Abstract
The role of chemokine (C-C motif) ligand 2 (CCL2) in peripheral artery disease is unclear. We measured the difference between serum and plasma levels of CCL2 in patients with chronic ischemia threatening the lower extremities following the observation that atypical chemokine receptors in blood and tissue cells may prevent CCL2 from entering the circulation and consequently modulate its function of attracting monocytes to the site of lesion. To identify the influence of CCL2, we compared the patients’ values to those in bio-banked samples from a control population. Further, we explored the association with the Asp42Gly polymorphism (rs12075) in Duffy antigen chemokine receptor; one of these atypical chemokine receptors. When possible, we evaluated in surgically excised normal and affected arteries the calcium burden as well as the expression of CCL2 and related receptors reflecting the inflammatory status. Our findings indicate that circulating CCL2 was significantly associated with the severity and presence of the disease (OR 0.966, 95% CI 0.944 to 0.988, p = 0.003). Circulating CCL2 was dependent on the rs12075 genotype (AA>AG>GG), which, probably, indicates a higher expression of chemokine receptor in the arteries of AA subjects. The associations with genetic variants and the over-expression of atypical chemokine receptors in diseased arteries may have potential implications and our data indicate that CCL2 may represent a previously unrecognized factor that needs to be considered in the screening of patients with risk factors for peripheral artery disease.
Keywords
Introduction
Lower extremity peripheral artery disease (PAD) is a major health concern that may be associated with catastrophic events in different arterial territories. The disease increases with age and, in people over the age of 55 years, the prevalence of PAD in the legs is approximately 20%.1–4 Paradoxically, although preventive measures are effective in early stages, PAD is under-diagnosed when asymptomatic and, consequently, prevention is either applied too late or not at all.1–6 Therefore, the search for laboratory surrogates is a relevant pursuit. We aimed to increase our understanding of the chemokine (C-C motif) ligand 2 (CCL2) in the overall inflammatory response observed in patients who develop chronic limb ischemia in order to identify associated factors7–12 and to test the hypothesis that the expression of this chemokine and its receptors is increased in diseased arteries. This is based on our previous findings indicating that CCL2 and CCR2 (its cell surface receptor) are widely distributed in mice. 13 The effect in human tissues is unknown. We also hypothesized that factors affecting the availability and/or systemic function of CCL2 may result in misinterpretation of clinical measurements.
We propose a plausible clinical impact of CCL2 in PAD because: first, the biological function of the CCL2/CCR2 pathway is its ability to induce cell migration; second, arteries with moderate atherosclerosis accumulate CCL2 in response to a variety of pro-inflammatory stimuli; and third, CCL2 may represent a potential therapeutic target.7,14–18 We have already shown that serum CCL2 levels are consistently higher than plasma CCL2 levels and that analytical variability is significantly higher in serum. 18 This finding may alter the relevance of the observed clinical associations. 19 Consequently, we reasoned that measurement of CCL2 should be performed in both types of biological samples and we arbitrarily defined circulating CCL2 as the difference between serum and plasma under identical conditions (ΔCCL2). A possible explanation for differences during laboratory analyses may lie in poorly understood chemokine receptors collectively known as ‘atypical’, ‘silent’, or ‘decoy’. These are G-protein coupled receptors that do not activate conventional signaling events (mainly, Duffy blood group chemokine receptor (DARC), D6, CCX-CKR1 and CXCR7). 20 Conversely, they may internalize, degrade or transport ligands (i.e. they have the potential to create clinically relevant chemokine patterns in tissues). Their levels of expression have not been explored previously in diseased arteries.
A recent genome-wide-analysis study identified a strong association between serum CCL2 levels and chromosome 1q22-q23, especially at the rs12075 marker in DARC. 17 This was not confirmed in plasma, and a recent study revealed complex associations that may be relevant in the study of atherosclerosis. 21 Here we present, for the first time in patients with PAD, the association between the Asp42Gly polymorphism (rs12075) in the DARC gene and circulating CCL2 levels, in relation to the relative expression of selected ligand and receptors in damaged arteries. Despite the apparent complexity in the interpretation, taken together our data suggest that circulating chemokines may play an important role in the pathogenesis of PAD.
Methods
Study population
This is an observational, cross-sectional study with a pre-set duration of 3 years (from January 2009 to December 2012). It involves data prospectively collected from selected patients referred to the vascular disease department of our hospital for the assessment of intermittent claudication. Diagnosis was with standard clinical assessments that included measurement of the ankle–brachial index (ABI), non-invasive imaging, and angiography when indicated. Our hospital’s ethics committee and institutional review board approved the procedures of the study protocol, and written informed consent was obtained from the participants prior to entry into the study (10-04-29/4proj3; 11-10-27/10proj1).
We recruited 174 patients aged between 55 and 85 years, in whom chronic limb ischemia was diagnosed (>2 weeks of resting pain or ulcers attributed to arterial occlusion disease). Staging III or IV was according to Fontaine, but the patients were only included if ulcers healed through medical therapy, irrespective of whether revascularization was further indicated to improve quality of life and to increase survival. These criteria were to exclude other factors such as infection, repeated trauma or significant neuropathy, and to ensure that the inclusion of diabetic patients does not constitute bias. 22 We did not find significant difficulties in recalling past events and main exclusion criteria were acute ischemia, signs of infection, renal failure, liver disease, cancer, or autoimmune diseases. The number of patients recruited within our fixed term of 3 years (n=174) was higher than the minimal sample size required to analyze biological markers with this design, and taking into account the incidence and laboratory variability6–8 (α=0.02, β=0.85; n=125). Diabetes, hypertension and dyslipidemia were defined according to established criteria.23–25 The comparison group was composed of bio-banked samples (n=154) representing the same general population from which the cohort of patients was selected. The controls were known to be unaffected (i.e. normal ABI values) and aged >50 years. To preserve the characteristics of the general population and to avoid population stratification, no other matching procedure was performed 26 and, consequently, there was a difference in age and gender distribution. However, these did not affect genetic studies and, presumably, did not skew the results of other comparisons. 7
Laboratory measurements
At the time of recruitment, fasting blood samples were drawn and serum and plasma obtained. Blood cell counts were performed immediately in an automatic cell counter (Coulter LH 750 Analyser; Beckman Coulter, Brea, CA, USA). Plasma concentrations of interleukin-8 (IL-8), soluble CD40 ligand (sCD40L) and soluble P-selectin (sP-selectin) were also measured immediately using a Bender MedSystems FlowCytomix detection on flow cytometry (Izasa, Barcelona, Spain). The remaining samples were frozen in aliquots at −80°C within 2 h of collection and stored until batched analyzed. Standard measurements were performed using enzymatic assays or chemiluminescent immunoassays.27,28 The updated homeostasis model assessment (HOMA2) estimates for insulin resistance were calculated. 14 Plasma β2-microglobulin and C-reactive protein were measured using turbidimetric reagents obtained from Biokit (Barcelona, Spain). Plasma and serum CCL2 were measured with an ELISA-based assay (PeproTech, London, UK) as described. 19 The difference between serum and plasma was expressed as ΔCCL2, or as a percentage [(serum – plasma) × (100 / serum)]. The calculated value was considered a measurement of circulating CCL2, and used to investigate cell–cell interactions during laboratory handling. DNA was extracted from EDTA blood using a column-based method from Qiagen (Izasa) and samples genotyped for rs12075 using the TaqMan 5’ allelic discrimination genotyping assay C_2493442_10 (Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s recommendations. 18
Immunohistochemical analyses
Portions of femoral and/or popliteal arteries from patients were obtained during surgical procedures for infra-inguinal limb revascularization (n=30). The same approved procedures had been used to generate non-affected donor tissue samples for banking (n=10) according to our Blood and Tissue Bank (www.bancsang.net/es/donants/donacio_teixits.html). After harvesting, the tissues were rinsed in phosphate buffer to remove residual blood and placed in at least 10 volumes of buffered formalin using a standard protocol for paraffin-embedded tissue samples. Three sections per slide were obtained and immunohistochemical studies were performed in batches, essentially as previously described. 29 Owing to differing manufacturing characteristics, and to ensure specific immunoreactivity, it is usual to test different antibodies from different suppliers before making the choice based on results obtained. A pre-requisite for an antibody is that it can be used for Western blot (WB) as well as immunochemical analyses, in order that the antibody can be checked against commercially supplied antigens. The optimal dilution and exposure time for each antibody were optimized for all subsequent analyses. In this particular study we used: D6, Abcam 1656, 1/1000; DARC, Abcam 58965, 1/500; CD68, Dako KP1, ready to use; CCR2, Abcam 21667, 1/50; and CCL2, Abcam (C-17) sc-1304, 1/500. The appropriate secondary antibodies (Vector Laboratories Inc., Burlingame, CA, USA) were used at a dilution of 1:200. Detection was performed with the ABC peroxidase system (Vector Laboratories, Inc.). For all the immunohistochemical measurements, negative controls were treated the same as test samples, but omitting the primary antibodies in the incubation. Also, arteries were routinely stained for calcium deposits with alizarin red S (Sigma, Madrid, Spain). Immunoreactivity was quantified automatically for each antibody using an image analysis system (AnaliSYS, Munster, Germany). 29
Statistical methods
The Kolmogorov–Smirnov test was used to check for normality of distribution of the variables. Values are presented either as mean (SEM) or median (interquartile range: the 1st quartile subtracted from the 3rd quartile). Comparisons between groups were made with Student’s unpaired t-test or Kruskal–Wallis one-way analysis. An allelic frequency description and Hardy–Weinberg equilibrium test were performed using SNPStats. The crude odds ratio (OR) and their 95% confidence intervals (95% CIs) for genotype associations were calculated using the inheritance model selected by the Akaike Information Criterion. 30 Logarithmic transformations were used in some cases to model nonlinear relationships. Multiple regression models were used to analyze the association between variables and results are provided only when the model fits the data (i.e. relatively low residuals). We included all variables in multivariable analyses as on theoretical grounds all have shown to be either risk factors or potential confounders. The number of variables was reduced, however, according to the explanatory or predictive purpose of the model. We routinely used the forward stepwise selection as the variable selection method in each analysis and at an arbitrarily specified significance level of p<0.05. Data were analyzed with SPSS version 18.0 (SPSS Inc., Chicago, IL, USA).
Results
Study population
We evaluated 174 patients (348 limbs) and identified 195 limbs with the diagnosis of chronic limb ischemia, as described above. Decreased, or absent, pulse was recorded in all patients and, in most, dry skin and coolness to palpation. Ulcers (Fontaine stage IV) were found in 120 limbs (61.5%) and pain at rest (Fontaine stage III) in 75 limbs (38.5%). The ankle systolic pressure ranged between 27 and 58 mmHg (41±9 mmHg) and the ABI values between 0.3 and 0.5 (0.39±0.17). Among the remaining limbs, the ABI was <0.9 in 143 and >0.9 in 10. Overall, there was no difference between right and left limbs with respect to ABI values (0.59±0.04 and 0.61±0.04, respectively). Other selected characteristics and descriptive variables of the patient cohort and population group are depicted in Table 1. Prescriptions are commonly obtained from the attending general practitioners. Medication use was recalled from questionnaires applied to the patients, but these probably indicate adherence rather than actual prescriptions. There was clinical evidence of concomitant cerebrovascular or coronary artery disease in 82 patients (47.1%). Most patients (n=141, 81.0%) were current or former smokers (27.3% in the population control group) and other co-morbidities were significantly higher (p<0.001) in patients than in the population control, including hypertension (66% vs 13.2%) and dyslipidemia (31.6% vs 10.0%). Of note, 15 patients were diabetic (n=1 in the population control) that were managed with either insulin (n=3) or metformin and, as mentioned above, the possibility of bias was considered negligible.
Selected descriptive characteristics and laboratory variables in participants.
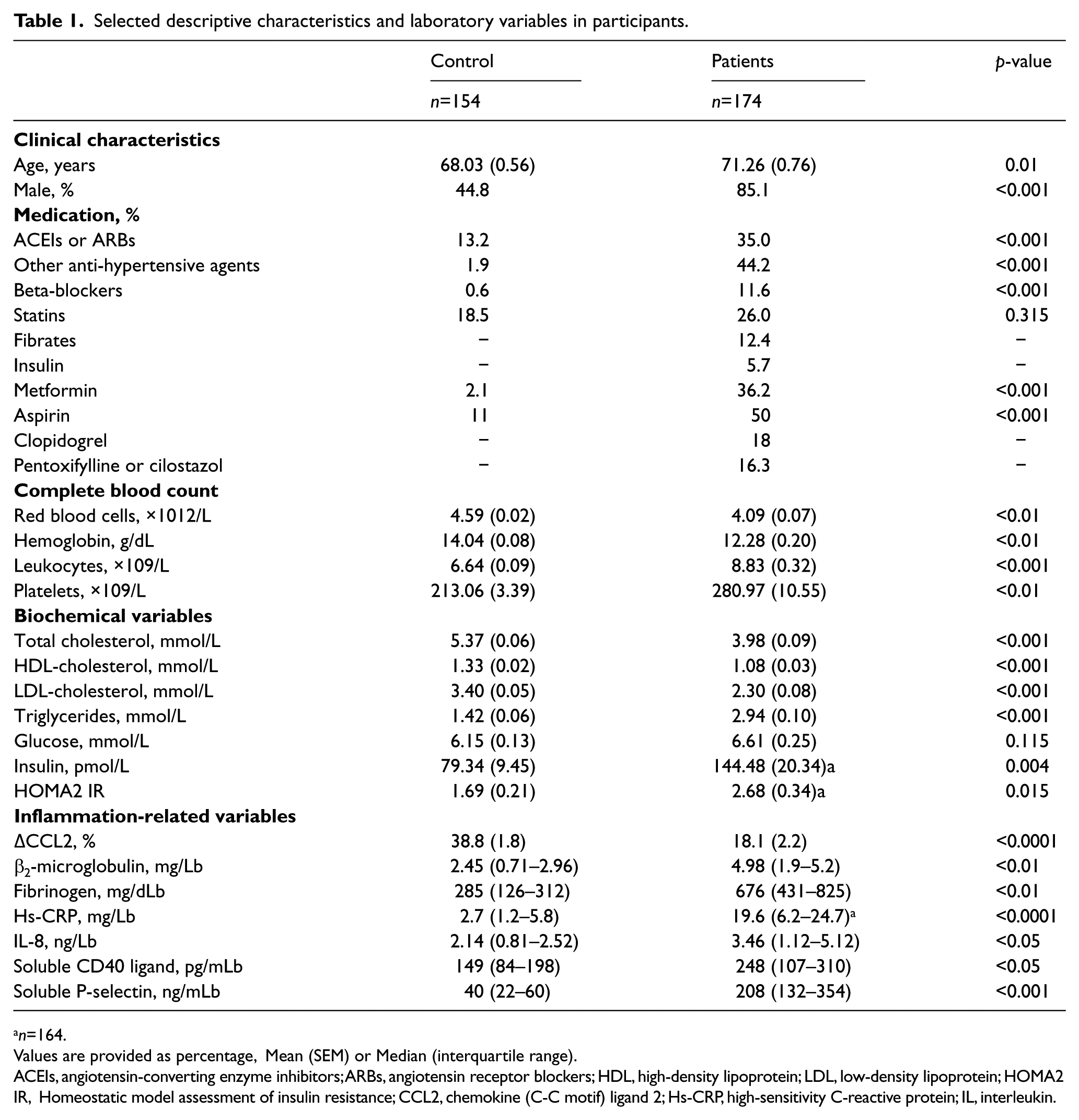
n=164.
Values are provided as percentage, Mean (SEM) or Median (interquartile range).
ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin receptor blockers; HDL, high-density lipoprotein; LDL, low-density lipoprotein; HOMA2 IR, Homeostatic model assessment of insulin resistance; CCL2, chemokine (C-C motif) ligand 2; Hs-CRP, high-sensitivity C-reactive protein; IL, interleukin.
Biological significance of CCL2 in PAD
Plasma and serum CCL2 concentrations were higher in patients than in controls and measurements in both samples were significantly correlated (Figure 1). The difference (ΔCCL2) between values in plasma and serum CCL2 was significantly (p<0.0001) lower in patients. We interpret the significant decrease in patients as an indication that CCL2 is either functioning or sequestered in affected arteries. A substantial amount of CCL2 may be normally bound to blood cells and, consequently, only fully detected in serum (i.e. after coagulation). The platelet count was higher in patients than in controls, and significantly correlated with ΔCCL2 (ρ = 0.214; p = 0.015). The design of the study only allows the computation of the odds ratio (OR). Despite being less sensitive than relative risk, we found (once adjusted for variables as outlined in Table 1) that plasma CCL2 values (OR 1.007, 95% CI 1.003 to 1.012; p = 0.001) and ΔCCL2 (OR 0.966, 95% CI 0.944 to 0.988; p = 0.003) were associated with the presence of the disease. This is probably useful diagnostic and prognostic information. This association was not observed for any other inflammation-related variable. Exploration of associations with other biological determinants showed that there were no significant associations with monocyte count, age, and smoking. However, diabetes mellitus affected the interpretation of serum and plasma CCL2. These measurements were associated with plasma insulin concentration and insulin resistance (data not shown). Because our method detects exogenous insulin, the association was calculated excluding patients taking insulin. Intriguingly, serum CCL2 concentrations were significantly lower in patients with diabetes mellitus than in those without diabetes [550.51 (40.07) vs 642.51 (69.31) pg/mL; p<0.001] and this difference disappeared when CCL2 was measured in plasma [441.26 (26.27) vs 435.46 (29.72) pg/mL]. These results reinforce the concept that ΔCCL2 is considerably more informative in these patients. To explore the effect of genetic variants with a potential impact on CCL2 we determined the relationships between the Asp42Gly polymorphism (rs12075) in the DARC gene and the circulating CCL2 levels in patients with PAD. The rate of successful genotyping was 96%. Genotype frequencies did not deviate from the Hardy–Weinberg equilibrium, either in patients or in controls. The A allele was more frequent (61%) than the G allele (35%) and there were no differences in distribution between patients and controls (Figure 2A). However, circulating CCL2 was clearly associated with the genotype (AA>AG>GG) both in population controls and in patients (Figure 2B). Following adjustment for clinical, laboratory and genetic variables and using the over-dominant model of inheritance, ΔCCL2 remained significantly associated with the presence of PAD (OR 0.965; 95% CI 0.942 to 0.989; p = 0.004). As such, these genotypes appear to play a role in CCL2 function in the tissue in PAD. A preliminary assessment of the expression of CCL2 and receptors in arteries is justified.
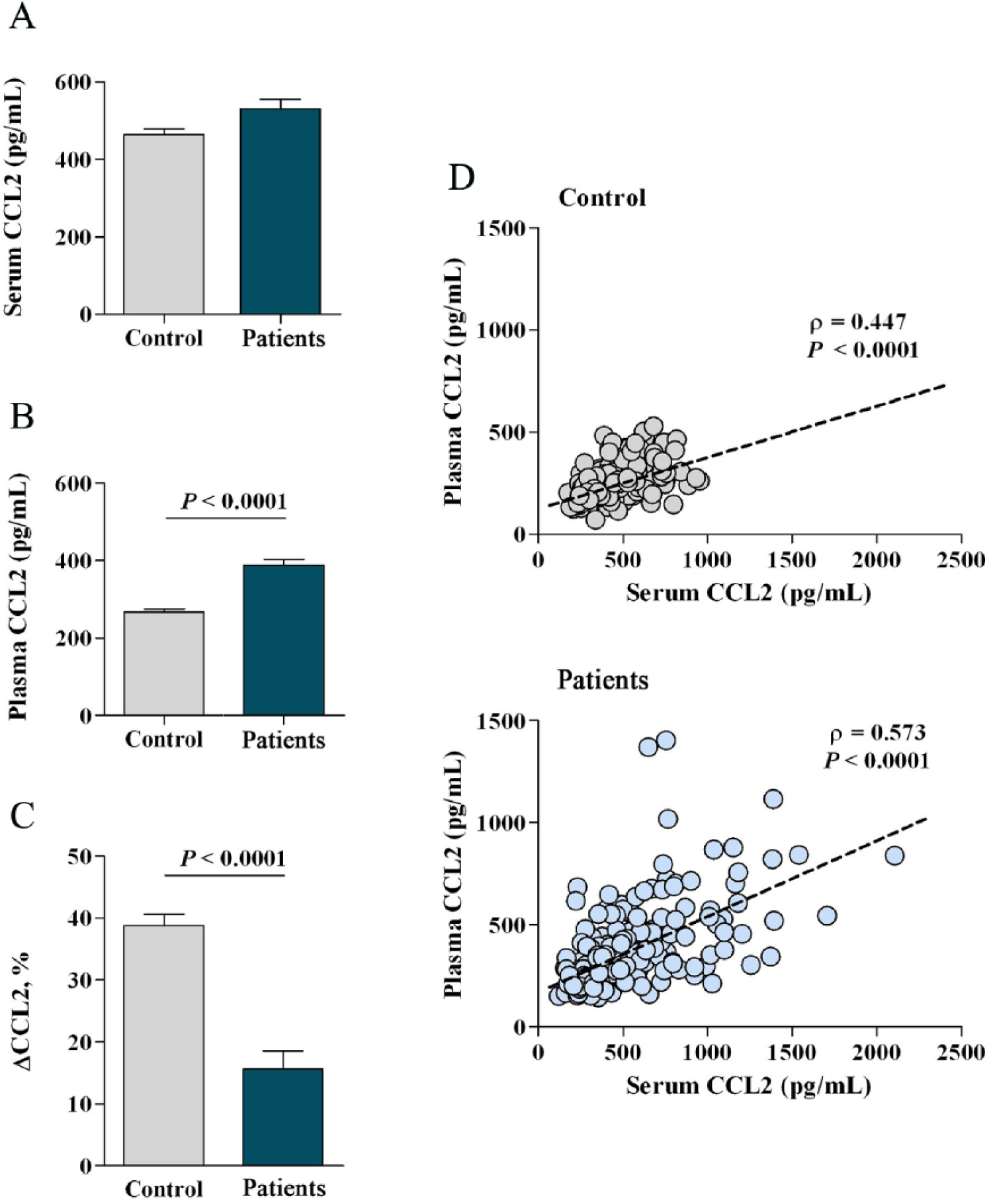
CCL2 concentrations (pg/mL) in serum (A) were uniformly higher than in plasma (B) in controls and patients. The differences between serum and plasma CCL2 (ΔCCL2) were calculated, and arbitrarily defined as the total amount of circulating CCL2 (C). Serum and plasma CCL2 concentrations maintained significant correlations in controls and patients (D).
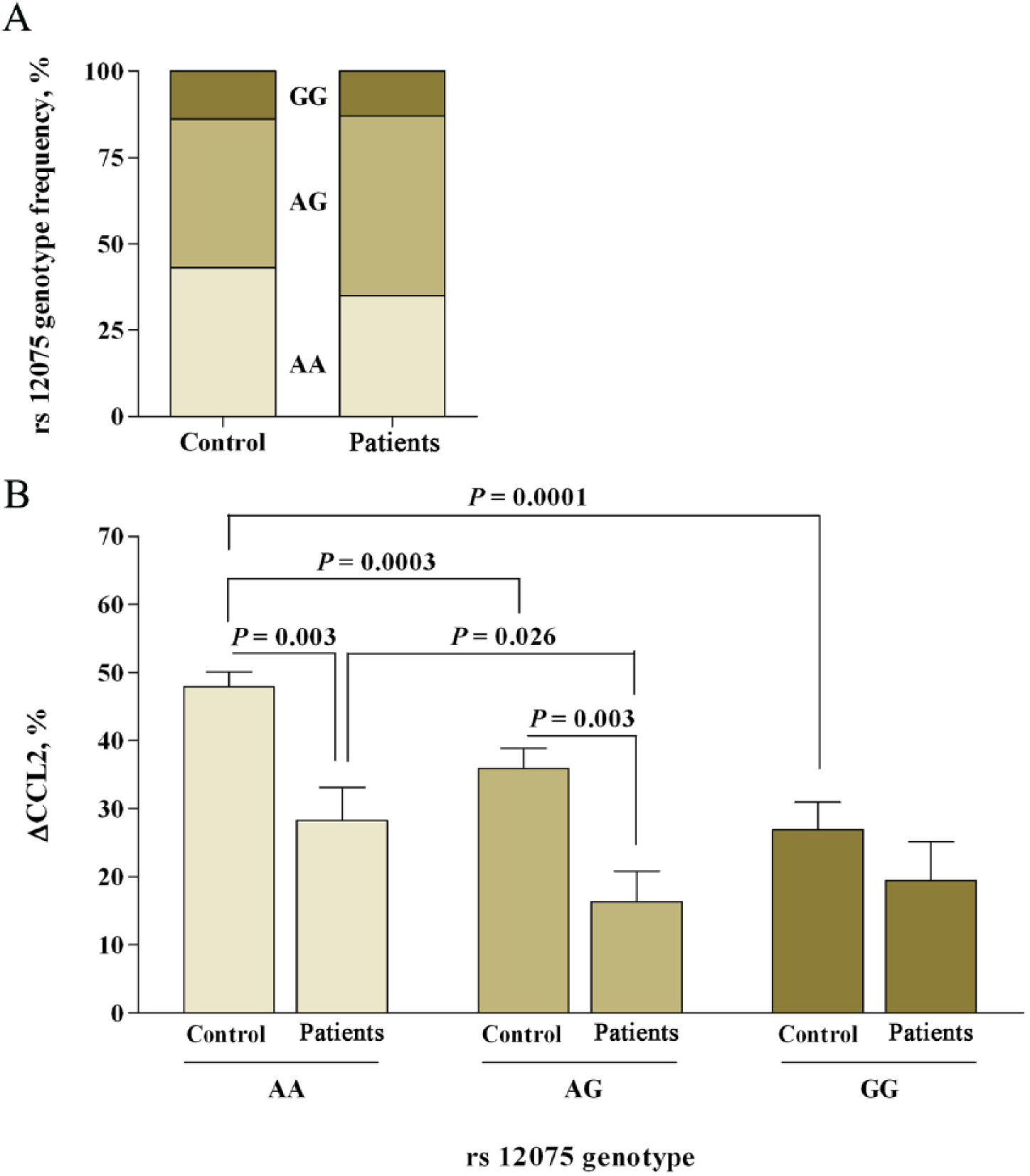
The rs12075 genotype distribution in patients compared to healthy participants from the same population (A). Differences between circulating CCL2 (ΔCCL2) were related to the rs12075 (Asp42Gly) genotype (B).
Histology observations
Representative results in affected arteries are summarized in Supplementary Figure 1 (all supplementary figures are available online with this article) in which the same artery stained with each described marker is depicted. Calcium deposition, found exclusively in the media, was a distinct finding in affected arteries, compared to controls. The circumferential distribution of calcium as a dark stripe in hematoxylin and eosin stained samples was a relatively frequent observation (Figure 3A). There was no correlation between alizarin staining (Figure 3B) and the expression of other markers. The CD68-positive cells (macrophages) are not observed in normal or minimally affected arteries (Supplementary Figure 2, A and B) but are predominant in newly formed intima and in the adventitia of diseased arteries (Supplementary Figure 2, C and D). In non-diseased arteries, the CCL2-stained cells were observed only in the adventitia endothelium of the vessels (Figures 4A and 4B). In affected arteries, the expression of CCL2 was intense in the adventitia and the distribution was not homogeneous in the intima and media (Figures 4C and 4D). Although there were some trends regarding this expression and circulating CCL2 or DARC genotypes, the low number of specimens available for analysis precluded further inferences. The expression of CCR2-stained cells was strong and limited to the media in normal arteries. The expression in affected arteries was uniform throughout, including intima and adventitia (Figure 5). The increased presence of these functional receptors (i.e. CCR2) indicates that, probably, most cells are receptive to the presence of circulating CCL2 and, consequently, may be more reactive in the case of disease progression. DARC-stained cells were not found in normal arteries. The expression in the intima of affected arteries appears related, albeit non-significantly, to the genotypes investigated; in most cases, staining was intense in media and adventitia. Curiously, the expression of DARC is more intense in atheroma, which is in contrast with the expression of other chemokine receptors. It was also evident that the expression of DARC in arteries with higher deposition of calcium is limited to endothelial cells in contact with the lumen (Supplementary Figure 3). The expression of D6-stained cells was practically absent in normal arteries, but intense and widely distributed throughout affected arteries. The expression of D6 in the intima was also limited, usually to cells in contact with the lumen (Supplementary Figure 4).
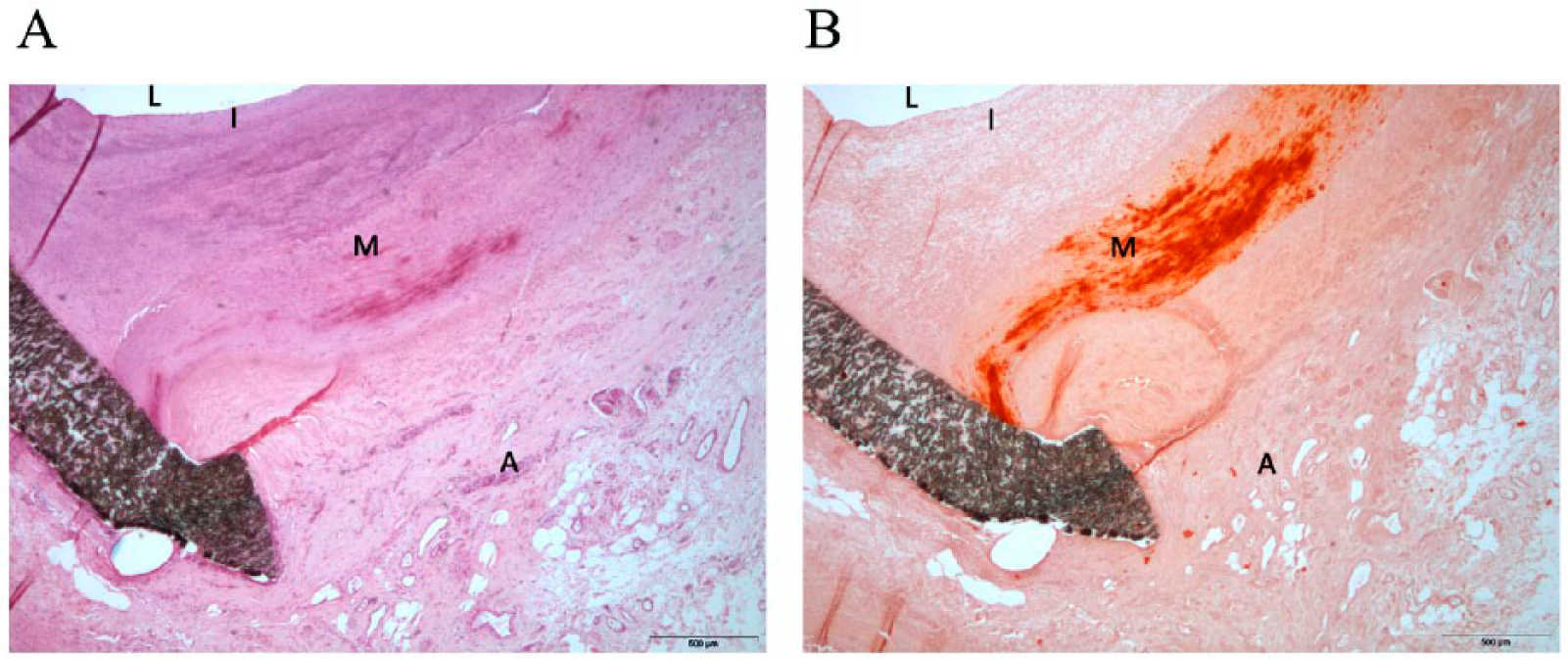
The presence of calcium in affected arteries is limited to the media, and unambiguous interpretation requires specific staining. Circumferential calcium deposition is observed as a dark stripe when stained with hematoxylin and eosin (A). Alizarin staining readily detects calcium without the formation of crystals (B). To clarify these findings, we have highlighted the approximate location of lumen (L), intima (I), media (M) and adventitia (A).
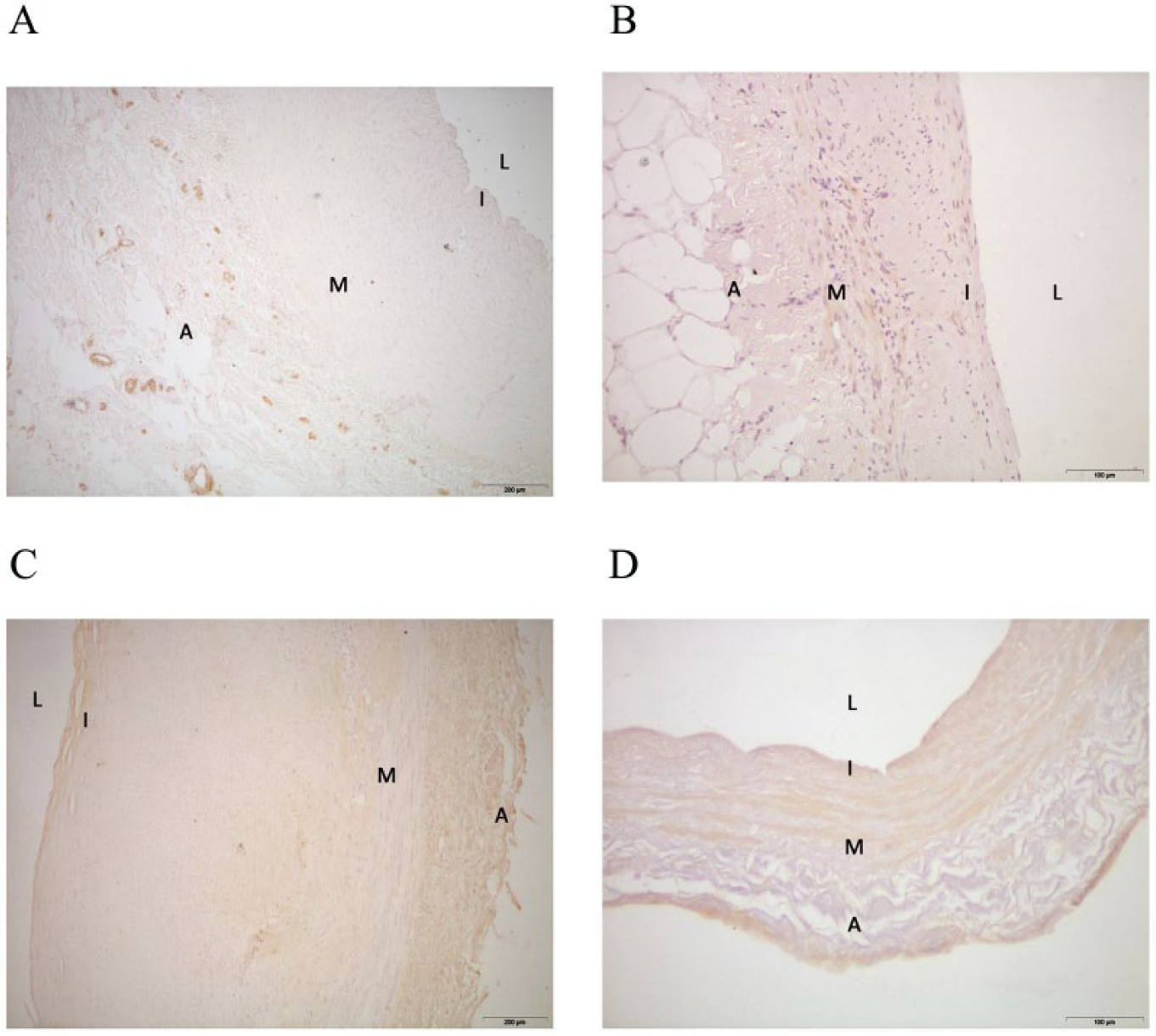
In normal or minimally affected arteries, CCL2 is only expressed in adventitia (A, B). Affected arteries depict widely heterogeneous expression throughout the arterial sites but, usually, are more intense in the adventitia (C, D). To clarify the findings, we have highlighted the approximate locations of lumen (L), intima (I), media (M) and adventitia (A). 200 μm, magnification ×40; 100 μm.
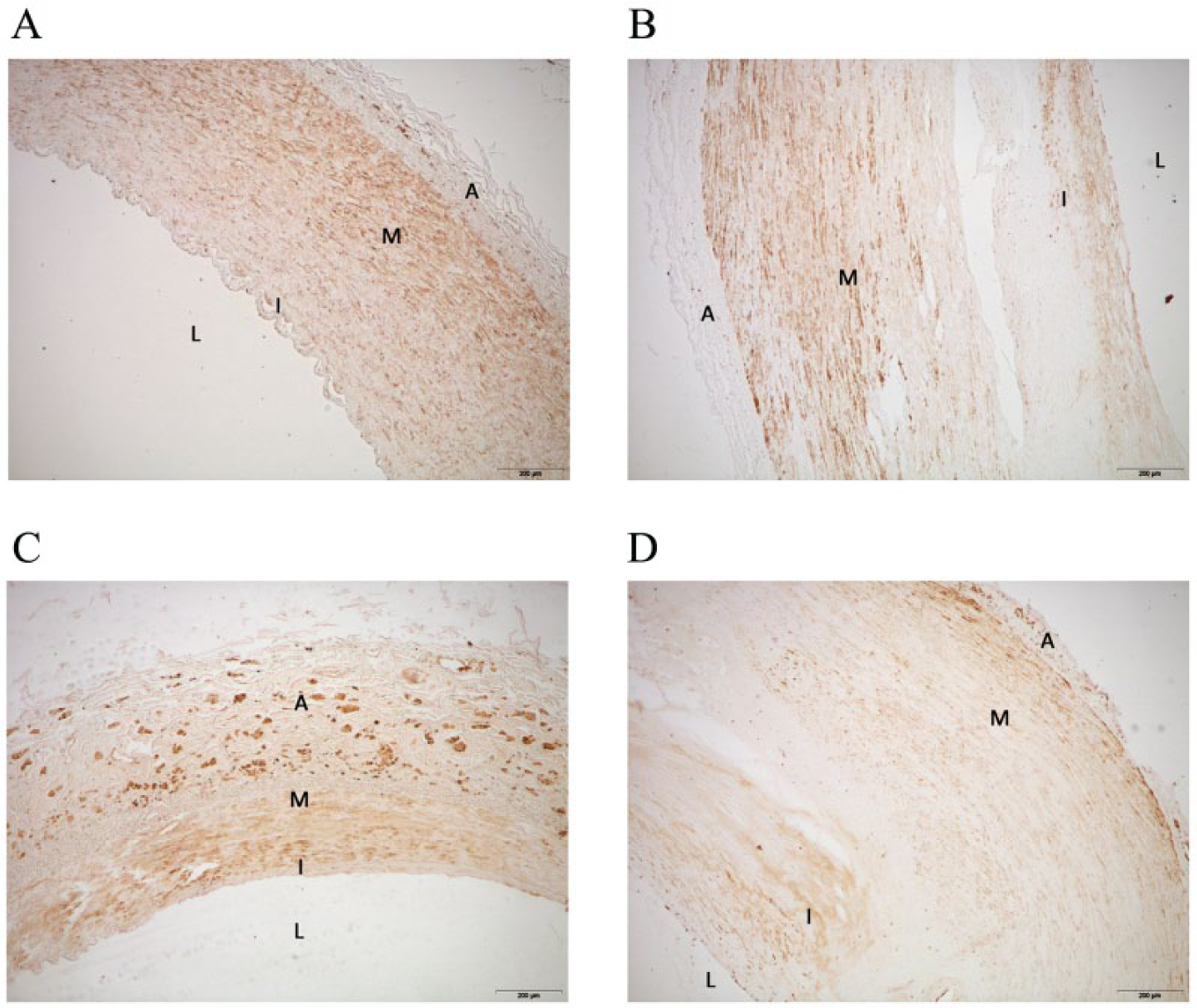
The expression of CCR2-stained cells was strong and limited, in normal arteries, to the media (A, B). In affected arteries the expression was uniform throughout the artery including intima and adventitia (C, D). To clarify findings, we have highlighted the approximate locations of lumen (L), intima (I), media (M) and adventitia (A). 200 μm, magnification ×40; 100 μm.
Discussion
The prevalence of PAD, a complex, multi-factorial and incompletely understood condition, is expected to rise. It carries significant morbidity and health care costs, especially in those developing critical limb ischemia. The available clinical management of these patients is poor, and prognosis is pessimistic.1–3 In our study, as in others, 31 it is distressing to note that risk factor modification is far from what could be considered optimal for such a condition. Atherosclerosis is increasingly being considered an inflammatory disease, and it is currently accepted that CCL2 is involved in its pathogenesis. In mouse models, overexpression of CCL2 increased the progression of atherosclerosis, deficiency of CCL2 reduced atherosclerosis, and plasma levels of CCL2 promote recruitment of monocytes and other immune cells to the vessel wall. 32 The value of CCL2 as a cardiovascular risk marker has been assessed, mainly in coronary heart disease.33–36 Although the findings are often contradictory, 16 there is no reason to exclude the relevance of lower extremities in atherosclerosis since there are limited, but consistent and suggestive, data to the contrary.37,38
We identified a few factors limiting the interpretation of our findings. For example, CCL2 concentration in plasma is consistently lower than in serum, and interpretation differs depending on which measurement is used.17–19 Our data show that this is not an anecdotal finding. Interpretation is not simple, especially if there are other possible biological determinants (e.g. diabetes mellitus). Therefore, we propose the use of the difference between serum and plasma values (ΔCCL2) as a classifier of severity. There are at least two advantages: (1) a decrease in the influence of laboratory mishandling; and (2) that chemokines bound to blood and endothelial cells are taken into account. Using this approach, the alternative measure of the total amount of circulating CCL2 (i.e. ΔCCL2) is strongly associated with critical limb ischemia, and remains a powerful predictor when adjusted for other confounding factors. Of note is that we observed that severe and extensive lesions are consistently associated with significantly decreased ΔCCL2 values. We interpret our data as being the result of a high burden of damaged tissue. Plasma CCL2 concentrations may vary but do not reflect CCL2 bound to blood cells such as platelets. 39 Serum CCL2 could represent a measurement of CCL2 post-coagulation. The combination of both measurements would indirectly represent the amount of CCL2 expressed, or transported, into the artery during protracted inflammatory stimuli (i.e. a measurement of chemokine availability and function in tissues). 40 More importantly, ΔCCL2 was not, in our study, associated with other inflammation-related variables. Further, preliminary results in an on-going prospective study (data not shown) suggest that these measurements may be useful for the stratification of the extent of PAD. The availability of CCL2 may be complicated by potential effects induced by differential expression of functional receptors (CCR2) and the presence of atypical chemokine receptors of unknown function in the regulatory network which enables the organism to modulate leukocyte migration. 40 For the first time in the current literature, we observed that a genetic polymorphism of DARC may be a contributing factor. Genotype frequencies in patients and in unaffected members of the same population are similar, but ΔCCL2 values are higher in genotype AA and lower in GG. Differences in ΔCCL2 between cases and controls were significant, and suggest a higher expression of these receptors in patients with AA. It is consequently a candidate genetic marker that warrants further investigation. Our histologic observations failed to show significant associations, but we found that the expression of CCR2, DARC and D6 was increased in diseased arteries, and that the distributions in the arteries are not uniform. A pathogenic role is likely, and data suggest that atypical chemokine receptors modify chemokine availability in PAD. Although these receptors have no involvement in cell migration, a modulation of the inflammatory response is likely. It should be mentioned that our finding of a significant calcium burden in the arterial tissue could be clinically important since patency rates with surgical interventions are low in cases of very calcified arteries. This high calcium load may represent an unrecognized confounding factor.
A number of limitations should be recognized in this study. First, despite our efforts to minimize the effect of diabetes mellitus (including neuropathy and difficulty in healing ulcers) it is not clear whether these patients should be included, according to the initial definition of critical limb ischemia. 22 Second, results obtained in a cross-sectional study do not quantify any improvement in clinical performance that can be gained from the addition of a biomarker to data on risk factors. Third, the genetic findings have, as yet, not been replicated in other studies and, as such, should be considered exploratory and requiring further validation. Fourth, the effects of suboptimal medical treatment the patients may have been receiving at the time of study, and the specific mechanisms of action of several drugs being administered, cannot be discarded as possible confounding factors.
Finally, PAD should be considered a clear example of a condition in which metabolism confronts inflammation. Recently, we developed a mouse model that over-expresses CCL2 and highlights an intriguing relationship between mitochondrial dysfunction, energy intake, and inflammation. 41 Patients with PAD appear to have abnormal mitochondrial function in diseased arteries, independent of tissue perfusion. 42 This would imply that, in PAD and other diseases of aging, a regulatory role of metabolic reprogramming in affected cells might be pivotal in determining the relationship with inflammation. 43
In conclusion, CCL2 is involved in the progression of PAD. Overall, our findings indicate that the availability of chemokines might represent a previously unrecognized factor in the development of chronic critical limb ischemia. To therapeutically influence ΔCCL2 to improve patient care is attractive.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by grants from the Instituto de Salud Carlos III (Carlos III Health Institute, Madrid, Spain) (PI08/1381, PI11/00130) and the Fondo Europeo de Desarrollo Regional (FEDER) (European Fund for Regional Development). Underlying research materials related to this manuscript are available on request from the corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
