Abstract
Chronic mesenteric artery ischemia can be difficult to evaluate after stent placement. This case describes the use of a pressure wire to demonstrate the hemodynamic significance of in-stent restenosis. Papaverine was used to vasodilate the mesenteric bed, mimicking the post-prandial state. The study (arteriography with pressure wire) and retreatment (covered balloon-expandable stent) was driven by recurrent symptoms avoiding unnecessary ancillary testing.
Introduction
Chronic mesenteric ischemia (CMI) is a treatable, yet possibly life-threatening vascular disease. Typically, two of three mesenteric arteries [celiac artery (CA), superior mesenteric artery (SMA) and inferior mesenteric artery (IMA)] need to be affected to cause symptoms of CMI, although this has been challenged. Lotun et al. described four patients with ischemia of the large intestine from isolated disease of the IMA who benefitted from percutaneous intervention of the IMA. 1 At the other extreme, there are patients with disease involving all three mesenteric arteries that remain asymptomatic. 2 The dilemma of equating an anatomic abnormality with clinical symptomatology becomes challenging. Therefore, clinicians rely on a variety of tests to support treatment decisions that include duplex ultrasound, computed tomography, magnetic resonance, and arteriography. Each of these modalities has limitations, particularly in patients who have previously had a stent placed in their mesenteric circulation. This case illustrates the utility of a pressure wire to assess a stented SMA, with vasodilation to simulate the post-ingestion state, at the time of repeat arteriography.
Case report
An 89-year-old female originally presented with a 30 pound (13.6 kg) weight loss, post-prandial abdominal pain, and evidence of a severe stenosis of her SMA on duplex ultrasound (Figure 1). Physical examination revealed a non-hypertensive, slender, elderly female. She had systolic bruits in the epigastrium that did not change with respiration and her lower extremity pulse examination was normal. Based upon the history, physical examination, and duplex ultrasound, arteriography with possible percutaneous intervention was recommended. The arteriogram showed a patent celiac, SMA stenosis of >70%, and occluded IMA. The SMA stenosis was treated with a balloon-expandable stent (6 mm × 19 mm Omnilink®; Abbott Vascular, Abbott Park, Illinois USA) (Figure 2A–C). Her post-prandial pain resolved and she gained weight.
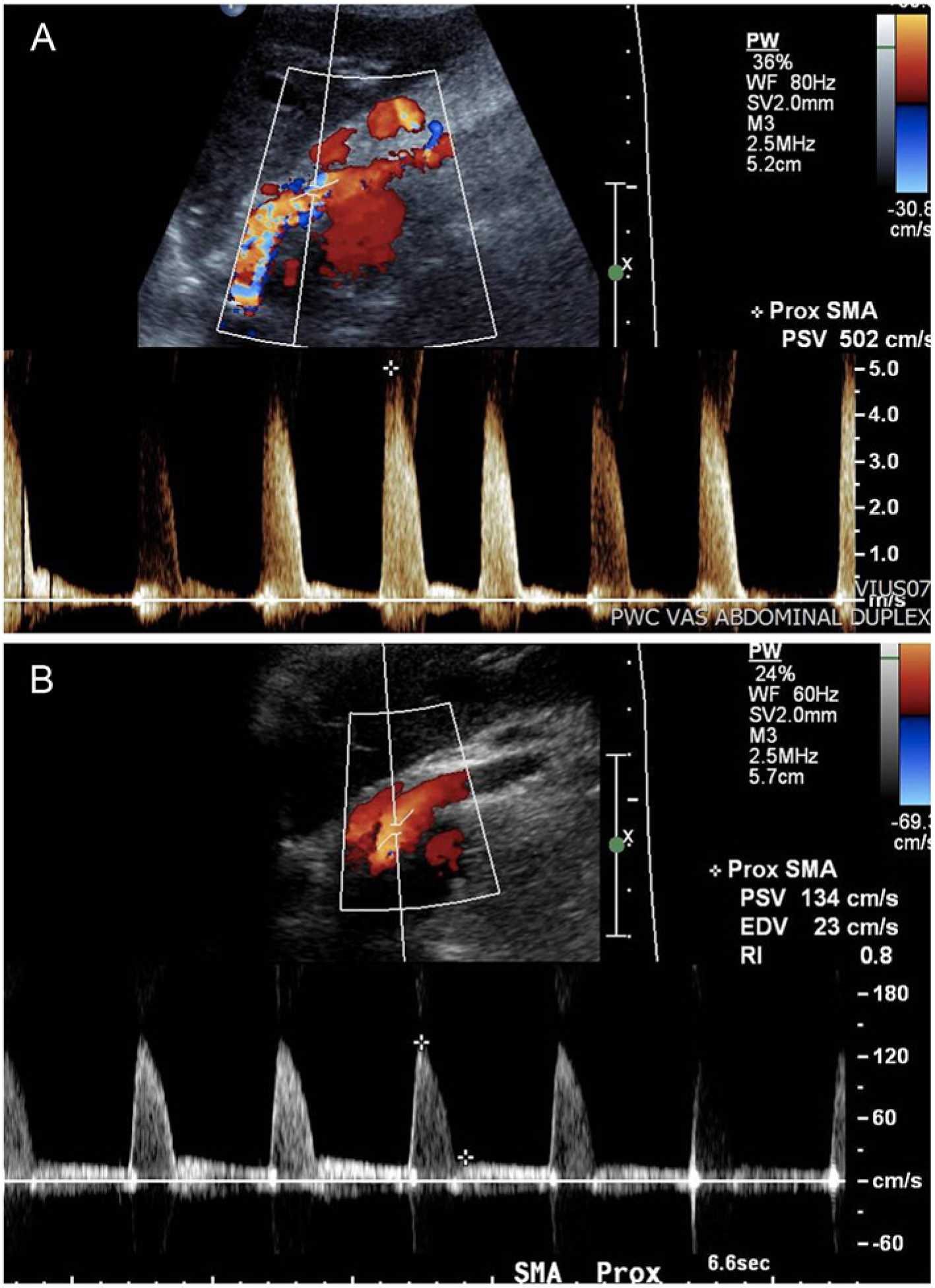
(A) Duplex ultrasound of SMA stenosis before stenting. Note the peak systolic velocity of 502 cm/sec. (B) Duplex ultrasound of the SMA after the second stenting procedure with a peak systolic velocity of 134 cm/sec.
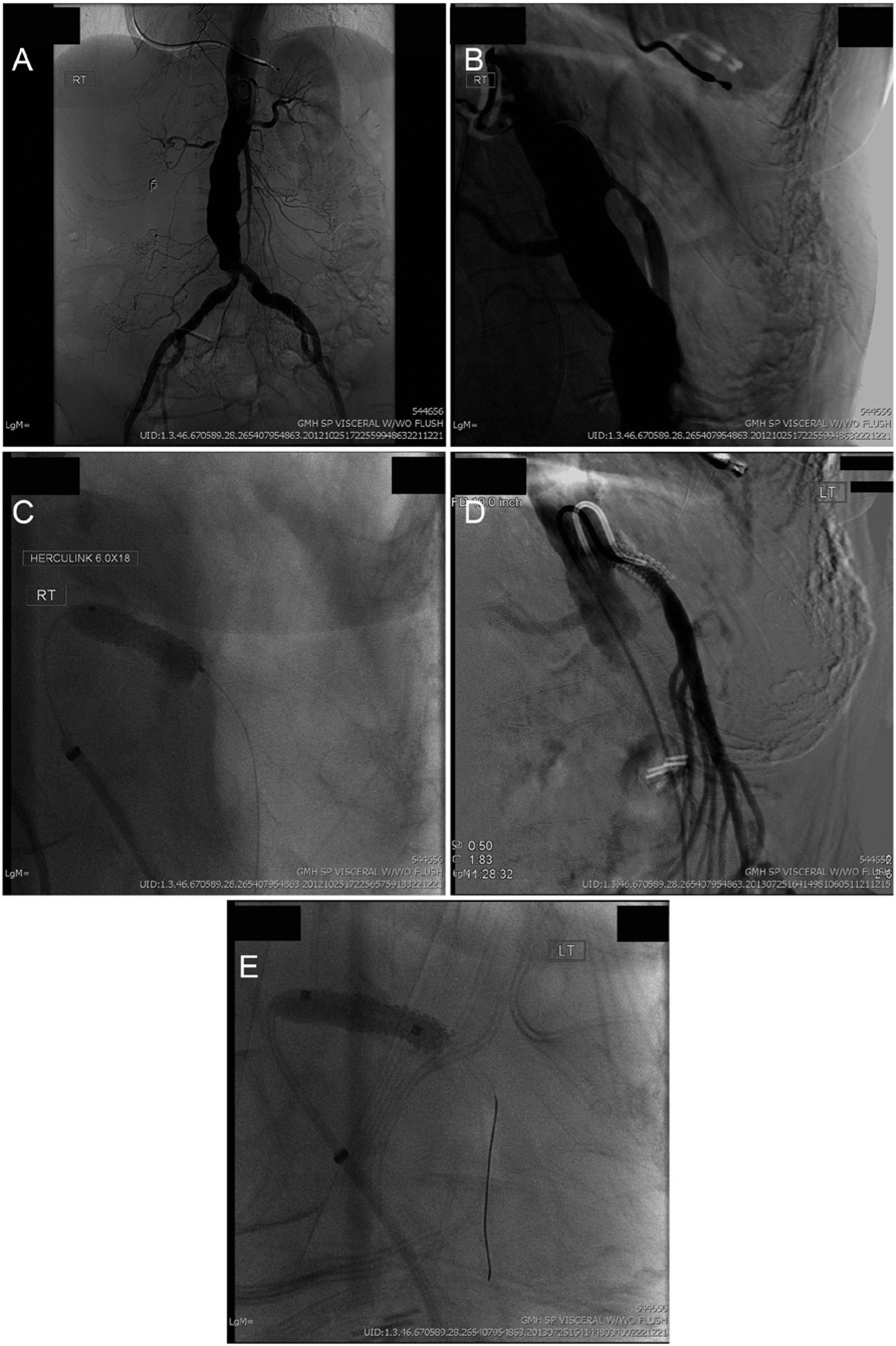
(A) Aortogram demonstrating the absence of the celiac and inferior mesenteric arteries, stenosis of the superior mesenteric artery (SMA) and right renal artery. The patient also has a small abdominal aortic aneurysm. (B) Lateral aortogram showing stenosis of the SMA. (C) Placement of a bare metal balloon-expandable stent to treat the SMA stenosis. (D) Re-angiogram of the bare metal stent (9 months later) in lateral projection. (E) Placement of a covered stent in the SMA to treat the in-stent restenosis.
Nine months later she complained of mild recurrent post-prandial pain without weight loss. She felt like ‘her stent wasn’t working’. Diagnostic testing with duplex ultrasound and computed tomography were considered, but repeat arteriography with hemodynamic evaluation of the stent was chosen. The study showed in-stent restenosis that was difficult to quantify angiographically (Figure 2D,E), but was studied hemodynamically with a 0.014” PrimeWire Prestige® PLUS (Volcano, San Diego, CA, USA). The initial systolic pressure gradient measured was unimpressive (Figure 3A); however, it was significantly enhanced with the addition of 30 mg of intra-arterial papaverine (Figure 3B,C). Papaverine caused significant vasodilation simulating the post-prandial state and established the clinical significance. The SMA was restented with a covered stent (iCAST™, 5 mm × 18 mm; Atrium, Hudson, NH, USA) then post-dilated to 6 mm. Repeat pressure assessment was made with papaverine with improvement of the Pd/Pa ratio to 0.95 and the Pa-Pd (p) gradient. She had resolution of her symptoms once again and follow-up duplex ultrasound showed mildly elevated flow velocity in the SMA.
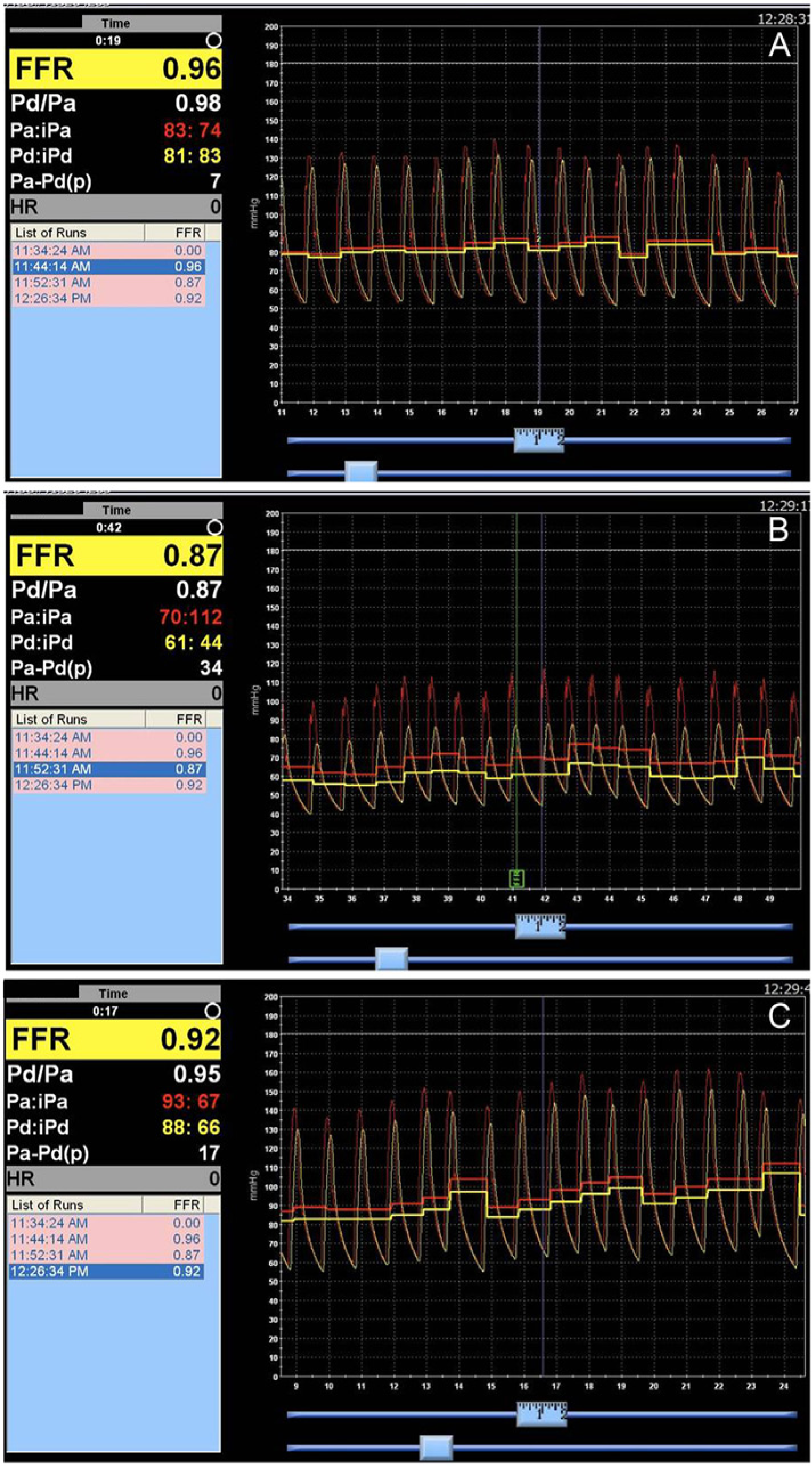
Pressure waveform of the superior mesenteric artery (with in-stent restenosis) before (A) and after (B) administration of papaverine. (C) Pressure waveform after restenting the superior mesenteric artery with papaverine.
Discussion
Duplex ultrasound is often the initial screening test for patients suspected of mesenteric artery stenosis. Others advocate the role of computed tomography with contrast (CTA) as the initial test of choice, 3 since a positive duplex study requires confirmation with an additional imaging study (CTA, magnetic resonance angiography or arteriography) prior to mesenteric artery reconstruction. Consensus does not exist for absolute duplex flow criteria to determine hemodynamic stenoses of the mesenteric arteries. Moneta’s criteria, published in 1991, uses a peak systolic velocity (PSV) of >200 cm/sec for a stenosis of >70% of the CA and >275 cm/sec for the SMA. 4 There is even less consensus as to the appropriate duplex criteria to identify restenosis of the CA or SMA after stenting since the criteria of Moneta tends to overestimate the degree of stenosis. 5 Several alternatives in the literature use a PSV of >330 cm/sec or even >400 cm/sec to identify hemodynamically significant in-stent restenosis of the SMA.6–8
There are limitations to other more advanced imaging methods as well. The metal artifact of a stent can hinder the clarity of not only magnetic resonance (MR) imaging, but CT as well. Calcification of the aorta and the mesenteric arteries are also limitations to the evaluation of native and stented mesenteric arteries. Arteriography alone can be limited by motion artifact, overlap of vessels, obesity, and inexact depiction of the stenosis. Many of these limitations can be overcome at the time of arteriography by assessing the hemodynamics of each vessel. Therefore, in our patient with recurrent symptoms, duplex ultrasound and CT were unnecessary prior to repeat arteriography with pressure gradient evaluation.
Pressures were obtained with the diagnostic catheter at the level of the aorta and compared to the pressure in the renal artery obtained with the pressure wire. Pressure wires are small (0.014”), have a transducer ~3 cm proximal to the tip of the wire and measure pressure that can be reported as a mean, peak systolic pressure, and/or a ratio to a reference pressure both before and after vasodilation. When used for evaluating a coronary stenosis, the term fractional flow reserve (FFR) is measured by using the ratio of the distal pressure (Pd) with the proximal pressure (Pa) after injecting a vasodilator. Papaverine is a short-acting vasodilator drug used most commonly in the treatment of arterial spasm and simulates the post-prandial state.9,10 The ‘normalization’ of the pressure variables after intervention is unknown, but we would advocate a Pd/Pa ratio >0.90 and Pa-Pd (p) value of <20 mmHg, as evidenced in this case. This technique provided the necessary information to evaluate the hemodynamic significance of flow in the SMA after vasodilation before and after intervention.
Conclusion
This case presented several challenges to the clinician: the need to equate anatomy to symptoms, the accurate evaluation of (re)stenosis, and the pertinence of the stenosis. The use of hemodynamic measurement is paramount and the pressure wire with vasodilation simulated the effect of eating on mesenteric flow. The ability to show resolution of the pressure gradient after reintervention was also important to ensure the quality of revascularization. Furthermore, as we become more cost conscious, sometimes going directly to a definitive test is prudent.
Footnotes
Declaration of conflicting interest
The authors have no conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
