Abstract
Postprandial lipemia has been associated with acute endothelial dysfunction. Endothelial dysfunction, in turn, is associated with increased arterial stiffness. However, the relationship between postprandial lipemia and acute changes in arterial stiffness has not been extensively investigated. Therefore, we conducted a pilot study on the effects of postprandial lipemia on arterial stiffness in 19 healthy young adults before and after consumption of a high-fat mixed meal. Arterial stiffness was assessed locally with echo-tracking carotid arterial strain (CAS) and globally with carotid–femoral pulse wave velocity (PWV). As assessed by these two benchmark parameters, arterial stiffness did not differ significantly postprandially. However, the arterial distension period (ADP) was significantly lower 2 hours after mixed meal ingestion. In addition, slopes of carotid artery area (CAA) curves were significantly steeper postprandially. Therefore, we concluded that ADP may be a more sensitive marker of arterial stiffness in healthy young adults when compared to PWV and CAS.
Background
Arterial stiffness is a well-established measure of vascular health with strong links to traditional cardiovascular risk factors such as obesity, smoking, hypertension, and diabetes.1–3 Endothelial function is also associated with traditional cardiovascular risk factors, and its diurnal changes have been studied extensively.4–6 However, there is currently no firm consensus regarding the nature of how acute changes in arterial stiffness occur or their relation to cardiovascular events.7,8
Postprandial lipemia is known to produce acute changes in endothelial function through a triglyceride-rich lipoprotein-mediated increase in oxidative stress. 9 Historically, endothelial function was thought to change acutely, while changes in augmentation index (AI), a known surrogate of arterial stiffness, were considered to be a chronic process. However, several studies have documented postprandial changes in AI, and it is now thought that changes in AI result not only from structural changes in the vessel wall but also from increased endothelial dysfunction and thus increased resistance in the peripheral arteriolar bed.10–13 Therefore, even though the structural changes that result in increased AI may take decades to occur, there are acute processes that may be clinically detectable after interventions that merely alter endothelial function. Moreover, AI is an important hemodynamic parameter. Despite being only an indirect measurement of arterial stiffness, it is a reflection of the net effect that changes in arterial stiffness, endothelial function and even left ventricular contractility have on the arterial vasculature. 14
Despite strong correlations between postprandial lipemia and endothelial function, as well as endothelial function and arterial stiffness, it is not known whether postprandial lipemia produces any acute detectable changes in arterial stiffness directly.9,15,16 Therefore, we sought to determine if acute changes in arterial stiffness occur in a postprandial state by assessing the effects of a mixed meal high in saturated fat (from here on referred to as a ‘mixed meal’) on healthy young adults using speckle-tracking ultrasound and carotid–femoral pulse wave velocity (PWV). We hypothesized that in healthy young adults arterial stiffness as assessed by these two parameters would increase significantly 2 hours after ingestion of a mixed meal.
Methods
Study population
In this pilot study, 19 healthy young adults aged over 18 years without any known cardiovascular risk factors were voluntarily recruited through the Baylor College of Medicine (Houston, TX, USA). Exclusion criteria included history of hypertension, diabetes, dyslipidemia or tobacco use. All studies were conducted in the morning at the noninvasive cardiac laboratory of the Ben Taub General Hospital (Houston, TX, USA) after a minimum 8-hour period free from food, alcohol, caffeine and other vasoactive substances. Carotid ultrasound, brachial blood pressure and arterial tonometry were assessed in each subject prior to and approximately 2 hours after ingestion of a breakfast sandwich containing sausage, egg and cheese on an English muffin (450 kilocalories, 27 grams of total fat (56%) (10 grams of saturated fat), 30 grams of carbohydrate (18%), 21 grams of protein (27%)) and 16 ounces of water. The study was approved by the Institutional Review Board of Baylor College of Medicine. Informed consent was obtained from all subjects.
Carotid arterial strain measurements
The methodology used in carotid arterial strain (CAS) imaging and analysis have been described previously. 17 Briefly, bilateral electrocardiography (ECG)-gated B-mode ultrasound scans of the carotid arteries (carotid artery presets, Vivid 7; GE Healthcare, Waukegan, IL, USA) were acquired for three cardiac cycles at 14 MHz with a vascular transducer. Image acquisition averaged 78 frames/s at a dynamic range of 72 dB. A single individual with experience in vascular ultrasound performed all CAS measurements.
With the probe located anterior to the carotid artery, the common carotid artery (CCA) was studied with a transverse (cross-sectional) image sweep from the proximal CCA to the carotid bifurcation to evaluate for the presence of carotid plaque. 18 We analyzed the distal CCA (approximately 1 cm inferior to the carotid bulb). Repeat image acquisition was attempted if motion artifacts were present. Images were anonymized with unique codes and exported in Digital Imaging and Communications in Medicine (DICOM) format for analysis such that whether the images were pre or post mixed meal ingestion could not be discerned.
Carotid artery time intervals
Three time intervals were measured from carotid ultrasound strain waves. The first was the peak carotid arterial strain time (PCAST), which was defined as the time from onset of electrical systole (beginning of the QRS complex on ECG) to the peak CAS (Figure 1). Pre-distension period (PDP), the second time interval measured, was defined as the time from electrical systole onset to the start of arterial expansion (upward deflection of the strain curve) (Figure 1). The third time interval, arterial distension period (ADP), was defined as the time difference between PCAST and PDP (Figure 1) and is the time interval in which the artery is actively expanding. All time intervals were corrected for heart rate prior to analysis.
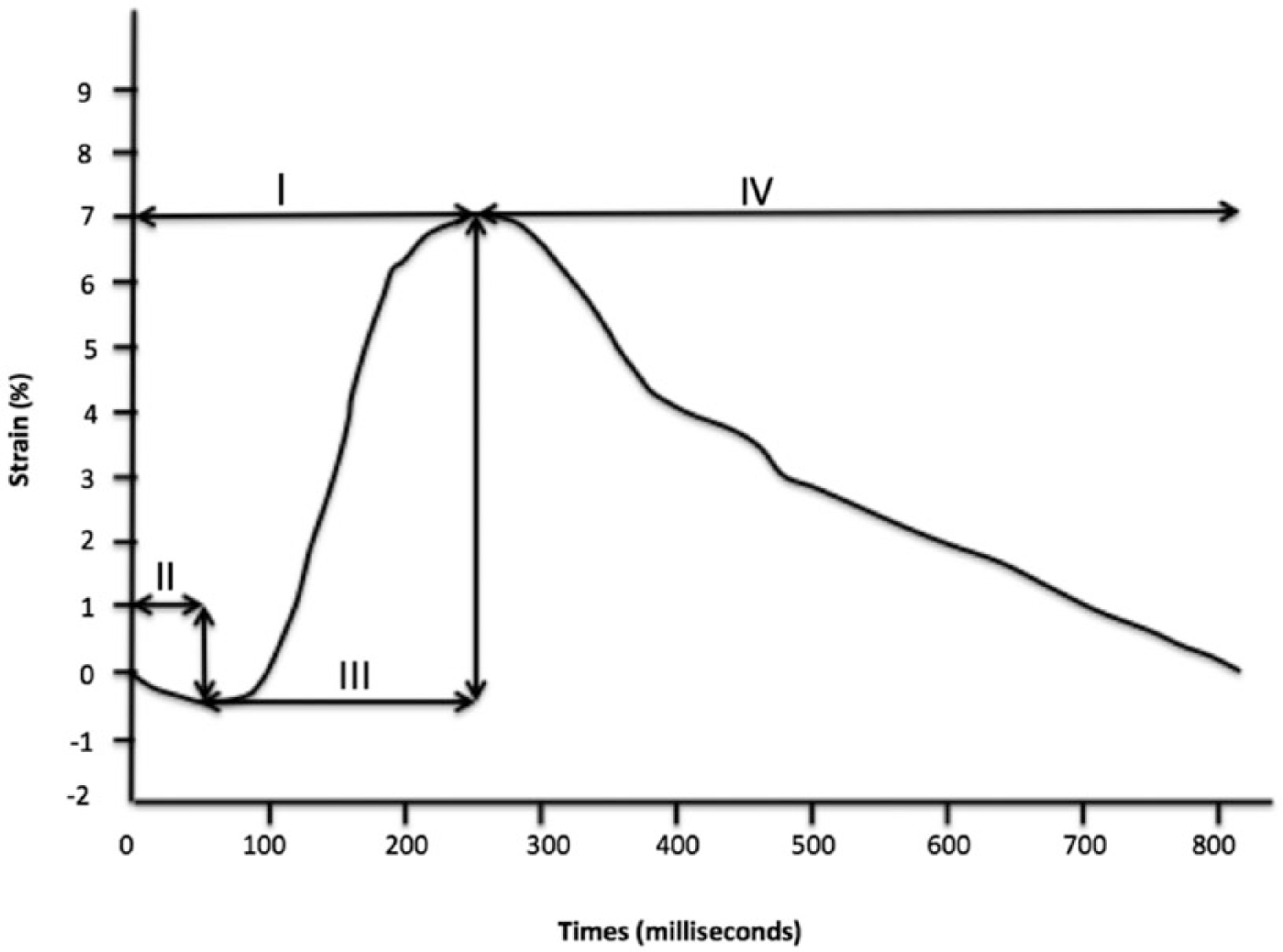
Carotid arterial strain curve with systolic time intervals defined relative to maximum and minimum carotid strain values. (I) Peak carotid arterial strain time; (II) arterial pre-distension period; (III) arterial distension period (ADP) relative to QRS onset on ECG (the far left side of the horizontal axis); and (IV) arterial diastolic time.
Pulse wave measurement and analysis
Bilateral systolic and diastolic brachial blood pressures (BP) were obtained prior to each set of pre- and postprandial PWV and carotid ultrasound studies and after at least 10 minutes of rest in the supine position (Welch Allyn 5200 Non-Invasive Blood Pressure (NIBP) cuff; Skaneateles Fall, NY, USA). Applanation tonometry was performed (SyphgmoCor device, Atcor Medical Inc., Itasca, IL, USA) according to the methodology described and validated previously. 19 Waveform quality was determined using a quality index percentage score (QI%), which is derived from an average pulse height, pulse height variation and diastolic variation. 20 Only waveforms with a QI greater than 80% were deemed satisfactory for analysis. Using a generalized transfer function, software converted radial waveforms into a central aortic waveform. This central waveform was in turn used to derive central aortic BP and central AI. Sequential applanation tonometry was then performed at common carotid and femoral artery pulse sites with ECG-gating. The sum of the distance from sternal notch-to-umbilicus plus umbilicus-to-femoral pulse, minus carotid pulse-to-sternal notch measured manually over the body surface was used to estimate the aortic carotid-to-femoral distance. 2 The time difference between R wave and the foot of the carotid waveform and R wave and the foot of the femoral waveform was calculated. The aortic carotid–femoral distance was divided by this time difference to obtain the carotid–femoral PWV.
Imaging analysis
Peak CAS and carotid artery time intervals were measured using an automated 2D strain computer software package (2D Cardiac Performance Analysis; TomTec Imaging, Munich, Germany) and computer-generated peak CAS averaged over the six measured carotid artery segments were recorded. Only global and mean far wall CAS were studied as these two measurements are the most reproducible of all CAS measurements with speckle-tracking ultrasound. 17 Carotid artery area (CAA) curves were generated using a previously described method, and their rates of change (slopes) were analyzed (Figure 2). Both carotid artery time intervals and CAA slopes were normalized to cardiac cycle length and are thus displayed as percentages. The slopes of CAA curves reflect the combined effects of pulse pressure (PP) and arterial elasticity on the rate of arterial distension. 21
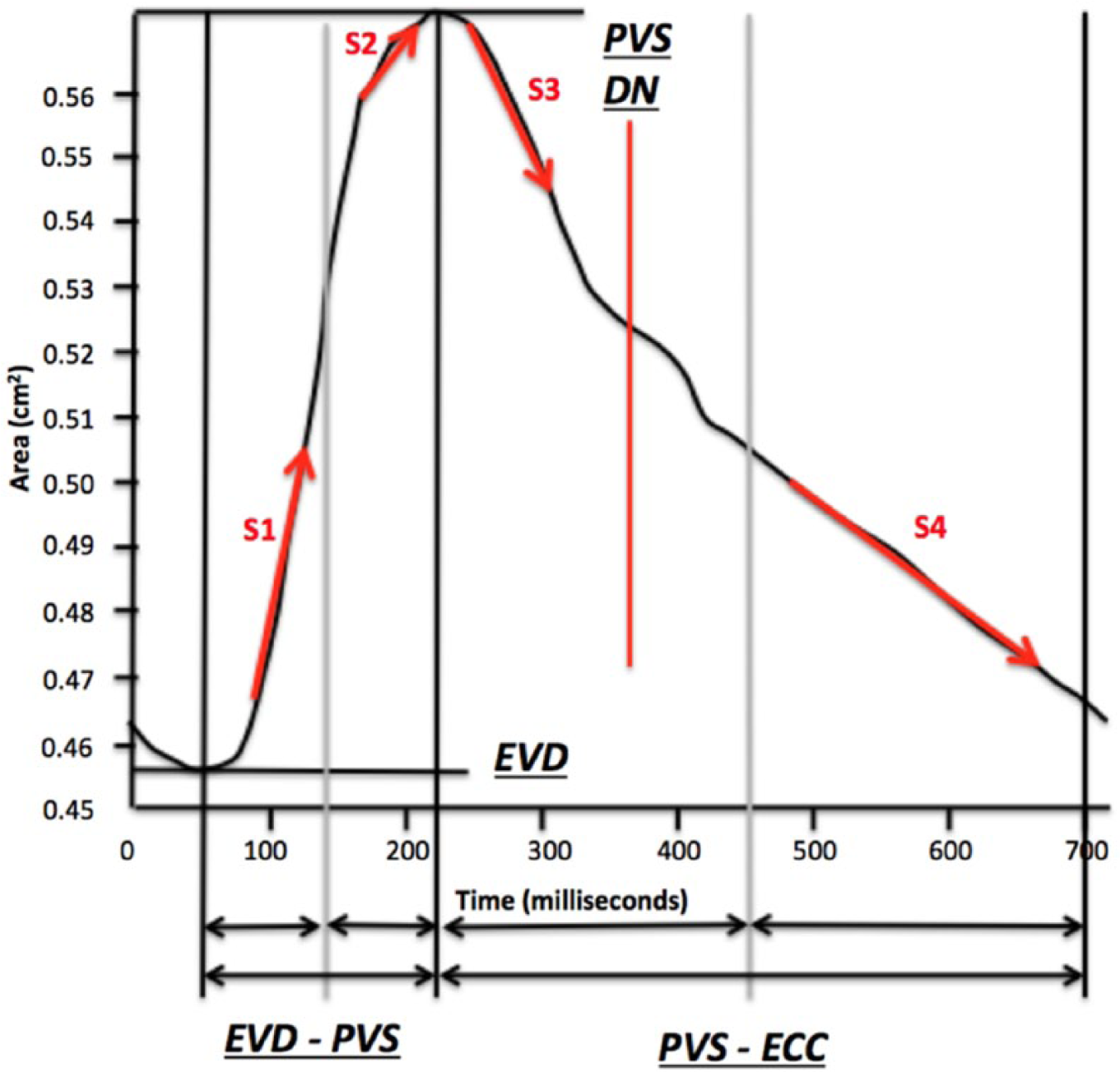
Carotid artery area curve from a healthy young adult subject with pertinent markers. All slopes (S1 to S4) were calculated with corrections for cardiac cycle length. (DN, dicrotic notch; ECC, end of the cardiac cycle; EVD, end vascular diastole; PVS, peak vascular systole.)
Statistical methods
Statistical analyses were performed using Stata version 11 (Stata Corp LP, College Station, TX, USA) or SPSS Statistics version 22 (IBM Corp., Armonk, NY, USA). Appropriate two-tailed parametric and non-parametric tests (e.g. paired t-test, Wilcoxon signed-rank test) were used to compare arterial stiffness parameters and carotid artery time intervals before and after mixed meal ingestion. Changes in CAA curves before and after intervention were compared with a repeated measure analysis of variance (RMANOVA) test. Change in PP and change in PWV were added as covariates to look for any potential interactions with CAA.
Results
Nineteen healthy young adults (nine women; mean age 25.6 years (standard deviation (SD) 6.06 years); mean body mass index 21.4 kg/m2 (SD 2.5 kg/m2)) were enrolled in and completed the study. One woman was taking glycopyrrolate; another woman was on warfarin for a hypercoagulability disorder. Six women were taking oral contraceptives and one man was taking cetirizine for seasonal allergies. The ethnic breakdown of the study subjects was 12 Caucasians, six Asians and one Hispanic. The average time between last meal and initial study was 13.2 hours (SD 0.56 hours). The average time between mixed meal intake and reassessment was 131 minutes (SD 6.4 minutes).
Arterial stiffness, as assessed locally by CAS and regionally by carotid–femoral PWV, was not significantly different 2 hours after intake of a mixed meal (Table 1). In contrast, carotid ADP decreased significantly from a preprandial average value of 27.65% (SD 4.47%) to a postprandial average value of 25.46% (SD 3.65%) (p=0.049). Similar but non-significant trends were seen in two other timing measures as well (Table 2).
Arterial stiffness parameters pre- and 2 hours post-ingestion of a mixed meal.
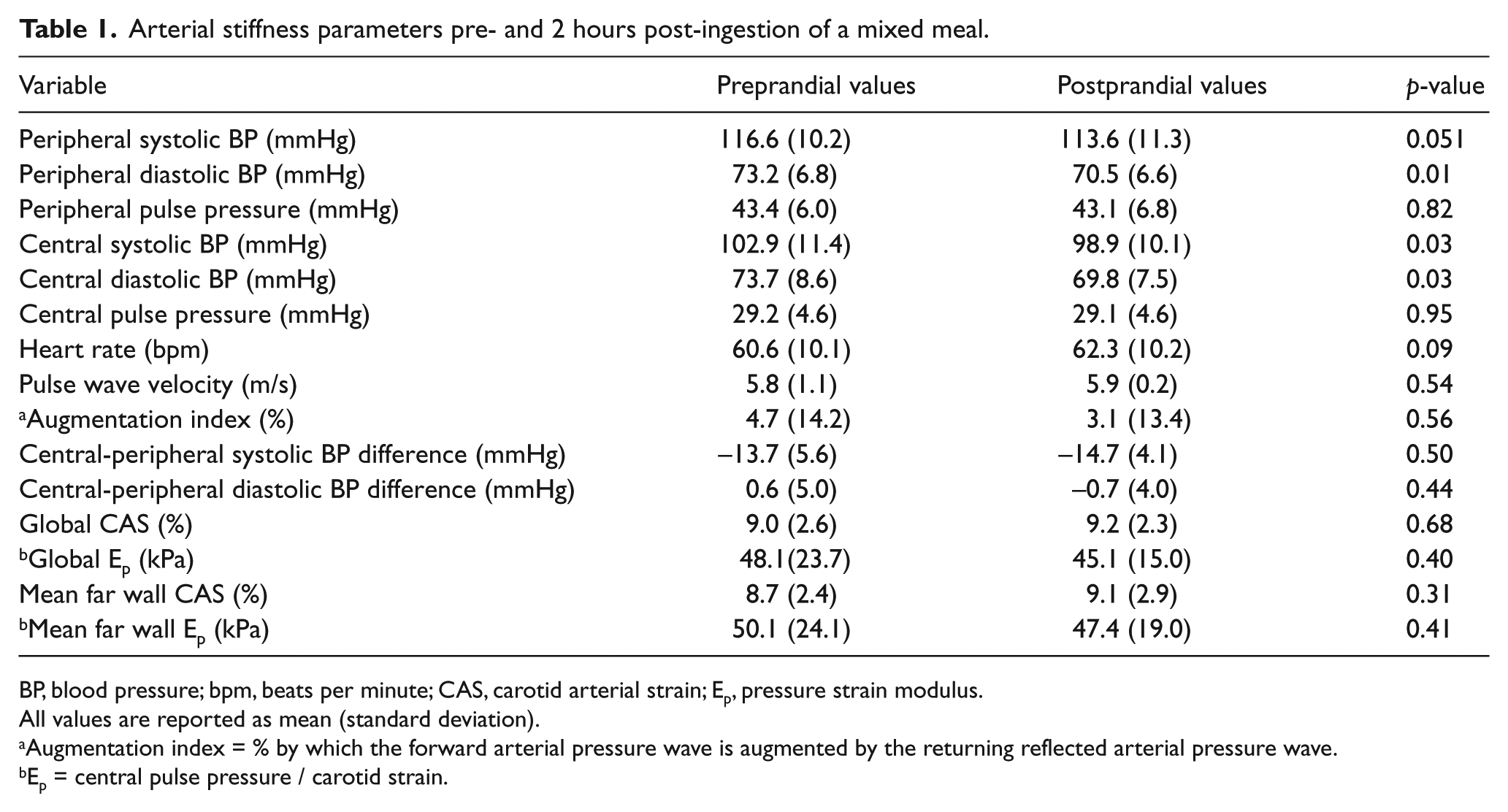
BP, blood pressure; bpm, beats per minute; CAS, carotid arterial strain; Ep, pressure strain modulus.
All values are reported as mean (standard deviation).
Augmentation index = % by which the forward arterial pressure wave is augmented by the returning reflected arterial pressure wave.
Ep = central pulse pressure / carotid strain.
Carotid artery time intervals pre- and 2 hours post-ingestion of a mixed meal.
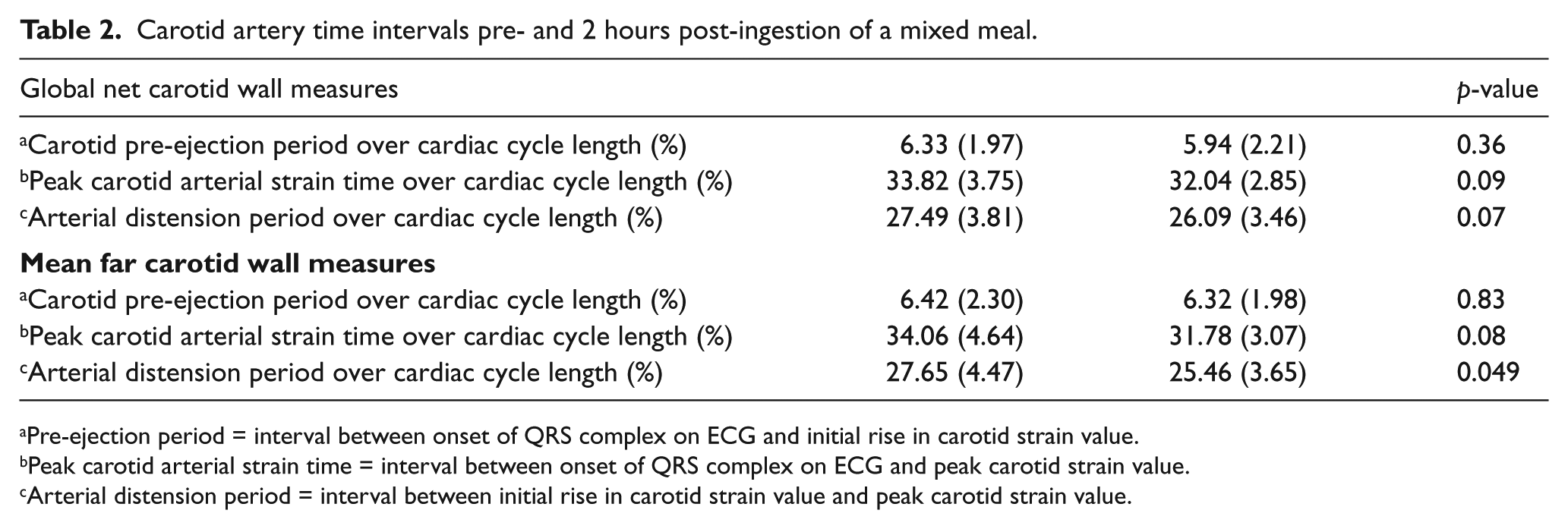
Pre-ejection period = interval between onset of QRS complex on ECG and initial rise in carotid strain value.
Peak carotid arterial strain time = interval between onset of QRS complex on ECG and peak carotid strain value.
Arterial distension period = interval between initial rise in carotid strain value and peak carotid strain value.
Significantly steeper CAA curve slopes were seen in segments S2 (p=0.01), S3 (p=0.03) and S4 (p=0.04) postprandially. Slopes of the S1 segment were not significantly affected by prandial state. The average mean slopes of segments S1 and S2 (p=0.01) and the average mean slopes of S3 and S4 (p=0.007) were both significantly steeper postprandially. The minimum-to-maximum change in CAA was significantly increased postprandially (p=0.03). The addition of change in PP and change in PWV as covariates in our model did not significantly alter these results (Table 3).
Salient markers from the ensemble-means carotid artery area curve in healthy young adults before and after oral fat load ingestion (n=19) controlled for change in pulse pressure and pulse wave velocity.
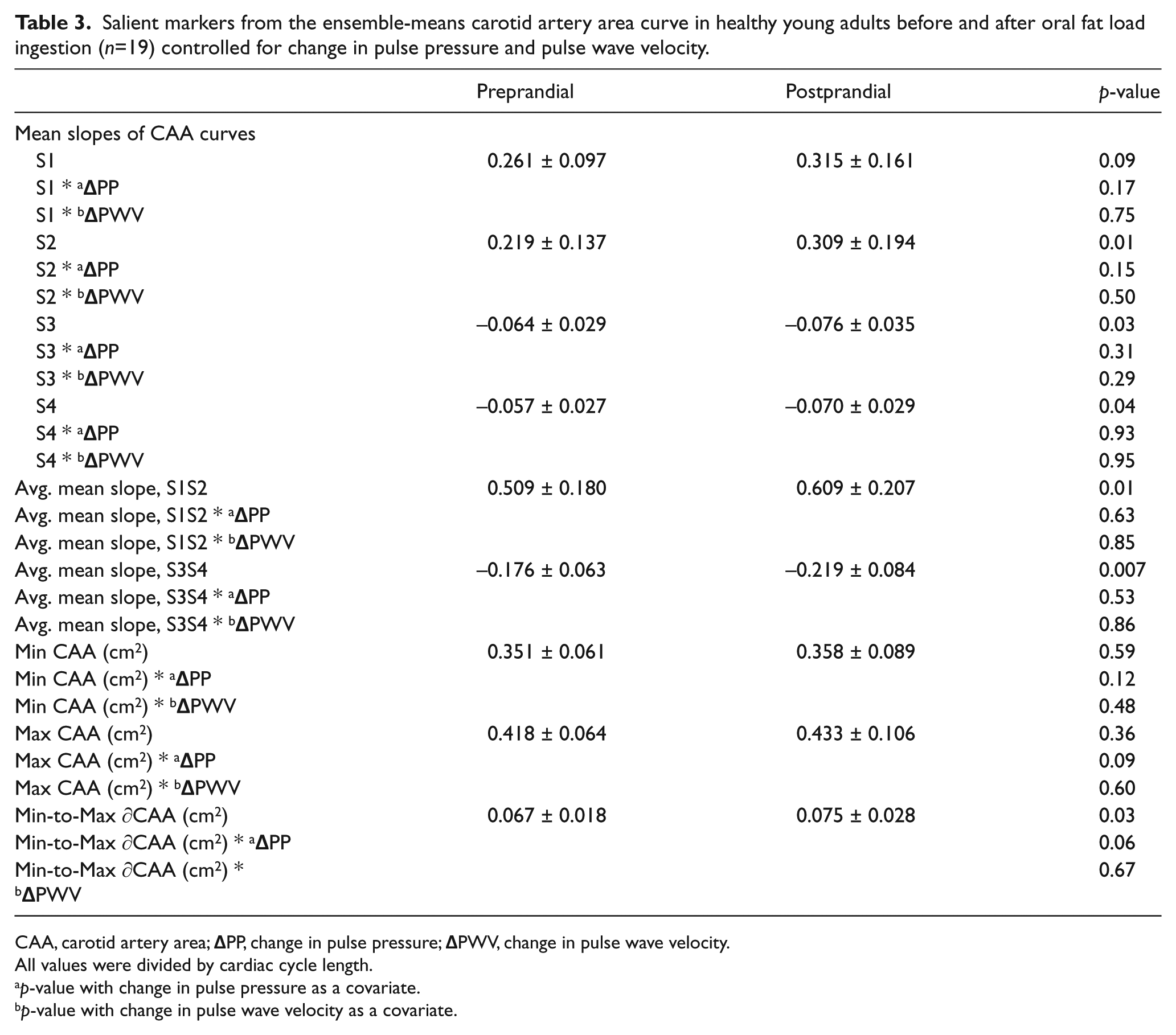
CAA, carotid artery area;
All values were divided by cardiac cycle length.
p-value with change in pulse pressure as a covariate.
p-value with change in pulse wave velocity as a covariate.
Postprandial central systolic and diastolic BP and peripheral diastolic BP were all significantly lower 2 hours after ingestion of a mixed meal. Central systolic BP decreased from 102.9 mmHg (SD 11.4 mmHg) preprandially to 98.9 mmHg (SD 10.1 mmHg) postprandially; p = 0.03. Central diastolic BP decreased from 73.7 mmHg (SD 8.6 mmHg) preprandially to 69.8 mmHg (SD 7.5 mmHg) postprandially; p = 0.03. Peripheral diastolic BP went from a preprandial value of 73.2 mmHg (SD 6.8 mmHg) to a postprandial value of 70.5 mmHg (SD 6.6 mmHg); p = 0.01. Peripheral systolic BP trended toward being significantly lower (p = 0.051). Central and peripheral pulse pressures and heart rate were not significantly different 2 hours after mixed meal ingestion (Table 1).
Discussion
In this pilot study, we aimed to study the effects of a meal high in saturated fat (known to affect endothelial function) on arterial stiffness in healthy young adults. As assessed regionally by PWV and locally by CAS, we were not able to detect any acute changes in arterial stiffness. As expected, central systolic and diastolic as well as peripheral diastolic BP were significantly lower postprandially. We did find that the ADP was significantly lower 2 hours after ingestion of a mixed meal, and several slope segments of the CAA curves were significantly steeper postprandially. Repeated measure ANOVA testing did not find PP or PWV to have any significant effect on CAA slopes, suggesting that changes seen postprandially are due to BP-independent changes in vascular function not detectable with applanation tonometry.
Conflicting results on the role of meals high in saturated fat on endothelial function (a determinant of arterial stiffness) have been reported previously.22,23 Despite strong associations between postprandial lipemia and oxidative stress, as well as oxidative stress and endothelial dysfunction, the direct association between high-fat meals, postprandial lipemia and endothelial dysfunction has been found to be rather weak. 9 A recent study of postprandial vascular changes in patients with metabolic syndrome found evidence of increased endothelial dysfunction after a meal high in saturated fat. The same study simultaneously documented a postprandial decrease in arterial stiffness in patients receiving an high-fat vs high-carbohydrate meal. 12 Such results suggest not only that endothelial dysfunction and arterial stiffness do not necessarily move in the same direction postprandially but also that they may be differentially affected by the type of meal ingested. 12
Previous studies of the effects of postprandial lipemia on BP and arterial stiffness have yielded confusing and inconsistent results. A study by Taylor et al. found decreased blood pressure and arterial stiffness after oral fat load ingestion in healthy young adults. 10 In contrast, a recent study by Lithander et al. that found increased central aortic and peripheral BP as well as increased arterial stiffness in healthy normotensive adults after both mono-unsaturated fatty acid (MUFA)-rich and saturated fatty acid (SFA)-rich meals. 11 Further adding a layer of complexity to our understanding of postprandial changes in arterial stiffness, a recent study by Dickinson et al. found arterial stiffness, as assessed by AI, was increased after a high-sodium meal (versus a low sodium meal) in healthy young adults despite no significant change in BP. 24 The Dickinson study is significant because it highlights that dietary factors other than triglycerides (e.g. sodium) may affect arterial stiffness postprandially.
In contrast to the abovementioned studies that used meals consisting almost entirely of saturated fat, we opted to use a high-fat mixed meal in order to reflect the day-to-day postprandial changes that occur with consumption of a diet not uncommon in the United States. While mixed meals may not produce an effect as profound on arterial stiffness, they provide data that are likely more reflective of changes occurring in the clinical setting. Several recent studies have also opted to use physiologic high-fat meals for similar reasons.23,25,26
In addition, repeat measurements of PWV and CAS could have been done too early to detect any postprandial changes that may have occurred. Previous studies looking at the effects of postprandial lipemia on arterial properties recorded measurements for up to 6 hours after fat load ingestion.22,23 Lastly, the acute postprandial perturbations of endothelial function seen in some studies may not be readily detectable with arterial stiffness, which may require chronic repeated exposure to hyperlipemic stimuli before detectable abnormalities appear.
The postprandial decreases in mean central and peripheral systolic and diastolic BP are consistent with an earlier study by Ahuja et al., which showed similar results in a group of reasonably healthy adults after ingestion of a light breakfast. 26 Although their study was not designed to determine how postprandial lipemia affects the cardiovascular system, Ahuja et al. attributed large declines in central aortic BP in their study to acute decreases in arterial stiffness resulting from rapid, reversible smooth muscle relaxation leading to splanchnic vasodilation in response to nutrient delivery. On a healthy vascular bed, the vasodilatory effects of postprandial lipemia may cancel out the effects that triglyceride-induced oxidative stress and subsequent vasoconstriction would otherwise have if they occurred in isolation. There is no known way to disaggregate and quantify these opposing vasodilatory and vasoconstricting mechanisms. Nonetheless, the balance between them is ultimately what determines arterial stiffness, tone and elasticity and may explain why this study failed to show any correlation between ingestion of a high-fat meal and increased arterial stiffness in healthy young adults. 27
Despite the lack of any association between physiologic fat intake and arterial stiffness as assessed by PWV and CAS, our findings of shorter ADPs after fat load ingestion suggest acute vascular changes occur in healthy young adults even while traditional benchmarks of arterial stiffness like applanation tonometry and carotid ultrasound remain unchanged. To date, no study has assessed postprandial changes in ADP. All previous studies of ADP have been cross-sectional in nature.21,28 In a previous study, ADP was found to be shortened in patients with hypertension and diabetes—conditions known to increase arterial stiffness and endothelial dysfunction—as compared to healthy controls. 21
This pilot study’s finding that ADP is shortened postprandially, even in healthy young adults, is important for three reasons. First, endothelial function and arterial stiffness both have strong correlations with cardiovascular risk.2,6,29 Second, ADP has been found to have associations with both arterial stiffness and subclinical atherosclerosis. 28 Third, arterial stiffness has diurnal variations that correspond to the times of day when cardiovascular events most commonly occur (the early morning and evening). 5 This relationship has led many to wonder whether acute changes in arterial stiffness are responsible for the temporal nature of cardiovascular events. In this study, the fact that ADP was significantly shorter in the morning after ingestion of a mixed meal may provide additional evidence to support the temporal nature of arterial stiffness; however, further investigation will be needed to determine the exact nature of the relationship of ADPs to arterial stiffness and cardiovascular risk. Given that the ADP changed significantly only 2 hours after ingestion of a mixed meal in healthy young adults while PWV and CAS did not, there is a case to be made that ADP may be an earlier and more sensitive marker of arterial stiffness when compared alongside the traditional benchmark measures of PWV and carotid strain. However, the paucity of research on the relationship between carotid artery time intervals like ADP and prandial state means that further study will be needed to confirm this finding.
In addition, our finding that slopes of CAA curves were significantly steeper postprandially is of interest because it suggests that the rate of change in the arterial area is significantly affected by prandial state. This finding persisted after controlling for BP and PWV. In a previous study, we found similar results in a comparison of the CAA curves of patients with hypertension and diabetes with those of healthy controls. 17 Along with the changes in ADP, these findings provide further evidence for the existence of vascular changes that occur after mixed meal ingestion in healthy young adults.
Limitations
This pilot study had several notable limitations. First, our analysis was performed with a system intended for studying myocardium, not peripheral vasculature, and on DICOM images instead of raw ultrasound data. To date, no dedicated platforms exist for studying arterial wall ultrasound images.
Second, we did not obtain serum lipid or glucose measurements to document the degree of postprandial lipemia and glycemia. While such measurements would help determine if postprandial changes in triglyceride levels were at all correlated with observed changes in ADP and CAA curves, such considerations may not be as relevant to this study given that all study subjects were young healthy adults without major co-morbidities in whom postprandial lipemia is expected to peak at between 2 and 3 hours after oral fat load ingestion. 30
Third, we did not assess the dietary patterns of study participants with either a food frequency questionnaire or dietary records. Such information would be helpful in delineating whether chronic dietary exposure to high fat is required for postprandial changes in arterial stiffness. A previous study by Fielding et al. showed in healthy young adults that fat from previous meals contributes to an early postprandial peak in triglyceride levels. 31 However, it should be noted that the results in the Fielding study were obtained between two meals (breakfast and lunch) separated by only 5 hours. All of the participants in this study had been fasting for at least 8 hours, thus mitigating some if not all of the effect of previous meals.
Fourth, this study is limited by the lack of a control group. Controls would have helped evaluate the contribution of diurnal changes in arterial stiffness to our results. A previous study by Bodlaj et al. found that AI in healthy young men were significantly lower at noon and the early evening when compared with 8:00 a.m. 7 In contrast, our study found no significant change in central AI postprandially. This raises the possibility that a normal physiologic decrease in AI, a marker of arterial stiffness, seen in healthy individuals was blunted by the effects of postprandial lipemia. Moreover, two randomized crossover studies in which each subject acted as his/her own control demonstrated significant albeit differing effects of high-fat, high-sodium and high-carbohydrate meals on arterial stiffness in healthy young adults.12,24 The results from these crossover studies and the study by Bodlaj et al. suggest that our findings with regards to ADP and CAA are more likely to be related to oral fat load ingestion than normal diurnal variation. However, without control subjects this claim cannot be verified with any degree of certainty. Future studies should include control subjects to reduce this limitation.
However, our study’s greatest limitation is a failing of all postprandial studies: a lack of standardized methodology. Despite the fact that postprandial lipemia is now directly linked to cardiovascular risk, the methods for studying it are highly variable and largely determined by study aims and available resources. 27 Such inconsistency not only calls the validity of individual study results into question, but also limits our ability to combine and compare data sets for more powerful analyses.
Conclusion
There appears to be no measurable increase in arterial stiffness as assessed by CAS with ultrasound or PWV with applanation tonometry after ingestion of a mixed meal in healthy young adults. There does, however, appear to be a significant effect on ADP, a novel time-sensitive marker of vascular function. Significantly steeper CAA curve slopes provide further evidence that changes in vascular function may be occurring even while arterial stiffness remains unchanged. Therefore, ADP may possibly be a more sensitive marker of arterial stiffness in healthy young adults when compared with PWV and CAS.
Footnotes
Acknowledgements
This study thanks Berthold Klas and TomTec Imaging (Munich, Germany) for supplying the speckle-tracking strain analysis software and associated documentation. We also thank the participants of this study for voluntarily contributing their time and effort.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
