Abstract
A retrospective analysis was conducted on the clinical data of 50 patients with acute mixed-type lower extremity deep vein thrombosis treated at the Affiliated People’s Hospital of Ningbo University. All patients underwent catheter-directed thrombolysis via small saphenous vein access. The thrombolytic catheter was inserted into the deep venous thrombus through either infrapopliteal deep veins or the popliteal vein. Continuous infusion of urokinase via a microinfusion pump was administered at a dose of (178.32 ± 79.45) × 104 U for direct clot dissolution. The efficacy was evaluated by comparing the pre-operative and post-operative circumferences of the upper and lower knees, venous patency scores, and thrombolysis rates. Post-operatively, all patients in the group showed significant improvement in symptoms after thrombolysis. Among the 50 patients who were treated, the pre-operative versus post-operative reduction in suprapatellar circumference difference was 74% ± 6% and the pre-operative versus post-operative reduction in infrapatellar circumference difference was 78% ± 6%. The venous patency score significantly improved (P < 0.01), with a thrombolysis rate of 78% ± 6%. No serious complications occurred during the treatment process. This study shows that direct thrombolysis through the saphenous vein catheter for acute mixed-type lower limb deep vein thrombosis is a safe, effective, and feasible treatment method.
Keywords
Introduction
Deep venous thrombosis (DVT) of the lower extremities, a common vascular surgical condition, involves a risk of life-threatening pulmonary embolism (PE) and may lead to severe post-thrombotic syndrome (PTS). 1 Conventional surgical interventions and systemic thrombolysis have demonstrated suboptimal efficacy. Recently developed catheter-directed thrombolysis (CDT) has emerged as a superior therapeutic option with favorable safety and efficacy profiles.2–4 AngioJet mechanical thrombectomy combines aspiration with local thrombolysis to rapidly remove thrombus, reduce the dosage of thrombolytic drugs, lower the risk of bleeding, and shorten the length of hospital stay; it is associated with a higher thrombus clearance rate and a lower incidence of complications than CDT treatment. In addition, the small saphenous vein (SSV), a part of the superficial venous system, typically drains into the popliteal vein at the popliteal fossa and serves as an access route for CDT in lower limb DVT. The SSV approach enables clear positioning of the external malleolus, causes few complications, and can address iliofemoral and lower leg DVT, with high thrombolysis efficiency. However, the tibial vein approach involves a deep vein puncture, which is difficult to locate and is easily damaged; in addition, the thrombolysis range is limited. From July 2019 to June 2024, the Vascular Surgery Department of Ningbo University Affiliated People’s Hospital treated 50 patients with acute lower extremity DVT using trans-SSV CDT, achieving favorable therapeutic outcomes.
Case presentation
General information
From July 2019 to June 2024, 50 patients with acute DVT were treated at our department. Among them, 31 were men and 19 were women; their age ranged from 21 to 79 years (mean age: 50.8 ± 8.6 years). Symptom onset occurred at 48.0–144.0 hours before admission (mean: 96.23 ± 32.48 hours). All cases involved unilateral DVT (40 left-sided and 10 right-sided). The number of Ileo-femoral/Fem-Pop DVT was three. Clinical manifestations included limb swelling, pain, and superficial varicosities consistent with acute venous occlusion. Four patients reported transient dyspnea (Table 1). The specific inclusion and exclusion criteria are as follows.
Demographic and clinical characteristics of the patients.

The inclusion criteria were acute iliofemoral vein/whole limb DVT, thrombus involving the iliofemoral vein or the deep vein of the whole lower limb, and requirement of rapid volume reduction to restore blood flow. In this study, all cases were those of provoked DVTs. The following inclusion criteria were also applied: low risk of bleeding (no recent history of bleeding and normal coagulation function), life expectancy ≥1 year, ability to tolerate thrombolytic drugs combined with mechanical aspiration, unobstructed SSV (diameter ≥3 mm, with no severe stenosis or thrombosis) that can be used as the access vessel, no contraindications to anticoagulation, and ability to tolerate pre-operative anticoagulation and intraoperative thrombolytic drugs (such as urokinase).
The exclusion criteria were as follows: combined presence of tumor thrombus or arteriovenous fistula, severe active bleeding (such as intracranial/gastrointestinal bleeding), expected survival <1 year, severe renal insufficiency (creatinine clearance rate <30 mL/min), light thrombus load (only thrombus in the intermuscular veins of the calf) or chronic fibrosis, poor access vessel conditions (narrowing/thrombus of the SSV), contraindications to anticoagulation (such as pregnancy and recent surgery), hemodynamic instability, and iodine contrast agent allergy. It is noteworthy that anticoagulation contraindications are not strict contraindications; permanent filters, such as the Beltran (Barcelona, Spain) filter, can resolve this issue.
Diagnostic methods
All 50 patients underwent duplex ultrasonography to confirm the diagnosis of DVT, identify the location of the thrombus, and assess its extent. Nine patients required to undergo computed tomography angiography for suspected concomitant PE. Pre-treatment circumference differences (measured 15 cm above and below the knee) were 5.1 ± 0.6 cm (thigh) and 3.6 ± 0.8 cm (calf).
Treatment methods
First, this study adopted a small-incision approach involving the great saphenous vein (GSV), and the key points are as follows. A scalp needle was used to puncture the distal superficial vein of the dorsum of the foot, and iohexol, a nonionic contrast agent, was continuously injected into the patient via a high-pressure syringe. Under fluoroscopic vision, positive and lateral calf, positive and lateral knee, positive thigh, and positive pelvic venous images were continuously taken. The site of thrombosis and the extent of luminal narrowing were determined and recorded (Figure 1(a)). All 50 patients underwent inferior vena cava filter placement; temporary filters (Bard (Murray Hill, New Jersey, USA) filter) were used in 39 cases, and permanent filters (Beltran filter) were used in 11 cases. In 46 cases, filters were placed via the healthy femoral vein, and in four cases, they were placed via the internal jugular vein. In addition, for all 50 patients, the AngioJet (Marlborough, Massachusetts, USA) thrombus aspiration device was used, and another cohort, clinically randomized from the remaining population, will be treated with CDT if clinically indicated, and the access route was thrombolysis by cannulation of the SSV. Under local anesthesia, a longitudinal incision was made at the midpoint of the line between the outer ankle and the Achilles tendon of the affected limb; the affected side of the SSV was located subcutaneously and then punctured. A 4-Fr catheter sheath was inserted, with a small amount of contrast pushed in by hand to observe and select the path. A 0.035-inch guidewire was used in conjunction with a Unifuse thrombolytic catheter (AngioDynamics, Inc., Latham, New York, USA) for gradual upstroke, and the Unifuse catheter was placed within the proximal end of the thrombus, with the tip of the catheter exceeding the thrombus-obstructed segment. The head end of the Unifuse catheter was then closed by placing the Unifuse catheter’s built-in wire. The skin was sutured after linking the T-connector at the caudal end, and the sheath was secured with pressure dressing. Intraoperatively, 100,000 to 200,000 U of urokinase was rapidly pushed as a starting dose (Figure 1(b)). Urokinase was pumped in at a dose of 20,000 to 40,000 U/h through the vascular pump, and the plasma fibrinogen concentration was rechecked every 6–8 hours; the thrombolytic effect was observed on repeat imaging every 24 hours; in order to prevent residual thrombus or secondary thrombus formation around the side-hole-less segment of the thrombolytic catheter, the catheter head was perioperatively retracted and repositioned. If the plasma fibrinogen concentration was <1.00 g/L, thrombolytic therapy was terminated. In this group, the dosage of urokinase was 60–440 (178.32 ± 79.45) million U, and the duration of thrombolysis was 20–132 hours (68.46 ± 46.41 hours). Low-molecular-weight heparin (0.4 mL) was injected subcutaneously every 12 hours, and warfarin was added as an anticoagulant from post-operative day 1; the low-molecular heparin was discontinued at the end of the thrombolysis, and the dosage of warfarin was adjusted for 24 months to maintain the internalized normalized ratio between 2 and 3 and continue the treatment. After the operation, the affected limbs were elevated by 30° and rested in a flat position as they were pre-operatively. Compression stockings were used after the end of thrombolytic therapy and removal of the thrombolytic catheter and catheter sheath, and long-term use of the affected limb was recommended.

Contrast image after thrombolysis of the saphenous vein cannula. (a) Fluoroscopic contrast shows acute mixed thrombosis of the left lower extremity, with the venous thrombosis characterized by segmental filling defects and contrast truncation. In addition, there is obstruction in the distal posterior tibial vein causing venous reflux and collateral circulation above the knee. (b) A filter umbrella is placed at the iliac vein followed by cannulation via the saphenous vein for thrombolysis and (c) if lower extremity DVT is accompanied with iliac vein stenosis, balloon dilatation of the stenotic segment can be performed, which typically resolves the structural obstruction of the iliac vein; further, the abnormal collateral circulation in the pelvis is significantly reduced or disappears. DVT: deep vein thrombosis.
Efficacy evaluation
We observed the improvement in lower limb swelling, soreness, and lung symptoms. We measured the circumference of the large and small legs at 10 cm above and below the knee, respectively, and calculated the difference between the circumference of the affected limb and that of the healthy side. Percentage of swelling reduction in the affected limb = (pre-thrombolysis circumference difference − post-thrombolysis circumference difference)/pre-thrombolysis circumference difference. Referring to the scoring criteria proposed by Zhang et al., 5 Qiu et al., 6 and others, the veins of the lower extremities were divided into the following seven segments: inferior vena cava, common iliac vein, external iliac vein, femoral vein, proximal segment of femoral vein, distal segment of femoral vein, and popliteal vein. Complete patency was scored as 0, partial patency as 1, and complete obstruction as 2. The scores of the seven segments were added to arrive at the total thrombolysis score. Thrombolysis rate = (total score before thrombolysis - total score after thrombolysis)/total score before thrombolysis. 7 Grade I was defined as <50% dissolution, grade II as 50%–90% dissolution, and grade III as complete dissolution. Additionally, we monitored patients for post-operative complications, including hemorrhage, fever, PE, and subcutaneous ecchymosis; we also measured fibrinogen (FIB) and D-dimer levels at 3, 6, and 12 months post-operatively to assess the risk of post-operative thrombus formation.
Statistical analyses
Data were analyzed using the Statistical Package for Social Sciences software, version 26.0. Pre/post-treatment circumference differences are expressed as mean ± SD values and were compared using paired t-test. Venous patency scores were analyzed using the Wilcoxon signed-rank test. P < 0.05 was considered statistically significant.
Outcomes and follow-up
All patients who underwent AngioJet thrombus aspiration showed significant improvement. Post-therapy circumference differences (thigh: P < 0.01; calf: P < 0.01) and reduction percentages (thigh: 74% ± 6%; calf: 75% ± 6%) demonstrated marked efficacy (Table 2). All 50 patients underwent contrast examinations before and after AngioJet thrombus aspiration and were assigned scores according to the contrast results; the mean venous patency score before thrombolysis by cannulation of the SSV was 7.8 ± 1.4, while the score after thrombolysis was 1.9 ± 0.6, representing a significant difference (P < 0.01). The average rate of thrombolysis was 78% ± 6% (Table 3). In the present study, 35 cases of left common iliac vein stenosis (May-Thurner syndrome) with pelvic collateral circulation were identified using venography after CDT, and 28 cases of ipsilateral femoral vein puncture were treated with balloon dilatation and molded stent placement. The balloon diameter was 8–12 mm, and the stent was pre-expanded. The diameter of the stent was 10–14 mm, and its length was 10–12 cm. After the treatment, the lumen of the iliac vein was smooth, the stenosis was eliminated, and the pelvic compensatory collateral branches were significantly reduced or had disappeared (Figure 1(c)). The AngioJet group required significantly less urokinase (P < 0.05), indicating reduced dependence on thrombolytic agents. There was no statistically significant difference in the thrombolysis time between the two groups. The AngioJet group exhibited shorter hospitalization duration (P < 0.05), consistent with faster clinical recovery (Table 4). The incidence of post-operative complications was not significantly different (P < 0.05) between the AngioJet and CDT groups (Table 5). The AngioJet group exhibited significantly lower FIB levels than the CDT group (P < 0.05). With respect to D-dimer levels, differences between the groups were highly significant (P < 0.001), indicating that the AngioJet group demonstrated superior efficacy in reducing D-dimer levels. Collectively, these findings suggest that AngioJet therapy outperforms CDT in improving both FIB and D-dimer profiles, with the improvements becoming increasingly pronounced over time, an observation that hints at its potential to accelerate patient recovery through a more effective thrombus clearance mechanism (Table 6).
Difference in the perimeter diameter before and after thrombolysis in 50 patients.

Venous patency score before and after surgery in 50 patients.

Comparison of thrombolysis time, urokinase dosage, and hospital stay between the two groups of patients.

CDT: catheter-directed thrombolysis.
Comparison of post-operative complications between the two groups of patients.
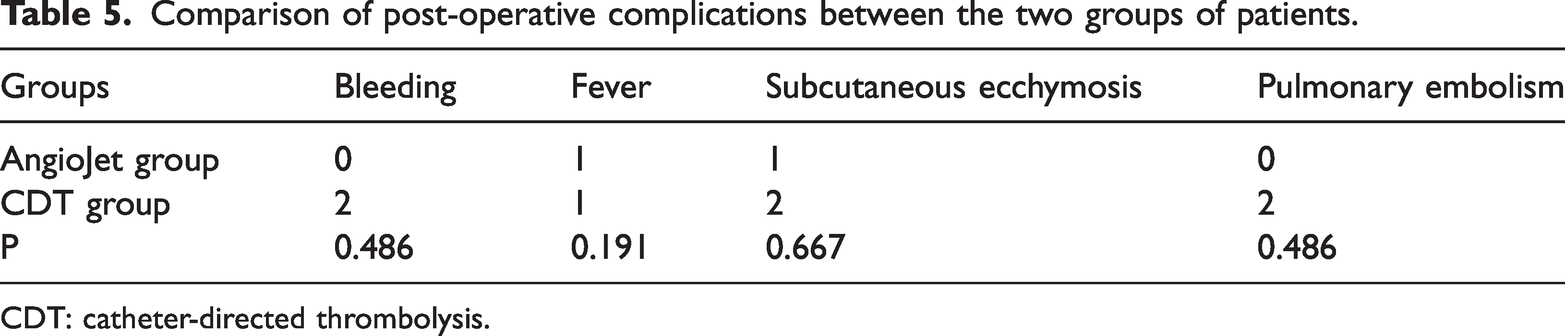
CDT: catheter-directed thrombolysis.
Comparison of post-operative follow-up results of FIB and D-dimer between the two groups of patients.

CDT: catheter-directed thrombolysis; FIB: fibrinogen.
Discussion
CDT enhances thrombolytic efficacy and speed through a targeted approach. Direct positioning of the thrombolytic catheter within the thrombus enables high-concentration thrombolytic agents to fully engage with the clot, accelerating dissolution. This method also prioritizes the preservation of deep venous valve function, enhancement of collateral vessel patency, and augmentation of venous return. Furthermore, by confining thrombolytic action to the local thrombus, CDT minimizes systemic drug exposure, thereby reducing the risk of bleeding complications. These combined advantages of superior clot resolution, valve protection, improved venous hemodynamics, and lower hemorrhagic risk have established CDT as the primary therapeutic choice for acute lower extremity DVT.8,9
The feasibility and advantages of SSV puncture and cannulation thrombolysis for acute mixed DVT are evident in specific clinical scenarios. When thrombus involvement extends to the popliteal vein and distal deep venous segments, the absence of effective blood flow in the femoropopliteal venous system and ambiguous ultrasonic localization increase the risk of adjacent tissue injury during surgical manipulation. Even in cases where successful puncture is achieved, the combined constraints of sheath length and the side-hole design of the thrombolytic catheter may prevent adequate advancement into the popliteal vein, limiting its ability to dissolve the thrombus within this critical segment. To solve this problem, Shan Haifeng et al. reported the success of direct thrombolysis by superficial calf vein puncture, selective cannulation through the superficial deep calf vein traffic branch, and placement of the thrombolytic catheter into the anterior tibial or posterior tibial vein and upstream in the deep vein. 10 In view of the anatomical characteristics of the SSV, the beginning of its external ankle segment is fixed; 70% of it converges to the popliteal vein near the popliteal fossa, 10% converges to the deep femoral vein, and 96.8% of the calf segments and deep veins have 1–3 traffic branches within 0–30 cm above the inter-ankle line. 11 In recent years, thrombolytic therapy via SSV access is being reported in China, although conventional approaches face limitations due to the narrow lumen of the SSV at the medial ankle, often requiring upward dissection to identify a thicker venous trunk for puncture. 12 To address this, our study refined the technique as follows: a 2-cm incision was made at the ankle to expose the proximal SSV, through which a sheath was placed for catheterization. Under digital subtraction angiography guidance, the thrombolytic catheter was then selectively advanced into the anterior or posterior tibial vein via the communicating branches in the lower leg or directly into the popliteal vein along the SSV. If successful, this method could dissolve the thrombus in the distal lower leg deep veins and their proximal segments; if the catheter fails to enter the deep veins via the communicating branches but reaches the popliteal vein through the SSV, it can still effectively lyse thrombus in the popliteal vein and its proximal segments, reflecting outcomes that are unachievable using conventional puncture methods targeting the popliteal or femoral veins. This improved approach not only addresses thrombi in the iliofemoral veins but also lyses most popliteal venous clots and portions of calf DVT. Thus, the advantages of SSV puncture thrombolysis include a simple surgical procedure; a fixed, anatomically distinct proximal SSV origin at the lateral ankle (unaffected by limb swelling); direct exposure of the SSV during incision to avoid vascular injury and bleeding complications common in “blind puncture” approaches; and distal catheter insertion into the deep veins of the affected limb, enabling thrombolysis of the popliteal vein, and even proximal calf deep veins to enhance overall thrombolytic efficiency. Additionally, downstream catheter placement minimizes guidewire and catheter-induced venous wall damage, preserving deep venous valve function. Higher the thrombolysis efficiency, shorter the thrombolysis time and lower the mechanical damage to the vein wall and valves. 13 During catheterization and indwelling thrombolysis, patients do not require to be in the prone position or follow specific postural restrictions, allowing unrestricted movement, an advantage particularly beneficial for individuals with obesity, severe affected limb edema, or limited mobility (e.g. elderly or frail patients); this enhances patient comfort during treatment. Collectively, these benefits position the trans-saphenous venous approach as an optimal thrombolytic strategy for lower extremity DVT. For cases involving occlusion or luminal narrowing of the infrapatellar anterior tibial, peroneal, or posterior tibial veins, the SSV is superficially located with a large lumen, facilitating clinical puncture and catheter placement for thrombolysis. Additionally, this approach minimizes the risk of accidental injury to adjacent veins during deep venous puncture procedures.
This study demonstrated significant reduction in post-operative circumference differences among patients, validating the feasibility of the “antegrade catheterization combined with low-access approach” technique. The SSV access enables coverage of the popliteal veins and even calf deep vein thrombi, achieving broader thrombolytic coverage. Anatomical advantages allow AngioJet thrombectomy to reach distal thrombus regions that conventional CDT struggles to access. Improvements in venous patency scores align with the discussed “localized high-drug-concentration contact” theory. Urokinase acts directly on thrombus interiors, overcoming the conventional systemic thrombolysis drug concentration limitations while reducing the bleeding risk at FIB thresholds. This technical refinement employs a lateral malleolar incision to expose the origin of the SSV, effectively addressing prior constraints while simultaneously circumventing blind puncture injuries and overcoming access challenges posed by the narrow diameter segments of the distal SSV.
Limitations of SSV puncture catheterization for thrombolysis in acute mixed DVT include the following. In cases of acute DVT with significant lower extremity swelling, increased exudation often occurs following skin incision, and meticulous dissection of the SSV is required due to its delicate nature. When the SSV converges with the GSV at a highly variable junction, 14 careful pre-procedural imaging assessment is essential to avoid guidewire/catheter perforation of the venous wall due to accidental trauma. Additionally, in patients with shorter stature, exposing the initial segment of the SSV may reveal that inserting a 4-Fr sheath is challenging, rendering interventional therapy unfeasible. In such scenarios, dorsalis pedis venography under roadmap guidance is employed to access the SSV or higher-level calf deep veins, allowing continuation of the interventional procedure. After the insertion of a 4-Fr sheath, difficulty in finding the thicker traffic branch under the roadmap or fluoroscopic view prevents the guidewire from advancing into the popliteal vein. This may indicate that the 4-Fr sheath has been inserted too deeply. If the 4-Fr single-curve catheter fails to advance under 0.035-inch exchange guidewire guidance at the popliteal fossa, it should be exchanged for a super-smooth, super-rigid guidewire to facilitate smooth passage of the 4-Fr single-curve catheter through the popliteal vein, potentially reaching the iliac vein, followed by the replacement of the 4-Fr thrombolytic catheter by an appropriate perfusion segment. If all these methods fail, the 4-Fr catheter can be replaced with a 4-Fr thrombolytic catheter via dorsalis pedis venography under popliteal vein guidance. Successful placement of the thrombolytic catheter through dorsalis pedis venography or ultrasound-guided popliteal vein puncture helps in opening the upper segments of the femoral, popliteal, and iliac veins, which aid in reducing limb swelling, limiting the progression of femoral cyanosis or leukoaraiosis, and mitigating post-thrombotic sequelae. During thrombolysis, dynamic monitoring of plasma FIB concentration (every 6–8 hours) and thrombolytic catheter angiography (at 12–24 hours) are performed to evaluate the efficacy, with external adjustment of the perfusion segment positioned within the thrombus. 15 Thrombolysis is terminated if angiography confirms satisfactory dissolution and deep venous patency, the plasma FIB level falls below 0.8 g/L, or serial angiograms show no progression. In this cohort, the urokinase dosage ranged from 60 to 440 (mean ± SD: 178.3 ± 79.5) million U, with thrombolysis duration spanning 20–132 hours (mean ± SD: 68.46 ± 46.41 hours).
This analysis underscores that direct thrombolytic therapy via selective cannulation of the SSV for acute mixed lower extremity DVT not only retains the core advantages of conventional CDT but also extends thrombolytic action to the popliteal vein and portions of calf deep venous thrombi. By delivering sustained thrombolytic exposure to the clot, this approach enhances thrombolytic efficacy, supports valvular function preservation, rapidly alleviates acute symptoms, and mitigates the PTS risk. It further distinguishes itself through minimally invasive characteristics, rapid recovery trajectories, and high therapeutic efficacy. Notably, the procedure imposes no specific positional limitations on the patient during treatment, exhibiting strong clinical adaptability. Collectively, these attributes position it as a clinically promising intervention worthy of broader exploration. This study has certain limitations with respect to the follow-up duration, and further research is needed to corroborate these findings.
Conclusions
This study shows that thrombolytic therapy via SSV catheterization effectively treats acute mixed lower extremity DVT. Its targeted drug delivery enhances precision, achieving greater reduction in systemic bleeding compared with that using conventional thrombolysis. Early use rapidly resolves edema, restores venous flow, and halts clot progression. By accessing deep veins through the superficial SSV, this technique avoids the challenges associated with deep trunk cannulation and offers a novel, minimally invasive approach, especially beneficial for those with complex vascular anomalies. Combining simplicity with targeted therapy, it sets a new standard for safer, more effective DVT management.
Footnotes
Acknowledgments
We thank the Department of Vascular Surgery, The Affiliated People’s Hospital of Ningbo University for providing the imaging data.
Author contributions
Haoxiang Zhu: Data curation, formal analysis, writing of the original draft of the manuscript, as well as review and editing of the manuscript. Huipeng Zhu: Study conceptualization, investigation, data visualization, writing of the original draft of the manuscript, as well as review and editing of the manuscript.
Consent to participate
The patients provided informed consent for undergoing the surgical procedure and utilization of clinical images for the present study.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
