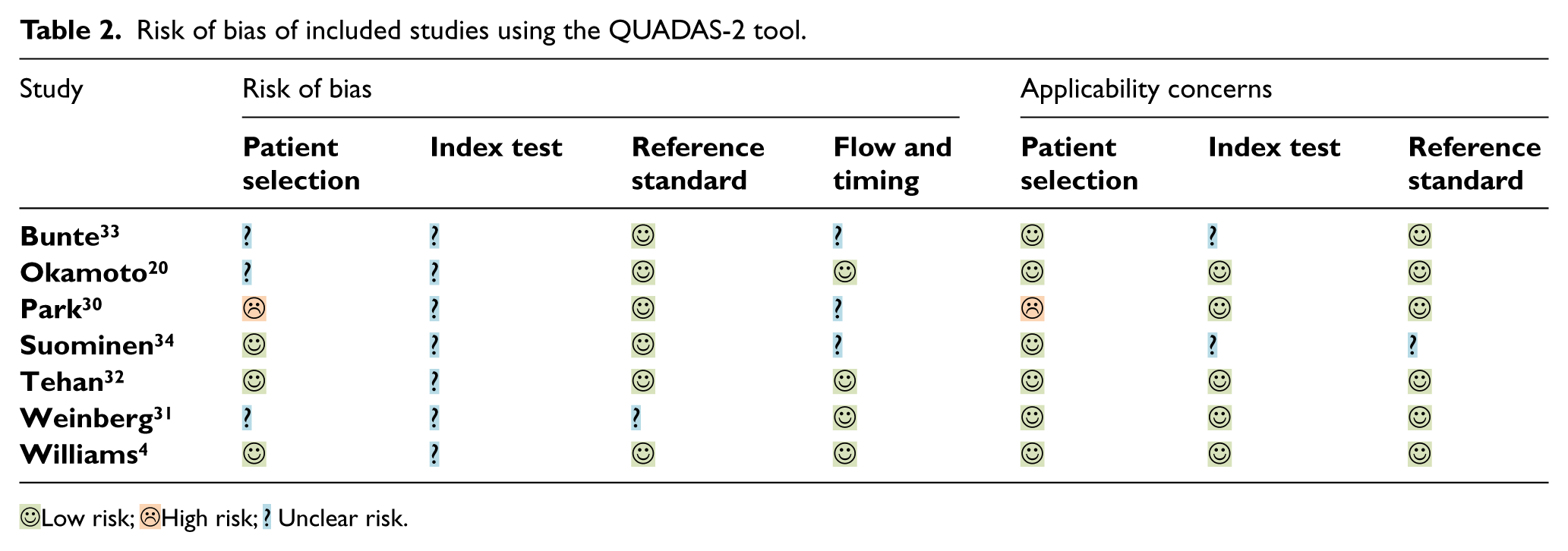
All seven included studies were appraised for risk of bias using the QUADAS-2 tool (Table 2, Figure 3).
Risk of bias of included studies using the QUADAS-2 tool.
 Low risk;
Low risk;  High risk;
High risk;  Unclear risk.
Unclear risk.
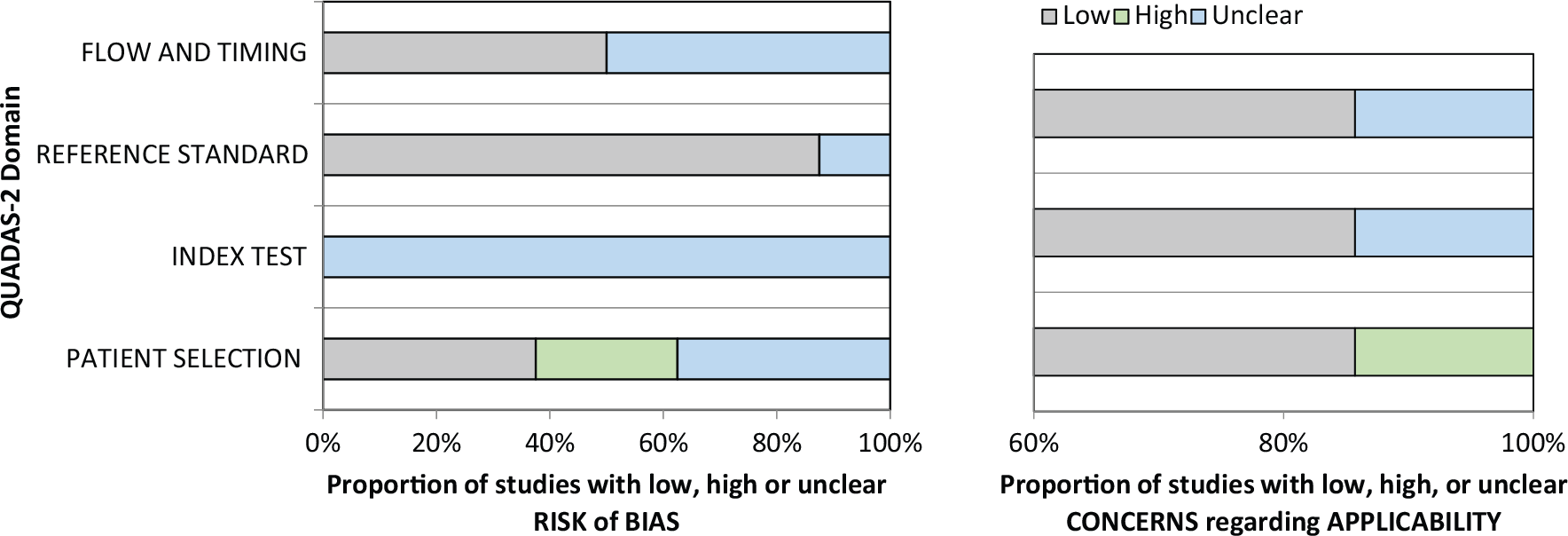
QUADAS-2 tool for assessing risk of bias of included studies.
Characteristics and overview of included studies
General
The studies included in this review examined sensitivity and specificity of TBI for detecting PAD in different cross sections of participants (Table 3). A total of 566 lower limbs were included in the seven studies. Of the 566 limbs, diagnostic imaging demonstrated 340 with PAD and 210 without PAD (16 limbs missing data 34 ). Reported participant age varied significantly (Table 3), with most of the studies examining an older age group – with the exception of one study 20 that had a range of 35–89 years and one study that did not report age at all. 30 Both men and women were included in most studies, with all reporting a higher number of male participants.4,30–34 One study did not specify the gender of participants. 20 Sample sizes varied amongst the seven studies ranging between 30 and 130 (Table 3). Most studies used paired data;4,20,30,31 one was unclear, 33 and two studies used one limb per person.32,34
Summary of studies of sensitivity and specificity of the toe–brachial index in detecting peripheral artery disease.
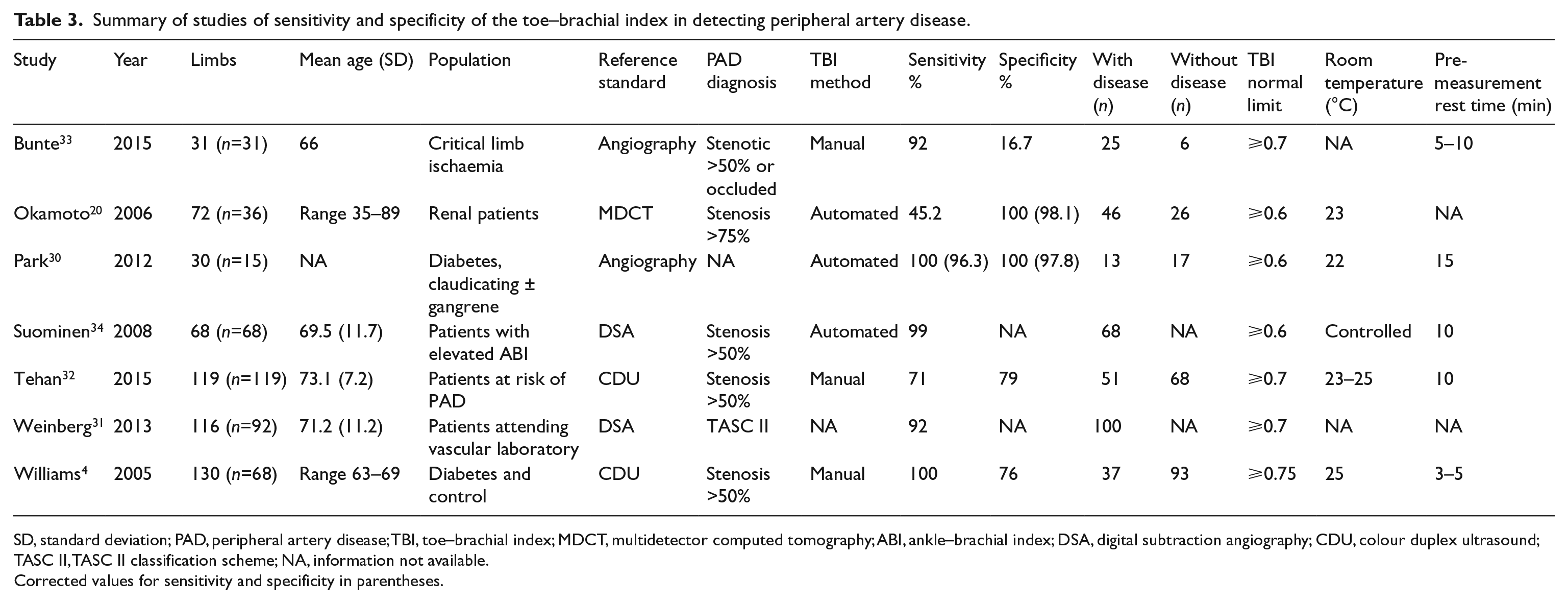
SD, standard deviation; PAD, peripheral artery disease; TBI, toe–brachial index; MDCT, multidetector computed tomography; ABI, ankle–brachial index; DSA, digital subtraction angiography; CDU, colour duplex ultrasound; TASC II, TASC II classification scheme; NA, information not available.
Corrected values for sensitivity and specificity in parentheses.
TBI method
Details of methodological procedures of included studies are provided in Table 3. All of the included studies used the photoplethysmography method to measure the toe pressure included in the TBI and included a mix of manual and automated measurements. The pre-measurement rest time varied between 3 and 15 minutes. Most studies used only one toe pressure measurement in the calculation of the TBI,20,29,30,32,34 whereas one study 4 took a mean of two measurements, taken at 3 and 5 minute intervals. Two studies did not describe the TBI method in sufficient detail to determine how many measurements were taken.31,33 Cut-offs for abnormal TBI values indicating PAD diagnosis also differed between the studies (<0.6, <0.7 and <0.75). Room temperature was controlled in most studies and only varied by a few degrees; two studies did not detail room temperature31,33 and one study stated it was controlled but did not specify the temperature. 34
Quality assessment
A QUADAS-2 checklist was used to assess methodological quality and risk of bias of the included studies (Table 2, Figure 3). In all of the included studies it was unclear if the results of the index test were interpreted without knowledge of the reference standard. It was also unclear in all of the studies if the reference standard results were interpreted without knowledge of the index test. Details of the methodological quality assessed by the QUADAS-2 tool are provided in Table 2 and Figure 3.
A range of different diagnostic imaging methods were utilised by the included studies to diagnose PAD, all of which have varying levels of diagnostic accuracy. Four of the included studies used the gold standard angiography as a reference standard, two used colour duplex ultrasound and one used multidetector-row computed tomography. The diagnosis of PAD using these reference standards also differed significantly between studies. Several different anatomic criteria for the diagnosis of PAD were used including >50% stenosis, >75% stenosis and one paper utilised the TASC classification system for interpretation of lower limb angiography. The index test, the TBI, was also interpreted differently between studies with definition of a normal value ranging from >0.6 to >0.75.
The sample populations studied all included groups representing people either at risk of, or with current PAD (Table 3). However, the diagnosis of haemodynamically significant PAD, disease severity and presence of underlying comorbidities varied significantly between studies. Only one of the included studies recruited a non-diseased control group with a further four studies including non-diseased single limbs and/or participants from different at-risk or symptomatic cohorts. Two studies stated they were performed retrospectively and included diseased limbs only. Underlying co-morbidities included diabetes, renal disease and mixed populations at risk of PAD that were symptomatic or non-symptomatic.
Sensitivity of TBI and specificity of TBI
Sensitivity was reported in all seven studies and ranged from 45% to 100% (Table 3), with the highest reported sensitivity by Park et al. 30 who demonstrated 100% sensitivity of the TBI for detecting PAD in a population of 30 claudicating limbs with and without gangrene. The lowest sensitivity was reported by Okamoto et al. 20 who demonstrated the TBI had 45% sensitivity for detecting PAD in a sample of 72 participants undergoing haemodialysis. Specificity of the TBI for diagnosing PAD was reported by five studies and ranged from 16% to 100% (Table 3). The highest reported specificity (100%) was also by Park et al. 30 and the lowest specificity (16%) was demonstrated by Bunte et al. 33
Discussion
This review assessed the sensitivity and specificity of the TBI in detecting PAD. Seven studies were included and these all examined the sensitivity of the TBI while five of the studies examined the specificity of the TBI for detecting PAD in a range of different populations. The TBI had varying degrees of sensitivity ranging from 45% to 100% and specificity from 16% to 100%, depending on the population studied. The heterogeneity of the included populations was notable. Overall, the TBI had good test performance in patients with diabetes, claudicants and those at risk of PAD, and therefore may be a useful adjunct for vascular screening in these cohorts. 9 Lower sensitivity was reported in a population with renal disease, and poor specificity in a cohort with critical limb ischaemia. Overall, the variable results of measures of diagnostic accuracy of the TBI for PAD in the existing literature make it difficult to determine the clinical utility of this test. The variable diagnostic accuracy reported in the included studies are likely to have been influenced by both the heterogeneity of included participants and the methodological differences between studies.
Methodological quality was varied across the seven studies, with a significant amount of heterogeneity across multiple domains. The QUADAS-2 assessment demonstrated that a large amount of information was unclear across the studies, particularly in relation to risk of bias with patient selection and the index test. Only one of the seven included studies recruited non-diseased participants, with two studies only including a diseased population. The lack of equitable non-diseased groups in the majority of studies creates significant spectrum bias. 35 In addition, it was unclear if there was appropriate operator blinding between the index and reference testing in all of the included studies, which was also likely to lead to an increased risk of bias.
The interpretation of the TBI value for normal was also a likely factor in the varying levels of reported accuracy. Studies that used the lower value for normal of >0.6 were likely to have overestimated the presence of disease compared to those using a much higher cut-off of >0.75. Unlike the ABI, the TBI does not have a well-established grading system or an agreed normal value which is correlated with gold standard diagnostic imaging. Currently there are discrepancies in the literature and any of the values used in the included studies (i.e. 0.6, 0.7 or 0.75) can be considered as a cut-off to differentiate normal and abnormal findings.4,12,14,24 Recent research has shown that in normal populations mean TBI values are 0.94–0.98, suggesting that the current cut-offs are too low and that underdiagnosis of PAD is likely. 16 The differing cut-offs used in the included studies are certain to have influenced the sensitivity and specificity.
The range of reference standards used by the included studies and differing anatomic criteria for diagnosis of PAD may also account for the varied levels of reported diagnostic accuracy. One study used multidetector-row computed tomography, 20 four used angiography30,31,33,34 and two used colour duplex ultrasound.4,32 Although angiography remains the gold standard in imaging for PAD, the studies using this method used differing criteria to diagnose PAD, making comparison between studies difficult. Whilst duplex ultrasound is the gold standard non-invasive imaging method for diagnosing PAD, and is used extensively clinically, it is operator-dependent. Both studies using duplex ultrasound reported high test-retest reliability; however, testing was conducted in a small sample and this form of imaging is known to have reduced diagnostic accuracy, particularly in infragenicular vessels 36 and those affected by extensive MAC. 37
Methodological differences in performing the TBI measurement between studies may also have had an effect on the reported sensitivity and specificity outcomes of the studies included in this review. The TBI is highly influenced by environmental factors and has limited utility in some populations such as those with vasospastic disorders. 12 External variables known to influence the toe pressure measurement, such as ambient temperature, varied in the included studies. Limb temperature, which is also known to influence the toe pressure measurement, was also not demonstrated by any of the included studies. 38 The included studies also reported differences in rest times prior to taking toe pressures, and use of serial (an average of two or more) and single measurements. There is evidence to suggest that toe pressures do not stabilise for the first 10 minutes, 39 possibly affecting the accuracy of studies using shorter pre-measurement rest time frames. Furthermore, use of one versus an average of two TBI measurements may also have affected measures of diagnostic accuracy. Although one measurement has been shown to have adequate diagnostic accuracy of the TBI for PAD, 32 there has been no comparative investigation of the effect of single or serial TBI measurements.
Our systematic review has demonstrated a paucity of data relating to the diagnostic accuracy of the TBI for PAD. Current international PAD screening guidelines recommend the TBI be used in the presence of an elevated ABI value. 10 It is possible the TBI can also provide additional information on small vessel PAD and disease below the ankle which is not detected by large vessel screening methods such as the ABI. Furthermore, co-existence of PAD and MAC have been demonstrated to reduce the ABI to a normal value, failing to detect either disease process 7 and may render the ABI less accurate in specific populations, including those with renal disease and diabetes. However, based on current literature, the value of the TBI for diagnosing PAD across populations at risk of the disease is inconclusive.
Limitations
We performed an exhaustive search for relevant literature; however, the volume of articles retrieved from database searches may have led to accidental omissions of relevant research. Six databases were utilised in the search; however, researchers in the field were not contacted for any unpublished work. Authors were only contacted where information from included articles was missing and in only one case responded. Furthermore, strict exclusion criteria meant that multiple studies were not included as they did not use valid diagnostic imaging as a reference standard or did not calculate sensitivity and specificity. Overall there was a lack of high-level evidence for determining the diagnostic accuracy of the TBI for PAD. All of the included studies had small sample sizes with large variations in methodology and very specific populations. More extensive investigation is required using larger sample sizes and including more general populations at risk of PAD in order to determine the true value of the TBI as a potential diagnostic tool.
Conclusions
This review highlights the lack of high-level evidence available for investigating the diagnostic accuracy of the TBI for PAD. Based on current literature it is not possible to determine the extent of the effectiveness of this test for diagnosing PAD in a clinical setting. We have also demonstrated there is a need for standardised normal values to be established for the TBI before diagnostic accuracy for PAD can be conclusively determined.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the national Research Training Scheme (Australia) and a University of Newcastle Faculty of Health and Medicine PhD exchange grant.
