Abstract
Retroperitoneal fibrosis (RPF) is a rare disease that is marked by systemic inflammation and the development of a periaortic fibroinflammatory mass. The fibroinflammatory infiltration can encase the abdominal aorta, ureters, and other abdominal organs. The clinical presentation often includes constitutional symptoms, abdominal pain, and signs of renal insufficiency or renal failure related to ureteral obstruction. Less frequently, RPF may present with vascular complications, such as venous thrombosis or claudication. The idiopathic form of RPF is most common but secondary forms have been described and are associated with malignancy and a variety of different medications. The pathophysiology is uncertain, but RPF has been linked with periaortitis and IgG4-related disease. Treatment centers on the relief of symptoms and complications associated with mass effects. Corticosteroids and other immunosuppressant therapies can improve constitutional symptoms, reduce infiltrate mass, and achieve disease remission, but a chronic relapsing course is not uncommon.
Keywords
Introduction
Retroperitoneal fibrosis (RPF) is a rare disease that is marked by systemic inflammation and the development of a periaortic fibroinflammatory mass. In this case report and topic review we will discuss the presentation, pathology, and treatment of RPF. Originally described by Ormond, 1 RPF is often linked with ureteral obstruction and associated urological complications. Our case report highlights the relationship of RPF with atherosclerotic disease and the infrequently reported symptoms of lower extremity claudication. We demonstrate how non-invasive physiologic vascular testing can be used to evaluate the functional significance of arterial insufficiency, help to distinguish the relative contributions of atherosclerotic disease compared to RPF-mediated arterial compression, and to monitor response to therapy.
Case history
A 57-year-old female with irritable bowel syndrome, dyslipidemia, and prior tobacco abuse (25 pack-years) presented to her primary care physician’s office with 5 months of left lower extremity pain with activity and 1 month of abdominal discomfort. Initially, her leg discomfort was noted during aerobic exercise classes and with prolonged sitting. It was described as dull and located in the groin and medial thigh. Subsequently, she also noticed left leg fatigue and calf discomfort when walking long distances or at a rapid pace. Her right lower extremity was unaffected.
Her abdominal pain was localized to the lower quadrants and was described as a dull ache that often woke her from sleep and was not associated with meals, bowel or bladder activity, or physical movement. The pain syndrome progressed and became more intense. While she reported having some non-bloody diarrhea and urinary frequency, she denied hematochezia or hematuria. She also denied fever, chills, or night sweats. She had mild unintentional weight loss (2–3 pounds). Other than a history of microscopic hematuria 9 years prior to presentation with negative urologic evaluation, past medical, social, and family medical histories were non-contributory.
On physical examination, vital signs were notable for blood pressure 138/70 mmHg, heart rate 64 beats per minute, respiratory rate 14 breaths per minute, and room air oxygen saturation 100%. She was 5 feet 4 inches tall (162.5 cm) and weighed 152 pounds (69 kg). The patient was a normal-appearing, middle-aged female in no acute distress. There was no evidence of lymphadenopathy or thyroid enlargement. Cardiac, pulmonary, dermatologic, and neurologic examinations were unremarkable. Abdominal examination demonstrated mild generalized discomfort within the lower quadrants and absence of rebound or guarding. Vascular examination demonstrated normal and symmetric upper extremity pulses, 2+ femoral and popliteal pulses bilaterally, and 1+ dorsalis pedis pulses bilaterally with intact capillary refill. Bilateral ileofemoral bruits were noted.
Laboratory studies were notable for elevated inflammatory markers, mild anemia, normal autoimmune studies, and normal serum IgG4 level (Table 1). Urinalysis demonstrated moderate heme. An abdominal computed tomography (CT) scan demonstrated the presence of a retroperitoneal mass encasing the mid to distal infrarenal aorta and common iliac arteries. The mass was located anterior and lateral to the aorta, sparring the retroaortic space, and was circumferential to the proximal common iliac arteries bilaterally (Figure 1A). There was diffuse vascular calcification and atherosclerosis of the infrarenal abdominal aorta and iliac arteries with moderate (50–70%) narrowing of the distal abdominal aorta and left common iliac artery. No vascular wall thickening was seen to suggest active vasculitis. Bilateral renal arteries and the superior mesenteric artery were widely patent.
Baseline Laboratory Values.

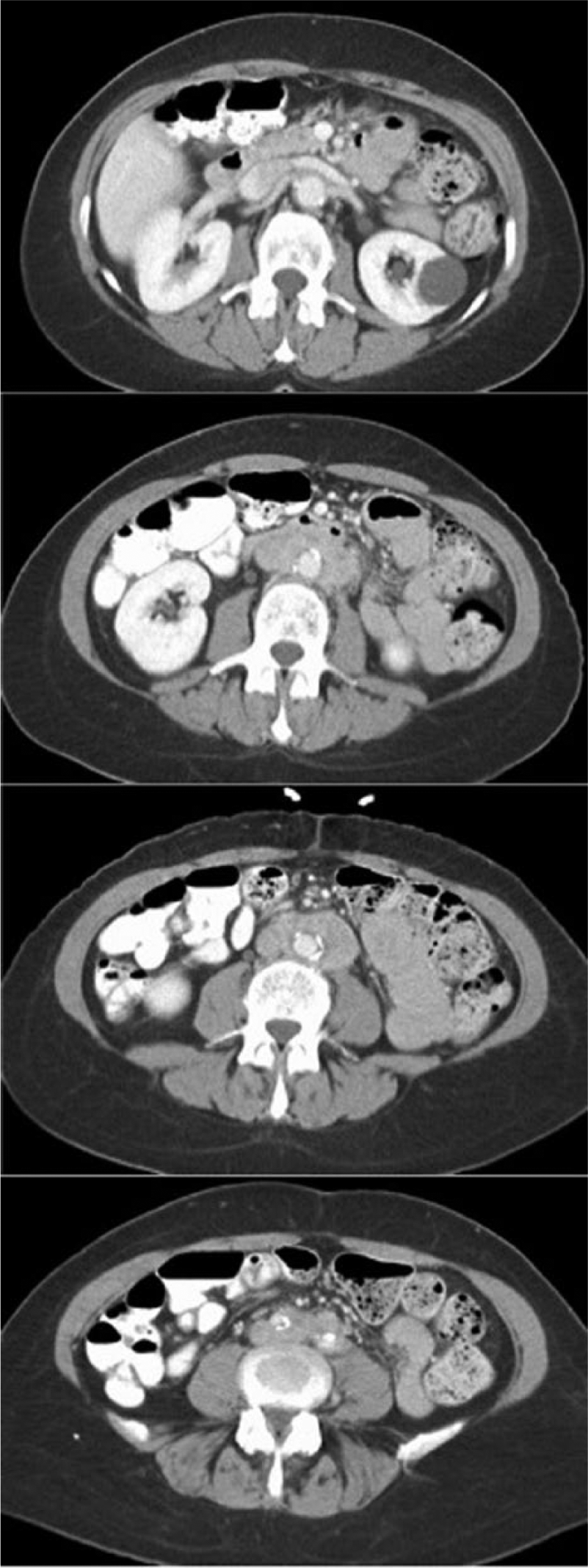
Abdominal computed tomography scan with intravenous contrast demonstrating a para-aortic mass located anterior and lateral to the aorta, sparring the retroaortic space, and circumferential to the proximal common iliac arteries bilaterally.
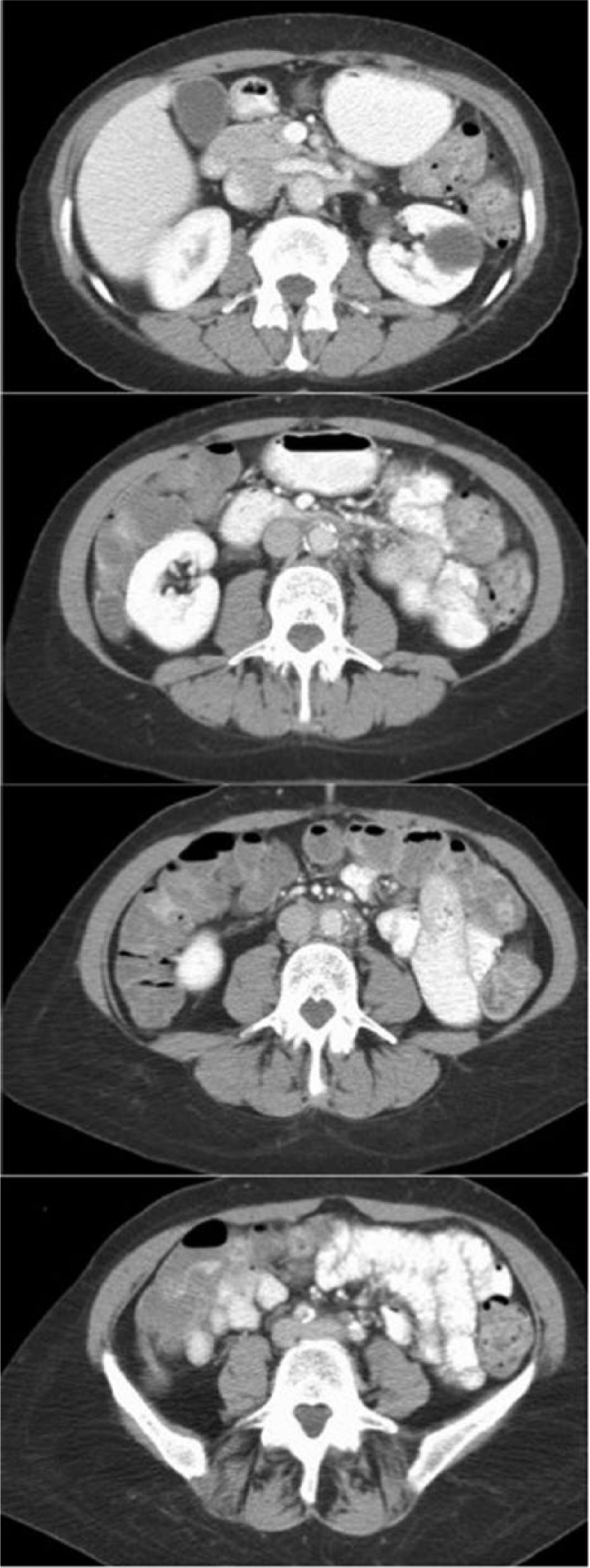
Abdominal computed tomography scan demonstrating significant regression in RPF mass after initiation of steroid therapy.
A CT-guided biopsy of the retroperitoneal mass was negative for malignancy, demonstrating dense fibrous tissue with scattered, benign-appearing lymphocytes. Immunohistochemical stains for CD3 and CD20 highlighted small, benign-appearing lymphoid aggregates with scattered B cells and T cells. CD34 staining was negative (Figure 2). IgG4 staining was not performed. Rheumatology and vascular medicine consultations were obtained.
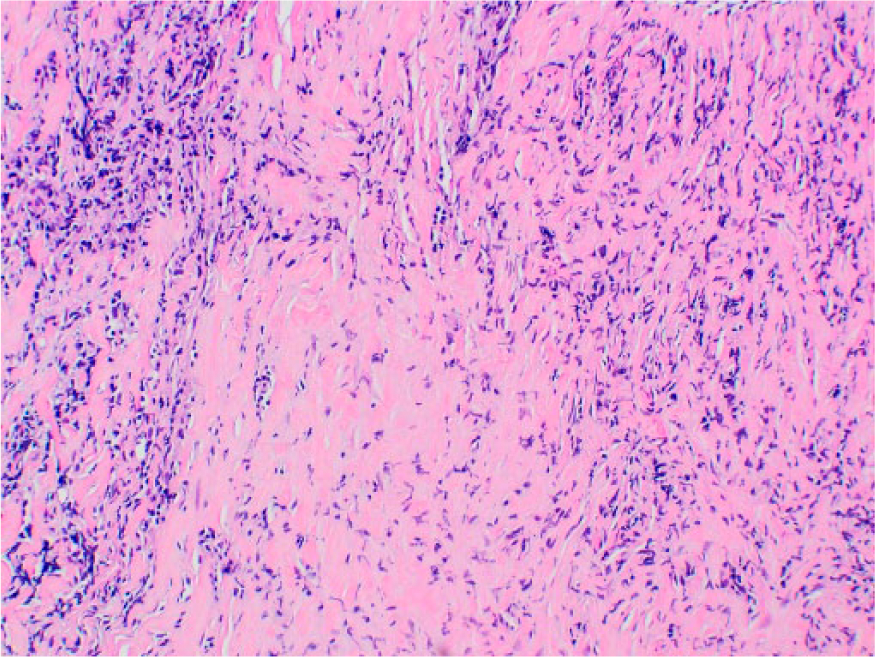
Retroperitoneal biopsy (hematoxylin and eosin) demonstrating dense fibrous tissue with scattered benign-appearing lymphocytes.
Resting and post-exercise ankle–brachial index (ABI) examination confirmed the presence of mild-to-moderate, left-sided, arterial insufficiency with abnormal resting and post-exercise ABI values (Table 2). A magnetic resonance angiogram (MRA) demonstrated evidence of moderate atherosclerosis, narrowing of bilateral common iliac arteries with the degree of luminal compromise greater on the left compared to the right, and complete occlusion of the left posterior tibial artery and near complete occlusion of the right posterior tibial artery. There were no findings to suggest vasculitis (Figure 3). With neither the CT or MRA scans demonstrating evidence of vasculitis, the impression was that of aortoiliac atherosclerosis with superimposed arterial compression from RPF, and further imaging (e.g. positron emission tomography) was not pursued.
Resting and post-exercise ankle–brachial index and segmental pressures at baseline and after 6 months of prednisone therapy.
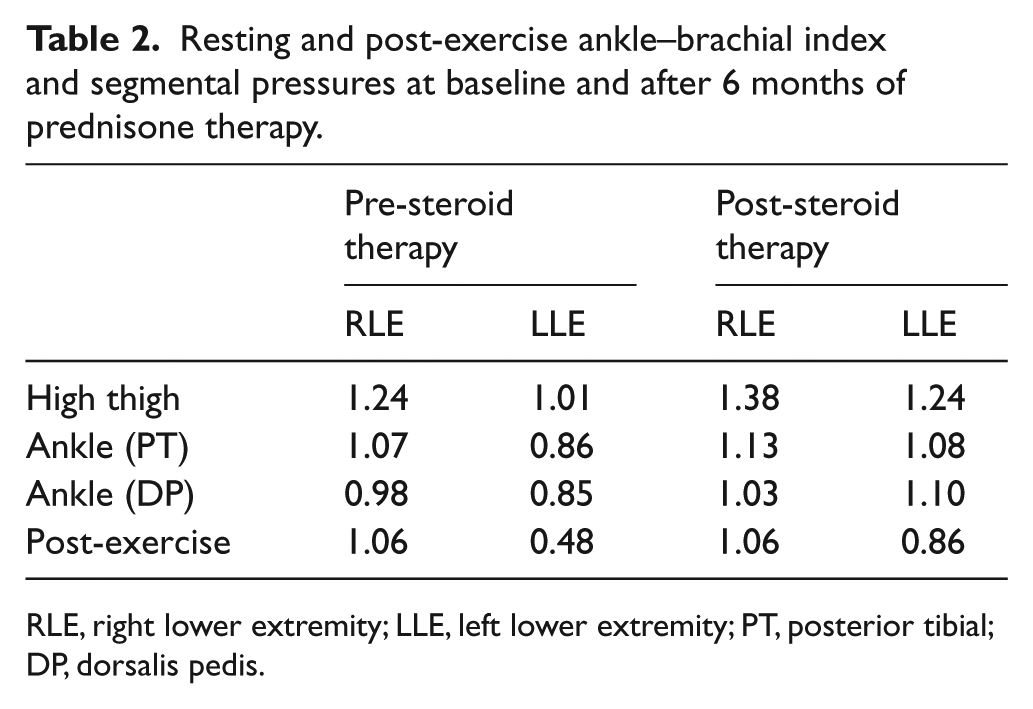
RLE, right lower extremity; LLE, left lower extremity; PT, posterior tibial; DP, dorsalis pedis.
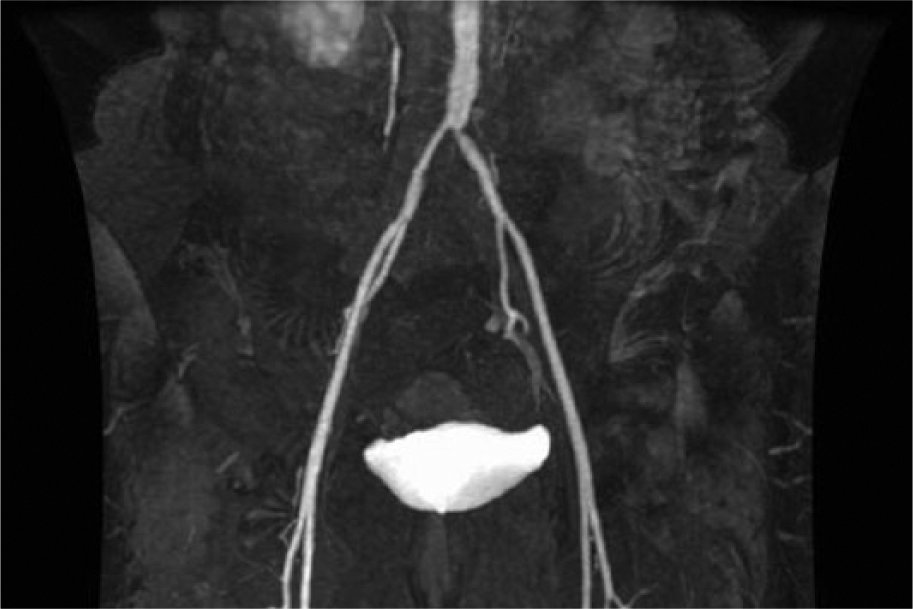
Magnetic resonance angiogram of the abdomen, pelvis and lower extremities demonstrating aortoiliac atherosclerotic disease, particularly affecting the ostial left common iliac artery. No findings of active vasculitis (T1 sequence mural contrast enhancement and vessel wall thickening or T2 sequence vessel wall edema) were identified.
The patient was started on prednisone for treatment of idiopathic retroperitoneal fibrosis. Her abdominal and left groin discomfort, which were felt to be related to mass effect, resolved and her claudication significantly improved within weeks of initiating steroid therapy. At the 2-month follow-up, she reported sustained relief from her abdominal discomfort and a stable non-lifestyle-limiting claudication. CT imaging demonstrated a dramatic interval improvement in retroperitoneal fibrosis; the periaortic mass thickness decreased from 1.5 cm to 0.5 cm (Figure 1B).
The patient had a chronic relapsing course and an inability to discontinue prednisone therapy. After 1 month of high-dose prednisone (60 mg daily) her dose was slowly tapered to a maintenance dose of 10 mg daily, and this was maintained for 6 months. At 9 months, attempts to taper prednisone to 5 mg daily resulted in disease relapse with complaints of recurrent abdominal pain and CT scan evidence of increased RPF mass thickness. Ultimately, oral methotrexate (10 mg weekly) was initiated as a steroid-sparing agent and at the time of publication the patient was asymptomatic but complete disease remission had not yet been achieved.
The patient’s complaint of left lower-extremity claudication also seemed to correlate with RPF burden and steroid therapy – markedly improving with high-dose steroids and disease regression and returning with steroid taper and an increase in RPF burden. ABI testing confirmed improvement, with normalization of resting left ABI values and an increase in post-exercise ABI values bilaterally in response to treatment (Table 2). Cilostazol was initiated, but not tolerated. Ultimately, once on steroid and methotrexate therapy, the patient’s claudication symptoms became mild, stable, and non-lifestyle-limiting. Overall, her presentation, clinical response, radiologic imaging, and physiologic non-invasive vascular testing were consistent with mild baseline arterial insufficiency that was exacerbated by RPF-mediated arterial compression. For this reason and the patient’s symptomatic response to medical therapy, revascularization was not pursued.
Pathology
First described in 1905 by French urologist Albarran 2 and later formally described by Ormond 1 in 1948, RPF was initially recognized as an extensive fibrosis which could cause ureteral obstruction necessitating surgery. Since then, RPF has been shown to be a chronic inflammatory process involving the infrarenal great vessels. It is a rare disease with an incidence of about 0.1 per 100,000 people and a prevalence of 1.4 per 100,000 people based on a study in Finland. 3 Histopathological features of RPF include inflammation of the adventitia and periadventitia, atherosclerosis and thinning of the media of the aorta.4–6 As a result, it has been considered a form of chronic periaortitis, a classification that includes abdominal aortic aneurysms and perianeurysmal retroperitoneal fibrosis. Differentiating RPF from these other forms of periaortitis essentially requires delineating the presence or absence of a dilated aorta, the latter of which is not a feature of RPF.7,8 Grossly, a firm, gray mass, extending from the renal arteries to the common iliac arteries, surrounds the abdominal aortal and iliac arteries. This mass may encase other retroperitoneal structures, such as the ureters. Microscopically, the mass consists of fibrotic tissue and inflammatory infiltration. The fibrotic component contains type I collagen and fibroblast- and myofibroblast-like cells.6,9 The inflammatory component consists of target-like-appearing aggregates of T cells around a B cell or germinal center core, IgG4-bearing plasma cells, macrophages and rare eosinophils.6,9 Additionally, Corradi et al. found vasculitis involving smaller retroperitoneal vessels in about 50% of RPF cases in a small case study. 6
Retroperitoneal fibrosis can be divided into an idiopathic form, which accounts for the majority of cases, and a secondary form, caused by factors such as malignancy, history of radiation, and medications. Secondary RPF will not be discussed in this case review. The pathogenesis of idiopathic RPF remains unclear. Mitchinson and Parums, who first characterized the changes in the vascular tissue in periaortitis, have theorized that localized inflammation occurs in response to oxidized components of atherosclerotic plaque.4,5,10 Specifically, they suggest that medial thinning permits exposure of oxidized low-density lipoprotein and ceroid (a lipoprotein resulting from low-density lipoprotein oxidation) to macrophages, stimulating a lymphocytic response that causes an inflammation of the aortic vascular wall.10–12 However, such a localized response does not explain the constitutional symptoms often described in RPF or the extension of histological changes seen in the thoracic aorta. Mitchinson noted these changes in autopsy and in live cases in 1970. 13 Salvarani et al. used positron emission tomography (PET) scans to demonstrate the pathologic uptake of 18F-fluorodeoxyglucose (FDG) not only in the abdominal aorta and iliac arteries, but also in the thoracic aorta and its main branches as well. 14 Research has indicated the possible role of genetic and environmental risk factors. In a small case–control study, Martorana et al. found an association between HLA-DRB1*03 and HLA-B*08 alleles and periaortitis. 15 Uibu et al. demonstrated an association between asbestos exposure and development of RPF. 3 Clinical data suggest a possible association with other connective tissue, and rheumatologic and/or vasculitic conditions. Though non-specific, up to 60% of patients with idiopathic RPF have tested positive for anti-nuclear antibodies (ANA). 16 In addition, these patients have tested positive for rheumatoid factor, smooth muscle antibodies, double-stranded DNA antibodies, and anti-neutrophil cytoplasmic antibodies (ANCA). Other researchers have found RPF associated with small to medium-vessel vasculitides, such as Wegener’s and polyarteritis nodosa.17–19 In a study of five patients with concomitant gastrointestinal vasculitis and chronic periaortitis, Salvarani et al. found that histological data and imaging indicate the possibility of chronic periaortitis and its separate disease entities as a type of large-vessel vasculitis. 20
More recently, researchers have found an association between RPF and IgG4-related systemic disease. In a review of 14 published cases of IgG4-related RPF, Stone found that all of the cases identified as IgG4-related, based on a serology, were male, which confirmed prior evidence of male predominance in IgG4-related systemic diseases. 21 Other studies have demonstrated a preponderance of IgG4-related RPF cases among those previously identified as ‘idiopathic’. In a prospective trial involving 14 patients with RPF, Vaglio et al. found 29% of RPF cases were due to IgG4-related disease. 22 Further, using a diagnostic criterion of a minimum of 40% as the cut-off of the IgG4/total IgG ratio, Khosroshahi et al. found that 57% of their RPF cases were IgG4-related. 23 This diagnostic criteria had been utilized in a prior study and was shown to be supported by histopathological biopsies in this particular study. Khosroshahi et al. noted that IgG4-related RPF biopsies showed a prominence of storiform fibrosis compared to the non-specific hyalinized fibrosis sheets seen in non-IgG4-related RPF. 23 Other findings notable for IgG4-related RPF include obliterative phlebitis and eosinophilia. In a study of clinical characteristics of IgG4-related RPF, Chiba et al. found that the majority of their patients complained of symptoms related to associated autoimmune illnesses rather than the more classic abdominal and/or back pain associated with idiopathic RPF. 24
Our patient’s retroperitoneal biopsy demonstrated histopathology typical of RPF with aggregates of lymphocytes and fibrotic tissue. The serum IgG4 level was not elevated and the patient did not have eosinophilia or other clinical features to suggest IgG4-related disease. IgG4 staining of the tissue biopsy would, however, have been helpful to definitively exclude this possibility. As Mitchinson and Parums previously reported, our patient had significant atherosclerotic disease of the abdominal aorta and this may have contributed to the underlying pathology behind the development of RPF.4,5 Although active vasculitis was not demonstrated in our patient, the use of a more sensitive measure such as FDG-PET may have been useful in further investigating this possibility.
Clinical presentation
Clinically, the most common primary complaint in RPF is abdominal pain. The pain may be dull and constant and is not worsened by movement, meals or palpation. 8 Constitutional symptoms, such as lower-extremity edema with possible deep venous thrombosis, constipation, scrotal swelling, etc., constipation, scrotal swelling, hydrocele or varicocele, polyuria or urinary frequency, hematuria, polyuria and, in late stages, evidence of ureteral obstruction. 8 In our case, the patient experienced vague but persistent, undulating abdominal pain associated with fatigue and claudication.
Arterial complications, such as claudication, can occur in up to 2% of cases as demonstrated in a recent large retrospective observational study conducted by Kermani et al.8,25,26 Case reports, however, have been infrequent and the true incidence of arterial compromise associated with RPF is uncertain. Arterial insufficiency may result from associated atherosclerosis, external compression from RPF mass effect, or from luminal compromise related to vasculitis. Arterial occlusion and acute limb ischemia are uncommon. Recently, there were two reported cases of perivascular inflammatory changes involving the coronary arteries in patients with idiopathic RPF. 27 Both cases had negative stress tests and were for the most part asymptomatic. 27
Venous complications related to RPF include deep venous thrombosis (DVT) and venous congestion. The incidence of venous complications is largely uncertain. The few published case reports suggest a predilection for venous complications among patients with advanced stages of RPF that encase the caval and iliac venous structures.28–30 For instance, in a case published by Agrawal et al., a 62-year-old with RPF initially treated with prednisone subsequently developed left-sided lower popliteal DVT and pulmonary emboli related to a periaortic retroperitoneal mass extending from the kidneys to the encase the left iliac veins. 29 Previous case reports have described the presentation of occult DVT as edema that is unresponsive to other treatment (e.g. relief of obstructive hydronephrosis, steroid therapy for mass compression) and venous claudication in chronic cases.28,31
Differential diagnosis
The differential diagnosis for a retroperitoneal mass encasing the abdominal aorta includes infection (e.g. histoplasmosis, tuberculosis), malignancy (e.g. lymphoma), sarcoidosis, history of abdominal or pelvic radiation therapy, postoperative changes, retroperitoneal hematoma, and RPF. Distinguishing these conditions depends on the clinical presentation and findings on physical examination, laboratory testing, and diagnostic imaging. Tissue biopsy is required to help determine the correct diagnosis and to exclude the possibility of malignancy.
Histoplasmosis and tuberculosis are usually manifest by fever, cough, and pulmonary involvement. Para-aortic lymphadenopathy can be seen in both conditions and can be confused with malignancy. However, the lack of fever, respiratory symptoms, or history of conspicuous travel or exposure made the diagnosis of histoplasmosis and tuberculosis unlikely in this patient. The presence of a large circumferential periaortic mass, particularly when in combination with mediastinal lymphadenopathy, is concerning for non-Hodgkin’s lymphoma. Circumferential involvement and anterior displacement of the aorta are more commonly seen with malignancy than with RPF. B symptoms (i.e. fever, night sweats, and unintentional weight loss) are variably present in patients with non-Hodgkin’s lymphoma. Metastases to the para-aortic lymph nodes can be seen in a variety of malignancies including, prostate, cervical, testicular, and renal cell carcinoma, but the appearance is more often nodular than confluent. Our patient did not have B symptoms, but the presence of a large, confluent para-aortic mass was concerning for malignancy. Sarcoidosis is an inflammatory disease of unknown origin that is characterized by the formation of non-caseating granulomas. Pulmonary and mediastinal involvement is most common, but occasionally extensive lymphadenopathy with periaortic involvement can be seen. In this patient, the lack of pulmonary or mediastinal involvement and the confluent appearance of the periaortic mass reduced the concern for sarcoidosis. Our patient did not have a history of prior abdominal surgery or radiation therapy and did not have risk factors that would predispose her to retroperitoneal hemorrhage. Finally, Erdheim-Chester disease is a rare non-Langerhans histiocytic disorder, most commonly seen in men, that has been associated with RPF. It is characterized by multifocal osteosclerotic lesions of long bones and usually presents with bone pain, neurologic features, diabetes insipidus, or constitutional symptoms.
This case report highlights the association of idiopathic RPF with atherosclerotic disease of the abdominal aorta and branch vessels, as well as the potential relationship of RPF with large-vessel vasculitis. While the presence of left groin pain was likely related to local mass effect, the complaint of left lower-extremity pain with exertion was suspicious for intermittent claudication. The mechanism for arterial insufficiency was likely related to underlying atherosclerotic disease and concomitant RPF-mediated arterial compression. The improvement in claudication with steroids is likely related to the regression of mass effect. However, as RPF has been strongly associated with chronic periaortitis and large-vessel vasculitis, it is also possible that vascular inflammation contributed to the patient’s arterial insufficiency. Adult-onset large-vessel vasculitides (e.g. Takayasu’s arteritis, giant cell arteritis) are more likely to manifest in the thoracic aorta and branches with cranio-ischemia and upper extremity claudication. Similarly, abdominal pain in the presence of mesenteric artery atherosclerotic disease required the consideration of mesenteric ischemia. However, the patient’s presentation and diagnostic study results were not otherwise typical of acute or chronic mesenteric ischemia.
Diagnostic testing
Laboratory testing
Initial laboratory abnormalities may include abnormal renal function and urinalysis findings secondary to urological complications, elevated acute phase reactants, and positive autoimmune studies. Kermani et al. found renal insufficiency among 42% of their tested population and an average hemoglobin level of 12.6 g/dL. 26 Similarly, Scheel and Feeley found that the average RPF patient was mildly anemic. 7 Though the majority of RPF patients may have elevated acute phase reactants, there remains a significant portion of the RPF population who may have normal C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) values, such as the 42% of patients in the Kermani et al. cohort. 26 Other laboratory values of interest may include eosinophil percentage and IgG4 levels due to the association of RPF with IgG4-related diseases. Laboratory studies are, however, non-specific and are not sufficient to establish the diagnosis of RPF.
Radiological imaging
Imaging studies are critical to the detection and monitoring of RPF disease course. Ultrasound and plain X-ray films may demonstrate findings secondary to the indirect effects of RPF, but are not usually the studies of choice for diagnosis. In some cases, ultrasonography can be used to monitor RPF progression indirectly via assessment of ureterohydronephrosis. 9
CT and magnetic resonance imaging (MRI) are considered the imaging studies of choice for diagnosis. Both modalities reveal RPF as a para-aortic mass around the anterior and lateral sides of the abdominal aorta, extending from the origin of the renal arteries to the common iliac arteries. 8 In some cases the inferior vena cava may be encased and/or compressed and often the ureters are encased and externally compressed.9,32,33 On CT, the mass should appear isodense to muscle and be enhanced with contrast, especially during early inflammatory stages. 8 On MRI, the fibrotic mass may appear hypointense on T1-weighted images but hyperintense on T2-weighted images, particularly in the early inflammatory stages.9,32 Additionally, these modalities may reveal not only the extent of fibrotic involvement (e.g. involvement of the thoracic aorta), they may also suggest secondary causes, such as malignancy. Arrive et al. found that the presence of non-homogenous signal on T2-weighted images of the fibrotic mass may suggest malignancy as the etiology.9,34 Kottra and Dunnick found that anterior displacement of the aorta and lateral displacement of the ureters occurred more often in cases of malignancy rather than idiopathic RPF. 32
CTA and MRA are particularly useful in identifying vascular complications associated with RPF. In addition to identification of underlying atherosclerotic disease and aneurysm, both CTA and MRA can demonstrate evidence of vasculitis. Arterial wall thickening, mural enhancement and the presence of low-attenuation rings on delayed images are CTA findings that can be seen in patients with active large-vessel vasculitis. MRA findings of active vasculitis include vessel wall edema on T2-sequence and mural contrast enhancement and vessel wall thickening on T1-sequences. MRA has also been shown to have particular application in patients with venous complications associated with RPF. One radiological study of venous thrombosis conducted by Finn et al. found that MRA imaging provided more comprehensive information compared to catheter venography. 35 This finding was subsequently echoed in a case report from Japan regarding large-vessel thrombosis in a patient with RPF. 28
Another potentially useful imaging modality is FDG-PET. Although its sensitivity is too low for it to be used as an initial diagnostic study, 36 numerous recent publications have reported its benefit as a functional imaging technique to detect metabolic activity of the retroperitoneal mass and the presence of active vasculitis.8,36 Additionally, the whole body image created by FDG-PET can help to evaluate the full extent of disease activity and the presence of other findings that might be characteristic of competing diagnoses (e.g. lymphoma, tuberculosis, metastatic cancer, and sarcoidosis).
In our patient, the diagnosis was first suggested by CTA imaging. MRA was secondarily obtained to determine the severity and location of arterial obstructive disease and to further assess the possibility of vasculitis. Serial CTA imaging proved helpful in determining response to therapy.
Retroperitoneal mass biopsy
Tissue biopsy is strongly encouraged to help establish the diagnosis of RPF and to exclude the possibility of malignancy or other competing diagnoses. This is particularly important in patients who have constitutional symptoms, lymphadenopathy, or an atypical appearance to the mass. The biopsy can also provide insight into potential etiology, such as IgG4-related disease, that may require further testing.
Non-invasive physiologic vascular testing
To our knowledge, there are no reported cases of RPF in which ABI testing was used to assess or monitor disease status. In our case, ABI testing was useful for several reasons. First, non-invasive physiologic testing allowed us to assess the extent of underlying stenotic disease and better understand the etiology of the patient’s leg discomfort. Second, ABI testing revealed the clinical significance of arterial flow obstruction as our patient had only mild peripheral artery disease at rest but clear evidence of activity-limiting flow reduction with exertion. Third, serial ABI testing in coordination with CTA imaging allowed us to quantify the relative contribution of RPF-mediated arterial compromise as compared to that caused by chronic underlying atherosclerotic narrowing.
Treatment and prognosis
The disease course for RPF is typically chronic-relapsing. The rarity of this condition precludes large randomized control trials. Treatment is primarily targeted toward the reduction of fibrotic mass thickness and its associated symptoms. Owing to the delayed recognition of many cases, invasive procedures meant to relieve ureteral obstruction and treat renal dysfunction are sometimes necessary. From a vascular standpoint, revascularization may be required in the management of patients with RPF. In advanced disease, arterial revascularization may be necessary for treatment of claudication, and thrombolysis or thrombectomy with adjuvant endovenous therapy may be required for management of acute venous thrombosis. In less advanced disease, it is our opinion that revascularization procedures should be avoided as first-line therapy as medical therapy is often sufficient to achieve relief of complications associated with RPF-mediated compression. Kermani et al. reported that only 6% of their cohort of 185 patients had vascular stenting or surgery. 26 In general, revascularization procedures should be avoided for the treatment of external arterial compression unless the condition is refractory to medical therapy and symptoms are severely lifestyle disabling, or there are complications of acute or chronic arterial insufficiency. A case study by Vorwerk et al. illustrated such a situation in which stenting of caval vessels resulted in temporary relief of symptoms with subsequent return due to thrombosis, requiring thrombectomy and further stenting to resolve. 37 Surgical interventions should be limited to repairs of the other forms of periaortitis involving aortic aneurysm rather than idiopathic RPF, or in specific patients who have failed medical therapy for venous claudication. In an older study published by Rhee et al., Mayo surgeons reported the benefit of venous reconstructive surgery in two patients who had chronic, disabling venous claudication. 31 However, the authors cautioned against using venous reconstructive therapy except in patients who have intractable edema or claudication despite medical therapy and who have not demonstrated adequate venous collateralization after at least 1 year from an acute thrombosis episode. 31
The mainstay of medical therapy has been systemic corticosteroids. On a case-to-case basis, patients receive steroids at doses varying between 30 and 60 mg daily, which is then tapered slowly with possible maintenance therapy – where as long as 2 years is recommended.38–40 The goals of steroid therapy are to stop progression of the fibrosis, reduce fibrotic mass effect, quell acute-phase reactions and subsequent systemic effects, and to prevent recurrence. 8 Oftentimes, adjuvant immunosuppressant therapy may be used, particularly in steroid-refractory patients and for the purpose of glucocorticoid sparing. There has been anecdotal success published using methotrexate, azathioprine, and cyclophosphamide. However, use of these immunosuppressive drugs is hampered by their side-effect profiles. Recently, the combination of prednisone and mycophenolate mofetil has been shown to be effective with fewer side effects than other immune-modulating medications.41–43 Scheel et al. found regression of periaortic masses and non-recurrence of ureteral obstruction following J-stent removal in a small case study of seven patients. 44 Another immune-modulating medication that has had success in managing some patients with RPF is tamoxifen.45–48 Vaglio et al. conducted an open-label randomized controlled trial that compared prednisone alone to tamoxifen alone for maintenance of remission of idiopathic RPF, including perianeurysmal RPF, following a 1-month induction with prednisone. 48 The trial demonstrated that prednisone was more effective in preventing relapse, leading to the recommendation that prednisone should be the initial treatment of choice in idiopathic RPF. 22
Conclusions
Our case of a middle-aged female who presented with abdominal discomfort and lower-extremity claudication secondary to idiopathic RPF and arterial obstructive disease highlights several issues regarding this rare, not well-defined disease process. First, it illustrates the complicated interplay of existing atherosclerotic disease and secondary compression from RPF mass effect in causing symptoms. Second, it demonstrates the potential role that non-invasive vascular assessment can play in monitoring therapy response. Finally, though it is rare for claudication to be a presenting symptom in patients with RPF, this case demonstrates the need to include RPF in the differential diagnosis for lower-extremity discomfort with ambulation as the treatment for RPF greatly differs from that of other causes of claudication.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
