Abstract
Arterial stiffness is associated with cardiovascular events and mortality. Lifestyle factors such as physical activity (PA) may reduce arterial stiffness. The purpose of this study is to determine the impact of change in PA on 1-year change in arterial stiffness in 274 overweight/obese sedentary young adults. The Slow Adverse Vascular Effects of excess weight (SAVE) trial was a study evaluating the relationships between weight loss, dietary sodium, and vascular health. PA was measured with the ActiGraph AM7164 accelerometer. Intensity of activity was determined using established cut-points. Arterial stiffness was assessed by brachial–ankle pulse wave velocity (baPWV) using an automated device. Analysis of covariance compared changes in total accelerometer counts, minutes/day in light-intensity PA (LPA), moderate-to-vigorous PA (MVPA), and sedentary time, by categories of change in baPWV. Models were adjusted for time since baseline visit, age, sex, race, homeostatis model of assessment of insulin resistance, mean arterial pressure, heart rate, and weight change. Total accelerometer counts and time spent in MVPA increased from baseline to 12 months while time spent in LPA significantly decreased. Mean baPWV was similar at each time point. Those who showed decreased baPWV also showed an increase in total accelerometer counts per day and time spent in MVPA in the fully adjusted models (p<0.001). Changes in sedentary time and time spent in LPA were not associated with changes in baPWV. These results indicate that even modest increases in MVPA can reduce arterial stiffness, a risk factor for future cardiovascular events.
Introduction
Stiffening of the arterial vessels is an age-related process associated with increased risk for cardiovascular events.1,2 While age-related increases in arterial stiffness have been observed even in healthy adults, modifiable lifestyles factors such as obesity and physical inactivity are associated with accelerated vascular aging.3,4 For example, obese individuals have been shown to have 5.2 cm/s higher pulse wave velocity (PWV) than non-obese individuals, 5 which is associated with 5–10 years of accelerated vascular aging. 6 Even among young adults free from overt conditions, obesity appears to be associated with arterial stiffness7–9 although not all studies agree. 10 Weight loss has been shown to reduce cardiovascular risk factors, including arterial stiffness.11–13 In addition, physically inactive individuals have been shown to have stiffer arterial vessels than physically active individuals.14,15
The Slow Adverse Vascular Effects of excess weight (SAVE) trial was a randomized controlled trial examining the effects of weight reduction, through diet (calorie restriction) and physical activity, and dietary sodium reduction on vascular health. The trial showed that sodium restriction offered no greater impact on arterial stiffness above weight loss through diet and physical activity alone. 16 However, the independent effects of physical activity are unknown.
Studies have shown that aerobic training can decrease arterial stiffness. 17 For example, in a study of 179 women randomized to either a low calorie diet or a structured moderate-intensity aerobic exercise intervention (90 minutes 3 days/week), 6 those randomized to the exercise arm had greater reductions in PWV (−1 cm/s vs −6 cm/s, p=0.004) compared to women in the diet arm. Other studies have found similar results; however, most have used structured exercise training regimens and did not assess total movement. Total movement consists largely of low-intensity, unplanned, and unstructured activities and has been associated with cardio-metabolic outcomes independent of physical activities of moderate intensity. 18 Since little is known about the association between total movement and arterial stiffness, the purpose of this study is to determine if change in total physical activity is associated with change in arterial stiffness independent of weight loss, and whether that association differs by intensity of activity, in young overweight or obese sedentary adults.
Methods
Study population
The Slow Adverse Vascular Effects of excess weight (SAVE) trial is a randomized controlled trial examining the effects of weight reduction, through dietary and physical activity changes, and dietary sodium reduction on improving vascular health (NCT00366990). A detailed description of the study design has been reported elsewhere. 19 In brief, the SAVE study included men and women aged 25–45 years who were physically inactive and overweight to class II obese. Participants were recruited from June 2007 through May 2009 in Pittsburgh, Pennsylvania using mass mailing. Individuals were excluded if they: (1) had known comorbid conditions (i.e. diabetes, hypertension, atherosclerotic disease, or inflammatory conditions); (2) were current users of cholesterol-lowering, antipsychotic, or vasoactive medicines or devices; or (3) were pregnant, breastfeeding, or had plans for pregnancy during the study period. For the current analysis, we excluded individuals who did not wear the accelerometer for ≥3 days for ≥10 hours per day or did not have data on arterial stiffness at baseline. Data for these analyses were collected at baseline, and at 6 and 12 months’ follow-up. However, the accelerometer was only provided at baseline and 12 months’ follow-up. All study participants signed an informed consent document approved by the University of Pittsburgh Institutional Review Board in March 2007.
Study design
Individuals were randomized into either a lifestyle or lifestyle plus intervention arm. The lifestyle arm received a 6-month diet and exercise intervention with a weight loss goal of 10% of their baseline weight. Participants were encouraged to increase their physical activity levels to 150–200 minutes per week and reduce their calorie intake to achieve weight loss of 1–2 pounds (0.5–0.9 kg) per week. The lifestyle plus arm received the same diet, physical activity, and weight loss goals, with an additional goal to reduce their sodium intake by 50%. Since both groups received the same physical activity intervention, we decided a priori to combine both groups for this analysis.
Arterial stiffness
Brachial–ankle pulse wave velocity (baPWV) is a mixed measure of both central and peripheral arterial stiffness; however, some studies have shown that baPWV is more strongly related to central than to peripheral PWV. 20 baPWV has been associated with cardiovascular disease (CVD) risk factors, events and mortality.21,22 baPWV was measured non-invasively using an automated device (Colin Co., Komaki, Japan).23,24 The validity and reliability of baPWV assessment with this device has been previously reported. 25 The Ultrasound Research Laboratory at the University of Pittsburgh has found measurement of baPWV with the Colin machine to be highly reproducible (ICC=0.97). All measures were performed in a quiet, temperature-controlled room after a 12-hour fast and abstinence from caffeine. Individuals were also asked to refrain from exercise 30 minutes prior to testing.
Resting blood pressure and heart rate were measured twice after a 10-minute rest using an automated device. To measure baPWV, appropriate-sized blood pressure cuffs were attached to both uncovered arms and ankles. Electrocardiogram clips were attached to both wrists. A phonocardiogram was held in place with a 2-pound (0.9 kg) weight at the fourth intercostal space to the left of the sternum.
PWV was calculated as the distance in centimeters between arterial sites of interest over difference in time (in seconds) that the pressure waveforms travel from the heart to the respective arterial sites. Distances between the brachial and ankle sites were calculated using a height-based algorithm. 24 Time was calculated using the foot-to-foot velocity method of waveforms measured at various sites. 23 The average of two runs was used in this analysis.
Physical activity
Physical activity was assessed objectively at baseline and 12 months using the ActiGraph AM7164 accelerometer (ActiGraph, Pensacola, FL, USA). At baseline, individuals were provided with this uniaxial accelerometer and instructed to wear the monitor on their hip for 7 days during all waking hours, removing during water-related activities. Participants returned the monitor to the University of Pittsburgh the following week at their next scheduled group session. At 12 months, only those participants who wore the monitor at baseline were given another accelerometer during their clinic visit and were provided with prepaid envelopes to mail the monitors back to the University of Pittsburgh. Data were screened for wear time using methods similar to those reported by Troiano et al. 26 A minimum of 10 hours of wear time per day on at least 3 of 7 days was required for data to be considered for further use in calculating weekly estimates of physical activity. 27
Activity counts are output from the accelerometer, which quantify the amplitude and frequency of detected accelerations, and were summed over a 60-second time interval (i.e. epoch). Total accelerometer counts per day (ct/d) were calculated using averaged daily counts detected over wear periods to provide an estimate of total movement volume. The sum of the activity counts in a given epoch is related to activity intensity and was classified based on validated activity count cut-points:28,29 sedentary (<100 counts), light intensity (LPA) (100–1951 counts), moderate (1952–5724 counts), and vigorous (>5725 counts). 29 Since little time was spent in activities of vigorous intensity, moderate- and vigorous-intensity activity (MVPA) were combined (≥1952). Each minute of MVPA, LPA, and sedentary activity were summed separately and divided by the number of valid days of wear time to obtain daily averages.
Covariates
Self-reported information was collected from participants regarding age, sex, race, and current smoking status. Staff measured height and weight using standardized protocols. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared (kg/m2). Mean arterial pressure (MAP) was calculated automatically from the Colin machine.
Laboratory assays were performed on fasting serum samples at the University of Pittsburgh’s Graduate School of Public Health Heinz Laboratory (Pittsburgh, PA, USA). Total cholesterol (TC) and high-density lipoprotein cholesterol (HDL-C) were determined using a enzymatic method that was previously described. 30 HDL-C was determined after selective precipitation by heparin/manganese chloride and removal by centrifugation of very-low-density lipoprotein and low-density lipoprotein cholesterol (LDL-C). 31 LDL-C was calculated indirectly using the Friedewald equation. Triglycerides were assessed enzymatically using a similar procedure to Bucolo and David. 32 Serum glucose was determined enzymatically with a procedure similar to that described by Bondar and Mead. 33 Insulin was measured using a radioimmunoassay developed by Linco Research, Inc. (St Charles, MO, USA). Insulin sensitivity was estimated using the homeostasis model assessment of insulin resistance index (HOMA) derived from the following equation: HOMA (mmol/L × μU/ml) = fasting glucose (mmol/L) × fasting insulin (μU/ml)/22.5. 34 C-reactive protein (CRP) was measured using an enzyme-linked immunoassay (Alpha Diagnostic International, Inc., San Antonio, TX, USA) using laboratory methods described elsewhere. 19
Statistical analysis
Descriptive statistics were computed to summarize study variables at baseline, and 6 and 12 months, and presented as median (interquartile range (IQR)) or mean (standrad errors) for continuous variables and percentages for categorical variables. Whether the changes in body size, cardiometabolic factors, hemodynamic factors, activity levels, and PWV were significantly different from baseline at 6 and 12 months’ follow-up were determined by testing the coefficient for time in a linear mixed model for the measure of interest. Non-normally distributed variables were transformed as necessary before modeling. The intervention arm was included as a covariate in every model for consistency with trial design. Interaction between the intervention arm and time since baseline was tested and included in models only when significant at p<0.10.
Analysis of covariance was used in the main analysis to examine the relationship between change in baPWV and changes in total accelerometer counts, by physical activity intensity (i.e. LPA and MVPA) and sedentary time. Since there are no established thresholds to indicate a clinically significant increase or decrease in baPWV, we created quartiles of change in baPWV from baseline to 12 months’ follow-up. Model 1 adjusted for potentially confounding factors including time (years since baseline), age, sex, race (black/non-black), smoking status (current vs past/never), and intervention group, for consistency with trial design; each of these covariates was included in models if significant at p<0.10. Model 2 further adjusted for MAP and heart rate, since previous analysis of SAVE indicated they mediate effects of weight loss on baPWV. 11 Additionally, we included other potentially mediating factors such as HOMA-IR, CRP, HDL-C, and triglycerides if they were significantly associated with both physical activity and baPWV at p<0.10. In model 3, weight change was added to each model to determine if physical activity was independently associated with changes in baPWV.
Results
The SAVE population consisted of 349 individuals at baseline, of which 339 had data on baPWV and 277 additionally had at least 3 days of accelerometer data with ≥10 hours of wear time. The median number of days with valid accelerometer data was 5.5, with approximately 20% having 7 days of accelerometer data. Individuals with missing accelerometer data at baseline did not significantly differ from those with accelerometer data with respect to any demographic or clinical characteristic, including baPWV.
Overall, SAVE study participants saw improvements from baseline to 6 months for BMI, MAP, CRP, HDL-C, triglycerides, and HOMA-IR (p<0.05) (Table 1). The observed improvements in these factors remained at 12 months with the exception of HOMA-IR. While mean baPWV decreased from baseline to 6 months by ~13 cm/s in the sample, these mean changes were not maintained at 12 months.
Characteristics of SAVE study participants at baseline, and 6-month and 12-month follow-up.
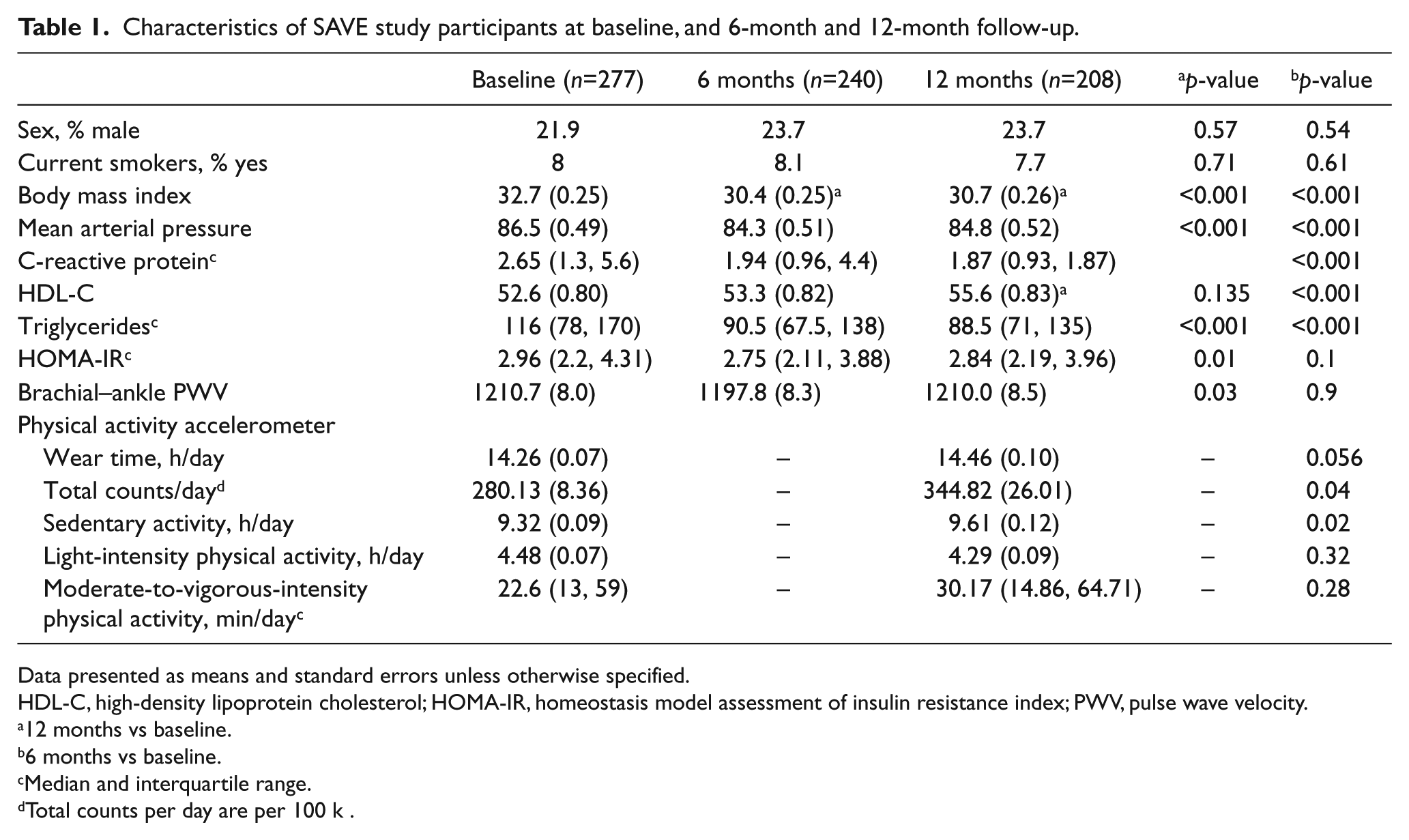
Data presented as means and standard errors unless otherwise specified.
HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostasis model assessment of insulin resistance index; PWV, pulse wave velocity.
12 months vs baseline.
6 months vs baseline.
Median and interquartile range.
Total counts per day are per 100 k .
From baseline to 12 months in the overall SAVE population, total accelerometer counts significantly increased (280.1 vs 344.8 counts/day, p=0.04) (Table 1). Change in total accelerometer counts was more strongly correlated with change in time spent in MVPA (r=0.79; p<0.01) than change in time spent in LPA (r=0.39; p<0.01), or sedentary activity (r=−0.21; p<0.01). Median time spent in MVPA saw a non-significant increase of ~8 minutes/day (p=0.28) (Table 1). LPA was similar at 12 months and baseline (p=0.32). Sedentary activity, however, slightly increased ~12 minutes/day from baseline to 12 months (p=0.02).
Change in baPWV from baseline to 12 months ranged from −293 cm/s to 381 cm/s and was inversely associated with changes in CRP, HOMA-IR, MAP, and heart rate, and positively associated with weight loss (p<0.05). Individuals who showed the biggest decreases in baPWV also showed the biggest increases in physical activity (Figure 1). Specifically, compared to individuals whose baPWV increased ≥53 cm/s, those whose baPWV decreased ≥70.5 cm/s had greater increases in total activity counts/day from baseline to 12 months’ follow-up (−109.8 vs 280.0 counts/day, p<0.001) after adjusting for age, race, intervention group, and time in the study. Further adjustment for MAP and heart rate, and weight loss did not attenuate these findings. Similarly, compared to individuals whose baPWV increased ≥53 cm/s, those whose baPWV decreased ≥70.5 cm/s had greater increases in MVPA from baseline to 12 months’ follow-up (−7.5 vs 12.7 min/day, p<0.001) in the fully adjusted models. Change in time spent in sedentary activity or LPA was not related to change in baPWV.
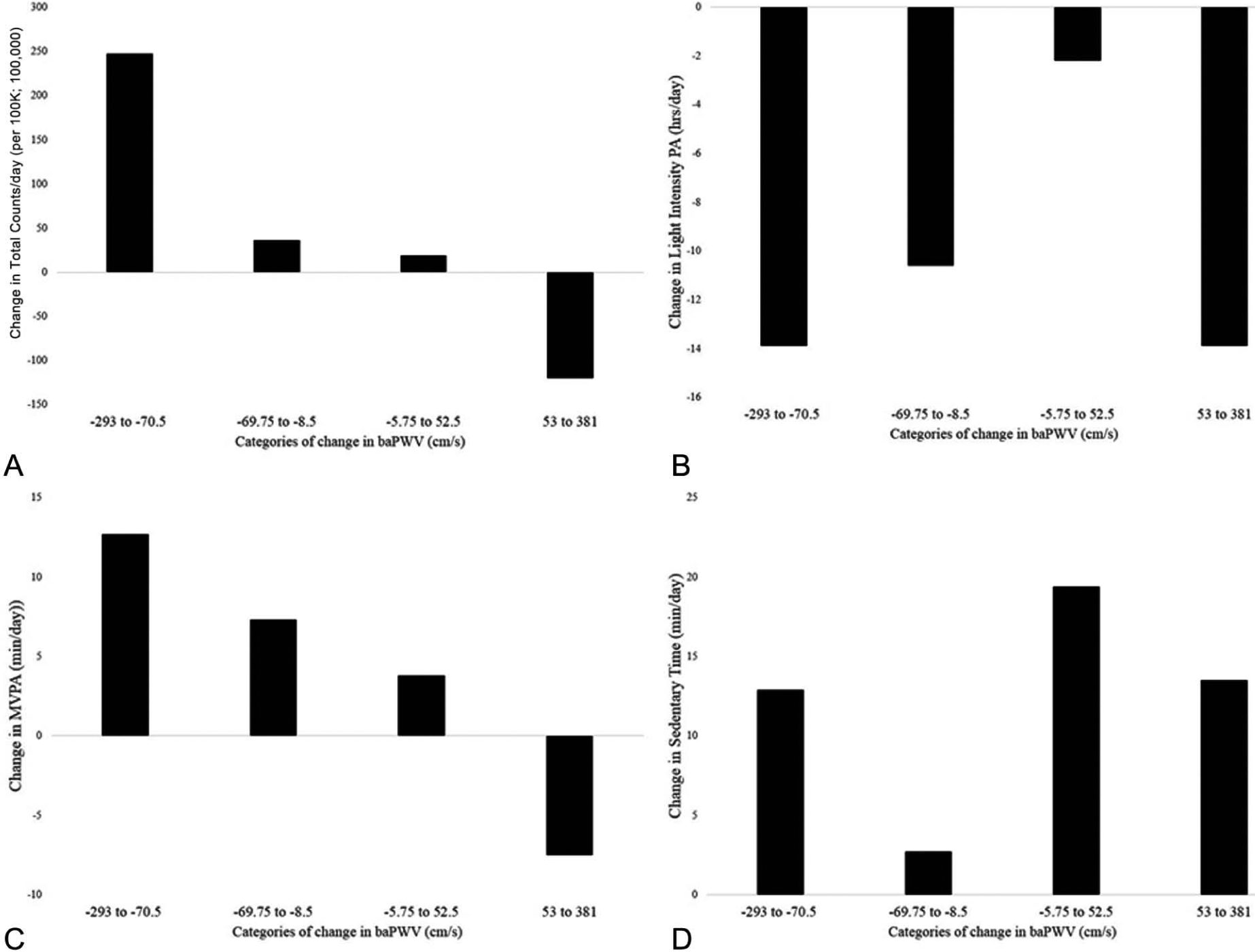
Categories of change in baPWV and change in total accelerometer counts/day (A), light-intensity physical activity (PA) (B), moderate-to-vigorous-intensity physical activity (MVPA) (C), and sedentary time (D) from baseline to the 12-month follow-up. The lowest quartile of change in baPWV compared to the highest quartile of change in baPWV adjusting for age, race, randomization group, time in the study, mean arterial pressure, heart rate, and change in weight: total accelerometer counts/day, p<0.001; light-intensity PA, p=0.99; MVPA, p<0.001; sedentary time, p=0.97.
Discussion
In this study of young overweight/obese sedentary adults, changes in physical activity were associated with changes in arterial stiffness. Specially, we showed that those with the greatest decreases in baPWV had the greatest increases in time spent in MVPA, independent of weight loss. Additionally, increased total volume of movement comprised of the combination of LPA and MVPA was also associated with reduced arterial stiffness. However, LPA or sedentary time was not independently associated with arterial stiffness.
Previous studies have shown that short-term exercise training can reduce arterial stiffness; 35 however, many of these studies were conducted in older adults or in individuals with comorbid conditions.36–39 Few studies have examined the relationship between physical activity and arterial stiffness in a young, normotensive population free from overt disease. The American Heart Association has highlighted in their 2020 strategic impact goals the importance of intervening in low-risk individuals. 40 Targeting young adults who are free from overt conditions or elevated risk factors may greatly impact on the development of future cardiovascular events. Consistent with this concept, we showed that even modest increases in MVPA could reduce arterial stiffness, a risk factor for future cardiovascular events. However, the long-term impact of these observed reductions in arterial stiffness on future CVD is unclear.
In addition to examining the effects of MVPA, we also examined the impact of increasing total movement, which includes LPA, in addition to MVPA, on arterial stiffness. To date, there have been only two reports that have examined the cross-sectional relationship between PWV and various intensities of physical activity.41,42 In both studies, LPA was inversely associated with arterial stiffness in older adults. The current analysis was the first to examine this relationship in a longitudinal study of young adults. We showed that increased total movement was inversely associated with change in arterial stiffness. When adjusting for change in MVPA, the relationship between total movement and arterial stiffness was attenuated, but still statistically significant. Although LPA did not independently associate with arterial stiffness, it modestly contributed to the effects of total movement.
The mechanisms by which physical activity impacts on arterial stiffness are not well understood. In this study, changes in arterial stiffness were significantly associated with changes in MAP, CRP, insulin resistance, heart rate, and weight loss. As previous studies have shown that physical activity can impact on these factors,43,44 we evaluated whether changes in these factors explained the relationship between MVPA and arterial stiffness. Even after adjustment for these factors, MVPA remained significantly related to reductions in arterial stiffness. Other potential mediating factors not measured in this study include oxidative stress, endothelial function and sympathetic nervous system activity. 45 A cross-sectional study by Moreau et al. showed a positive relationship between physical activity and arterial compliance was mediated by lower oxidative stress which may in turn increase nitric oxide availability. 46 Increased nitric oxide availability through regular activity may also attenuate age-related impairment of endothelium-dependent dilation, which may prevent endothelial dysfunction. 47 Likewise, sympathetic nervous system activity has been linked to vascular health. 48 Unfortunately we did not measure these factors in the SAVE study to determine if they mediated the relationship between MVPA and reductions in PWV.
This study has several limitations. First, accelerometer data was not available at 6 months, when activity change was likely maximal. If the changes in activity/inactivity from baseline to 6 months were greater than those observed at 12 months, we would have had more power to investigate the effect of the various components of activity/inactivity on arterial stiffness. Second, nearly half of the study participants with baseline data on arterial stiffness and physical activity did not have physical activity data at 12 months’ follow-up. Additionally, while waist-worn, uni-axial accelerometers provide an accurate measure of ambulatory activities, they fail to capture all physical activities that may contribute to an improvement in health outcomes. 26 Potentially, some light-intensity physical activities, which incorporate upper limb movement, were not assessed in this population, which may have attenuated the association of physical activity with arterial stiffness. Lastly, baPWV is not the gold standard measure of central arterial stiffness as it also includes information from the peripheral arteries. However, baPWV has been shown to be strongly correlated with measures of central arterial stiffness. 25 Furthermore, studies have found that change in baPWV has been shown to be more strongly associated with change in central versus peripheral stiffness. 20 In addition, baPWV is highly reproducible, making it a good measure to use in studies measuring change.
In conclusion, even modest increases in time spent in MVPA can reduce arterial stiffness in a young, overweight, or obese, sedentary population free from overt conditions. This has important implications for primordial prevention, which may greatly reduce future rates of CVD. Since the activity goal used in the SAVE study was to increase time spent in MVPA, with no attempts to change light-intensity activity or sedentary time, future interventions which encourage increases in total movement and reducing sedentary time are needed to determine the impact of these other activity/inactivity components on vascular health.
Footnotes
Acknowledgements
Registry name: Clinical Trial to Reverse Early Arterial Stiffening; Registry number: NCT00366990.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The SAVE study was supported by the NHBI (R01 HL077525-01A2).
