Abstract
Lower extremity deep venous thrombosis (DVT) is uncommon in the pediatric population, but it can be associated with severe symptoms and potential long-term morbidity secondary to post-thrombotic syndrome. Inferior vena cava (IVC) atresia can predispose a patient to the development of extremity DVT. There is no clear consensus on optimal management of extensive extremity DVT in pediatric patients, especially in patients with IVC anomalies. We report a case of iliofemoral DVT in a 9-year-old boy with IVC atresia and presumed protein S deficiency that was treated successfully using pharmacomechanical thrombectomy and catheter-directed thrombolysis. He was maintained on long-term anticoagulation and remained symptom free at 6 months’ follow-up.
Background
Lower extremity deep venous thrombosis (DVT) is uncommon in the pediatric population, and the reported incidence is around 0.07 per 10,000. 1 The importance of extremity DVT is increasingly being recognized due to the potential of severe symptoms and risk of post-thrombotic syndrome (PTS) in children and adolescents. 2 Inferior vena cava (IVC) atresia is an uncommon but recognized anatomical venous anomaly that can predispose a patient to the development of lower extremity DVT. 3 Atresia of the IVC can be asymptomatic or diagnosed in patients with extensive iliac and femoral DVT. 3
There is no clear consensus on the optimal management of extensive lower extremity DVT in the pediatric population. In addition to standard anticoagulation, new endovascular techniques including pharmacomechanical thrombectomy (PMT) and catheter-directed thrombolysis (CDT) are increasingly used to treat extensive extremity DVT in both pediatric and adult patients. 4
We report a case of extensive popliteal, femoral and iliac DVT in a 9-year-old boy with IVC atresia that was managed successfully using PMT and CDT.
Case report
A previously healthy 9-year-old African American boy presented to the emergency room with a 2-week history of severe pain and swelling in his left leg. His family history was significant for venous thromboembolism in his mother and grandmother with known protein S deficiency treated with long-term anticoagulation. On examination, his left leg was tender to palpation with significant swelling, but no evidence of neurological deficit was present. The pedal pulses were palpable and there was no evidence of ulceration. Hypercoagulable workup revealed a low functional protein S assay. Other hematological testing including protein C assay and factor V Leiden were otherwise unremarkable.
Duplex ultrasound findings were suggestive of acute left femoral-popliteal and iliofemoral vein DVT with possible thrombus extension into his IVC. A computed tomographic (CT) venogram confirmed the findings of the duplex ultrasound with absence of flow in the IVC, which indicated either thrombosis or congenital atresia (Figure 1). The patient was started on systemic anticoagulation with unfractionated heparin, and the decision was made to proceed with PMT of his left leg DVT due to his severe symptoms.
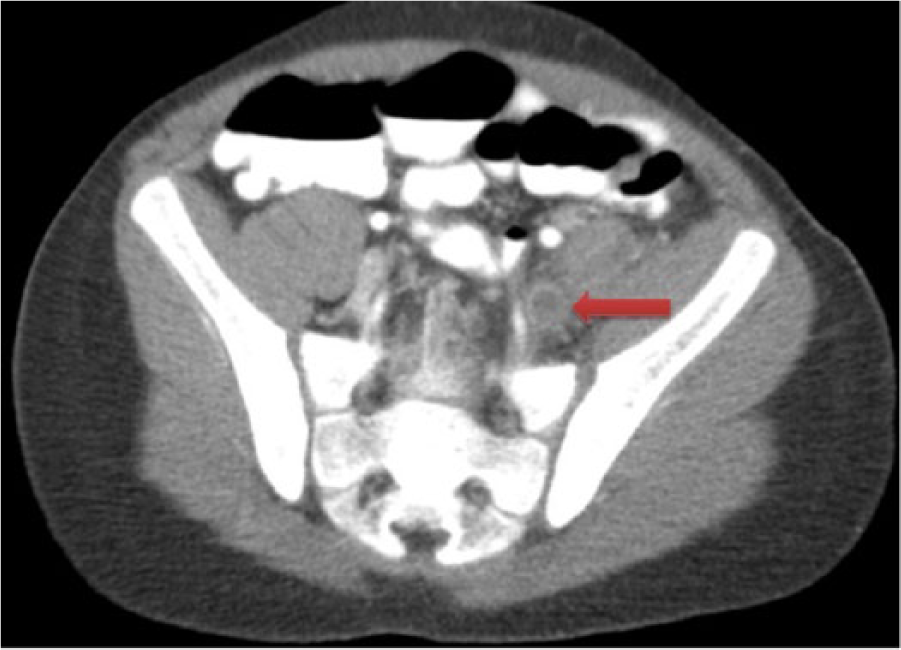
Computed tomographic venogram showed thrombosis in the left iliac vein (arrow).
The patient was placed in the prone position after induction of general endotracheal anesthesia. The left popliteal vein was accessed under ultrasound guidance and a 6-Fr sheath was inserted into the popliteal vein. An intra-operative venogram revealed extensive femoral-popliteal and iliac DVT with possible thrombus extension to the IVC (Figure 2). Pulse spray infusion of tissue plasminogen activator (TPA) and mechanical thrombectomy was performed using an Angiojet® rheolytic thrombectomy catheter (Possis Medical Inc., Minneapolis, MN, USA) after systemic anticoagulation. The stenoses in the femoral and iliac veins were treated with balloon venoplasty using 6 mm and 8 mm balloons. As there was suggestion of residual thrombus in the IVC, the decision was made to proceed with CDT. A 5-Fr multiple side-hole catheter (Unifuse®; AngioDynamics, Queensbury, NY, USA) was inserted from the popliteal vein and advanced up to the iliac vein. TPA was infused at a rate of 0.1 mg/kg and low-dose unfractionated heparin was infused through the access sheath. The status of the patient was monitored closely by clinical examination and laboratory studies including complete blood count, activated partial thromboplasmin time (aPTT) and fibrinogen level in the pediatric intensive care unit.
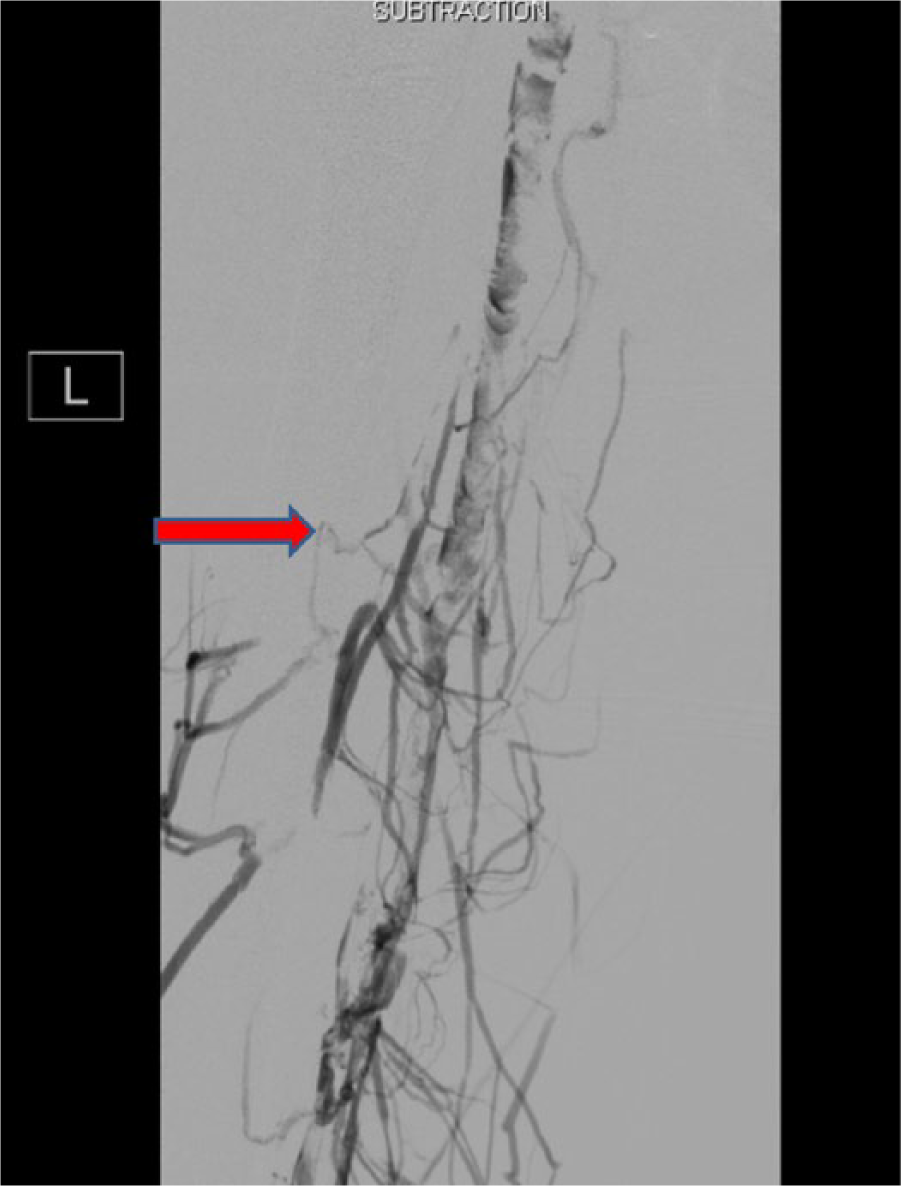
An intra-operative venogram showed thrombosis of the left femoral and iliac veins (arrow).
The patient was brought back to the operating room after 12 hours of thrombolysis, and the post-lysis venogram revealed evidence of IVC atresia (Figure 3). Femoral, popliteal and iliac veins remained patent with no residual thrombus or stenosis. The catheter, wire and sheath were removed and manual pressure was held at the puncture site for hemostasis. He was started on intravenous unfractionated heparin infusion 6 hours after the surgery, and his anticoagulation regimen was subsequently transitioned to oral warfarin with bridging therapy using low molecular weight heparin (LMWH). His left leg pain and swelling improved significantly, and he was discharged home on postoperative day 8 when his anticoagulation was therapeutic with an international normalized ratio (INR) > 2.
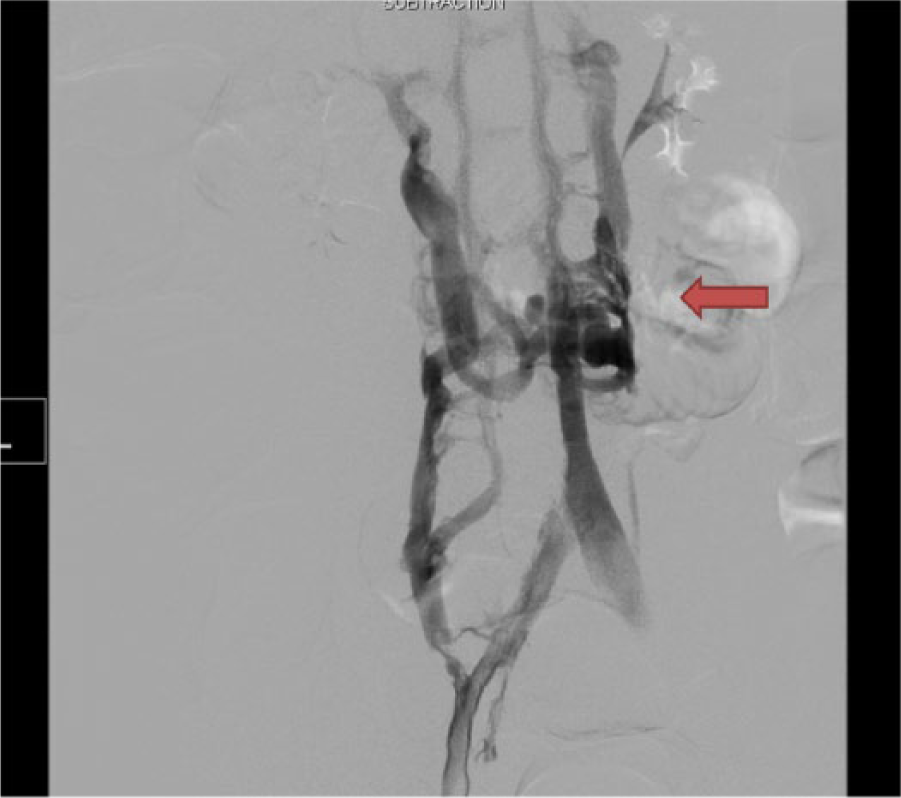
Follow-up venogram after catheter-directed thrombolysis showed a patent left iliac vein and inferior vena cava atresia. There were significant collateral veins present due to IVC atresia (arrow).
The patient remained asymptomatic during his 6 months’ follow-up, and he was maintained on long-term anticoagulation with warfarin. A follow-up duplex ultrasound showed that iliac, femoral and popliteal veins were patent with no evidence of recurrent venous thrombosis.
Discussion
DVT in an extremity is increasingly recognized as a source of long-term morbidity in the pediatric population due to risk of PTS. 2 PTS can develop in up to 20% of children with symptomatic DVT. 2 It is characterized by persistent pain, swelling, discoloration, tenderness, varicosities and ulcers. 5 Important risk factors for PTS are persistent thrombus despite treatment, extent of DVT especially when involving central veins, delay in treatment and recurrence of DVT.2,5 Early treatment initiation and measures to prevent recurrence may minimize the risk of PTS. 2
IVC atresia is a rare but well-recognized anomaly, and it can predispose the patient to the development of lower extremity DVT.3,6 It is controversial as to whether IVC atresia is congenital or caused by early vena cava thrombosis. It can be found incidentally in asymptomatic patients or is occasionally revealed in patients with extensive proximal DVT. 3 The conventional treatment of DVT in patients with IVC anomalies is anticoagulation therapy, but new techniques, including CDT, have been used successfully in these patients.7–9 A recent report by Ganguli et al. has demonstrated the safety and effectiveness of PMT in adult and pediatric patients with acute DVT and IVC anomalies. 9 Their series included a pediatric patient (aged 16 years) and adult patients with an average age of 21 years. 9 There was no recurrence of DVT or PTS during the average 2-year follow-up among the six patients in this series who underwent the procedure successfully. 9
The current standard treatment of extremity DVT in children is anticoagulation. Pediatric patients with thromboembolism should be managed with a multidisciplinary approach involving a neonatologist, pediatrician, pediatric or adult hematologist, and vascular medicine and/or vascular surgeons with special interests in pediatric patients, supported by an experienced pediatric hematologist. Children should undergo extensive hypercoagulable workup to determine the cause of the thromboembolism, and they are usually treated with either continuous unfractionated heparin infusion or LMWH. 10 Unfractionated heparin is titrated to achieve an anti-Xa range of 0.3–0.7 units/ml. For patients treated with LMWH, the therapeutic range should be monitored with an anti-Xa range of 0.5–1.0 units/ml. Children should subsequently be transitioned to outpatient warfarin with an INR goal of 2.5 (range 2–3).
Similar to adults, endovascular techniques including CDT and PMT are increasingly used in the pediatric population to treat extremity DVT.4,7–9,11,12 The largest series was reported by Goldenberg et al., which included 16 adolescent patients who underwent intervention for extremity DVT. 11 Patients underwent PMT and CDT initially, followed by transluminal angioplasty and deployment of a bare-metal stent for residual stenosis. Technical success in this series was 94%. Forty percent of patients developed early recurrent DVT (30 days) and most were treated successfully with re-intervention. Late recurrence was reported in up to 27% of the patients. 11 Kukreja et al. recently reported their experience in 11 very young pediatric patients who underwent endovascular intervention for upper and lower extremity DVT. 12 The most common risk factor in this series was placement of central venous catheters; the procedure being performed to preserve venous access. The techniques used were CDT, PMT and balloon angioplasty. No recurrence of DVT was found on follow-up. 12
The techniques of CDT and PMT in pediatric patients need to be modified according to the patient’s size and available equipment, which is conventionally designed for use in adult patients. The diameter of the target vessels should be measured and the size of instruments carefully chosen. Other potential complications in pediatric patients, including the long-term risks of radiation exposure, renal failure due to contrast nephropathy, 13 hemolysis, hemoglobinuria, cell lysis and a sudden increase in serum uric acid, are rare but important considerations. The optimal dosage for TPA in children for CDT is unknown. Low-dose TPA (0.01–0.06 mg/kg per hour) has been shown to be as effective as a higher dosage (0.1–0.5 mg/kg per hour) in children in some reports for CDT. 4
The patient is maintained on long-term anticoagulation due to a combination of the IVC atresia and the presence of a hypercoagulable state. Other pediatric patients have been similarly treated with chronic anticoagulation by others.7,14 The optimal duration of treatment in these patients is not well studied and the risk of long-term anticoagulation is weighted carefully against the risk of recurrence.
We presented a case of successful percutaneous thrombectomy and thrombolysis in a pediatric patient with underlying IVC atresia. Our patient had extensive spontaneous left femoral-popliteal and iliac DVT with underlying IVC atresia and presumed protein S deficiency. He hadsignificant lower extremity symptoms that were managed successfully using PMT and CDT.
Conclusion
Proximal lower extremity DVT in the pediatric population can lead to significant long-term morbidity. Our case is an example to show that endovascular techniques including PMT and CDT can be used in pediatric patients with proximal DVT and vena cava anomalies for symptom relief and in an attempt to minimize the risk of PTS.
Footnotes
Acknowledgements
The authors would like to thank Mr John Cyrus who provided us with editorial assistance.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
