Abstract
Abnormalities of the microvasculature are linked to major cardiac events, but their role in the development of atrioventricular conduction abnormalities (AVCA) is unknown. We examined the association between central retinal arteriolar equivalent (CRAE), a measure of the microvasculature, and incident AVCA. This analysis included 3975 participants free of AVCA at baseline from the Multi-Ethnic Study of Atherosclerosis (MESA). Incident AVCA was defined as a composite of new heart rate-adjusted PR interval ⩾ 200 ms (first-degree AV block) and advanced block (second-degree or complete AV block) detected from the MESA exam 5 electrocardiogram (ECG). CRAE was measured from retinal photographs at exam 2. Both ECGs and retinal photographs were collected using standardized methods and read and graded at central core labs. Incident AVCA were present in 7.4% (n=290) of the participants, of which 94% were first-degree AV block. Incident AVCA were increasingly more common in participants with narrower CRAE (4.6% in Q4, 6.4% in Q3, 7.0% in Q2 and 10.8% in Q1, p-value for trend < 0.0001). The socio-demographic and cardiovascular disease risk-adjusted odds of incident AVCA in the Q1 group (the group with the narrowest retinal arteriolar diameter) was nearly twice the odds in the Q4 group (OR: 1.68, 95% CI: 1.15–2.51). This association remained significant after adjustment for major ECG abnormalities and incident cardiovascular disease (Q1 vs Q4, OR: 1.65, 95% CI: 1.01–2.71). In conclusion, narrower retinal arteriolar caliber is associated with development of new AV conduction abnormalities.
Introduction
The duet of aging and hypertension contribute to abnormalities in atrioventricular (AV) conduction 1–3 and the microvasculature.4,5 The retinal arteriole, a widely used and easily accessible window to the microvasculature, can develop microaneurysms, and arteriovenous nicking. Digital photography also documents narrowing in retinal arterioles by the software-assisted measure of central retinal arteriolar equivalent (CRAE). Such abnormalities are independently linked to coronary heart disease and cardiovascular mortality and known to result from the chronic effects of this duet.6–8 Further, the traditional thinking of the benign nature of first-degree AV block has been recently challenged by reports of increased risk of atrial fibrillation, pacemaker implantation, heart failure, cardiovascular mortality and all-cause mortality among community-dwelling adults with a prolonged PR interval.3,9,10 Cardiovascular mortality and morbidity appear to be heralded by a narrow retinal arteriolar caliber and first-degree AV block. These primarily asymptomatic clinical diagnoses are detected by widely used, non-invasive technology. Real-time data capture along with remote data transmission and interpretation capability have potential implications for population health.
Clinical and epidemiologic data suggest a potential role for microvascular abnormalities in developing various cardiovascular diseases; it is unknown whether this applies to AV conduction defects or not. Hence, we examined the association between baseline retinal arteriolar caliber and incident AV conduction abnormalities in the Multi-Ethnic Study of Atherosclerosis (MESA), a population-based cohort study.
Methods
Study participants
The MESA, sponsored by the National Heart, Lung, and Blood Institute is a longitudinal, population-based study of 6814 men and women, aged 45–84 years, from four ethnic groups (white, black, Hispanic and Chinese American) recruited from six field centers (Baltimore, MD; Chicago, IL; Forsyth County, NC; Los Angeles, CA; New York, NY; and St Paul, MN) during the years 2000–2002. MESA is a study of the characteristics of subclinical cardiovascular disease (disease detected non-invasively before it has produced clinical signs and symptoms) and the risk factors that predict progression to clinically overt cardiovascular disease. A detailed description of the MESA sampling and recruitment procedures has been published. 11 Institutional review board approval was obtained at all MESA sites. All participants provided written informed consent.
For this analysis we first created a dataset of 4489 participants (Figure 1) with all relevant variables including exams 1 (electrocardiogram; ECG), 2 (retinal examination) and 5 (ECG), and then excluded participants with incomplete data, poor quality ECG, prevalent first-degree AV block (heart rate-adjusted PR interval ⩾ 200 ms) or advanced AV block (second-degree or complete AV block), wandering atrial pacemaker, Wolff–Parkinson–White syndrome, and atrial fibrillation or flutter on ECG obtained at exam 1. After all exclusions, 3975 participants remained and were included in this analysis. The mean interval was 1.9 years between exams 1 and 2, and 7.7 years from exam 2 to 5.
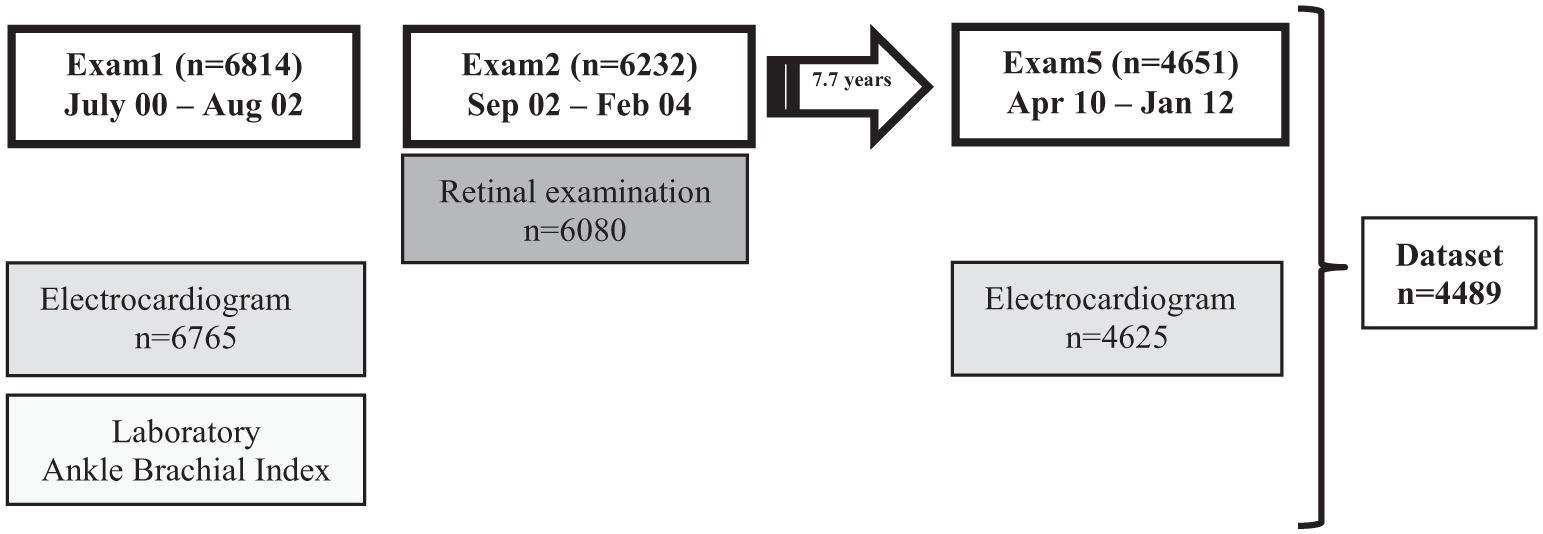
MESA timeline.
Data collection
At the baseline examination, standardized questionnaires were used to obtain demographic information and level of education, annual household income, smoking history, and medication usage for high blood pressure, high cholesterol, or diabetes.
Cigarette smoking was calculated in pack-years and defined as current, former, or never. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. Resting seated systolic and diastolic blood pressure was measured three times using an automated oscillometric sphygmomanometer (DINAMAP PRO 100; GE Medical Systems Information Technologies, Inc., Milwaukee, Wisconsin, USA), and the average of the last two measurements was used for analysis. Hypertension was defined as systolic blood pressure ⩾ 140 mmHg, diastolic blood pressure ⩾ 90 mmHg or history of using blood pressure lowering medication. Pre-hypertension was defined as a systolic blood pressure between 120 and 139 mmHg, or diastolic blood pressure between 80 and 89 mmHg. Peripheral artery disease was defined as an ankle–brachial index < 0.9.
Laboratory
Total and high-density lipoprotein (HDL) cholesterol, triglycerides, and glucose levels were measured from blood samples obtained after a 12-hour fast. Low-density lipoprotein (LDL) cholesterol was calculated by the Friedewald equation. Diabetes was defined as fasting glucose ⩾ 126 mg/dl or use of hypoglycemic medication. Using fasting blood samples from the baseline exam, the following factors were selected based on their association with the CRAE in prior analyses: high-sensitivity plasma C-reactive protein (CRP), estimated glomerular filtration rate (eGFR), and ABI. The method for measuring these variables has been published. 11
Electrocardiography
Standard 12-lead ECGs were digitally acquired using a Marquette MAC-PC electrocardiograph (GE Medical Systems Information Technologies, Inc., Milwaukee, Wisconsin, USA) at 10 mm/mV calibration and a speed of 25 mm/s. ECG tracings were centrally read at the EPICARE Center (Wake Forest School of Medicine, Winston-Salem, NC, USA), where they were initially visually inspected for technical errors and inadequate quality then automatically processed using a GE Marquette 12SL (version 2001) (GE Medical Systems Information Technologies, Inc., Milwaukee, Wisconsin, USA). The PR duration (PR-dur), defined as the duration between the start of P wave until the start of QRS complex, was measured automatically. Given the automated process of measuring the PR interval from a digital ECG, expectedly there was perfect reproducibility. We computed a heart rate-adjusted PR-dur (PRa) using the Soliman–Rautaharjue formula, as follows: 12 [PRa = PR-dur + 0.26 (heart rate − 70) in individuals < 60 years, and PRa = PR-dur + 0.42 (heart rate − 70) in individuals ⩾ 60 years].
Prolonged PR duration (first-degree AV block) was defined as PR ⩾ 200 ms in the main analysis, and 220 ms in additional analysis. The earlier cut-point is the most commonly used cut-point in clinical practice, and the latter is the definition of first-degree AV block by the Minnesota ECG classification. 13 Advanced AV block was defined as the presence of one of the following: Mobitz type II, partial (second-degree) AV block, Wenckebach’s phenomenon or complete (third degree) AV block.
Retinal photography and measurement of retinal arteriolar caliber
Retinal photography was performed at each study center during exam 2, using a standardized protocol by trained fundus photographers with a 45-degree 6.3-megapixel digital non-mydriatic camera (Canon, Lake Success, NY, USA) in a darkened room. Two photographic fields were obtained for each eye, one centered on the optic disc and the other on the fovea. All images acquired and archived using standard software (Eye QSL; Digital Healthcare Inc., Cambridge, England) were then encrypted and electronically transmitted to be evaluated by trained graders masked to patients’ characteristics at the Retinal Reading Center, University of Wisconsin, Madison. Retinopathy signs were graded in both eyes as prescribed by the Early Treatment Diabetic Retinopathy Study (ETDRS) protocol. 14 Retinopathy was considered to be present if any characteristic lesions as defined by the ETDRS severity scale were present: microaneurysms (MA), hemorrhages, cotton wool spots (CWS), intra-retinal microvascular abnormalities (IRMA), hard exudates (HE), venous beading, and new vessels. These lesions were defined as present if graded as either definite or probable. The left-eye photographs were used only if the optic disc-centered right-eye photographs did not produce a retinal vascular caliber measurement. For each photograph, all arterioles coursing through an area of 0.5–1 disc in diameter from the optic disc margin were measured and the largest six arteriolar calibers were used in estimating the CRAE using formulae developed by Hubbard and modified by Knudtson.14,15 Reproducibility of retinal vascular measurements have been reported previously, with intra-grader and inter-grader intra-class correlation coefficients ranging from 0.78 to 0.99.14,15
Outcome
Atrioventricular conduction abnormality (AVCA) was defined as a composite of first-degree AV block (heart rate-adjusted PR interval ⩾ 200 ms) and advanced AV block (second-degree or complete AV block). Incident AVCA was defined as detection of that composite outcome in MESA exam 5 ECGs in those who were free of AVCA at exam 2. In additional analyses, prolonged PR duration (using PR ⩾ 200 ms and 220 ms, separately) and advanced AV block were used separately.
Statistical analysis
Frequency distributions of all variables were first inspected to identify anomalies and outliers possibly caused by measurement artifacts. Characteristics of the study population were examined and distribution of AV block was reported by quartiles of CRAE. With Q4 (widest quartile of retinal arteriolar caliber) as the reference group, the odds ratios (ORs) and 95% confidence intervals (CIs) of incident AVCA associated with the other three quartiles of CRAE were calculated using the following incremental models. Model 1: adjusted for demographics and socio-economics (age, sex, race, education and income); Model 2: Model 1 variables + cardiovascular disease risk factors (smoking, HDL and total cholesterol, hypertension status including hypertension medication use and diabetes status); Model 3: Model 2 + C-reactive protein, peripheral artery disease, estimated glomerular filtration rate (CKD-EPI), anti-arrhythmic drug use, any major ECG abnormalities including left ventricular hypertrophy; Model 4: Model 3 + baseline and retinal venular caliber and baseline PR duration; and Model 5: Model 4 + intervening cardiovascular disease events. Interactions were tested by gender, race and diabetes and hypertension status (as present/absent and normal/pre-hypertension/hypertension, separately) by both product method and stratification. Using these models, ORs and 95% CIs were computed to examine the relationship between retinopathy markers and incident AVCA.
A two-tailed probability value < 0.05 was considered significant. The JMP (John’s Mathematical Program), version 10.1 (SAS Institute, Cary, NC, USA) was used to perform all computations.
Results
Table 1 shows the baseline characteristics of the study participants stratified by quartiles of CRAE. As shown, participants with the narrowest retinal arteriolar caliber (quartile 1) were older and more likely to be male, have hypertension, and be an ex-smoker; whereas diabetes and current smoking was increasingly noted among participants with wider retinal arterioles.
Baseline characteristics stratified by retinal arteriolar diameter quartiles.
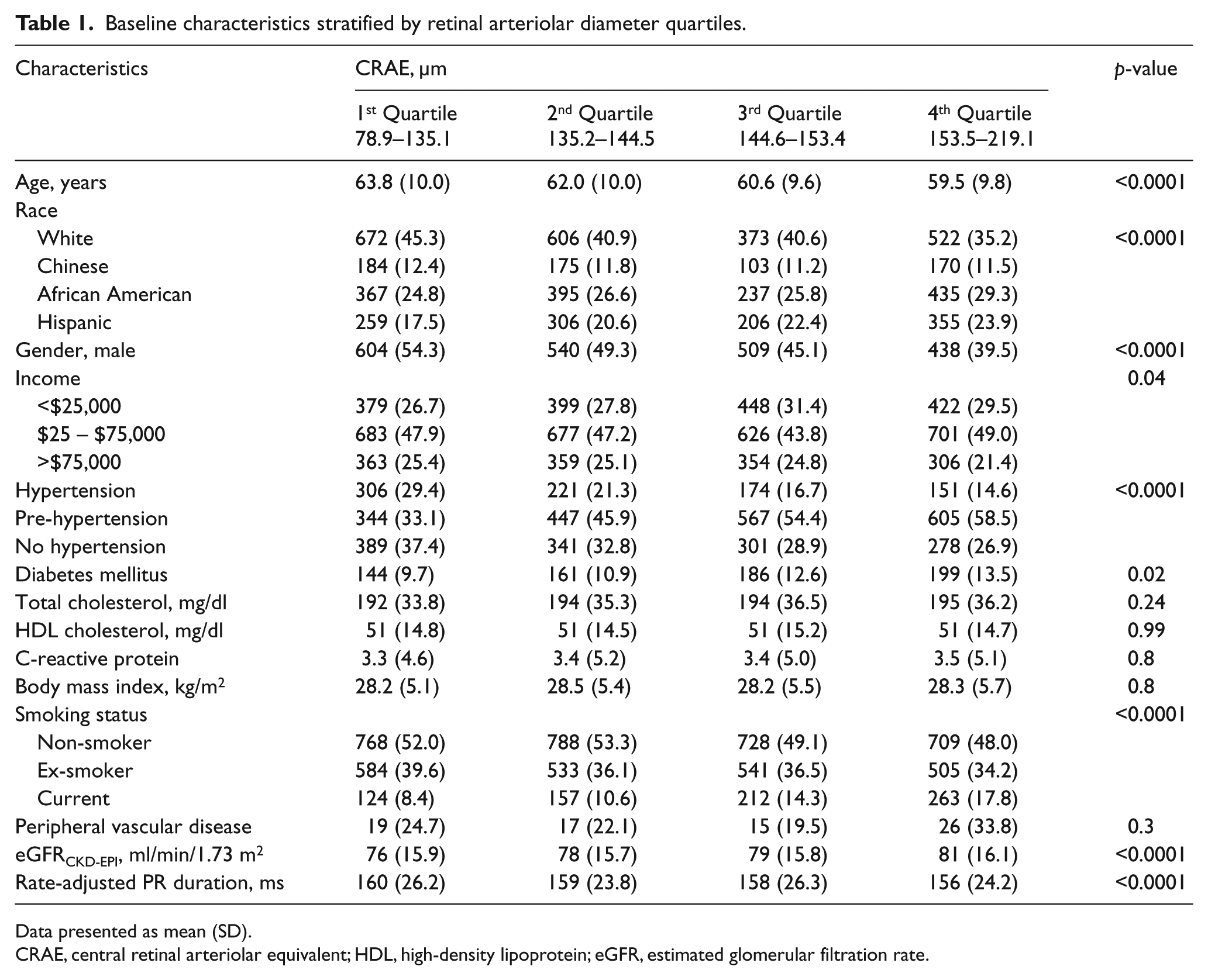
Data presented as mean (SD).
CRAE, central retinal arteriolar equivalent; HDL, high-density lipoprotein; eGFR, estimated glomerular filtration rate.
Incident AVCA was detected in 290 participants (7.4%). Of those who developed incident AVCA, advanced AV block was present in 6.4%. The greater the retinal arteriolar narrowing was at baseline the more frequently incident AVCA occurred (4.6% in Q4, 6.4% in Q3, 7.0% in Q2 and 10.8% in Q1; p-value for trend < 0.0001). This pattern was mainly driven by the first-degree AV block whether using 200 ms or 220 ms as cut-points (Figure 2).
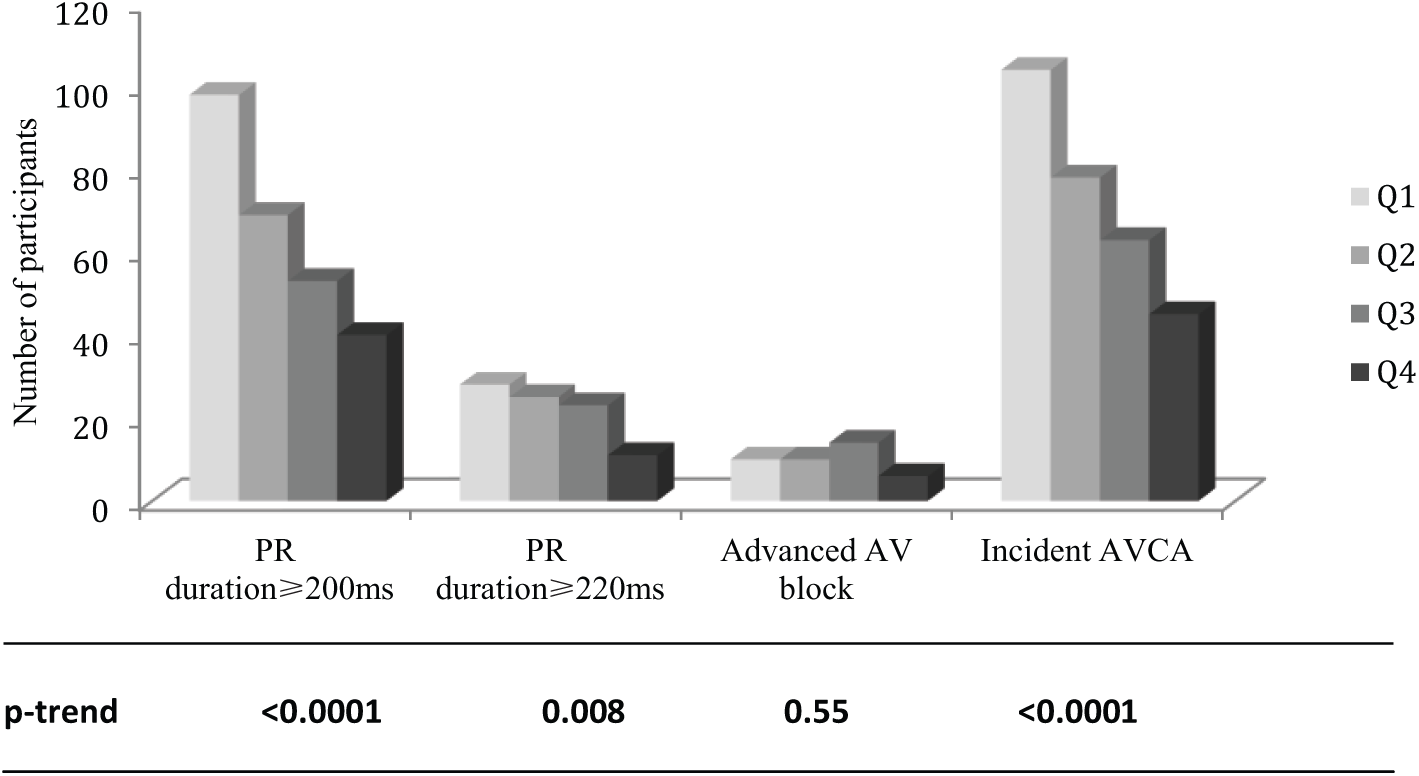
Distribution of atrioventricular block by CRAE quartile.
The socio-demographic-adjusted odds of incident AVCA in the Q1 group (the group with the narrowest retinal arteriolar diameter) was nearly twice the odds in the Q4 group (Table 2). This association remained strong (OR 1.65) and significant after further adjustment for cardiovascular risk factors, major ECG abnormalities and incident cardiovascular disease (Table 2). Similar results were obtained when systolic blood pressure (as a continuous variable) replaced hypertension in model 2 (OR: 1.65, 95% CI: 1.12–2.46, p=0.01). There were no significant multiplicative interactions by race, gender, diabetes or hypertension status (present/absent or normal/pre-hypertension/hypertension). After stratification, the odds of incident AVCA in the Q1 group compared to Q4 was 1.71 (95% CI: 0.59–5.12, p=0.3) among non-hypertensives and 6.24 (95% CI: 0.68–74.2, p=0.10) in hypertensives.
Presence of incident AVCA (first-degree AV block and advanced AV block) by quartiles of retinal arteriolar caliber.
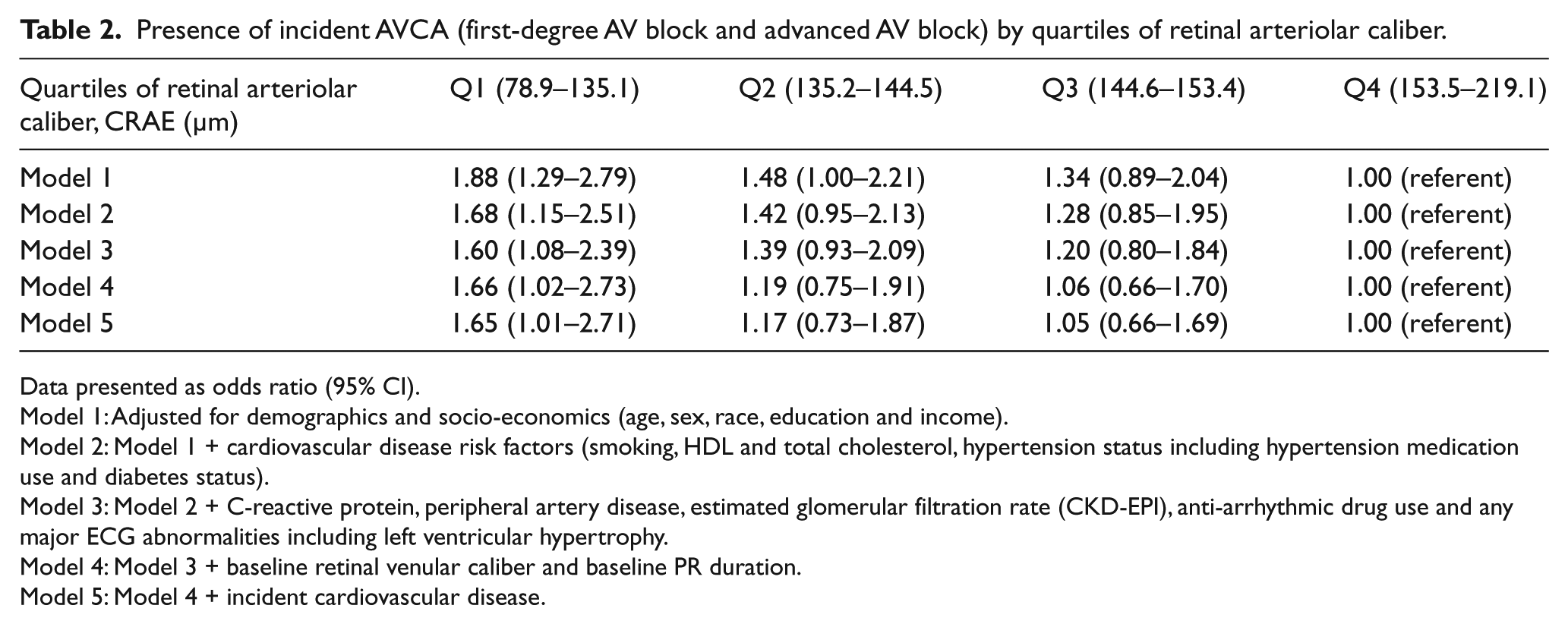
Data presented as odds ratio (95% CI).
Model 1: Adjusted for demographics and socio-economics (age, sex, race, education and income).
Model 2: Model 1 + cardiovascular disease risk factors (smoking, HDL and total cholesterol, hypertension status including hypertension medication use and diabetes status).
Model 3: Model 2 + C-reactive protein, peripheral artery disease, estimated glomerular filtration rate (CKD-EPI), anti-arrhythmic drug use and any major ECG abnormalities including left ventricular hypertrophy.
Model 4: Model 3 + baseline retinal venular caliber and baseline PR duration.
Model 5: Model 4 + incident cardiovascular disease.
The retinopathy markers (hard exudates, soft exudates, microaneurysms with or without hemorrhage, focal arteriolar narrowing), both individually and together, were not associated with incident AVCA.
We also attempted to investigate alternative definitions and subgrouping of AV conduction abnormalities (Supplementary Table 1). As shown in Supplementary Table 1 and Figure 2, the relationship between arteriolar narrowing and first-degree AV block (whether defined as PR ⩾ 200 or 220 ms) was significant after accounting for demographics and other potential confounders. However, the small number of advanced AV block cases did not allow for enough statistical power to examine this group separately.
Discussion
In this large, prospective, population-based study of adults free of cardiovascular disease, we found evidence suggesting an association between narrow retinal arteriolar caliber and incident atrioventricular conduction abnormality. This association persisted after adjustment for cardiovascular risk factors and potential confounders.
Wong et al. 6 described the link between retinal microvascular abnormalities and cardiovascular mortality and we now provide the first evidence that retinal arteriolar narrowing is correlated with AV conduction abnormality.
Several potential mechanisms may account for the association of retinal arteriolar narrowing and AV conduction abnormalities. First, the increased risk might be due in part to associated asymptomatic myocardial ischemia. Following thrombolysis for acute myocardial infarction, reperfusion injury and myocardial ischemia involve conduction tissue and results in both AV block and atrial arrhythmias.16,17 The arterial supply to the nodal tissue frequently arises from the right coronary artery and measures 1–3.5 mm in external diameter at its origin. 18 Narrowing of the AV nodal artery is linked to increase in fibrous and fatty contents on the AV node and these histopathologic changes are seen in idiopathic degenerative nodal disease, the most common cause of nodal dysfunction.19,20 Finally, retinal arteriolar narrowing may be associated with the presence and severity of coronary artery disease defined on angiography and cardiac magnetic resonance imaging.21,22 However, our study population comprised individuals free of cardiovascular disease at baseline, we excluded those with baseline AV conduction disorders, and accounted for incident major cardiovascular events. This makes it unlikely that the observed associations in our study are due to coronary disease.
Uncommon conditions such as myotonic dystrophy and cardiac syndrome X are associated with conduction defects and offer insight into the role of the microvasculature. In myotonic dystrophy, microvascular dysfunction leads to ischemia and conduction disturbances, whereas in cardiac syndrome X, cardiac microvascular function shows striking abnormalities.23–26
However, stronger evidence suggests that increased risk of AV conduction abnormalities may be due to microvascular dysfunction wherein narrowed retinal arterioles serve as a surrogate for abnormal systemic microvascular circulation with related findings of myocardial perfusion defects and lacunar strokes.27,28 Retinal arteriolar narrowing is strongly associated with hypertension. Hypertension in turn is an important contributor to cardiovascular disease and chronic kidney disease and the latter is associated with cardiac conduction disorders.4,6,9 This could partially explain our findings of the association between microvasculature and AV conduction abnormality. This link remains subclinical and direct correlation with hard clinical outcomes needs to be explored in the future. In our study, smoking and diabetes were linked to a wider retinal arteriolar diameter, an effect consistent with previously reported associations.7,29–32 Possible explanations include pseudodilation caused by endothelial disruption due to inflammation, elastic tissue degeneration and, more likely, increased nitrous oxide production and potassium channel activation.30–32 C-reactive protein was not linked to retinal arteriolar caliber, as reported previously. 22
Our findings should be interpreted in the context of certain limitations. Similar to all studies with a similar design and despite adjusting for several factors, we cannot completely exclude the possibility of residual confounding of the association between retinal arteriolar narrowing and an AV conduction abnormality by unmeasured variables. Also, retinal photographs and ECGs were not done at the same time, creating the potential for lag time bias. Selection bias may have resulted from the length of time between the measurement of CRAE (exam 2) and outcome ECG (exam 5). Despite these limitations, this is the first report examining the association between microvasculature and AV conduction in a large, population-based, multi-ethnic cohort with the use of quantitative, masked, valid and reliable assessment of retinal arteriolar caliber and virtually complete outcome ascertainment.
Conclusion
In conclusion, we showed that retinal arteriolar narrowing is associated with incident AV conduction abnormality. These findings suggest that microvascular changes may occur simultaneously in separate anatomical beds and play a role in the pathophysiology of AV conduction abnormalities in multi-ethnic adults free of apparent cardiovascular disease. Further research is warranted to clarify the underlying pathophysiological link and identify longitudinal clinical outcomes.
Footnotes
Acknowledgements
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The MESA was supported by contracts N01-HC-95159 through N01-HC-95169 from the National Heart, Lung, and Blood Institute. This work was supported in part by 5K12HL083763 (BC) and a Ruth L Kirschstein NRSA Institutional Training Grant 5T32HL076132-10 (WQ).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
