Abstract
Deep vein thrombosis related to immobilization is a common and extensively studied disorder, particularly in hospitalized patients. However, the phenomenon of iatrogenic venous compression (IAVC) and related deep venous thrombosis (DVT) is under-recognized and under-reported. In the absence of relieving the compressive pathology, the recanalization rates are expected to be very low – thereby putting patients at a significant risk for the development of post-thrombotic syndrome (PTS). In this report, we describe two cases of DVT related to IAVC, and review similar cases that have been previously reported in the literature. With advancements in catheter-based technology, patients with IAVC (with or without DVT) may now be offered advanced endovascular treatment options such as catheter-directed pharmacomechanical thrombolysis (PMT) and percutaneous venoplasty and/or stenting. Hence, timely recognition and treatment is essential in the prevention of disabling PTS or life-threatening pulmonary embolism.
Keywords
Introduction
Venous thromboembolism is the most common cause of cardiovascular morbidity and mortality after coronary artery disease and stroke1,2 with an incidence of 1.0 per 1000 population per year.3,4 In 1856, Virchow 5 postulated three contributive factors to vascular thrombosis (Virchow’s triad): endothelial damage, hypercoagulability and stasis. Since veins are thin-walled and low-pressure structures, they are easily compressed by external forces, making them very susceptible to stasis and thrombosis (Figure 1).
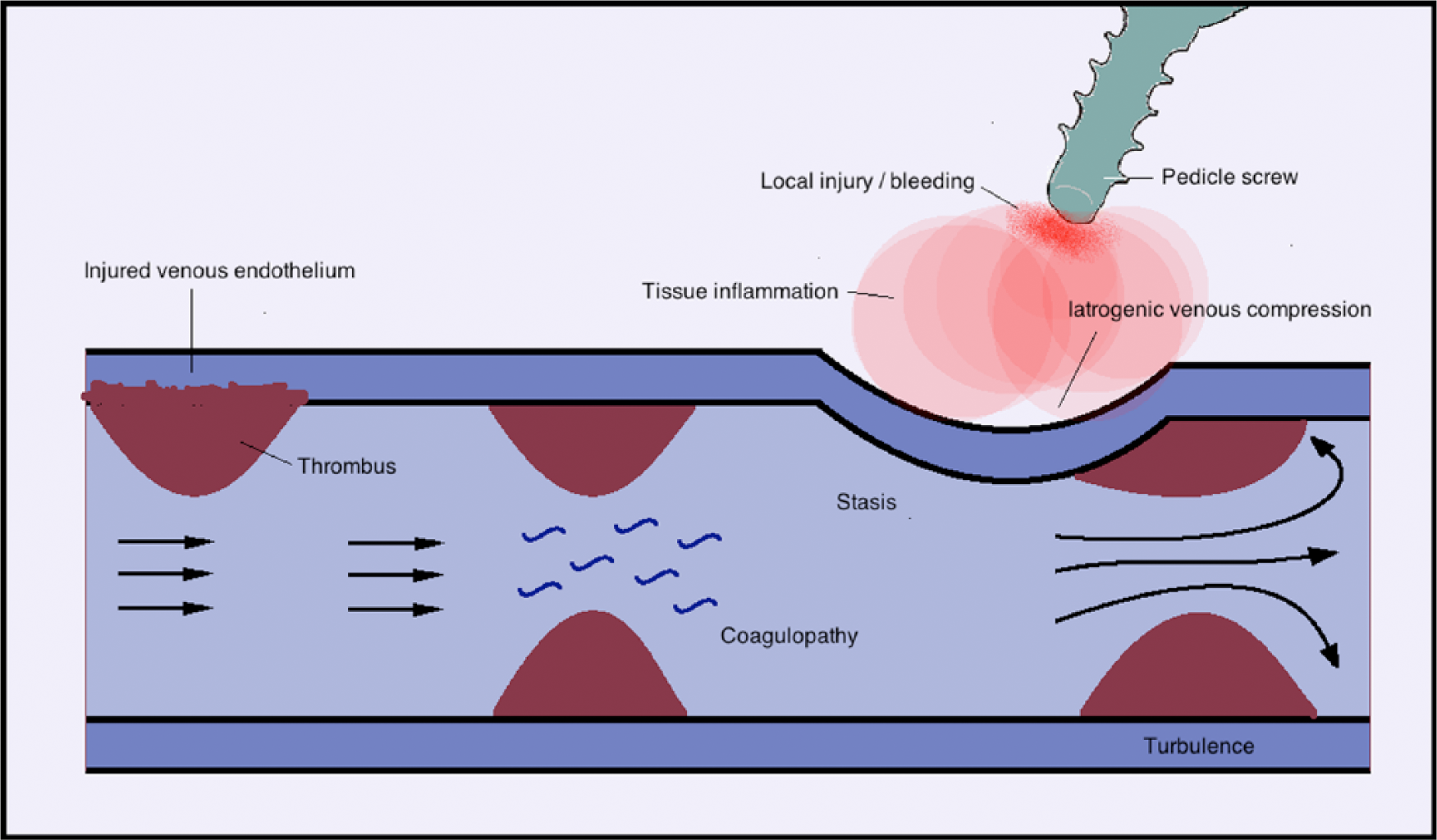
Schematic diagram illustrating the components of Virchow’s triad: endothelial damage, coagulopathy and stasis. This illustrates a possible mechanism by which iatrogenic venous compression could result in stasis and thrombus, even without direct physical contact between iatrogenic hardware and deep veins.
Venous compression syndromes due to abnormal anatomy of adjacent structures have been described in multiple venous beds such as thoracic outlet syndrome, May–Thurner syndrome and Nutcracker syndrome.6–9 In addition, venous compression by enlarging cysts, tumors, or abdominal aortic aneurysms has also been described.10–12 However, iatrogenic causes of venous compression are rarely reported.13–15 In this article, we provide a description of two cases of deep venous thrombosis (DVT) resulting from iatrogenic venous compression (IAVC) and review other such cases previously reported in the literature.
Case 1
A 16-year-old male with congenital scoliosis underwent spinal fusion and decompression laminectomy, complicated by paraplegia. His initial physical examination (PE) showed bilateral lower extremity (LE) paraplegia with hyperreflexia. He exhibited bilateral upgoing Babinski’s sign (BS), consistent with baseline upper motor neuron dysfunction. Prophylactic enoxaparin was held prior to his computer tomographic (CT) myelogram, following which he developed increased left calf and thigh swelling. Duplex ultrasonography revealed DVT extending from the calf veins to the left common femoral (CF) vein. Repeat PE now showed a complete loss of left-sided Babinski’s reflex (flaccid response). This was suggestive of concurrent lower motor neuron (LMN) dysfunction, thought to be secondary to nerve compression from engorged vertebral collaterals. He was initiated on intravenous heparin and he was referred for catheter-directed thrombolysis (CDT).
A left LE venogram via a transpopliteal access showed occlusive thrombosis of the left iliofemoral and popliteal veins. An infusion catheter was placed from the bifurcation of the iliac veins to the left popliteal vein and 24-hour tPa/alteplase infusion was initiated at 1 mg/h. Repeat venography revealed >50% lysis of the iliofemoral DVT, as well as 99% stenosis of the left common iliac (LCI) vein underneath a pedicle screw (Figure 2A). The residual thrombosis was treated with isolated pharmacomechanical thrombolysis (PMT) with a Trellis catheter (Covidien, Mansfield, MA, USA). The LCI vein stenosis was treated with balloon angioplasty and stenting using a self-expanding Wallstent (Boston Scientific, Natick, MA, USA) with an excellent angiographic result (Figures 2B–2E). Within 48 hours, the patient had marked reduction in LE swelling and resolution of LMN dysfunction (he had return of baseline upgoing BS).
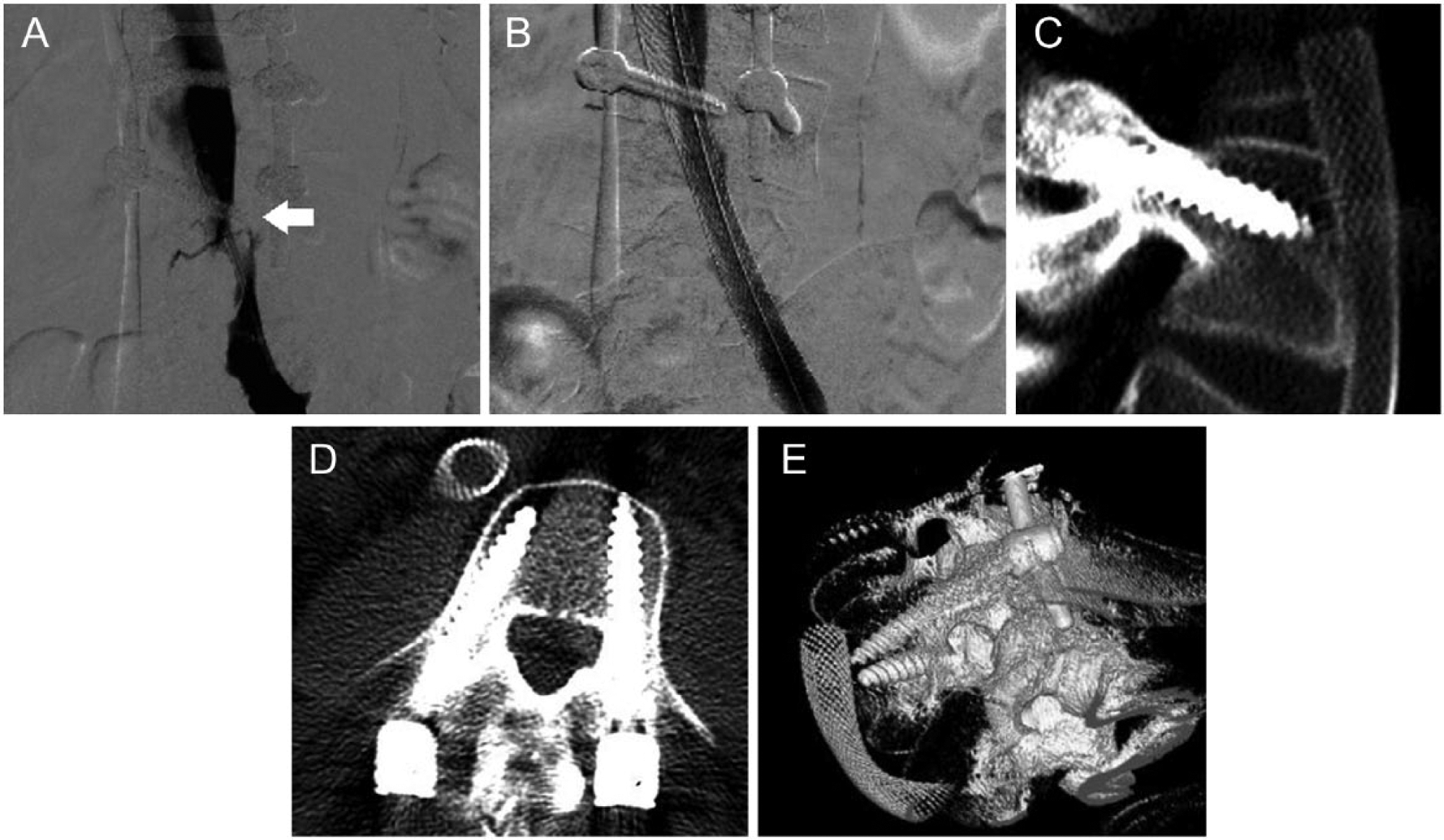
(A) Left iliofemoral venogram showing iatrogenic venous compression from the overlying pedicle screw (arrow). (B) Result following intervention showing resolution of stenosis. (C/D) Post-procedure CT images (thick Multiplanar reconstruction (MPR) 8 mm) demonstrating the spatial relationship of the pedicle screw to the left iliofemoral vein. (E) Post-procedure CT volume rendering.
The patient was discharged on dual anti-platelet therapy (DAPT) and warfarin. At the 1-year follow-up, he had no LE edema or any signs of post-thrombotic syndrome (PTS). A CT venogram abdomen at this time showed patent iliac stents as well as a patent inferior vena cava (IVC).
Case 2
A 53-year-old female with colorectal cancer with metastases to the spine underwent L4 corpectomy (with expandable cage placement) to treat L4 cord compression. In the postoperative course, she developed extensive DVT involving her LCI vein, left external iliac (EI) vein, and IVC. A venogram revealed LCI vein occlusion, with thrombus extending towards the distal IVC. She therefore underwent LCI vein stenting with IVC filter placement within the IVC, above the level of the thrombus. Postoperative imaging showed suboptimal positioning of the expandable cage, and she was therefore referred for revision surgery. In the week following this revision, her left LE edema continued to worsen, despite therapeutic enoxaparin. She was therefore referred for CDT.
Left femoral venography revealed occlusion of the left CF and iliac veins, as well as significant thrombus burden below the level of the IVC filter with an occluded LCI vein stent. Moreover, severe stenosis of the LCI vein was noted within the area of prior LCI vein stenting, coinciding with an overlying spinal pedicle screw (Figure 3A).
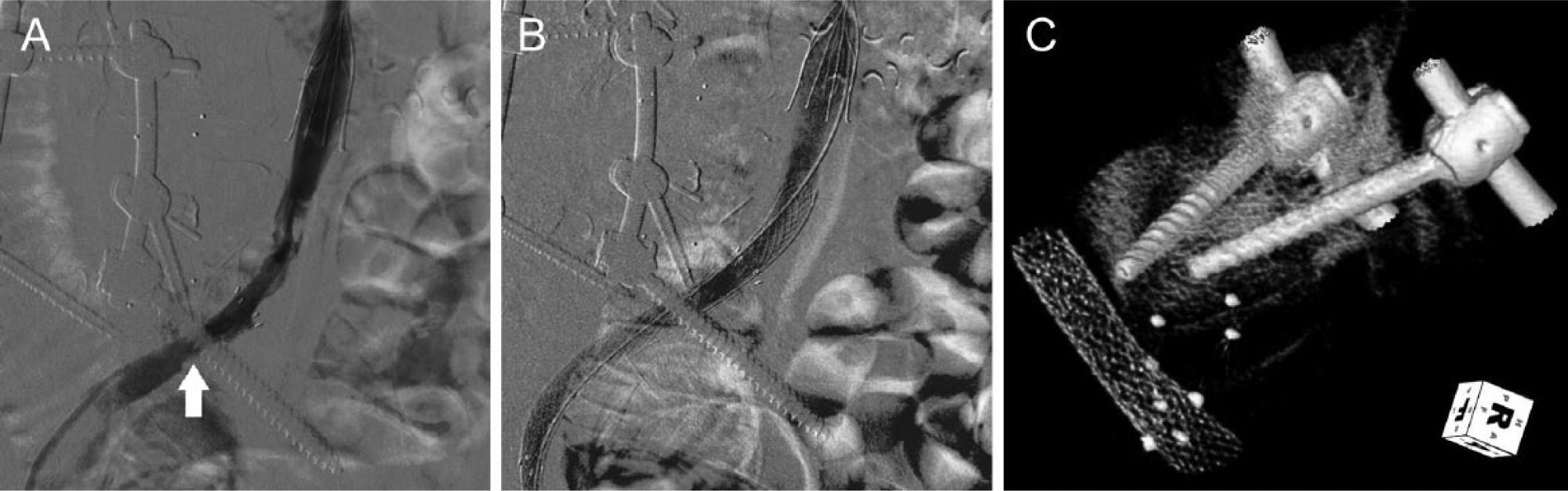
(A) Left iliocaval venogram showing severe stenosis of the left common iliac vein from the overlying pedicle screw (arrow). (B) Venography depicting resolution of stenosis, post-stenting of the stenotic (compressed) left common iliac vein. (C) Post-procedure CT volume rendering demonstrating the spatial relationship of the pedicle screw to the left common iliac vein.
She underwent isolated PMT with a Trellis catheter using 17 mg of tPa/alteplase. Given a large residual clot burden, a Uni*Fuse catheter (AngioDynamics, Latham, NY, USA) was placed across the iliac and caval segments for additional CDT with tPa/alteplase at 1 mg/h. Venography 8 hours later showed 90% clot lysis. Her residual venous stenoses were treated with three stents in the LCI, EI and CF veins (Figures 3B and 3C).
In the days following this procedure, her LE swelling improved significantly. After medical stabilization, she was discharged on therapeutic enoxaparin and DAPT.
Discussion
IAVC is an under-recognized and under-reported cause of DVT and its associated complications such as PTS and pulmonary embolism. Our literature search revealed 17 prior cases of surgical-related IAVC (summarized below).
Of the 17 cases we found, most cases (n=14, 82%) were associated with orthopedic procedures (Table 1). Out of the 14 orthopedic procedures, seven were associated with hip arthroplasty hardware or cement. In these cases, displacement of hardware caused external compression of iliac veins, resulting in acute DVT.12,14–18 The DVT resolved following decompression of the vein in all seven cases. Casts and hardware used for the treatment of scoliosis were implicated in four cases. In three of these cases, the edge of the cast caused venous compression leading to acute DVT of the right lower, left lower, and right upper extremities, respectively. Treatment in these cases ranged from cast removal and anticoagulation, cast removal alone, and anticoagulation alone. In another case, the construction of a pelvic ring led to compression of the left CF vein. Here, the DVT resolved following anticoagulation alone. 19 Two cases of thoracic outlet syndrome were noted following surgical repair of clavicular fractures. One case resolved after surgical decompression (first rib removal and scalenectomy) in combination with anticoagulation. The other case resolved with anticoagulation alone.20,21 In one case, a LCI vein DVT was noted secondary to compression from a migrated bone graft fragment. DVT resolved after removal of the bone fragment, IVC filter placement and anticoagulation. 22
A summary of reported cases of iatrogenic venous compression-related DVT.
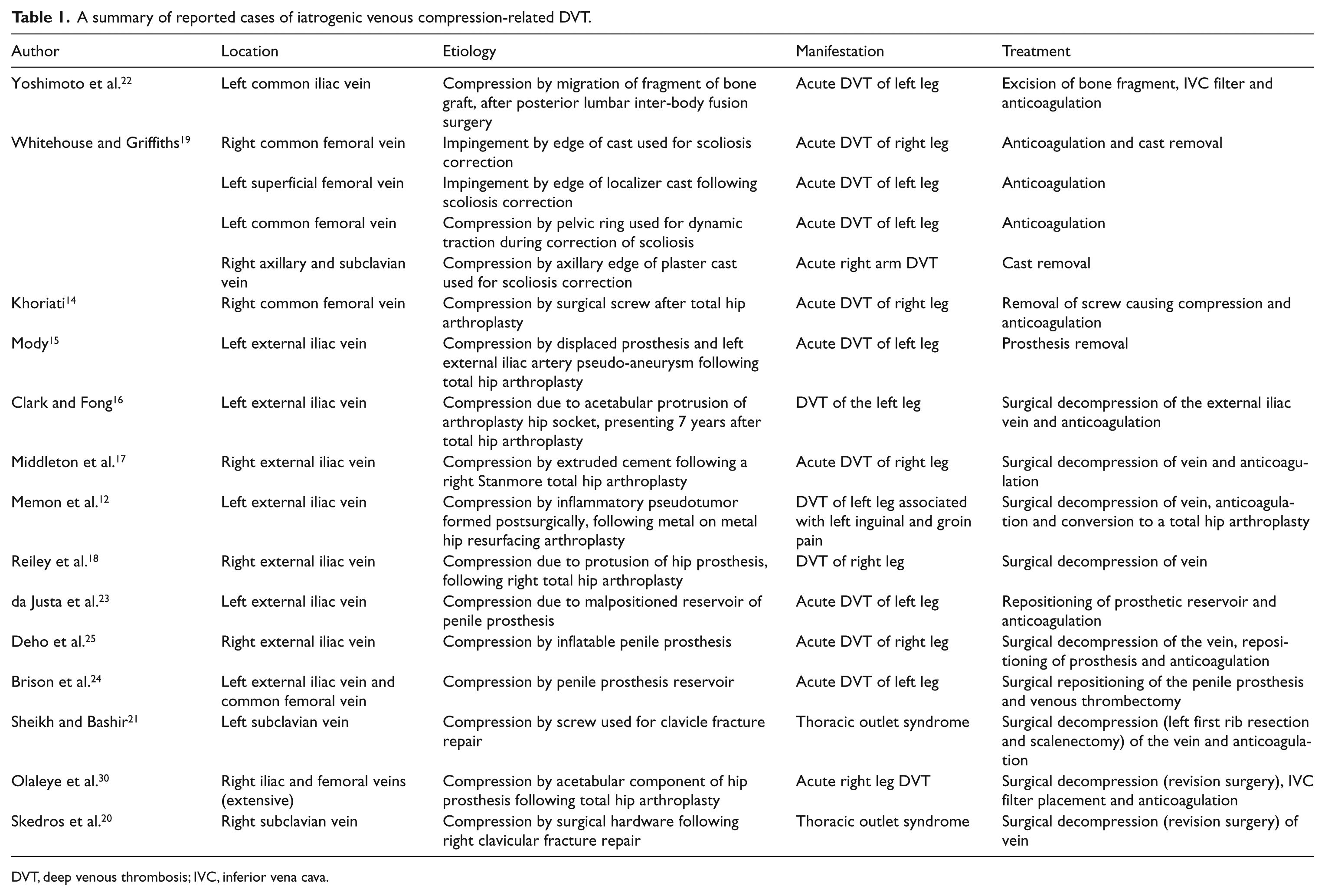
DVT, deep venous thrombosis; IVC, inferior vena cava.
Urological procedures were the second leading cause of IAVC and DVT (n=3, 17%). In three patients, malposition of penile prostheses led to EI vein compression and development of proximal DVT.23–25 All three cases were treated by repositioning of the prosthesis (with subsequent decompression of the vein). In two cases, adjunctive anticoagulation was utilized. The third case underwent venous thrombectomy.
In our ‘Case 1’, the patient underwent spinal fusion and decompressive laminectomy. His postoperative LE edema and neurological changes were thought to be secondary to peripheral nerve compression from vertebral venous collaterals and their engorgement from occlusive acute DVT. The timing of the DVT to the placement of a surgical screw and the angiographic appearance of focal critical stenosis underneath the screw both suggest an iatrogenic compression as an etiology of his DVT. The spatial proximity of the distal tip of the pedicle screw to the course of the vein is demonstrated in Figure 2C. Even though direct venous compression by the screw is not visualized, it is plausible that venous compression could nevertheless easily occur due to localized bleeding and inflammation following iatrogenic perturbation of the vertebral body cortex. After CDT and Wallstent placement, our patient’s LMN function recovered, as evidenced by the return of his upwards BS. Interestingly, his DVT has not recurred since treatment, despite paraplegia.
Similarly, in ‘Case 2’, a patient with metastatic colorectal cancer developed DVT acutely following spinal surgery. A severe stenosis of her LCI vein was seen in association with an overlying spinal pedicle screw. Again, the timing and a focal critical stenosis underneath the pedicle screw both point to an iatrogenic factor in the development of her recurrent DVT (Figure 3C). Once again, symptoms were dramatically reduced following CDT and stenting of the compressed segment.
Major orthopedic and neurosurgical procedures are associated with a high incidence of DVT due to extended immobility and the hypercogulable state in the postoperative period.14,19,22,26 The guidelines regarding the prevention and management of perioperative DVT in these patients are comprehensive. However, the treatment of DVT due to external compression of the veins by the implanted hardware is less well addressed. Anticoagulation is the standard treatment for LE DVT. 27 However, the underlying cause of DVT in venous compression syndrome (external compression of the vein) is not definitively treated with anticoagulation alone. Hence, these patients are potentially at greater risk for developing PTS than those patients without any compression. As seen in Table 1, treatment in most cases is surgical decompression of the involved vein, accompanied sometimes with anticoagulation. In cases where direct surgical decompression is not an option, advanced catheter-based therapies such as CDT, followed by endovenous angioplasty and stenting could be a viable treatment solution to prevent PTS and recurrent DVT in these patients.
CDT aims at achieving thrombus removal by local delivery of thrombolytic agents with or without mechanical fragmentation of the clot. This treatment modality has been shown to reduce the incidence of PTS in patients with proximal DVT. 28 However, residual venous stenosis after thrombus removal is associated with a high incidence of recurrent DVT. 29 In these cases, balloon venoplasty and/or stenting can significantly reduce the rate of recurrent thrombosis. Our patients with residual stenosis from IAVC were free of recurrent DVT at the 1-year follow-up after successful venoplasty and stenting of the compressed vein segments.
Conclusion
The possibility of an iatrogenic complication exists with almost every procedure in medicine. However, unlike most complications, the entity of IAVC leading to DVT is under-recognized and under-reported. In this article, we highlight this entity as an important cause of DVT, particularly in the post-surgical population, where there may be a higher probability of venous compression secondary to implanted hardware. This etiology may be particularly relevant if the occurrence of acute DVT is in anatomic proximity and ipsilateral to newly implanted surgical hardware. With a high clinical suspicion, venography may be considered to confirm the diagnosis.
As seen from our summary of previously reported cases (Table 1), resolution in the majority of the cases (n=15; 88%) required some form of venous decompression (via repositioning, or removal of surgical hardware). For those patients in who direct surgical decompression may not be optimal in the immediate postoperative setting, catheter-based therapies and venous stenting (highlighted above) should be considered. With a high index of suspicion, prompt recognition and timely management of surgical hardware-related IAVC, disabling PTS or life-threatening pulmonary embolism may be prevented in post-surgical patients.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
