Abstract
The purpose of this study is to describe 10 cases of symptomatic isolated spontaneous celiac trunk dissection (ISCTD) in order to evaluate the initial clinical presentation, diagnosis, treatment modalities and outcomes. A retrospective search was performed from 2009 to 2014 and 10 patients with ISCTD were included in the study. Patients with associated aortic and/or other visceral artery dissection were excluded. The following information was collected from each case: sex, age, associated risk factors, symptoms, diagnostic method, anatomic dissection pattern, treatment modality and outcome. Most patients were male (90%), with an average age of 44.8 years, and the most common symptom was abdominal pain (100%). Hypertension and vasculitis (polyarteritis nodosa) were the most frequent risk factors (40% and 30%, respectively). Diagnosis was made in all patients with computed tomography. Dissection was limited to the celiac trunk in three patients and extended to celiac branches in the other seven. Initial conservative treatment was attempted in every case and was successful in nine patients. In one case, initial conservative treatment was unsuccessful and arterial stenting with coil embolization of the false lumen was performed. After successful initial treatment, late progression of the dissection to aneurysmal dilatation was observed in two patients and it was decided to perform endovascular treatment. Mean follow-up was 19 months, ranging from 2 to 59 months. In conclusion, initial conservative treatment seems adequate for most patients with ISCTD. Long-term follow-up is mandatory, owing to the risk of later progression to aneurysm.
Introduction
Isolated spontaneous celiac trunk dissection (ISCTD) without associated aortic disease is uncommon and only case reports and small case series have been described in the literature.1–3 The exact pathogenesis of ISCTD remains unclear, but risk factors for visceral artery dissection include atherosclerotic disease, hypertension, fibromuscular dysplasia, cystic medial necrosis, segmental arterial mediolysis, pregnancy and connective tissue disorders.2,4–6
Initial clinical presentation is variable. While the most common symptoms are abdominal pain and weight loss, some patients may be asymptomatic and others may present with severe complications, such as arterial rupture, acute pancreatitis, obstructive jaundice or intestinal angina.1,6 Several treatment modalities were proposed for this condition, including conservative management, anticoagulation, endovascular interventions and open surgery; however, there is no consensus regarding the best approach.2,7,8
The purpose of this study is to describe 10 cases of symptomatic ISCTD in order to identify optimal management of this condition.
Materials and methods
Ten symptomatic patients with ISCTD were referred to the Vascular Interventional Radiology Department and Vascular and Endovascular Department of our institution between 2009 and 2014 for evaluation of endovascular treatment and were included in this retrospective study. Patients with associated aortic and/or other visceral artery dissection (superior mesenteric artery and renal artery) were excluded from the study.
The following information was collected from each case: sex, age, associated risk factor, symptoms, diagnostic method, anatomic dissection pattern, treatment modality and outcomes.
This study was reviewed and approved by the institutional review board. The requirement for patient informed consent was waived.
Results
In our series of 10 patients, nine were male (90%) and one was female, with an average age of 44.8 years (range 35–55), and the most common symptom was abdominal pain, reported in all cases (100%), without signs of peritonitis. There were no asymptomatic patients (Table 1). Hypertension and vasculitis (polyarteritis nodosa) were the most frequent risk factors (40% and 30%, respectively).
Celiac trunk dissection patients’ demographics and outcomes.
PAN, polyarteritis nodosa.
All 10 patients underwent multidetector computed tomography (MDCT) and diagnosis was made, identifying an intimal flap. Dissection was limited to the celiac trunk in three patients and extended to celiac branches in the other seven. The mean follow-up was 19 months, ranging from 2 to 59 months.
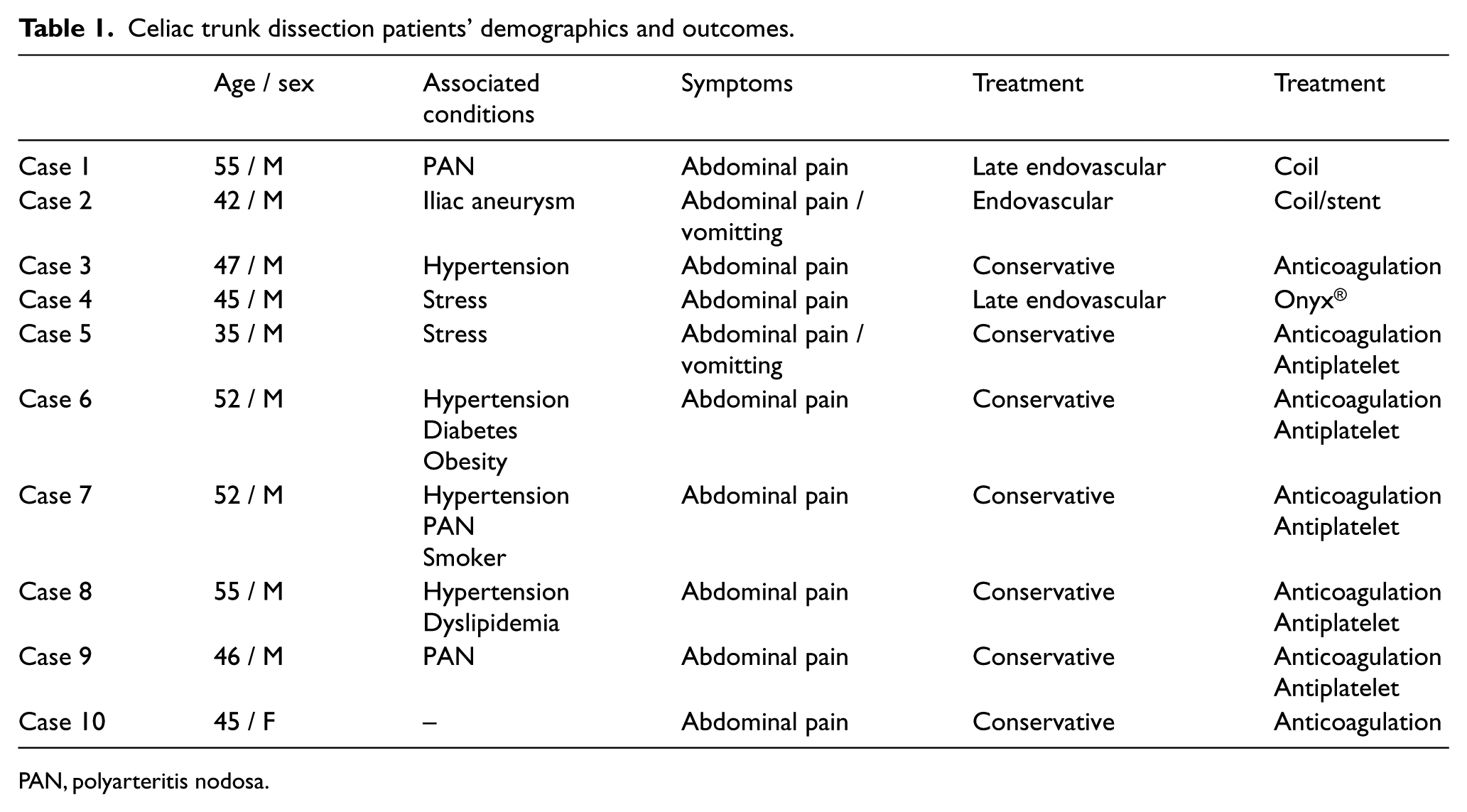
Initial conservative treatment, with anticoagulation, pain and hypertension control, was attempted in every case and was successful in nine patients. In one case (Case 2), initial conservative treatment was unsuccessful and the patient maintained abdominal pain despite medication. Therefore, endovascular intervention was indicated, with stent deployment and coil embolization of the false lumen (Figure 1).
Imaging exams of a patient with ISCTD. (A) Abdominal computed tomography (CT) showing celiac trunk aneurysm and occlusion of the hepatic and splenic arteries. (B) Angiography reveals celiac trunk aneurysm, with occlusion of the hepatic and splenic arteries. (C) Celiac trunk angiogram after stent deployment and coil embolization. Celiac trunk: white arrow; left gastric artery: black arrow.
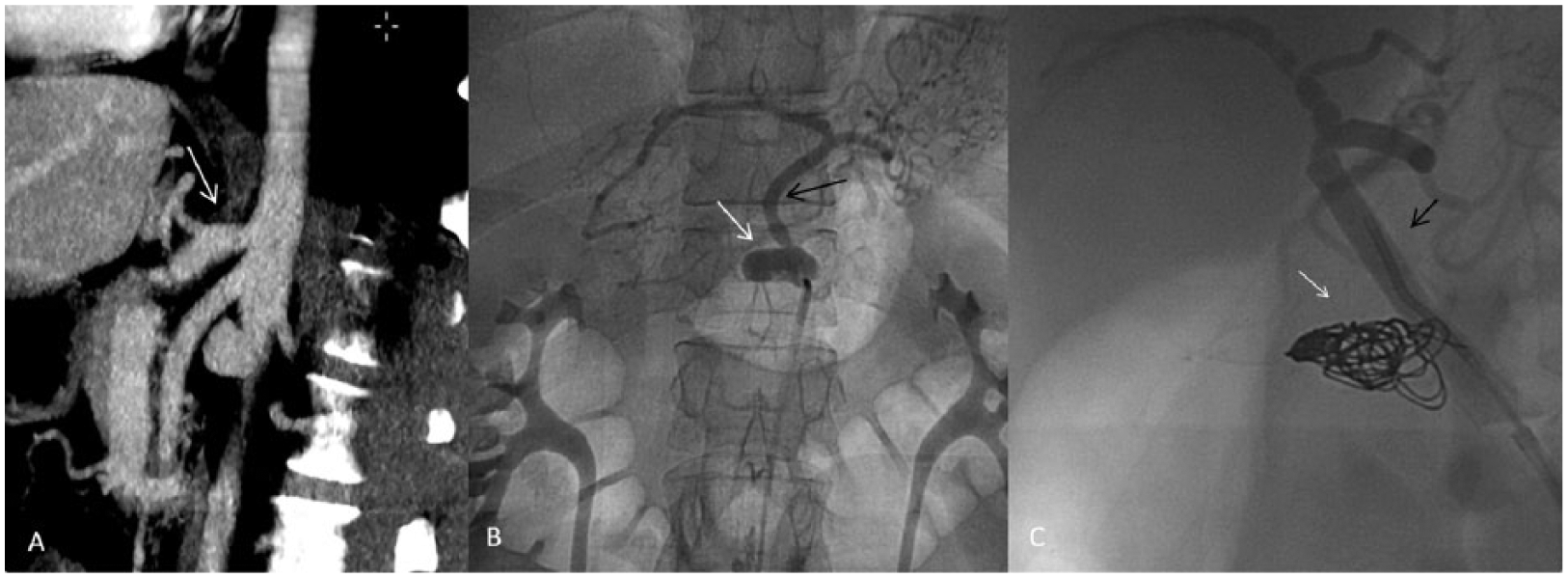
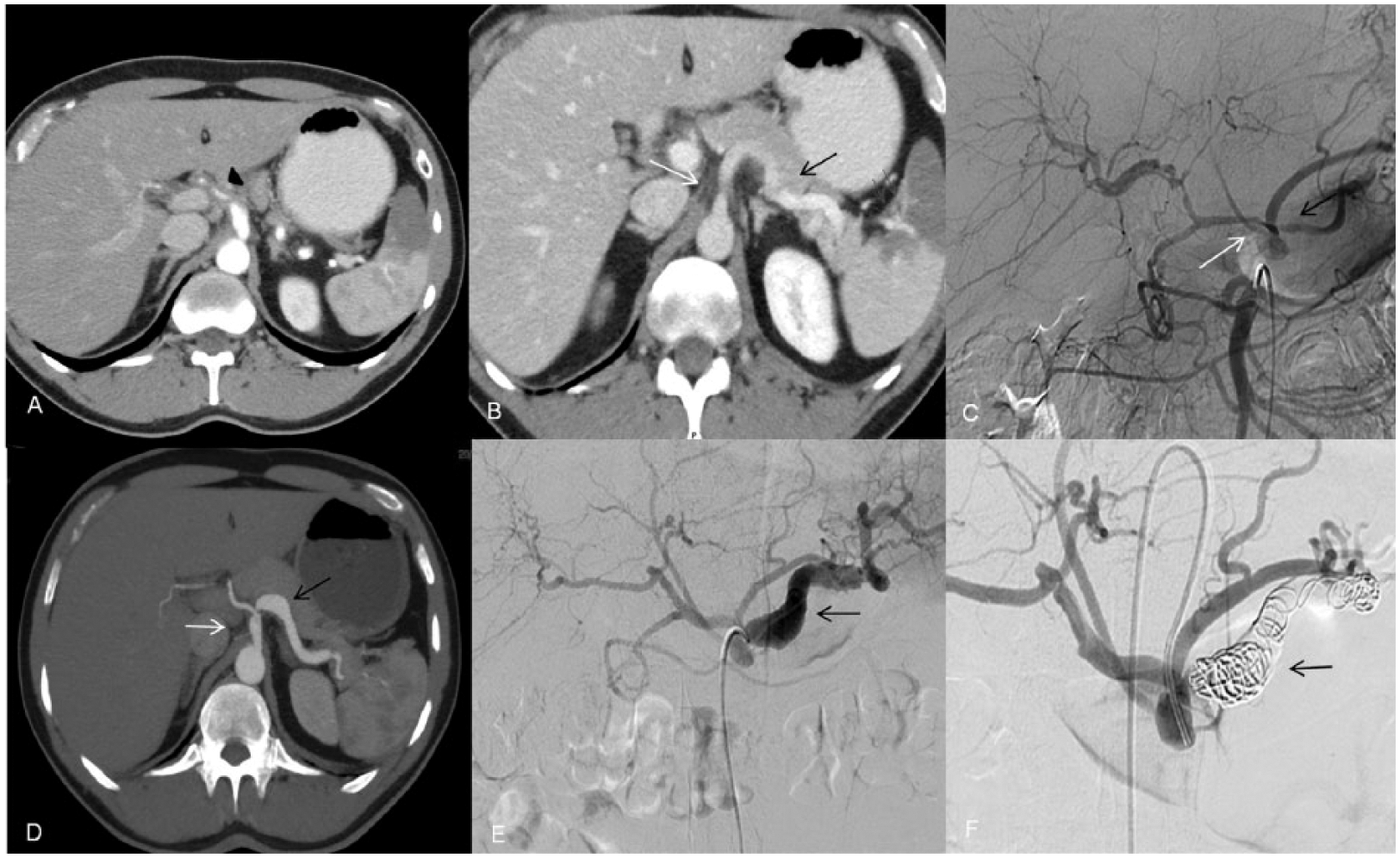
After successful initial treatment, late progression (more than 30 days) of the dissection to aneurysmal dilatation was observed in two patients and it was decided to perform endovascular treatment. Both patients were then asymptomatic and the aneurysm diagnosis was made on follow-up CT. One of these patients (Case 1), with celiac trunk dissection extending to the hepatic and splenic arteries, developed a splenic artery aneurysm 44 months after the initial diagnosis and underwent coil embolization (Figure 2). The other patient (Case 4) had celiac trunk dissection that evolved into aneurysmal dilatation of the false lumen after 6 months of conservative treatment and underwent endovascular embolization with Onyx® (ev3 Neurovascular, Irvine, CA, USA) (Figure 3).
Imaging exams of a patient with ISCTD. (A) Abdominal CT showing celiac trunk dissection extending to the hepatic artery. (B) Abdominal CT showing celiac trunk dissection extending to the splenic artery. (C) Celiac trunk angiogram depicting ISCTD. (D) Abdominal CT after 44 months demonstrating splenic artery aneurysm. (E) Celiac trunk angiogram demonstrating splenic artery aneurysm. (F) Celiac trunk angiogram after splenic artery embolization with coils. Celiac trunk: white arrow; hepatic artery: black arrowhead; splenic artery: black arrow; area of splenic malperfusion: gray arrow.
Imaging exams of a patient with ISCTD. (A) Abdominal CT showing celiac trunk aneurysm 6 months after initial dissection. (B) Angiography demonstrating celiac trunk aneurysm. (C) Celiac trunk angiogram demonstrating occlusion of the celiac trunk with Onyx. Celiac trunk: white arrow; hepatic artery: black arrowhead; coil embolization of left gastric artery: black arrow.
Discussion
Isolated dissection of a splanchnic artery is uncommon. Most reports of this condition are regarding superior mesenteric artery dissection, and ISCTD is an even rarer event.9–12
Despite several risk factors, such as hypertension and vasculitis, which have been associated with visceral artery spontaneous dissection, the real causes are still unclear. 13 In our series, hypertension and polyarteritis nodosa were important associated conditions, found in 40% and 30% of patients, respectively. The occurrence of polyarteritis nodosa as a risk factor for ISCTD was not previously reported in the literature, but was observed in a high percentage of our patients. Although we cannot explain the reasons for the high frequency of this condition in our series, it is known that polyarteritis nodosa is a vasculitis that is more frequent in men between 40 and 60 years of age and is associated with hypertension, which is also a condition frequently observed in celiac trunk dissection. 14
All of our 10 patients were admitted to hospital with abdominal pain and two of them also suffered vomiting. This is similar to reports in the literature, where abdominal pain is also the most common symptom.3,6,12 In our series, however, there were no asymptomatic patients, unlike in previous studies that reported an incidence of 19–50% who were incidentally diagnosed from imaging exams requested for other reasons.3,4,8
Most cases of ISCTD have been reported in the last 5 years. This increase in incidence reflects the wider use of MDCT for evaluation of abdominal pain or low-grade abdominal symptoms.12,15 The diagnostic method of choice for ISCTD is MDCT because it is fast, minimally invasive and reliable, and provides good evaluation of the vasculature, including the course and extension of the dissection, the patency of the false lumen and the presence of anatomic variation.3,6,9,10,15–17 Also, MDCT allows evaluation of differential diagnosis and visualization of ischemic bowel or parenchymal infarction. 13 Although MDCT is the primary imaging method for ISCTD, magnetic resonance imaging, duplex scanning and endoscopic ultrasound can also be used for diagnostic investigation.15,18–20 In our series, MDCT was also the main imaging method for ISCTD diagnosis, performed in all patients and capable of demonstrating an intimal flap and the extension of the dissection to celiac branches.
Whereas the diagnosis of ISCTD is one of exclusion, the initial evaluation should be based on clinical, laboratory and imaging exams, focusing on abdominal pain investigation (which is the most frequent symptom). More frequent conditions that may cause abdominal pain should be excluded. The specific management of patients with ISCTD will start from identification of the dissection on imaging exams such as computed tomography. Thereafter, the patient may follow one of four routes. If there is evidence of intestinal necrosis associated with dissection, the indication is exploratory laparotomy. In cases of dissections associated with ruptured aneurysms, open surgical correction or endovascular treatment should be performed, according to the team’s experience and availability of materials. If the patient has refractory abdominal pain, aneurysm ⩾2 cm, true lumen compression or intestinal ischemia suspicion, he/she should undergo endovascular treatment. Otherwise, the patient should receive clinical care with control of risk factors and periodic imaging. An algorithm for ISCTD management is proposed in Figure 4.
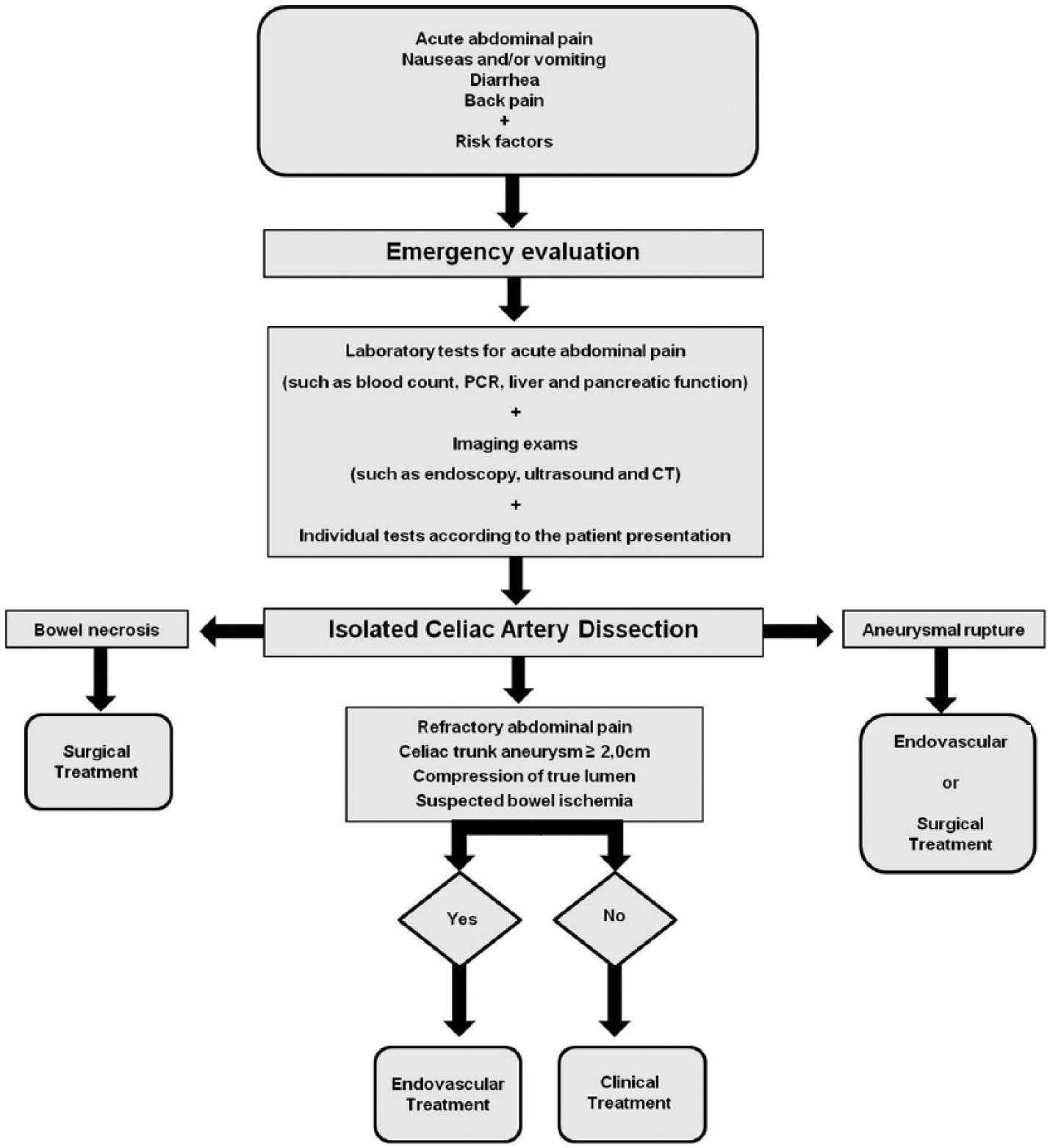
Management algorithm for ISCTD.
The best management strategy for patients with ISCTD remains controversial. 21 Most authors recommend initial conservative treatment in the absence of visceral malperfusion, with anticoagulation and control of risk factors.3,7,8,21
The best medical treatment for hypertension includes rigorous control of blood pressure, ranging from oral antihypertensives to admission to ICU with the use of intravenous antihypertensives. For vasculitis, the specific treatment includes corticosteroids and immunosuppression. In our series, all patients with polyarteritis nodosa had a diagnosis made during their admission for abdominal pain due to celiac dissection and other reported symptoms, such as weight loss, testicular pain, myalgia and/or leg weakness and polyarthralgia, associated to negative anti-neutrophil cytoplasmic antibody (ANCA) and increased levels of C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR); therefore they did not have any previous treatment for their condition.
There is no consensus about the best medical treatment for polyarteritis nodosa. There are only a few recommendations, so each institution or physician does what is deemed to be the best medical treatment. In our group, when there is visceral involvement, all patients are initially treated with oral prednisone at a dosage of 1 mg/kg/day for 30 consecutive days; after that the dosage is reduced by 10 mg every 15 days to a dosage of 20 mg/day; then the dosage is reduced by 5 mg every 15 days to a dosage of 10 mg/day; finally, by 2.5 mg every 15 days to the minimal effective dose or, when possible, until definitive withdrawal. Prednisone is used in association with pulses of cyclophosphamide 0.5–1.0 g/m2/month (normally we start with 0.7 g) for 6 months, then cyclophosphamide is replaced by oral azathioprine 2–3 mg/kg/day for at least 2 years. Adjuvant treatments are compulsory, such as potassium, calcium, vitamin D3 and bisphosphonate supplementation to prevent corticosteroid-induced osteoporosis.
One of the patients suspended corticosteroid use without medical recommendation and evolved to aneurysmal dilatation of the false lumen 44 months after the initial event. The other two patients maintained corticosteroid use and remained uneventful. None of the patients had other visceral aneurysms.
To avoid thromboembolic complications, the duration of anticoagulation should be extended to 3–6 months.7,22 Reviewing the literature, we observed that conservative treatment was the modality of choice in more than 70% of patients. In our series, the initial treatment in all 10 patients was anticoagulation, starting with an intravenous 80 UI/kg bolus of unfractionated heparin and then 16 UI/kg/hour was used to maintain full anticoagulation during hospitalization. After hospital discharge, anticoagulation was maintained with warfarin (international normalized ratio (INR) goal between 2 and 3) for a period of at least 3 months. In five cases, acetylsalicylic acid (100 mg/day) was used in association with warfarin. Conservative treatment was successful for pain relief in nine cases. One fail – due to persistent pain – was treated with stent deployment and coil embolization of the false lumen. Patients who underwent endovascular treatment (coil, stent or Onyx) were discharged only with antiplatelet therapy (acetylsalicylic acid, 100 mg/day).
After successful conservative treatment, 26% of patients with a type B thoracic dissection develop aortic aneurysm during follow-up due to dilatation of the dissected aorta. 23 The same seems to happen in long-term celiac trunk dissection, but the frequency is unclear. There are previous reports of late celiac trunk or hepatic artery dilatation in patients with ISCTD after successful initial conservative management.6,24–26
In our series, 20% of patients treated conservatively developed aneurysmal dilatation requiring intervention due to fast expansion of the false lumen: one patient evolved to splenic artery aneurysm and underwent embolization with coils. Splenic artery aneurysm could be a complication of celiac trunk dissection and when the aneurysm is larger than 2 cm it should be treated. One of the techniques described to treat this condition is splenic artery coiling – this embolization can be performed without causing splenic ischemia due to collateral irrigation provided by short gastric arteries and the left gastroepiploic artery. One patient evolved to celiac trunk aneurysm and underwent embolization of the false lumen with Onyx. To our knowledge, this is the first reported case of Onyx embolization in this situation.
These two cases from our series, in addition to four more cases from the literature,6,24–26 suggest that in a similar way to what happens in thoracic dissection, progression to aneurysm may also happen in ISCTD patients and therefore these patients may require imaging follow-up for a long period to monitor the progression of the disease and assess for aneurysms. Of these six cases, arterial dilatation occurred in two of them within 30 days and in five of six (83%) within 6 months. These findings lead us to suggest a follow-up protocol with clinical evaluation and MDCT at 30 days after the initial event, every 3 months in the first year until complete regression or thrombosis of the false lumen, and then annually after the first year.
Intervention – open surgery or an endovascular approach – is recommended when the patient is hemodynamically unstable, when there is visceral ischemia, when there is progression of the dissection on follow-up exam or on the failure of conventional medical therapy for pain.1,22 In patients requiring intervention, less invasive endovascular techniques have been proposed as the preferential method. 25 Two endovascular approaches are commonly used in these cases. Stenting is preferred when malperfusion due to near occlusion of the false lumen occurs and isolated coiling or coiling associated with stenting are chosen when false lumen dilatation is observed. 12 Nowadays, open surgery is only recommended when bowel necrosis or peritoneal irritation is found12,26 and when endovascular treatment fails.
In our series, severe complications, such as acute bleeding and liver ischemia, were not observed. These complications are poor prognostic factors for patients with ISCTD, and a mortality of 40% is observed in these cases. 20
Our study has the advantage of being the largest series of patients with symptomatic ISCTD; however, it has the drawbacks of being a single center and retrospective study.
In conclusion, our results suggest that initial conservative treatment may be adequate for patients with ISCTD and that long-term follow-up is mandatory, owing to the risk of later progression to aneurysm.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
