Abstract
Venous leg ulceration (VLU) is one of the most predominate medical disorders worldwide and in the western hemisphere it affects around 1.5% of the general population and up to 5% of the elderly population. Unfortunately, this trend will only increase given the growth of an aging population worldwide. Understanding its pathophysiology that begins with venous hemodynamic abnormalities and leads to inflammatory alterations with microcirculatory changes is critical to delivering effective curative therapy. As such, the main component to treatment is reversing the underlying venous hypertension and pro-inflammatory milieu using compression treatment along with various adjuvant therapies. Given its impact, a comprehensive multi-pronged approach to care, treatment and prevention is required to reverse the increasing trend that is observed worldwide.
Keywords
Introduction
Chronic venous leg ulceration (VLU) occurs in up to 5% of the population over 65 years of age 1 and in 1.5% of the general population. 2 VLU represents a significant health care burden in the western hemisphere. This trend will only increase given the growth of the aging population worldwide. In the United States, this translates to approximately 0.5–1 million individuals affected by the condition. 2 Moreover, the estimated cost of treating VLU is approximately $3 billion/year. 3 The cost does not take into account the indirect (non-medical) costs such as disability and lost work days, which can reach up to 2 million days per year. 4 Many studies have suggested a higher prevalence of VLU in women, 5 but this disparity decreased with age,6,7 and in fact a more recent cross-sectional study suggested male gender carried a higher risk to develop VLU. 8 Nevertheless, the overall economic and social burden worldwide is huge.
Pathophysiology
Inflammation
Both reflux and obstruction account for the pathophysiology of VLU, with a higher prevalence of reflux in patients presenting with VLU, while obstruction has a higher rate of patients developing VLU and has a much more rapid progression of disease.9 –11 Whether reflux or obstruction is the cause for the patients’ clinical presentation and symptomatology, both conditions lead to increased ambulatory venous pressure. The fundamental pathological process in VLU is inflammation within the venous circulation that is subjected to increased hydrostatic pressure; therefore resulting in increased ambulatory venous pressure 12 (Figure 1). The inflammatory response involves leukocytes, particularly macrophages and monocytes, as well as T-lymphocytes, inflammatory modulators and chemokines, cytokine expression, growth factors, metalloproteinase activity and many regulatory pathways that perpetuate inflammation.13 –16
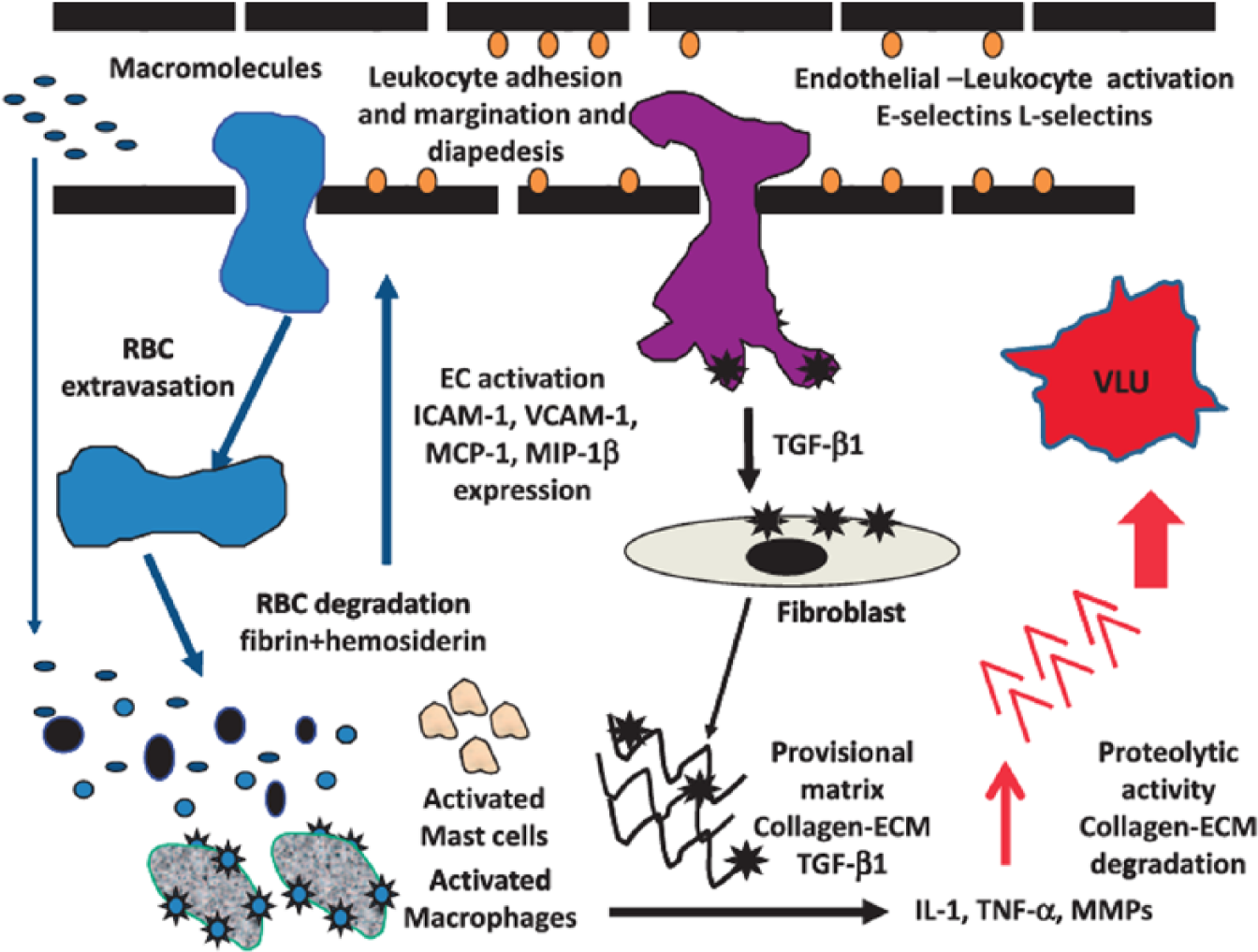
Schematic representation of endothelial cell activation, leukocyte activation and expression of chemoattractants, cytokines, and matrix metalloproteinases (MMPs) leading to the progression of a venous leg ulcer. (Reproduced from Raffetto JD.
Genetic influences
A growing body of knowledge is assessing the genetic variants that are associated with VLU.
13
Further studies have focused on genetic polymorphism in populations with chronic venous disease (CVD) in the development and healing potential of VLU.
17
Cytokines and related chemoattractants (Figure 1)
It is clear from biochemical, immune-histochemical, and functional studies that both the vein wall and the valve are involved in the primary events leading to venous disease. Whether it is the vein wall changes preceding valve insufficiency or the valve insufficiency causing wall distension and wall changes, is a moot point.
13
Importantly, evidence has demonstrated that both the vein wall and the vein valve are pathologically altered to cause primary venous disease. The perturbation in microcirculation is a critical component in the pathophysiology of CVD. The endothelium is a key regulator of vascular tone, hemostasis and coagulation. In CVD, the sine qua non is persistent elevated ambulatory venous pressure. The effect on the microcirculation begins with altered shear stress on the endothelial cells, causing endothelial cells to release vasoactive agents, express E-selectin, inflammatory molecules, chemokines, and prothrombotic precursors.23,24 Mechanical forces, low shear stress, and endothelial-smooth muscle stretch are sensed by the endothelial cells via intercellular adhesion molecule-1 (ICAM-1, CD54) and the mechano-sensitive transient receptor potential vanilloid channels (TRPVs) that are present in the endothelium.24,25 It is well known that patients with CVD have increased expression of ICAM-1, which is expressed on endothelial cells and activates the recruitment of leukocytes and initiates endothelial transmigration, setting up an inflammatory cascade.26,27 Initiating events likely involved altered shear stress and mechanical stress forces on the endothelium and its glycocalyx (glycosaminoglycan on the surface of endothelial cells) with perturbations on nitric oxide production, vasoactive substance release, expression of macrophage chemoattractant protein-1, vascular cell adhesion molecule-1 (VCAM-1, CD-106),
A key component of inflammation in VLU is the expression of MMPs and cytokines, which have significant deleterious effects on the vein wall, venous valve, endothelium, the glycocalyx, and surrounding tissues, including destruction of the dermis with eventual skin changes and ulcer formation. 15 Experimentally in rat inferior vena cava, MMPs can be released as a result of mechanical stretch (and hence during the condition of venous hypertension) and have significant effects on the endothelium, venous smooth muscle, and adventitia. 30 MMPs have been found to be present in high quantities in VLU and in the VLU wound fluid, and correlation of increased expression of MMP activity is associated with poor healing.15,29,31 The regulation of MMP production and function in VLU pathogenesis is not fully understood, but is likely linked to inflammatory cells, cytokines, urokinase-type plasminogen activator (uPA), extracellular MMP inducer CD147 (EMMPRIN), platelet-derived growth factor (PDGF-AA), and the mitogen-activated protein kinase pathway (MAPK).15,32 –41 MMPs exert many of their effects in degrading the extracellular matrix and chronic inflammation within the VLU environment, perpetuating MMP degradation of the provisional matrix and persistent VLU.16,31 Pro-inflammatory cytokines are considered as potent inducers of MMP expression in chronic wounds. In addition, they have been shown to downregulate the expression of tissue inhibitors of MMP (TIMP), thus creating an environment with a relative excess of MMP activity. 42 Unrestrained protease activity is one of the major underlying pathologic mechanisms of non-healing wounds and leads to degradation of provisional matrix. Ulcer formation may be favored by enhanced turnover of the extracellular matrix (ECM) mediated by unrestrained activity of specific MMPs. Collagen turnover and MMP activity are also important elements in VLU healing. In an interesting study, biopsies were taken from 12 patients whose VLU went on to heal; 15 patients whose VLU failed to heal despite 12 months of compression bandaging; and 15 controls. 39 Collagen III N-terminal pro-peptide (PIIINP, pro-peptide extensions at both ends of pro-collagen III and released during synthesis, deposition, and degradation of type III collagen) and degraded collagen (collagen turnover) levels were higher in ulcers that healed compared with ulcers that failed to heal. Collagen type III turnover appeared to be an important biological event that was necessary during the healing of VLU. MMP-1 activity was also higher in healing ulcers than in resistant ulcers and normal skin. Rapidly healing VLU had increased collagen turnover and MMP-1 activity, which differentiated them from those that failed to heal within 12 months. 39
Cytokines have important roles throughout the course of CVD, beginning with inflammation, leukocyte activation, expression of cytokines in the interstitial space, and MMP activation in the VLU wound bed that cause tissue wound degradation. 43 One of the most important cytokines studied in VLU is TGF-β1. The role of TGF-β1 in VLU healing was investigated in a study comprising 80 patients treated with multilayer compression bandaging. In the wound fluid and venous blood, cytokines reflecting inflammation (IL-1β, TNF-α), proteolysis (MMP-2 and MMP-9), angiogenesis (basic fibroblast growth factor (bFGF), vascular endothelial growth factor (VEGF)), and matrix deposition/proliferation/fibrosis (TGF-β1) were measured at 0 and 5 weeks post-compression. Although there was a significant positive correlation between ulcer size and initial concentration of wound fluid bFGF (larger VLU had increased levels of bFGF), ulcer healing only correlated with increased concentrations of TGF-β1 in the VLU wound fluid, indicating the multiple roles of TGF-β1 involved in fibrogenesis, matrix deposition, and proliferation. 34 In a non-randomized trial that evaluated various cytokine levels and VLU healing in patients undergoing compression therapy, it determined that untreated ulcers typically displayed high levels of pro-inflammatory cytokines including several interleukins, TNF-α, and interferon-γ (IFN-γ). After 4 weeks of compression therapy, the levels of pro-inflammatory cytokines decreased significantly and the wounds began to heal. After compression, levels of TGF-β1 increased significantly as the ulcers improved. When specific cytokine levels were related to the percentage of healing, it was found that those with higher levels of pro-inflammatory cytokines, including IL-1 and IFN-γ, healed significantly better than those with lower levels of these cytokines prior to compression. 44 The inflammatory cells identified in the venous microcirculation were macrophages and mast cells. The result was a major abnormality in dermal microcirculation that formed the post-capillary cuff composed of collagen I and III, fibronectin, vitronectin, laminin, tenascin, fibrin, TGF-β1, and α2-macroglobulin.16,45 The change in the post-capillary venule was an adaptation to maintain the structure and integrity in response to increased venous pressure.
Microvalves
Other interesting venous microstructural changes are venous micro-valves, which have been identified in both control specimens and in patients with CVD. 46 The noteworthy aspect was a system of sequentially smaller generations of tributaries leading to a small venous network with competent and incompetent micro-valves. The regions were divided into six generations before reaching the small venous network. In regions where incompetence existed in micro-valves out to the third generation tributary (the boundary), the resin was able to penetrate deeper into the micro-venous networks of the dermis. In limbs with varicose veins and VLU, reflux into the small venous networks and capillary loops were more extensive with more dense networks and greater tortuosity. In addition to superficial axial saphenous vein insufficiency, micro-valve insufficiency also existed. Once it compromised the third generation set of micro-valves, there was a greater risk for the development of dermal VLU. These finding may help explain why some patients with longstanding varicose veins did not develop VLU, since the micro-valves may be intact at the third generation network, preventing clinical deterioration. 46 In addition, these findings may explain why skin changes consistent with venous disease (hyperpigmentation and even small skin ulcerations) were seen clinically in patients with normal duplex ultrasound of the superficial, deep and perforator venous systems.
Further research on the factors responsible for initiating the altered shear stress and stretch on vein walls will make it possible to have specific pharmaceutical targets to restore the integrity of the microcirculation and treat the spectrum of CVD, especially VLU. Understanding both cellular and bio-molecular mechanisms involved in VLU initiation and progression will allow us to identify new targets for possible future pharmacological intervention, which are mainly related to the cellular and bio-molecular aspects of disease. A proactive approach to the treatment of the early and late stages of CVD may be focused on the inflammation-related and MMP-dependent proteolysis. In fact, the inhibition of MMPs may represent a realistic, novel and possible therapeutic intervention to limit the progression of VLU.
Venous disease classification and reporting
Classification of venous disease is important for standardization of venous disease reporting and characterization as well as for assessment of treatment effectiveness. Based on the most recent VLU care guideline published by the Society for Vascular Surgery and the American Venous Forum 47 (SVS-AVF guideline), the Clinical, Etiology, Anatomy, and Pathophysiology classification system (CEAP) (Table 1) is recommended for all patients with VLU for disease characterization. For a patient with active VLU, he/she is characterized as CEAP-6 and, after healing, as CEAP-5. Additionally, the Venous Clinical Severity Score (VCSS) (Table 2) should be used for outcome assessment. It includes 10 clinical descriptors scored from 0 to 3 including pain, varicose veins, venous edema, skin pigmentation, inflammation, induration, number of active ulcers, duration of active ulceration, size of ulcer and compressive therapy use. As the total number increases, disease severity increases as well. Moreover, additional venous disease evaluation should include patient-oriented quality of life (QOL) assessment. 47 QOL assessment has become a standard tool in any disease intervention and natural history study. 48 Surrogate markers such as anatomical and hemodynamic parameters may not necessarily reflect overall patient condition. Both generic tools, such as the Short Form 36-item Health Survey (SF-36), SF-12, SF-6D and EuroQol 5D-5L, as well as disease-specific questionnaires (the Venous Insufficiency Epidemiologic and Economic Study of Quality of Life/Symptoms questionnaire (VEINES-QOL/Sym), the Chronic Venous Insufficiency Questionnaire (CIVIQ), the Aberdeen Varicose Vein Questionnaire (AVVQ), the Specific Quality-of-life and Outcome Response-Venous questionnaire (SQOR-V) and the Charing Cross Venous Ulcer Questionnaire (CXVUQ)), have been validated for patients with venous disease.47,48
Clinical, Etiology, Anatomy and Pathophysiology (CEAP) classification.

Permission obtained from Eklof B, Rutherford RD, Bergan JJ, et al. Revision of the CEAP classification for chronic venous disorders: Consensus statement.
Venous Clinical Severity Score (VCSS).
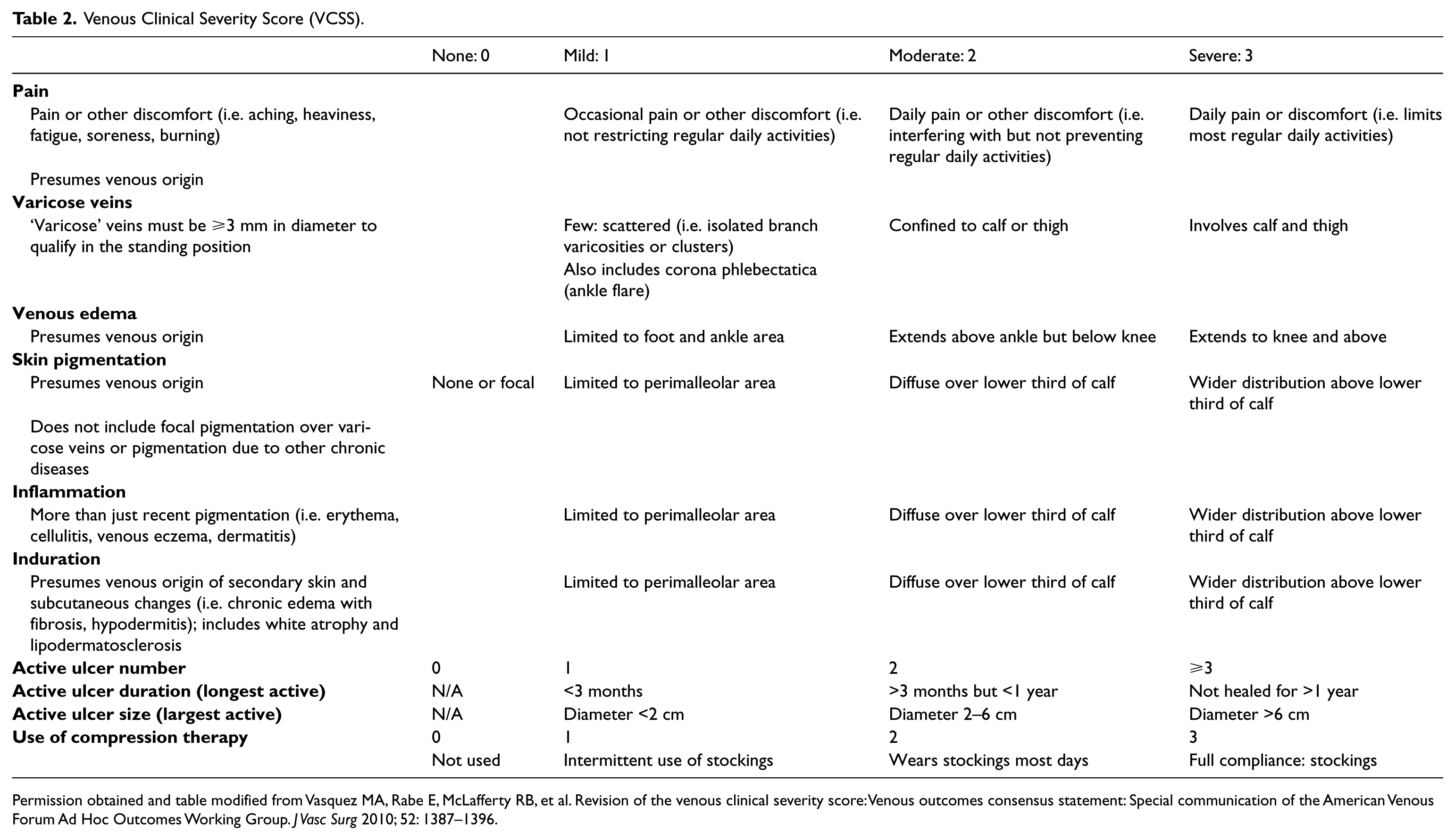
Permission obtained and table modified from Vasquez MA, Rabe E, McLafferty RB, et al. Revision of the venous clinical severity score: Venous outcomes consensus statement: Special communication of the American Venous Forum Ad Hoc Outcomes Working Group.
As for venous procedure outcome assessment, use of recommended reporting standards as well as surrogate markers is crucial. 48 For example, VCSS and individual venous disease-specific questionnaires (VEINES-QOL/Sym, CIVIQ, AVVQ, SQOR-V, and CXVUQ) can be tabulated and the total score may be used to demonstrate the efficacy of any intervention objectively. Reporting standards have been discussed priori, and surrogate makers should include anatomic parameters, venous hemodynamic success, procedure-related complications and impact on VLU healing. 48 Lastly, participation in a venous disease registry can facilitate tracking of procedure outcome.47,49
Treatment (Table 3)
Compression
Compression therapy is the most widely used treatment for VLU and its usage had been documented over 300 years ago 50 (Figure 2(a) and (b)). The mode of action is yet to be clarified but it is postulated that the application of external pressure to the leg and calf raises the interstitial pressure, decreases the hydrostatic venous pressure in both the superficial and deep venous systems, improves the venous return and causes a reduction in venous hypertension, which allows healing to occur. 51 The two main compression therapies are compression bandaging and compression hosiery. However, compression can also be achieved using inflatable boots and other systems. From available published trials, high-grade compression (> 30 mmHg) and multilayer compression bandages were more effective in healing VLU. 52 Inelastic compression garments maintained limb size and venous volume better than graduated compression stockings over time. 53 Based on published data, short stretch bandage provided higher stiffness and interface pressure compared to long stretch bandage. 54 Double short stretch bandage demonstrated higher stiffness and interface pressure than short stretch bandage over short stretch stockings, which in turn provided higher stiffness than double short stretch stockings. 54 Interestingly, a recent study suggested two-layer compression hosiery was just as effective in healing VLU as compared to four-layer bandage. 55 Unfortunately, measuring interface pressure during wound dressing and compression bandage changes has not been routine, yet such information will provide the much-needed objective evidence to treatment efficacy. From the provider’s view, the sine qua non of VLU treatment is compression. Documenting the optimal interface pressure amongst the CVD population with various clinical features will not only improve care but also deliver crucial basic science to the pathophysiological process amongst the disease population.
Venous leg ulceration (VLU) treatment based on the Society for Vascular Surgery and the American Venous Forum (SVS-AVF) guideline. 47

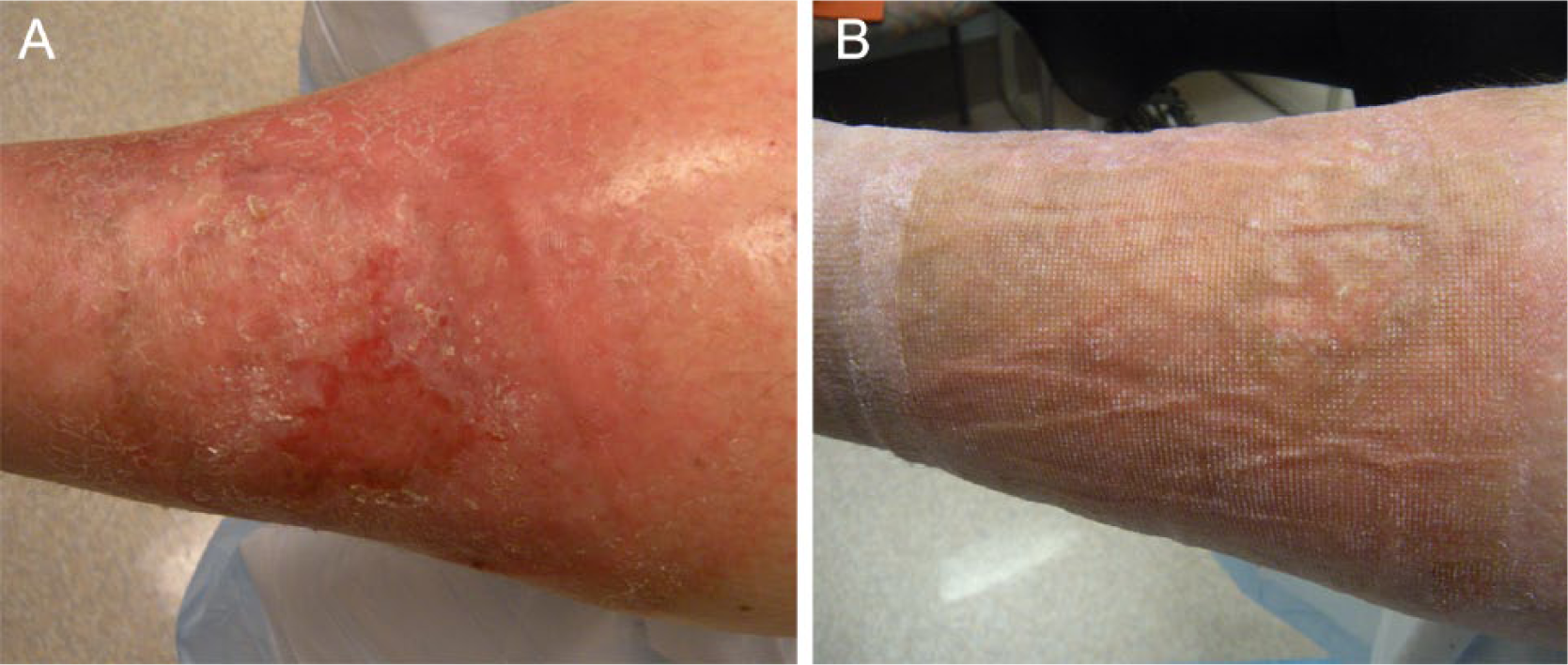
(A) Pre-compression therapy; (B) Post-compression therapy.
Intermittent pneumatic compression has traditionally been used for the prevention of deep venous thrombosis.56 –58 However, studies have been performed on its use in treating VLU.59,60 Compression is achieved by using inflatable, below-knee boots, which produces a pressure gradient between 30 mmHg and 70 mmHg. Its use is advocated for those patients who are unsuitable for compression bandaging or hosiery. 61
Many studies including the Cochrane database demonstrated the beneficial effects of compression therapy on rate of ulcer healing and preventing of recurrence.62,63 Unfortunately, for those who were non-compliant with compression therapy only 55% healed their VLU and the recurrence rate was close to 100% in 36 months. 64 For those who were compliant, the rate of healing was above 90% with a recurrence rate <20% in this meta-analysis. 64
In the past, the focus of research on compression therapy has been on its hemodynamic effects, but molecular changes in the VLU microenvironment are pivotal in the understanding of the pathophysiology and cure. It is well known that VLU is an inflammatory condition that involves a significant number of inflammatory agents in and around the ulcer bed as discussed priori. Beidler et al. demonstrated that compression therapy lowered many of the inflammatory cytokines as outlined in detail in the pathophysiology segment 44 (Figure 3). The underlying message was that inflammatory and anti-inflammatory cytokines were present in VLU and compression therapy was able to modulate their expression and facilitate ulcer healing.
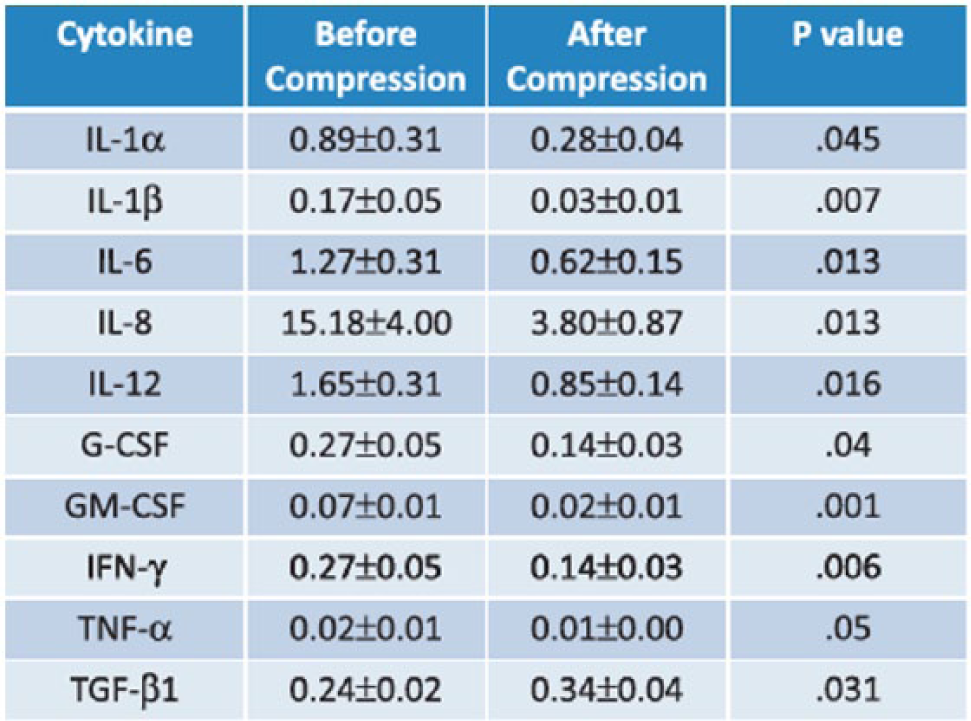
Cytokine profile before and after compression at 4 weeks in 30 limbs with venous insufficiency and active venous leg ulcers. (Reproduced from Raffetto JD.
Overall, using various methods of compression, ulcer healing rates have consistently averaged 60–70% at 12–24 weeks of treatment.16,65 This translates to a significant number of patients who do not heal despite best efforts. Although VLU treatment and care require a multi-modal approach, advances in compression therapy research are much needed. Examples such as interface pressure documentation via pressure measurement devices, development in fabric polymer nanotechnology and defining patient obstacles to compression therapy in VLU treatment are some of the potential areas of research, which may lead to improvement in treatment outcome. As the science of VLU care evolves, the economics to deliver such care is also important. The recent paradigm shift in healthcare delivery in the US from a provider centric to that of consumer-based care, shifting some of the VLU care from the medical establishment to a patient’s comfort zone, such as the home, may provide not only significant healthcare dollar savings but at the same time deliver high-quality care. As a case in point, often patients return two to three times a week to their provider’s office for dressing and compression change. If tools are available through advances in compression fabric, simplified dressing changes, interface pressure monitoring devices and tele-health capabilities, many basic VLU care functions may be performed at home by the patients or caregivers. Patients will not only save gas money and time but the providers will be geared toward more urgent and critical issues rather than routine wound checking.
Wound dressing and debridement
It is important to acknowledge that VLU care requires a multi-level approach. Besides adequate compression therapy, wound dressing is also imperative to the care plan. The optimal VLU wound should have a good granulated matrix bed, free of infection, necrotic tissue and wound exudates to promote healing. Debridement to remove necrotic tissue should be performed during visits. Wound dressings are classified into non-occlusive (simple non-adhering), semi-occlusive/occlusive, and advanced (either growth factor or human dermal equivalent) 66 (Figure 4). A review article by O’Donnell and Balk summarized the essential requirements of wound dressing using a scientific approach. 66 The type of wound dressing employed should depend on the characteristics of the wound. A ‘moist wound environment’ was preferred. Hydrocolloid was recommended for wounds with granulating bases, while in highly exudative wounds, alginate or other highly absorptive primary dressings were preferred. As discussed priori, the significance of inflammation and associated biological markers, such as inter-leukin, interferon, MMPs, etc., were crucial to the VLU pathophysiology and formation. Linking basic science to wound dressing and debridement methods will help to further discern the molecular mechanisms as to how each therapy interferes and counteracts the pro-inflammatory cascade, which may lead to new dressing and debridement approaches to promote timely healing.
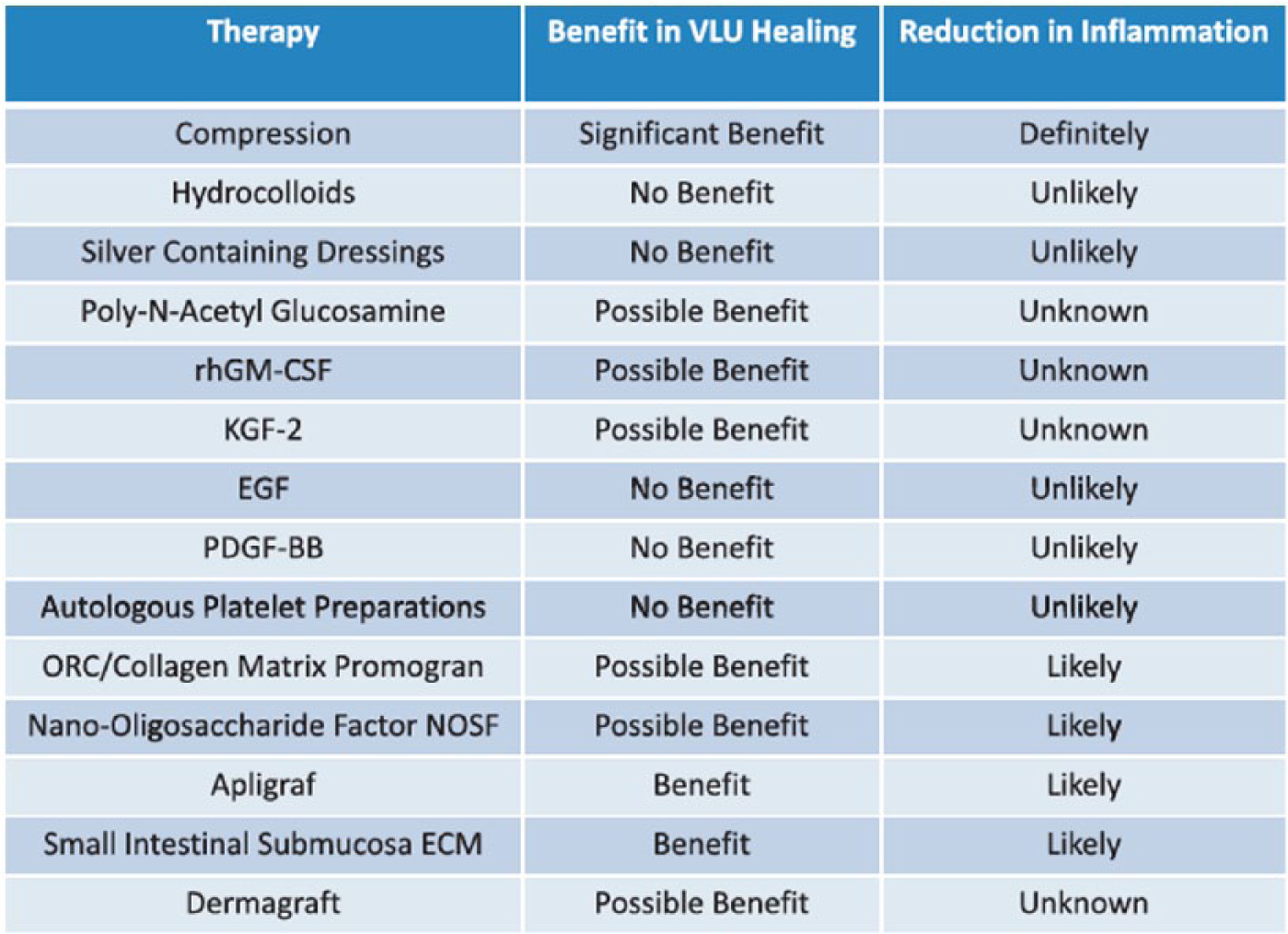
Summary of treatments that may be useful in venous ulcer healing and reduction in inflammation. (Reproduced from Raffetto JD.
Human skin equivalents, biologics, and skin grafts
Adjunctive therapies for VLU healing can be considered after all standard of care measures have been performed (Figure 4). Large VLU that persist or show lack of improvement with a decrease in size over 3–4 weeks of treatment, or that have been present for greater than 6 months, may be considered for adjunctive healing methods such as Apligraf®, Dermagraft®, and OASIS®. Apligraft is able to provide many of the growth factors and cytokines to help stimulate the wound bed provisional matrix. Based on evidence, Apligraf has demonstrated a higher rate of healing compared to those wounds that did not use Apligraf (57% vs 40%,
Dermagraft, which is a human fibroblast-derived dermal replacement, demonstrated efficacy similar to Apligraf. In a recent VLU randomized controlled trial (RCT), 186 patients treated with Dermagraft with compression were compared to 189 patients treated with compression alone. The primary outcome of complete healing at 12 weeks was 34% in the Dermagraft group and 31% in the control group (
An extracellular matrix-based biological intervention such as SIS OASIS has also been proven efficacious in VLU healing. It is derived from porcine small intestine mucosa and provides scaffolding for the fibroblast and keratinocyte to exert wound closure. Based on a controlled trial of 120 patients randomized to either SIS OASIS plus compression or compression alone, the healing rate was 55% versus 34%, respectively (
As the field of biologic innovation in VLU continues to evolve, ECM and human skin equivalent will play an important role, especially in those with persistently hard to heal VLU in addition to compression therapy. These biologics provide scaffold for cell attachment, migration and secretion of growth factors such as FGF, keratinocyte growth factor (KGF), granulocyte-macrophage colony-stimulating factor (GM-CSF), epidermal growth factor (EGF) and TGF, which ultimately facilitate the natural cellular healing function.
Autologous skin grafting, as a primary therapy for VLU, has suffered from a lack of sufficient data due to study bias, small sample size and indirect comparators. A Cochrane review published by Jones and Nelson failed to reach a definitive recommendation on the use of skin grafting in those with VLU. 70
In summary, human and synthetic skin equivalents have proven to be viable adjunctive tools for those hard to heal VLUs. As genetic and molecular factors have been found to play various roles in VLU, specific gene and molecular targets via skin equivalents may achieve a more dominant position in the overall treatment of VLU.
Endovascular and surgical management
Endovascular and surgical management of VLU target two specific outcomes: ulcer healing and prevention of ulcer recurrence. The managements are categorized anatomically as superficial venous disease, perforator venous disease, deep infrainguinal disease and iliocaval disease (Figure 5). Overall, the quality of evidence is mostly limited to level C as a result of a lack of comparative prospective RCTs. The exceptions are the superficial venous treatments and the endovascular management of inferior vena cava and/or iliac vein chronic total occlusion or severe stenosis, which have demonstrated clinical effectiveness and safety in RCTs. Mauck et al. 71 conducted a systematic review and meta-analysis of surgical intervention versus conservative therapy, including seven RCTs and four observational studies showing superiority of surgery to compression therapy for ulcer healing. The result was hampered by the low quality of evidence. As for recurrence, based on available evidence, surgery plus compression demonstrated higher statistical significance in ulcer prevention than compression alone.62,72,73 Multiple RCTs consistently demonstrated that modern open surgery, radio frequency and laser ablation were equivalent in both effectiveness and safety in saphenous vein intervention.74 –78 Moreover, foam sclerotherapy was shown to be an effective saphenous ablative procedure but had a significantly higher recanalization rate on the treated saphenous segments compared to other ablative or surgical methods. 79
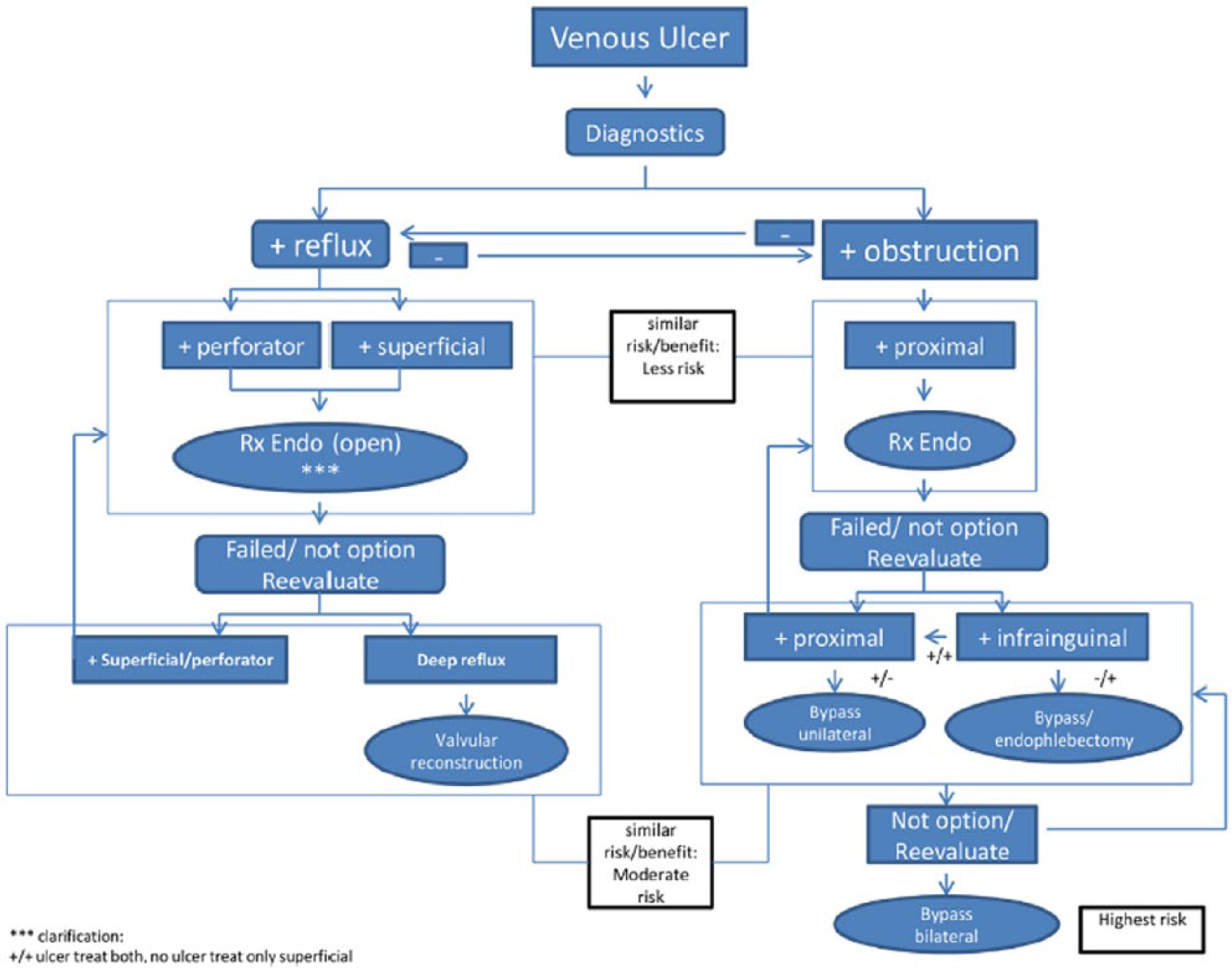
Proposed algorithm for endovenous and operative treatments of patients with venous leg ulceration. (Modified from O’Donnell TF, Passman MA, Marston WA, et al.
Perforator venous reflux demonstrated its role in VLU formation based on the literature. However, the evidence regarding management of perforator abnormality was complicated by combining its treatment with that of superficial venous reflux.80,81 Similar to saphenous vein intervention, the quality of evidence for the surgical management of perforator vein abnormality in VLU healing and prevention of recurrence was mostly limited to level C. However, endovenous methods, including radio frequency and laser ablation as well as foam sclerotherapy of perforator veins, have gained acceptance due to the minimally invasive nature of these interventions. In one large series of endovenous ablation procedures on 110 VLUs in 88 limbs (74 saphenous and 66 perforator veins), there was a significant reduction in ulcer size and expedited healing in those who failed standard of care, including compression therapy. 82 Moreover, endovenous ablation of incompetent perforator and saphenous veins along with compression therapy had proven efficacy to reduce ulcer recurrence in those with a previously healed ulcer (CEAP C5 venous disease). 83
Surgical intervention to reverse a deep system abnormality such as open surgical bypass,84,85 autologous valve substitute,86,87 valve transposition or transplantation88,89 for VLU healing is mostly limited to level C evidence. The exception is endovascular repair of chronic total occlusion or stenosis of the inferior vena cava or iliac veins, which have shown a cumulative patency rate of 90%, with a 60–80% ulcer healing rate.90 –92
In summary, surgical and endovenous intervention play an important role in the overall multi-modality approach to VLU care and management. As evidence has indicated,it alone cannot guarantee the desired outcome of VLU healing. Furthermore, development in molecular and inflammatory markers may shine more light as to how invasive interventions hinder and interject the inflammatory cascade to promote healing. More importantly, venous disease classifications (CEAP and VCSS) and various QOL assessments in addition to surrogate targets are essential to prove the efficacy of these interventions and should serve as the main end points in addition to wound healing for future comparative trials in VLU care.
Biophysical intervention
Biophysical interventions via ultrasound at both high (⩾1 MHz) and low (<1 MHz) frequencies have been shown to enhance VLU healing based on a few non-randomized studies.93,94 Other trials, including a RCT of 337 VLU patients given weekly treatment with high-frequency ultrasound, did not result in an increased healing rate or improved quality of life compared with standard VLU care. 95 Unfortunately, most of the trials related to therapeutic ultrasound for the healing of VLU lacked standardized protocols and relatively few studies focused on VLU alone.
Negative pressure for wound healing has had a positive effect in general but its application specifically to VLU has been lacking, with most studies reporting on mixed wound causes. 96 Other biophysical agents, such as phototherapy, whirlpool, and hyperbaric oxygen therapy were assigned level C evidence for VLU healing by the Association for the Advancement of Wound Care (AAWC) and the Wound Healing Society (WHS).97,98 Overall, the SVS-AVF guideline 47 does not recommend the routine use of the above ancillary measures for VLU healing or prevention of ulcer recurrence.
Unfortunately, despite early enthusiasm from various small non-randomized trials, biophysical interventions have not been accepted as the standard of care in VLU. Many lessons can be learned from such demise, including trial design and appropriate clinical end points. Therapies with minimal or limited potential in reversing or counteracting the basic pathophysiology of VLU should be avoided.
Summary
A multi-pronged and inter-professional approach to VLU care is vital to a successful treatment outcome. Regrettably, the current management of VLU has been hampered by a narrowed focus on only ulcer healing or recurrence rather than on the continuum of care, including public education and prevention. While there are numerous evidence-based guidelines for VLU care, a number of these guideline elements need further study and refinement. Overall, the evidence level for VLU management is mostly moderate strength. This further justifies the need for well-done RCTs on the treatment of VLU. As genetic and molecular mechanisms play more dominant roles in VLU pathophysiology, therapies including compression therapy, wound dressing, human skin equivalents, invasive intervention and other adjunctive modalities can be better studied and understood. In time, these efforts will lead to better cure. At present, available devices to measure interface pressure should become a part of routine VLU management. Tele-health capabilities can further facilitate VLU care and at the same time decrease economic burden, which translate to healthcare dollar savings. Ultimately, it is imperative for specialized societies with an interest in VLU to collaborate and develop consensus documentation to improve and facilitate patient care in the most cost-effective way.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
