Abstract
Venous thromboembolism (VTE) is a prevalent multifactorial health condition associated with significant morbidity and mortality. Population-based epidemiological studies have revealed an association between systemic autoimmune diseases and deep venous thrombosis (DVT)/VTE. The etiopathogenesis of increased risk of VTE in systemic autoimmune diseases is not entirely clear but multiple contributors have been explored, especially in the context of systemic inflammation and disordered thrombogenesis. Epidemiologic data on increased risk of VTE in patients with primary systemic vasculitides (PSV) have accumulated in recent years and some of these studies suggest the increased risk while patients have active diseases. This could lead us to hypothesize that venous vascular inflammation has a role to play in this phenomenon, but this is unproven. The role of immunosuppressive agents in modulating the risk of VTE in patients with PSV is not yet clear except for Behçet’s disease, where most of the studies are retrospective. Sensitizing physicians to this complication has implications for prevention and optimal management of patients with these complex diseases. This review will focus on the epidemiology and available evidence regarding pathogenesis, and will attempt to summarize the best available data regarding evaluation and treatment of these patients.
Introduction
The term venous thromboembolism (VTE) includes deep venous thrombosis (DVT) and pulmonary embolism (PE). The accurate incidence of VTE is not known but approximately 300,000 to 600,000 individuals are affected in the US each year. 1 PE is the third most common “cardiovascular disease”, following myocardial infarction and stroke. 2 VTE is a multifactorial disease contributed to by genetic as well as environmental factors. Thromboembolism is also influenced by the affected vascular bed. 3 The common inherited thrombophilias include factor V Leiden mutation, prothrombin gene mutation, protein S deficiency, protein C deficiency, and antithrombin III deficiency. 4 In practice, acquired risk factors are much more common than inherited thrombophilia. Advanced age, immobility, trauma, surgery, hospitalization, long-distance air travel and pregnancy are well-established risk factors for VTE. Chronic medical conditions such as cancer, congestive heart failure, chronic obstructive pulmonary disease, and chronic kidney disease, especially with nephrotic syndrome, also increase chances of VTE. 5
Recent large-scale epidemiological studies have highlighted systemic autoimmune diseases as a risk factor for VTE.6–8 This article aims to touch upon VTE among patients with systemic autoimmune diseases and to discuss the occurrence of this condition among patients with primary systemic vasculitides (PSV). Since the literature on VTE in patients with PSV is scarce, mechanisms pertaining to other systemic autoimmune diseases have been hypothetically extrapolated to patients with PSV.
Epidemiology of VTE in systemic autoimmune diseases
VTE in patients with systemic autoimmune diseases has been increasingly recognized recently. A few population-based epidemiological studies have been performed in Europe and the United States. Ramagopalan et al. published a study on risk of VTE in people admitted to hospital with immune-mediated diseases using the complete dataset of the England National Hospital Episode Statistics from 1999 to 2008. Compared to controls, patients with various autoimmune rheumatologic conditions showed a statistically higher rate ratio (systemic lupus erythematosus (SLE) 3.71 [95% confidence interval (CI); 3.43–4.02, P<0.001], Sjögren’s syndrome 2.02 [95% CI; 1.80–2.26, P<0.001], scleroderma 1.97 [95% CI; 1.73–2.23, P<0.001], rheumatoid arthritis (RA) 1.75 [95% CI; 1.70–1.80, P<0.001], polyarteritis nodosa 3.53 [95% CI; 2.76–4.44, P<0.001], dermatomyositis/polymyositis 3.04 [95% CI; 2.60–3.54, P<0.001], and ankylosing spondylitis 1.93 [95% CI; 1.74–2.14, P<0.0001]). 8
Zöller et al. reported a study on risk of PE in patients with autoimmune disease from Sweden. The MigMed2 database containing information about all registered residents of Sweden from 1964–2008 was used. Standardized incidence ratios (SIRs) were adjusted for sex, age, time, and comorbidity. The results showed all 33 autoimmune diseases included in the study had higher incidence of PE within the first year of the first hospital admission. Among rheumatologic diseases, particularly polyarteritis nodosa (SIR 13.26, 95% CI; 9.33–18.29), polymyositis/dermatomyositis (SIR 16.44, 95% CI; 11.57–22.69), and SLE (SIR 10.23, 95% CI; 8.31–12.45) were associated with greater risk. The overall risk of VTE in patients with the 33 autoimmune diseases reduced over time from 6.38 (95% CI; 6.19–6.57) during the first year of follow up to 1.53 (95% CI; 1.48–1.57) at 1–5 years follow up, 1.15 (95% CI; 1.11–1.20) at 5–10 years follow up, and 1.04 (95% CI; 1.00–1.07) at ≥10 years follow up. 7
Yusuf et al. conducted a study regarding risk of VTE in four selected autoimmune diseases including RA, SLE, immune thrombocytopenic purpura (ITP) and autoimmune hemolytic anemia (AIHA), using the 2010 National Inpatient Sample of the Healthcare Cost and Utilization Project (HCUP) (sponsored by the Agency for Healthcare Research and Quality) in the United States. The adjusted odds ratio for recorded diagnoses of VTE with hospitalization for the diagnosis of SLE was 1.23 (95% CI; 1.15–1.32) and for RA was 1.17 (95% CI; 1.13–1.21). 6
A summary of the above studies is shown in Table 1. The data consistently show an increased risk of VTE associated with autoimmune diseases, especially related to hospitalization.
Risk of VTE among patients with systemic rheumatologic diseases. *
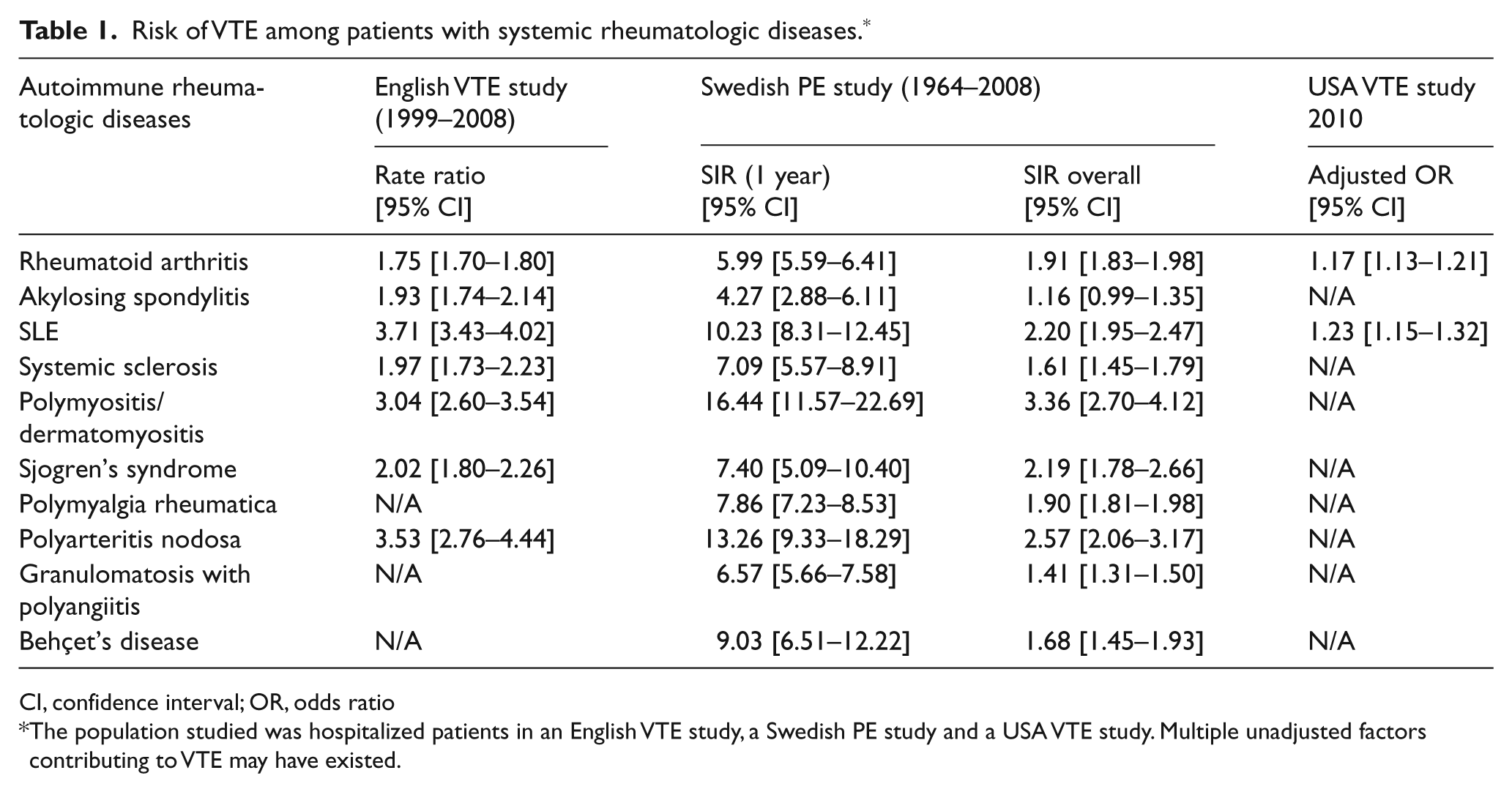
CI, confidence interval; OR, odds ratio
The population studied was hospitalized patients in an English VTE study, a Swedish PE study and a USA VTE study. Multiple unadjusted factors contributing to VTE may have existed.
Mechanisms linking inflammation and venous thrombosis
VTE is a typical multifactorial disease, contributed to by inherited, acquired and environmental risk factors. These factors could contribute to a part of Virchow’s triad, which is classically used to explain pathophysiology of venous thrombosis. The triad (and abnormalities of each) consists of blood flow (stasis), constituents of blood (hypercoagulability), and vessel wall (endothelial injury). 9
Inflammation is a key feature of systemic autoimmune diseases. Many studies have been performed to establish the association between inflammation and the hypercoagulable state or between inflammation and endothelial dysfunction.
Three major natural anticoagulant mechanisms exist including tissue factor pathway inhibitor (TFPI), heparin-anti-thrombin III pathway and the protein C anticoagulant pathway. 10
These mechanisms exert a potent anticoagulant effect under conditions of health. However, inflammation can shift this homeostatic balance to a procoagulant state via many different pathways.
Tissue factor (TF), a membrane-bound glycoprotein, is a trigger of the extrinsic coagulation pathway. It is expressed on cells such as monocytes, endothelial cells, and arterial smooth muscle cells. Exposed TF forms a complex with factor VIIa on a phospholipid surface, and further activates the downstream coagulation pathway. TFPI specifically blocks the complex of factor VIIa and TF (TF-FVIIa). 11
Interleukin 6 (IL-6), an important molecule mediating many inflammatory processes, is reported to increase TF expression on monocytes. This increased expression may be related to C reactive protein (CRP), another consequence of IL-6 action.12,13
The complex of TF-FVIIa activates factor X, which binds to factor Va. The complex of factor Va and factor Xa converts prothrombin to thrombin. Thrombin can bind to thrombomodulin, which activates protein C. This protein C activation is augmented when protein C is coupled with endothelial cell protein C receptor (EPCR). Activated protein C (APC) blocks the coagulation system by inactivating factor Va and factor VIIIa. 14
Inflammation can reduce the expression of anticoagulant factors on the endothelium through thrombomodulin and heparin proteoglycans, and can increase the expression of procoagulant factors through leukocyte adhesion molecules. 15
Thrombomodulin exists on most endothelial cells. Thrombomodulin binds thrombin, which prevents thrombin from effecting clot formation due to overlapping binding sites of thrombin to thrombomodulin and fibrinogen. In in-vitro studies of human and bovine coagulation pathways, tumor necrosis factor alpha (TNF-α), reduces thrombomodulin levels by inhibiting its transcription.16–18
Another inflammatory cytokine, interleukin 1 has also been shown to reduce thrombomodulin activity on endothelial cells.19,20
Another important pathway linking inflammation and coagulation is the fibrinolytic system. Plasminogen is converted to plasmin by tissue-type plasminogen activator (t-PA) or urokinase-type plasminogen activator (u-PA), both of which are regulated by plasminogen activator inhibitor-1 (PAI-1). Plasmin degrades fibrin to soluble fibrin degradation products (FDP). This process is regulated by α2-antiplasminogen. Thrombin activates thrombin-activatable fibrinolysis inhibitor, which inhibits fibrinolysis. 21
In patients with RA and ankylosing spondylitis, TNF-α inhibition reduced PAI-1 and decreased the PAI-1/t-PA ratio. This means that TNF-α is likely involved in inhibition of the fibrinolytic system among patients with chronic rheumatologic conditions. 22
Subtypes of TNF-α receptor (TNFR) may play an important role for thrombogenesis. TNFR1 is expressed ubiquitously but TNFR2 is expressed mostly on immune and endothelial cells. In a study with mice the time to complete thrombotic arterial occlusion followed by vascular wall injury was accelerated with TNFR1-deficient mice but not with TNFR2-deficient or TNFR1/TNFR2-deficient mice when TNF-α was given. This suggests that a TNF-α-induced hypercoagulable state requires TNFR2. 23
The endothelial tyrosine phosphatase SHP-1 may be another important player in vascular hemostasis in TNF-α-induced inflammation. Platelet–endothelium interaction and arterial thrombus formation were enhanced by inhibition of tyrosine phosphatase SHP-1 and this effect is further potentiated with inflammation introduced by TNF-α. This suggests the possible role of SHP-1 as an autoinhibitory feedback mechanism. 24
Inflammation also increases fibrinogen level, which is an acute phase reactant. Elevation in fibrinogen level was reported to be associated with an increased risk of venous thrombosis. 25
Although TNF-α seems to contribute to thrombosis, currently available TNF-α inhibitors may also cause thrombosis. TNF-α inhibitor associated lupus-like syndrome is an emerging disease entity after TNF-α inhibitors have been widely used for autoimmune diseases. It is now known to induce autoantibodies such as antinuclear antibody, anti-double strand DNA antibody and antiphospholipid antibodies. There have been clinical reports of thrombosis associated with TNF-α inhibitor use and antiphospholipid antibodies. 26
Microparticles, small membrane-bound vesicles, can be released from many different types of cell such as platelets, lymphocytes, monocytes, and endothelial cells. Microparticles derived from platelets have been the most investigated regarding thrombosis. They interact with factor Va, factor VIII and factor IXa to play a role in the coagulation cascade. TF and phosphatidylserine are exposed on the outer membranes of microparticles. In systemic autoimmune conditions such as RA, SLE and vasculitis, levels of platelet-derived microparticles were reported to be elevated. 27
A possible role of neutrophil extracellular traps (NETs) is being explored in the pathogenesis of rheumatologic conditions such as SLE. 28 NETs are released during pathogen-induced neutrophil death by a specific pathway referred to as NETosis. 29 NETosis has been implicated in the pathogenesis of anti-neutrophil cytoplasmic antibody (ANCA) associated vasculitis (AAV). Kessenbrock et al. reported robust NET formation in neutrophils incubated with ANCA Immunoglobulin Gs (IgGs). NETs were also induced with a proteinase 3 (PR3)-specific mouse monoclonal antibody. They also found the typical components of NETs containing PR3 and myeloperoxidase (MPO) in kidney biopsy specimens from patients with active AAV where they co-localized with ANCA antigens in cellular crescents. 30
Nakazawa et al. reported that immunization of rats with propylthiouracil-induced NETs resulted in typical features of small vessel vasculitis and production of MPO-ANCA. 31
In an ex-vivo study, NETs have been shown to stimulate platelet binding and aggregation and induce formation of red blood cell rich thrombus. 32
Recently, loss-of-function mutations in the CECR1 (cat eye syndrome chromosome region, candidate1) gene encoding adenosine deaminase 2 (ADA2) have been reported to be associated with inflammatory vasculopathy.33,34 It is unclear whether this new subset of inflammatory vasculitis caused by genetic mutations is associated with venous thrombosis. Another recent exciting discovery related to gene mutations and inflammatory vasculopathy is STING-associated vasclulopathy. 35 The gene mutations in exon 5 of TMEM173, encoding the stimulator of interferon genes (STING) were found in six patients with systemic inflammation, peripheral vascular inflammation and pulmonary manifestations. DVT was not observed in the six patients studied. However, the innate immune system stands at the crossroads of defense against infection, thrombosis, and inflammation, and future research will unveil mechanisms through which thrombosis may be a direct consequence or byproduct of activation of the innate immune system. Table 2 details proposed mechanisms to account for increased VTE risk in PSV.
Proposed mechanisms to account for increased VTE risk in primary systemic vasculitides.
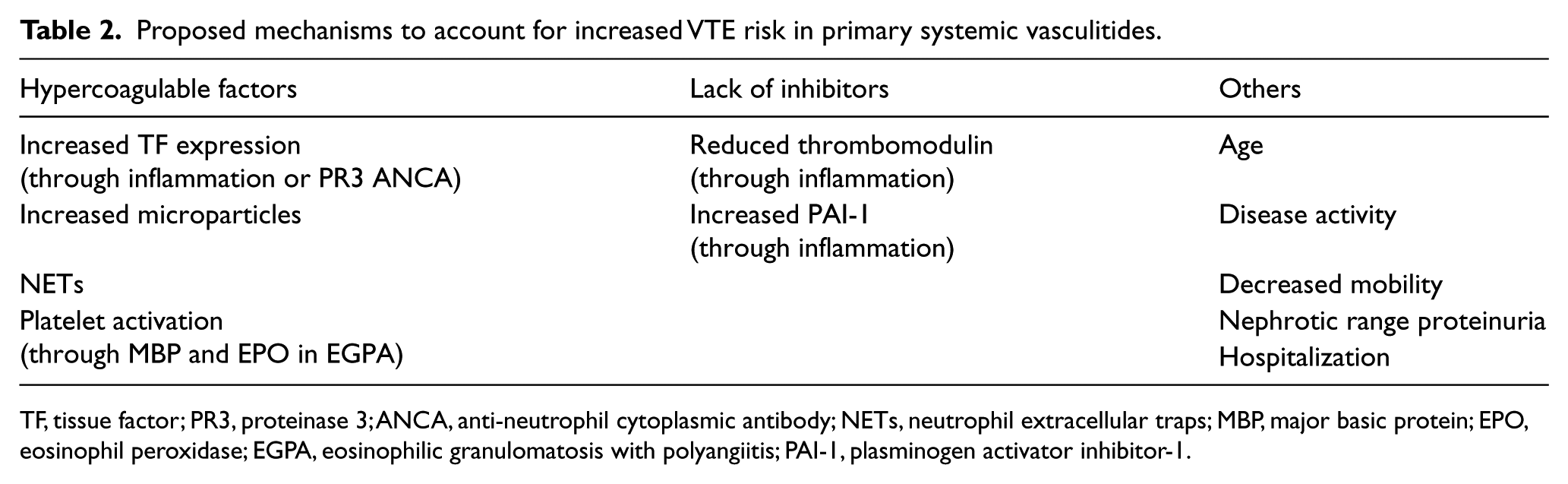
TF, tissue factor; PR3, proteinase 3; ANCA, anti-neutrophil cytoplasmic antibody; NETs, neutrophil extracellular traps; MBP, major basic protein; EPO, eosinophil peroxidase; EGPA, eosinophilic granulomatosis with polyangiitis; PAI-1, plasminogen activator inhibitor-1.
Venous thromboembolism in systemic vasculitides
Primary systemic vasculitides
The association between VTE and PSV has been increasingly described recently. A systemic retrospective study from the French Vasculitis Study Group cohort including 1130 patients with polyarteritis nodosa (PAN), granulomatosis with polyangiitis (GPA, formerly known as Wegener’s granulomatosis), microscopic polyangiitis (MPA) and eosinophilic granulomatosis with polyangiitis (EGPA, formerly known as Churg-Strauss syndrome) revealed that 2.5% of patients with PAN, 8% of those with GPA, 7.6% of those with MPA, and 8.2% of those with EGPA had experienced VTE during a mean follow-up period of 58.4 months. The multivariate analysis showed older age at the diagnosis of vasculitis, male gender, previous VTE, and presentation with stroke with motor deficit were significantly associated with VTE. Motor neuropathy in the lower extremity was associated with a lower risk of VTE. 36
Another study from the Netherlands reported a VTE incidence of 1.8 per 100 person-years, increasing to 6.7 per 100 person-years during active disease defined as two months before and after diagnosis or relapse in patients with AAV, excluding EGPA. There were no significant differences in the prevalence of classic risk factors including immobilization, trauma, major surgery, malignancy, pregnancy, use of oral contraceptives, use of hormone replacement therapy, heart failure, hematological disease, diabetes mellitus, smoking, and obesity between the VTE and non-VTE groups in their cohort. 37
Merkel et al. conducted the Wegener’s clinical occurrence of thrombosis (WeCLOT) study. 38 This study was prospective observational cohort study. Patients were recruited from WEGET, a multicenter, randomized, double-blind, placebo-controlled study to evaluate the efficacy of etanercept in GPA. The study included 180 patients with GPA. Of these, 13 patients had a previous history of VTE. Of the 167 patients without a history of VTE, 16 patients developed VTE during 228 person-years of prospective follow up. The incidence of VTE in patients with GPA was 7.0 per 100 person-years (95% CI: 4.0–11.4). There was no difference in the rate of VTE between the etanercept and placebo groups.
Sebastian et al. reported the frequency of anticardiolipin antibodies and genetic mutations related to hypercoagulability in patients with GPA in the same cohort. 39 Among 180 patients with GPA (29 patients with a history of VTE, the rest without), there were no differences in prevalence of anticardiolipin antibodies (IgG, IgM, and IgA), anti-β2-Glycoprotein I antibodies (IgG and IgM), factor V Leiden mutation, prothrombin gene mutation (G20210) and methylenetetrahydrofolate reductase (MTHFR) gene mutation.
The etiology of higher incidence of VTE in PSV is not well understood to date; however, some studies suggest insights into the etiology. Anti-PR3 antibodies are often present in patients with systemic GPA. In an ex-vivo study, human umbilical vein endothelial cells (HUVECs) stimulated with anti-PR3 antibody resulted in TF generation and activity, and increased TF messenger RNA levels compared to control cells stimulated by endotoxin. Interleukin 1 receptor antagonist inhibited the induced TF messenger RNA expression. 40 In another ex-vivo study, HUVECs were stimulated by MPO, PR3 and elastase. MPO did not lead to increased TF expression but PR3 and elastase increased TF messenger RNA expression and activity. 41
These data may suggest a possible pathological role specific to PR3 in VTE in GPA. Although these vasculitides may demonstrate venulitis at histopathology, inflammation of large veins remains a plausible hypothesis at this time. 42
The increased risk during active disease observed in GPA and MPA also suggests that systemic inflammation plays a role, although the presence of other softer risk factors such as possibly decreased mobility (during severe acute illness) and nephrotic range proteinuria, which have not been rigorously studied in the context of time (since disease onset), cannot be completely excluded. It is also unclear whether immunosuppressive treatment influences the occurrence of VTE. In relation to chemotherapy, the potential risk of venous thromboembolism has been raised in cancer treatment regimens containing cyclophosphamide (CYC).43,44 While the use of CYC may also explain, in part, the more frequent VTE during active disease in GPA and MPA, the increased risk of VTE among patients with AAV was observed even before treatment, and the confounding effect of the fact that CYC is administered to the sickest patients makes this a complicated inference to make from the study.
The prevalence of venous thrombosis in patients with EGPA has been reported to be between 5.8% and 30%. 45 In a retrospective study of 80 patients with EGPA and thrombosis, 57% of patients had only venous thrombosis, of whom 60.9% had DVT and 15.2% had pulmonary thromboembolic disease. 46
In patients with EGPA there may be different factors contributing to VTE. Eosinophilia is a distinct feature in EGPA. Several hypotheses have been proposed to link eosinophilia and thrombosis. Eosinophil specific proteins including major basic protein (MBP), eosinophil peroxidase (EPO), and eosinophil cationic protein (ECP) were implicated as participating in thromboembolic diathesis. MBP and EPO can stimulate platelet activation. 47 These proteins inhibit the capacity of endothelial cells to produce activated protein C. 48 Eosinophils contain TF in their granules, which are translocated to the cell membrane via platelet-activating factor and/or granulocyte macrophage colony stimulating factor. 49
Data regarding VTE are lacking in large vessel vasculitis such as giant cell arteritis (GCA) or Takayasu arteritis. Avina-Zubieta et al. reported increased risk of VTE in patients with GCA at the 2012 American College of Rheumatology annual meeting. 50 The health data of all residents in British Columbia, Canada from 1990–2007 were examined. The incidence rate of PE was 5.2 per 1000 person-years and that of VTE was 5.8 per 1000 person-years. The multivariate relative risk ratio of PE was 3.1 (95% CI: 1.9–5.1) and that of DVT was 2.4 (95% CI: 1.5–3.9). The risks of PE and DVT were higher within the first year of disease onset (10.4, 95% CI: 4.6–23.4 and 7.7 95% CI: 3.6–16.2, respectively).
Headache is a cardinal feature of GCA, often used as a surrogate marker for relapse. Headache in GCA needs to be interpreted cautiously. A case report suggested that scalp vein thrombosis could mimic GCA relapse. 51
Eighty consecutive patients with GCA were examined in Spain to investigate thrombophilic risk factors. There was no correlation between ischemic manifestations and thrombotic risk factors such as antiphospholipid antibodies, protein C deficiency, protein S deficiency, or antithrombin deficiency, factor V Leiden or prothrombin gene G20210A mutation. 52
Only a few case reports have been published regarding Takayasu arteritis with central venous thrombosis or retinal vein thrombosis. It is unclear whether the findings of these studies are coincidental or not.53,54
There is no specific comment on management of venous thrombosis in patients with systemic vasculitis in the American College of Chest Physicians guidelines on management of VTE, published in 2012. 55
Behçet’s disease
Behçet’s disease (BD) is a systemic inflammatory disease characterized by recurrent oral and genital ulcers. It is most prevalent along the Old Silk Route, where ancient merchants or traders traveled between China and the Mediterranean Sea. The etiology of this disease is unknown but vasculitis in BD can affect arteries of all sizes as well as veins. 56 The frequency of vascular involvement in BD in various studies ranges from 1.8% to 51.6%. 57
Venous disease in BD can manifest as deep vein thrombosis, superficial thrombophlebitis, cerebral venous sinus thrombosis, vena cava thrombosis, Budd-Chiari syndrome, or pulmonary artery thrombosis. 57 Venous involvement in BD also varies by the series studied. Of 6500 Iranian patients with BD, 1.1% had vena cava thrombosis. 58 The rate of venous involvement varies between 4% and 38%. 59 Certain combinations of symptoms occur more frequently than others in patients with BD. DVT often co-exists with superficial vein thrombosis. 60
In a study done in Turkey, 74 % of patients with pulmonary artery aneurysm due to BD had venous thrombosis in other locations. Of these patients 88% had lower extremity DVT, 32% had inferior vena cava thrombosis, 16% superior vena cava thrombosis, and 12% dural sinus thrombosis. 61
The pathophysiology of venous thrombosis in patients with BD is unclear. Thrombophilic factors such as protein C level, protein S level, antithrombin III activity, APC resistance, lupus anticoagulant, anti-cardiolipin IgG and IgM antibodies were investigated in one study but none of these were found to be related to thrombotic episodes in patients with BD. 62
A study from Spain compared APC levels in 39 BD patients with 78 age- and gender- matched healthy individuals. Among the 39 patients with BD, 12 patients suffered from VTE. Interestingly, at the time of the study, only two patients were felt to have active disease. APC and thrombomodulin were significantly lower in patients with BD. Specifically, patients with BD and a history of VTE had the lowest APC level. 63
In another study from Turkey, APC resistance level was significantly lower in 116 patients with BD when compared with 70 healthy controls. Thrombophlebitis was present in 56 patients (48.3%) out of 116 patients and 11 patients (9.5%) had DVT. There was no statistically significant association between APC resistance and thrombophlebitis. 64
Eight autopsy cases of BD with large vessel involvement from Japan were reported in 1991. Three cases of venous occlusion were observed including bilateral common iliac veins, bilateral renal veins, superior vena cava, inferior vena cava and hepatic vein. One patient had both arterial involvement (left pulmonary artery aneurysm and bilateral pulmonary artery occlusion) and venous occlusion, whereas two patients only had venous involvement. These venous occlusions were thought to be due to thrombophlebitis. 65
Treatment of venous thrombosis associated with BD is controversial and no prospective randomized controlled trials have been conducted. A small retrospective study from South Korea compared immunosuppression medication alone to immunosuppression with anticoagulation medication in patients with venous thrombosis and BD. Sixteen patients received immunosuppression medication alone and 17 patients received both immunosuppression and anticoagulation medication. There was no statistically significant difference in the rate of relapse of thrombosis. 66
Another retrospective study from France analyzed 807 patients with BD, of whom 296 (36.7%) had experienced venous thrombosis. Anticoagulation medication was administered to 98.6% of patients, 46.8% received immunosuppressive agents and 62.7% received only glucocorticoids. In patients treated with immunosuppressive medications, 69.3% received azathioprine, 34.3% received IV CYC, and 5.8% received methotrexate. Of the patients who received immunosuppressive agents, 20% had relapses of thrombosis, whereas 56% patients who did not receive immunosuppressive medications developed relapses of thrombosis. In multivariate analysis the use of immunosuppressive medications reduced the risk of recurrent thrombosis (Hazard Ratio [HR] 0.27 [95% CI: 0.14–0.52], P<0.0001). Glucocorticoid use also showed a trend toward prevention of recurrence but this did not reach statistical significance (HR 0.71 [95% CI: 0.14–1.07), P=0.099). 67
The European League Against Rheumatism (EULAR) recommendations for management of BD published in 2008 admit no firm evidence to guide management of major vessel disease in BD. Glucocorticoids, azathioprine, CYC or cyclosporine A are recommended for management of acute DVT in BD. They also comment on the absence of controlled data regarding the use of anticoagulants, antiplatelet agents or antifibrinolytic agents in the management of DVT. 68
Anticoagulation in patients with BD could be potentially hazardous given that these patients may have concomitant pulmonary artery aneurysms, the rupture of which may prove fatal. Pulmonary artery aneurysms typically require medical management with or without embolization, coiling or surgical resection. A regimen including pulses of methylprednisolone followed by a combination of high dose prednisone and CYC was used traditionally in Turkey. 69 Cases of successful treatment with TNF-α inhibitor for patients with BD and pulmonary artery aneurysm have been reported.70–72
There is a case report of a patient with BD, DVT and pulmonary artery aneurysm treated with inferior vena cava (IVC) filter, coil embolization for pulmonary artery aneurysm, anticoagulation and immunosuppression. 73 The data for the use of infra vena cava (IVC) filter in patients with BD are very limited. In a retrospective study from Turkey, one patient out of 52 patients with BD and venous thrombosis had IVC filter. Pulmonary embolism did not occur up to 28 months of follow up. 74
Interestingly, the management of major vessel thrombosis could be different depending on where it is managed. Tayer-Shifman et al. reported results of questionnaires, to which rheumatologists in Turkey, Israel and the United States responded. 75 Of American and Israeli rheumatologists, 97% and 87% respectively would anticoagulate for sagittal vein thrombosis and superior vena cava (SVC) syndrome in patients with BD, whereas only 40–44% of Turkish rheumatologists would treat these conditions with anticoagulation. Fewer Turkish rheumatologists would also give life-long anticoagulation for sagittal vein thrombosis and SVC syndrome in patients with BD. 75
Clinical applicability of these data and remaining research gaps
The data on VTE in systemic autoimmune diseases had been scarce in the literature, but in the past few years, multiple studies suggesting an association between systemic autoimmune diseases and VTE have been emerging. There are still no data available in some forms of PSV, but the current available data suggest increased risk of VTE among patients with PSV, especially during the acute phase of active disease and peri-hospitalization period. Multiple factors could contribute to development of VTE, especially during the peri-hospitalization period, and the data shown above may have overestimated the risk of VTE associated with PSV. But there may continue to be a finite risk for VTE many years after hospitalization. Patients with PSV, especially AAV, can have symptoms mimicking VTE such as pedal edema for many different reasons such as high dose glucocorticoid, chronic kidney disease, or medications such as amlodipine, but clinicians should maintain high suspicion of VTE, especially of lower extremity DVT.
The rate of VTE recurrence has not been studied well among patients with PSV. It is unclear whether controlling active vasculitis can lead to normalization of risk for VTE or whether past diagnosis of PSV puts patients at high risk of VTE regardless of disease activity. There are still many questions to be answered. At this time, owing to lack of data, no uniform recommendation can be made regarding duration of anticoagulation. Patients should be assessed individually and a clinical judgment should be made. In patients with BD, the limited data available suggested the benefits of immunosuppressive agents and no benefit of anticoagulation medication. Practice in terms of use of anticoagulation medication in this population varies depending on geographic location as mentioned earlier. Prospective randomized trials are necessary to clarify the benefits of anticoagulation medication in this setting.
Conclusion
Epidemiologic evidence regarding the increased incidence of VTE among patients with systemic autoimmune diseases is accumulating. In particular, large population based studies consistently show elevated incidence of VTE. Similar data for patients with PSV are also slowly emerging to suggest higher incidence of VTE especially during active small vessel vasculitis. The etiology and mechanism of VTE in patients with vasculitis still remain unclear and need to be explored and better understood; however, multiple factors are likely to contribute. The role of immunosuppression in preventing venous thrombosis in patients with vasculitis is unclear at this point, except in patients with Behçet’s disease. Appropriate clinical suspicion and DVT prophylaxis is necessary, especially in hospitalized patients with PSV and patients with AAV with active disease.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
