Abstract
Renal artery fibromuscular dysplasia (FMD) may be underdiagnosed. We evaluated the prevalence of FMD in CORAL (Cardiovascular Outcomes in Renal Atherosclerotic Lesions) renal artery stent trial participants, in which FMD was an exclusion criterion for inclusion. We also evaluated the prevalence of FMD in a relatively healthy population of patients undergoing computed tomographic angiographic (CTA) screening for renal donor evaluation. All renal donor CTAs performed at our institution from January 2003 through November 2011 were retrospectively reviewed for the presence of FMD along with patient sex and age. These results were compared to angiographic core lab (ACL) findings for the CORAL trial. The CORAL ACL database contained 997 patients (mean age 69.3 years; 50% female). Fifty-eight (5.8%) CORAL trial patients (mean age 71.8 years; 75.9% female) demonstrated incidental FMD. The renal donor cohort included 220 patients (mean age 40.5 years; 64.5% female). Five (2.3%) demonstrated FMD (mean age 48.6 years; all female). The odds of FMD in the CORAL cohort were 2.65 times that seen in the renal donor cohort (95% CI: 1.12, 7.57). In
Introduction
Fibromuscular dysplasia (FMD) is a vascular disease of medium-sized arteries, which most commonly affects the renal, extra-cranial carotid, and vertebral arteries, but has also been reported to involve the brachial, mesenteric, hepatic, iliac and coronary arteries. 1 Clinical manifestations of FMD reflect the severity of the disease and the vascular bed involved. The most frequent subtype of FMD is multifocal, which is described as having a ‘string of beads’ appearance on angiography.2,3 FMD has been associated with the development of arterial stenosis, occlusions, and dissections as well as aneurysm formation. FMD is a non-inflammatory and non-atherosclerotic vascular disease for which the exact pathogenesis, natural history and prevalence is not fully known.1,3
Indeed, there is considerable uncertainty of the prevalence and age distribution of FMD in the general population. Prior reports on FMD prevalence range between 1% and 6.6%.4–9 It is even less clear as to the prevalence of renal artery FMD in older patients. We sought to compare the prevalence of FMD in two discrete populations: a younger and healthier population, and an older population with a higher prevalence of atherosclerosis and hypertension.
Materials and methods
Renal donor cohort
Institutional review board approval was obtained. For evaluation of the younger, healthier population, we retrospectively reviewed the reports of all computed tomographic angiographic (CTA) studies at a single institution performed on renal donor candidates. The medical imaging database was queried for CTA studies with the indication of ‘potential renal donor’ at our institution between January 2003 and November 2011. These studies were initially performed and interpreted by fellowship-trained cardiovascular radiologists. The diagnosis was made using the classic description of multifocal FMD (medial fibroplasia) seen as non-ostial alternating webs and stenosis with areas of post-stenotic dilation, or focal FMD (non-medial) with a single web or tubular stenosis. The presence or absence of FMD was noted for each study at the initial interpretation as part of our institutions reporting protocol for renal donor candidates. These studies were performed on either GE (16-row detectors) or Siemens (64-row detectors) CT scanners at a slice thickness of 0.625 mm. All studies were performed in the pre-contrast, arterial, and 5-minute delayed phases. Multi-planar reformatted (MPR), maximum intensity projection (MIP) reconstructions, and source images were used for interpretation. The patients were evaluated for the presence or absence of FMD, age, and sex.
CORAL trial cohort
The older, hypertensive and more atherosclerotic population was represented by patients participating in the CORAL (Cardiovascular Outcomes in Renal Atherosclerotic Lesions) trial. CORAL was a randomized, multicenter clinical trial that compared optimal medical therapy alone versus renal artery stent placement plus optimal medical therapy in the management of patients with atherosclerotic renal artery stenosis equal to or greater than 60%. 10 The CORAL trial population consisted of patients 18 years or older with either documented hypertension (systolic ≥ 155 mmHg) on greater than two antihypertensive medications or chronic kidney disease. 11
The investigational sites initially evaluated the symptomatic patients for renal artery stenosis using conventional angiography and determined study eligibility based upon clinical and imaging criteria. An exclusion criterion for enrollment into the CORAL trial was the presence of renal artery FMD. 11 Patients who were randomized for stent treatment received the intervention at this time. The images were then sent to the CORAL trial angiographic core lab (ACL). These angiograms were then interpreted by one of two interventional radiologists. The CORAL ACL provided blinded interpretations of the angiograms submitted by the investigational sites without access to any associated clinical information. The presence or absence of FMD was noted for each catheter-based angiogram and was recorded in the ACL database.
We retrospectively reviewed the ACL database of renal artery angiograms submitted for inclusion in the roll-in and randomized phases of the CORAL trial. All patients with sex and age data with angiograms submitted to ACL were included in this study. Patients enrolled in the CORAL trial via the non-invasive pathways (duplex ultrasound, CTA, or MRA) who did not have a catheter-based angiogram or any patient for whom we had no sex or age data were excluded from this study. The original angiographic images were not re-reviewed for the current study.
Statistical methods
Patient sex and FMD status in each cohort were summarized by frequencies and percentages. The exact methods were used for construction of 95% confidence intervals for percentages of patients with FMD. The log-odds ratio for FMD was the statistic of comparison, and the exact statement of the LOGISTIC procedure of SAS version 9.2 (SAS Institute Inc., Cary, NC, USA) was utilized to estimate the log-odds ratio and to generate an exact 95% confidence interval for the log-odds ratio.
Results
Renal donor cohort
CTAs were performed on 220 potential renal donor patients from January 2003 through November 2011 (Table 1). None of these patients had a history of hypertension. There were slightly more females (142 or 64.5%) compared to males (78 or 35.5%). The mean age of these patients was 40.5 years, with a range of 18–73 years. A total of five patients in this population had imaging findings suggestive of FMD. Two CTA studies could not rule out FMD. These two patients went on to catheter-based selective renal angiography, which was negative for FMD. Of the five patients positive for FMD, two were bilateral, one was on the left, and two were on right. One of the right-sided renal artery FMD patients had additional involvement of an accessory right renal artery. The prevalence of FMD in this population was 2.3%. All five positive FMD studies were female, which led to a female prevalence of 3.5%. The mean age of the females with FMD was 48.6 years, with a range of 40–60 years.
Prevalence of renal FMD in a cohort of potential renal donors and in a population of patients with symptomatic renal artery stenosis in the CORAL ACL database.
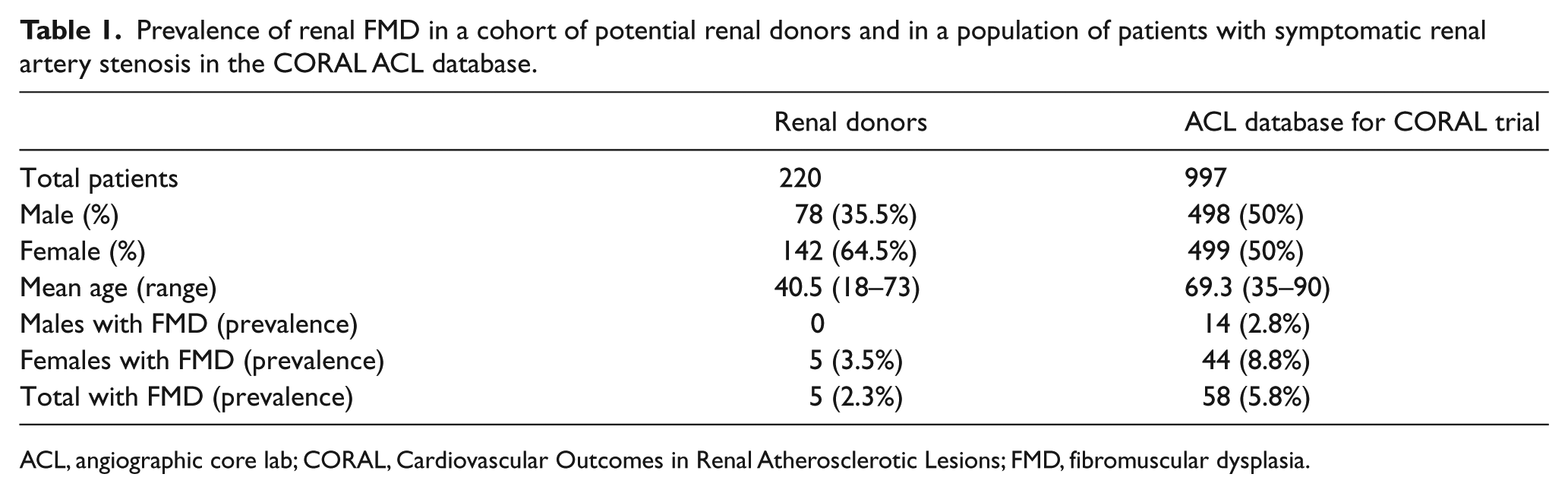
ACL, angiographic core lab; CORAL, Cardiovascular Outcomes in Renal Atherosclerotic Lesions; FMD, fibromuscular dysplasia.
CORAL trial cohort
The ACL database for the CORAL trial consisted of 997 patients (enrolled in CORAL trial = 769, roll-in patients = 228) who qualified for analysis for this study (Table 1). There were two patients who were excluded owing to lack of sex and age data. The sex distribution was 499 females (50.0%) and 498 males. The mean age of these patients was 69.3 years, with a range of 35–90 years. A total of 58 patients had imaging findings suggestive of FMD, for a prevalence of 5.8%. FMD was seen in 44/499 female patients, for a prevalence of 8.8% (95% CI 6.5, 11.7). FMD was seen in 14/498 male patients, for a prevalence of 2.8% (95% CI 1.5, 4.7). The odds for FMD were greater in the CORAL cohort than in the renal donor cohort (odds ratio: 2.65, 95% CI: 1.12, 7.57).
Discussion
As described above, when a patient presented for initial conventional angiographic evaluation for renal artery stenosis, the investigational site made a judgment as to whether the patient met criteria for enrollment (or roll-in) in the CORAL trial. 12 FMD was listed as an exclusionary criterion for enrollment. Of the 4375 patients that where initially screened for the CORAL trial, 87 were not enrolled due to the presence of FMD. 12 Following randomization, patients received medical treatment alone versus stent placement and the images were sent to the ACL for review. It was at this time that the images were re-evaluated for the presence of FMD as well as the other criteria, as specified by the CORAL trial. All of the patients in the ACL database had ostial renal artery atherosclerotic lesions. Additionally, it was interesting to find that there was a 5.8% prevalence of FMD in this cohort of patients in which the physician performing the procedure (either interventional cardiology, interventional radiology, or vascular surgery) did not exclude the patient from trial entry. An example from the CORAL trial ACL with both FMD and atherosclerotic lesions is seen in Figure 1.
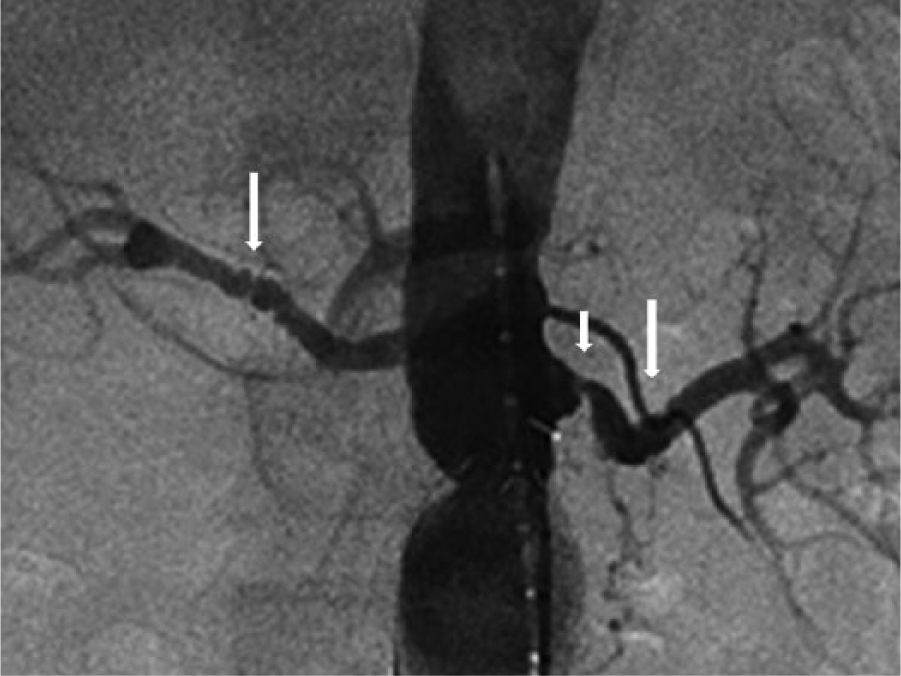
Angiogram from the CORAL trial demonstrating bilateral renal artery FMD (long white arrows) and left renal artery ostial atherosclerotic-related stenosis (short white arrow).
The younger and presumably healthier population of renal donor candidates had a FMD prevalence of 2.3%. This prevalence was similar to a recent study using preoperative CTA in renal donors in which they found a FMD prevalence of 2.6% in 2640 patients. 8 Another large study of renal donor candidates using conventional angiography found a FMD prevalence of 3.8% in 1862 patients. 4 Combining five prior studies4–8 in renal donor candidates (using either CT or conventional angiography) yielded a FMD prevalence of 3.6% in a total of 5478 patients, with a prevalence range of 2.6–6.6% (Table 2).
Prior reports of the prevalence of FMD in the renal donor population.
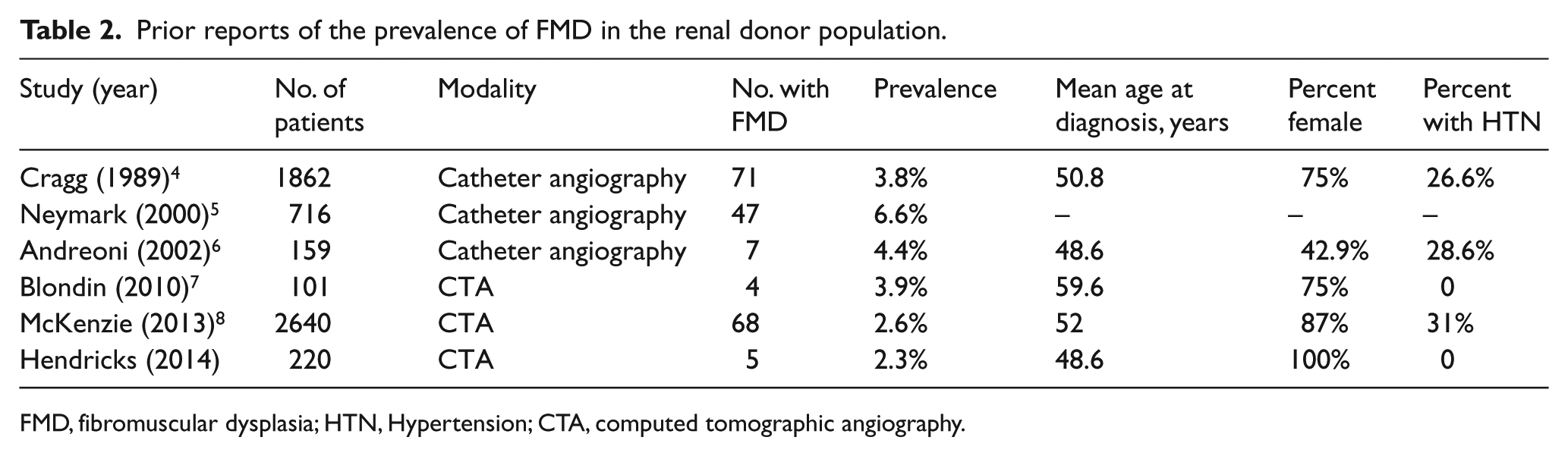
FMD, fibromuscular dysplasia; HTN, Hypertension; CTA, computed tomographic angiography.
It was interesting to find that there was a significantly higher prevalence of FMD (5.8% vs 2.3%, p=0.023) in the older, hypertensive, and atherosclerotic ACL cohort as compared to the younger, healthier renal donor population. This finding is suggestive that there may be underdiagnosis of FMD in older, hypertensive patients with atherosclerotic renal artery stenosis. This finding also raises questions about the natural history of the disease. Currently, it is not clear if FMD arises de novo in the elderly population or whether it is a progressive lesion. There are several prior reports suggesting angiographic progression13–18 based on imaging appearance only. However, these relatively old studies are limited due to the lack of trans-lesion pressure measurements or IVUS. Today we have a poor understanding of the natural progression of FMD, which is one of the 11 research priorities suggested in a recent article by Olin et al. 19
The differential diagnosis of renal artery FMD includes vasculitis, segmental arterial mediolysis, connective tissue diseases, and neurofibromatosis. 3 Vasculitis demonstrates inflammation of the vessel wall along with elevated acute phase reactants, but could be confused with FMD due to its propensity to affect multiple vascular beds and the presence of hypertension in this patient population. Segmental arterial mediolysis (SAM) is a non-inflammatory vascular disorder characterized by lysis of the medial layer of the arterial wall. 20 As best as could be determined from the data analysis, none of the cases in either of our cohorts had any of the other vascular entities for the cause of the findings in the renal arteries.
There are multiple limitations to this study. It is difficult to make direct comparisons of two populations with differing ages and states of health. In addition, for the CORAL trial patients, the ACL had no clinical information to supplement the interpretations of the angiographic images. Another complicating factor in this study is the use of differing imaging modalities for each study cohort for determining the presence of FMD (CTA versus catheter-based angiography). However, several prior studies21–23 have shown that CTA is a reliable diagnostic modality for detecting renal atherosclerotic and FMD lesions. A recent study 23 demonstrated that CTA detected 100% of the 40 FMD lesions that were detected by catheter-based angiography in a population of 21 hypertensive FMD patients. Nevertheless, mild non-symptomatic cases of FMD may not be detected on CTA. Catheter-based angiography remains the gold standard as it allows for anatomic and physiologic evaluation of the degree of stenosis. Additionally, the argument was made that the ACL cohort was ‘biased against’ the detection of FMD, since FMD was supposed to be an exclusion criterion for trial enrollment. However, an argument could also be made that since FMD is the second leading cause of renovascular hypertension, a ‘bias for’ the presence of FMD may be present.
In conclusion, we found a 5.8% prevalence of FMD in our older, hypertensive, and atherosclerotic population represented by patients in the roll-in and enrollment phases of the CORAL trial. The younger, healthier, renal donor population demonstrated a FMD prevalence of 2.3%, which is very similar to the largest series published to date. 8 This current study provides important insight into the prevalence of FMD in two differing patient populations. It also raises questions about the natural history of FMD and diagnostic awareness of this disease among physicians treating older patients with renovascular hypertension. Lastly, the Fibromuscular Dysplasia Society of America is sponsoring a national multicenter registry of patients with FMD. 1 The longitudinal data from this registry should provide ongoing and important insights into the manifestations, outcomes, and natural history of FMD.
Footnotes
Declaration of conflicting interest
Dr Matsumoto reports support from Medtronic, Cook, WL Gore, Boston Scientific, and The Medicines Company; Dr Cooper reports support from Pfizer, AstraZeneca, and the Cordis Corporation; Dr Murphy reports support from Summa Therapeutics; and Dr Cutlip reports support from Medtronic, Boston Scientific, and Abbott Vascular.
Funding
Research reported in this article was supported by the National Heart, Lung and Blood Institute of the National Institutes of Health under Award Numbers U01HL071556, U01HL072734, U01HL072735, U01HL072736, and U01HL072737. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Drug for this study was provided by AstraZeneca (Wilmington, Delaware), device support was provided by Cordis/Johnson&Johnson (Bridgewater, New Jersey), and supplemental financial support was granted by both Cordis/Johnson&Johnson and Pfizer, Inc.
